Peptide Sciences shut down on March 6, 2026. The company voluntarily ceased all operations, taking offline one of the largest grey-market research peptide vendors in the United States. No orders are being processed. No refunds have been confirmed. Finnrick, the independent testing platform, warned that any site still selling under the Peptide Sciences name is fraudulent.
> Status (last verified March 13, 2026): Peptide Sciences is permanently closed. If you need peptides, compounding pharmacies and legal alternatives are the safest pathway. The February 2026 reclassification restored 14 peptides to Category 1 compounding status.
The closure removes a supplier that sold BPC-157, TB-500, retatrutide, and dozens of other peptides labeled "for research use only" to tens of thousands of customers. Peptide Sciences called it voluntary. But the timing coincides with FDA warning letters to four vendors, ITC exclusion orders blocking tirzepatide imports, and Finnrick testing that gave their retatrutide an E rating across 37 samples.
Key facts:
| Detail | Information |
|---|---|
| Company | Peptide Sciences (peptidesciences.com) |
| Status | Permanently closed (voluntary shutdown) |
| Date confirmed | March 6, 2026 |
| Products affected | BPC-157, TB-500, retatrutide, semaglutide, tirzepatide, and 50+ other peptides |
| Finnrick safety rating | 123 tests across 10 peptides; retatrutide rated E (Bad) across 37 samples |
| Warning from Finnrick | Further purchases under this brand name are fraudulent |
| Legal alternatives | Compounding pharmacies, telehealth clinics |
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Happened to Peptide Sciences
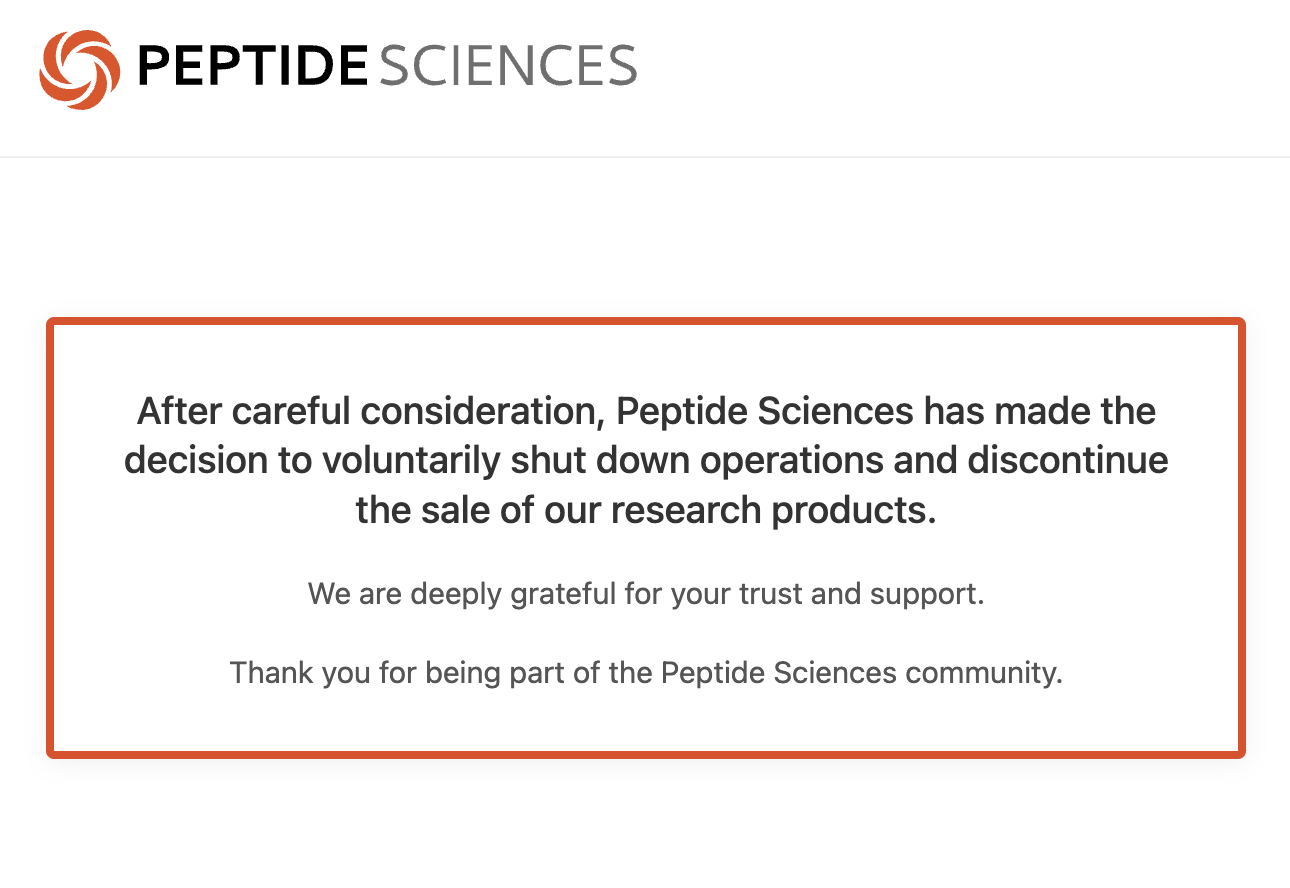
Peptide Sciences posted a notice on its website stating the company had made the decision to voluntarily shut down operations and discontinue the sale of all research products. The site went dark shortly after. The brand's reach was substantial: "peptide sciences" generates over 110,000 Google searches per month, making it one of the most searched peptide vendor names in the United States.
Hims House broke the news on X, noting that Peptide Sciences "was a leading vendor of peptides like BPC-157, TB-500, and retatrutide" and that the website announcement described the decision as voluntary. The post tagged Eli Lilly, Novo Nordisk, and Hims, connecting the shutdown to the broader pharmaceutical landscape around GLP-1 drugs.
