
You search "are peptides safe" and get contradictory answers. One forum post calls BPC-157 a miracle. A news article warns that research peptides are unregulated poison. Neither is accurate.
Most research peptides have favorable safety profiles in published studies. BPC-157, GHK-Cu, and ipamorelin show minimal adverse events across hundreds of animal trials and emerging human data. Others, like melanotan-2, carry documented risks including mole changes and nausea. The real variables are compound selection, source quality, and your individual health history. Check specific interactions between any peptides you plan to use with our peptide interaction checker.
This guide covers safety data for 15 peptides, side effects organized by class, contraindications, drug interactions, and practical steps to reduce risk. All claims reference published research. This is educational content for research purposes only. Consult a healthcare provider before starting any peptide protocol.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Peptide Safety Overview: Quick-Reference Table
The table below rates 15 commonly researched peptides on safety profile, most frequent side effect, serious risk potential, and whether controlled human trial data exists. Safety ratings reflect the weight of published evidence, not personal opinion.
| Peptide | Safety Rating | Most Common Side Effect | Serious Risk Level | Human Trial Data |
|---|---|---|---|---|
| BPC-157 | 4/5 | Nausea, injection site redness | Low | No (animal only) |
| TB-500 | 4/5 | Headache, temporary lethargy | Low | No (animal only) |
| GHK-Cu | 5/5 | Mild skin irritation (topical) | Very Low | Yes (topical) |
| Semaglutide | 4/5 | Nausea (44% at full dose) | Moderate | Yes (STEP trials) |
| Tirzepatide | 4/5 | Nausea (31%), diarrhea (23%) | Moderate | Yes (SURMOUNT trials) |
| Retatrutide | 3/5 | Nausea (26-43%), dysesthesia (4-21%) | Moderate | Yes (Phase 2/3) |
| Ipamorelin | 4/5 | Transient water retention | Low | Yes (limited) |
| CJC-1295 | 3/5 | Flushing, water retention | Low-Moderate | Yes (limited) |
| Melanotan-2 | 2/5 | Nausea, flushing, mole darkening | Moderate-High | Yes (limited) |
| KPV | 4/5 | Injection site reaction | Low | No (preclinical) |
| MOTS-c | 4/5 | Mild injection site pain | Low | Yes (limited) |
| LL-37 | 3/5 | Injection site pain, redness | Low-Moderate | Yes (2 RCTs) |
| Thymosin Alpha-1 | 5/5 | Injection site discomfort | Very Low | Yes (11,000+ patients) |
| Follistatin-344 | 2/5 | Joint pain, swelling | Moderate | No (gene therapy only) |
| Hexarelin | 3/5 | Cortisol elevation, hunger | Moderate | Yes (limited) |
How to read this table: A 5/5 rating means extensive data with very few adverse events. A 2/5 means limited data, notable side effects, or unresolved safety questions. No peptide on this list earns a perfect safety score across all categories because long-term human data remains sparse for most compounds.