Within hours, Finnrick, the independent peptide testing platform that has analyzed 5,521 samples from 177 vendors, updated its Peptide Sciences vendor page with a warning: further purchases from this brand are fraudulent. Any website still claiming to sell Peptide Sciences products after this date is not the original company.
Doctor Murphy, a physician who covers peptide regulation on Substack, published an analysis titled "Grey Market Peptide Giant Disappears." He described Peptide Sciences as "one of the largest and most recognizable suppliers of research peptides" and framed the closure as a signal that the grey market model may no longer be sustainable.
Why Did Peptide Sciences Shut Down
No single event explains the shutdown. Three forces converged: FDA enforcement that escalated from warnings to raids, quality failures exposed by independent testing, and the structural collapse of the "research use only" business model.
FDA Enforcement Reached a Breaking Point
The regulatory crackdown built over two years, accelerating through 2024 and 2025.
In late 2023, the FDA moved 19 widely used peptides, including BPC-157, to its Category 2 list, effectively prohibiting compounding pharmacies from preparing them. Around the same time, Eli Lilly filed an International Trade Commission complaint against 12 vendors selling imported tirzepatide.
In December 2024, the FDA issued warning letters to four peptide vendors: Prime Peptides, Xcel Peptides, SwissChems, and Summit Research. By January 2025, the ITC issued a General Exclusion Order blocking all tirzepatide imports that infringed Lilly's trademark.
By February 2025, the FDA declared the semaglutide shortage resolved, which ended the legal basis for pharmacies to compound it. The SAFE Drugs Act introduced further restrictions. Vendors began scrubbing GLP-1 agonists from their catalogs.
Grey-market vendors like Peptide Sciences operated under a "research use only" label. That legal fiction depended on regulators looking the other way. They stopped looking the other way.
Quality Problems Surfaced in Third-Party Testing
Finnrick tested 123 samples of Peptide Sciences products across 10 peptides. The results painted an uneven picture.
Some products scored well. Ipamorelin earned an A rating across 9 tests with an average score of 9.2/10. PT-141 also earned an A. BPC-157 scored an A with a 7.8/10 average.
Other products failed. CJC-1295 received an E rating (4.3/10 across 10 tests). Tesamorelin also scored E. Most notably, retatrutide received an E rating based on 37 samples tested between December 2024 and March 2026, with test scores averaging below 4. Finnrick assigned a confident rating of E (Bad), their lowest grade. The platform flagged a counterfeit detection among retatrutide samples in November 2025.

Hims House highlighted this data in a follow-up post on X: "Not sure if this was part of the reason they shut down... but worth noting that Finnrick tested 37 samples of Peptide Sciences' retatrutide and gave it a score of E (bad)."
| Peptide | Finnrick Rating | Tests | Avg Score |
|---|---|---|---|
| Ipamorelin | A | 9 | 9.2/10 |
| PT-141 | A | 6 | 9.1/10 |
| BPC-157 | A | 13 | 7.8/10 |
| Semaglutide | A | 14 | 7.7/10 |
| GHK-Cu | B | 6 | 8.1/10 |
| Melanotan II | C | 10 | 6.4/10 |
| Tirzepatide | D | 14 | 6.4/10 |
| CJC-1295 | E | 10 | 4.3/10 |
| Retatrutide | E | 37 | 6.1/10 |
| Tesamorelin | E | 2 | 4.9/10 |
A company selling peptides for "research" can survive regulatory ambiguity. Surviving regulatory ambiguity and documented quality failures at the same time is harder.
The Grey Market Business Model Is Breaking
Peptide Sciences operated in what Doctor Murphy describes as a three-tier peptide ecosystem:
Tier 1: Pharmaceutical peptides. Molecules that completed full FDA approval, manufactured under pharmaceutical-grade standards. Examples: semaglutide (Ozempic), bremelanotide (Vyleesi).
Tier 2: Compounded peptide medicine. Pharmacies producing customized formulations under federal compounding laws, supervised by physicians.
Tier 3: Research peptide market. Companies synthesizing peptides labeled "for research use only," sold directly to consumers online. This is where Peptide Sciences lived.
The third tier existed because demand outran regulation. Online communities on Reddit, Discord, and Telegram built informal knowledge ecosystems around these molecules. Vendors filled the gap between what people wanted and what the medical system offered.
That gap is closing. The FDA has shown it will enforce against research peptide vendors. Eli Lilly and Novo Nordisk have demonstrated they will use the ITC and courts to protect their products. In June 2025, the FDA raided Amino Asylum's warehouse in Memphis, Tennessee. The site went offline within days. Amino Asylum had been the second-most-recognized research peptide vendor in the United States after Peptide Sciences.
Peptide Sciences read the room and walked away before the room collapsed.
The Regulatory Landscape Has Shifted
The Peptide Sciences shutdown did not happen in a vacuum. The entire regulatory framework around peptides shifted in 2025 and early 2026.
In late 2023, the FDA moved 19 widely used peptides to its Category 2 list, effectively banning compounding pharmacies from preparing them. The FDA cited concerns about immunogenicity, manufacturing impurities, and insufficient human clinical trial data.
Then came the reversal. On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced that approximately 14 of the 19 restricted peptides would move back to Category 1, restoring legal access through licensed compounding pharmacies with a physician's prescription.
This is significant. The Category 2 restrictions had pushed patients toward unregulated grey-market sources. The reversal reopens the legitimate compounding pathway for peptides like thymosin alpha-1 and others.
But reclassification is not the same as deregulation. Compounded peptides still require a prescription. They still require a licensed pharmacy. And the FDA's enforcement posture toward unlicensed "research use only" vendors has only intensified.
The message from regulators is clear: peptides are not being banned. The unregulated channel for obtaining them is being shut down.
What This Means for People Who Used Peptide Sciences
If you were a Peptide Sciences customer, here is what changes.
No more orders. The site is down. The company is not processing orders, issuing refunds, or responding to support tickets in any confirmed capacity.
Remaining stock carries uncertainty. If you have unopened vials purchased before the shutdown, those vials are what they always were: unregulated products that were never manufactured under pharmaceutical oversight. Finnrick's testing showed that quality varied significantly by product. Some peptides tested well. Others did not.
Scam sites will appear. Finnrick has already warned that any site selling under the Peptide Sciences name after March 6, 2026 is fraudulent. The brand carried trust in the grey-market community. Scammers will exploit that trust. Do not purchase from any site claiming to be Peptide Sciences.
For guidance on proper peptide storage if you have existing supply, see our storage guide.
What Options Remain
The Peptide Sciences closure does not mean peptides are gone. It means one access channel closed. Others remain, and some are expanding.
| Factor | Grey-Market Vendor | Compounding Pharmacy |
|---|---|---|
| Prescription required | No | Yes |
| Quality testing | Varies (Finnrick: A to E range) | USP standards required |
| Legal status | Increasingly prosecuted | Legal with prescription |
| Typical cost (BPC-157/month) | $40-60 | $150-300 |
| Purity verification | Optional third-party | Mandatory in-house |
| FDA oversight | None | Licensed and inspected |
Compounding Pharmacies with a Prescription
This is the most regulated path. A licensed physician writes a prescription. A 503A or 503B compounding pharmacy fills it. The peptide is manufactured under pharmacy oversight with quality testing.
The February 2026 reclassification restored approximately 14 peptides to Category 1 status, meaning compounding pharmacies can legally prepare them again. This includes several compounds that were previously only available through grey-market vendors.
Cost is higher than grey-market pricing. A month of BPC-157 through a compounding pharmacy typically runs $150-300 versus $40-60 from a research vendor. The difference buys verified purity, accurate dosing, and legal protection.
Telehealth Peptide Clinics
Several telehealth platforms now offer peptide protocols supervised by physicians. You complete a medical intake, consult with a provider (often via video), and receive peptides shipped from a licensed pharmacy.
This model has grown rapidly since the FDA tightened enforcement on research vendors. It combines the convenience of online ordering with the medical oversight that grey-market vendors lacked.
The tradeoff: you need a legitimate medical reason. "I want to try BPC-157 for my tennis elbow" is a valid clinical conversation. "I want to experiment with 12 different peptides" is not something most telehealth providers will support.
Education First
The grey market trained people to think of peptides as consumer products you add to a cart. They are not. They are bioactive compounds with specific mechanisms, contraindications, and dose-response curves.
Before choosing any access pathway, understand what you are taking. Know the mechanism of action. Know the dosing protocols. Know the interactions. Know the storage requirements. Know how to reconstitute your peptides properly.
PeptidesExplorer exists for this purpose. We do not sell peptides. We provide the research, calculators, and educational resources that help you make informed decisions regardless of how the regulatory landscape shifts.
Mistakes to Avoid Right Now
The Peptide Sciences shutdown will push some people toward bad decisions. Here are the ones to avoid.
Panic-Buying from Unknown Vendors
When a major vendor disappears, smaller vendors see a surge in orders. Some of those vendors are legitimate. Many are not. Finnrick's database shows that across 177 tested vendors, product quality varies enormously. The average retatrutide purity ranges from 98.76% to 99.95%, but quantity diverges by up to 50% from the advertised value at the 95th percentile.
Buying from an untested vendor because you are afraid of losing access is how people end up injecting underdosed, mislabeled, or contaminated products.
Ignoring the Shift Toward Clinical Access
The February 2026 reclassification restored legal compounding access for most popular peptides. This is the biggest positive development for peptide access in two years. If you were using grey-market peptides because compounding was restricted, that barrier has largely been removed.
Talk to a physician. Get a prescription. Use a licensed pharmacy. The cost is higher. The peace of mind is worth it.
Trusting Any Site That Claims to Be Peptide Sciences
Finnrick confirmed on March 6, 2026 that further purchases under the Peptide Sciences brand are fraudulent. The company is gone. Anyone selling under that name is a scammer capitalizing on brand recognition. Do not enter payment information on any site claiming to be Peptide Sciences.
Frequently Asked Questions
Is Peptide Sciences permanently shut down?
Yes. Peptide Sciences announced a voluntary shutdown of operations on March 6, 2026. The company has ceased selling all research products. Finnrick, the independent testing platform, confirmed the closure and warned that any site still selling under the Peptide Sciences name is fraudulent.
Why did Peptide Sciences shut down?
The company described the shutdown as voluntary. The likely causes include escalating FDA enforcement against research peptide vendors, ITC actions blocking peptide imports, quality concerns revealed by Finnrick testing (E-rated retatrutide across 37 samples), and the broader collapse of the grey-market business model.
Are peptides illegal now?
No. Peptides themselves are not illegal. In February 2026, HHS Secretary RFK Jr. restored approximately 14 peptides to Category 1 status, allowing licensed compounding pharmacies to prepare them with a physician's prescription. What is increasingly illegal is selling unregulated peptides labeled 'for research use only' directly to consumers.
Can I still get BPC-157 after the Peptide Sciences shutdown?
Yes. BPC-157 remains available through licensed compounding pharmacies with a valid prescription, typically $150-300 per month. Telehealth platforms also offer supervised BPC-157 protocols. Since February 2026, BPC-157 is back on the Category 1 list, meaning any licensed pharmacy can compound it. Learn more on our BPC-157 profile page.
What happened to pending Peptide Sciences orders?
The company has not publicly addressed pending orders or refunds as of March 6, 2026. Customers with outstanding orders should contact their payment provider to dispute charges if products are not delivered. Credit card chargebacks typically have a 60-120 day window depending on the issuer.
Is it safe to use Peptide Sciences products I already have?
Existing vials are the same unregulated products they always were. Finnrick testing showed quality varied by product: BPC-157 scored A (7.8/10) while retatrutide scored E (6.1/10). Proper storage matters. Reconstituted peptides degrade within 30 days even when refrigerated. Unreconstituted lyophilized powder stored at proper temperatures retains potency longer.
Will other peptide vendors shut down too?
The trend points in that direction. Amino Asylum was raided by the FDA in June 2025. Four vendors received warning letters in December 2024. Since late 2023, the FDA has restricted 19 peptides and taken enforcement action against 5+ vendors. The 'research use only' label no longer provides legal cover.
How do I find a legitimate peptide provider?
Start with a physician consultation. A licensed doctor can prescribe peptides filled by a 503A or 503B compounding pharmacy. Telehealth platforms have made this process more accessible. The cost is higher than grey-market pricing, typically $150-300 per month for BPC-157 versus $40-60 from a research vendor, but you receive verified purity and legal protection.
The Bottom Line
The Peptide Sciences shutdown marks the end of one chapter in the peptide market. The largest grey-market vendor is gone, and the regulatory pressure that drove it out is not easing.
This is not the end of peptide access. The February 2026 reclassification restored compounding rights for most popular peptides. Clinical pathways through physicians and licensed pharmacies remain open and are expanding. The grey-market model, built on "research use only" labels and regulatory ambiguity, is the part that is contracting.
PeptidesExplorer will continue providing the research, dosage guides, calculators, and educational resources you need to understand these compounds. The access pathways are changing. The science is not. For those wondering whether the company might reopen, read our analysis: Is Peptide Sciences Coming Back?. For the full regulatory picture, see FDA peptide crackdown 2026. For sourcing, see where to buy peptides in 2026. For safe handling, see our peptide safety guide and getting started with peptides. Stay informed at peptidesexplorer.com.
Related Articles: - Peptide Sciences Alternatives - Is Peptide Sciences Coming Back? - FDA Peptide Crackdown 2026 - Where to Buy Peptides in 2026 - How to Store Peptides
Helpful Tools
Related Articles
Is Peptide Sciences Coming Back?
Is Peptide Sciences coming back? No. FDA regulatory analysis, company status update, and where to get research peptides now. Updated March 2026.
Where to Buy Peptides in 2026
Legitimate routes to buy research peptides in 2026: telehealth, compounding pharmacies, quality verification, and legal status.
Peptide Sciences Alternatives 2026: Where to Buy Now
Peptide Sciences shut down March 2026. Safest alternatives: telehealth clinics ($150-500/mo), 503B compounding pharmacies, and clinical trials.
FDA Peptide Crackdown 2026
Timeline of FDA actions against peptide vendors in 2025-2026. Why Peptide Sciences shut down, which peptides are banned, and what options remain.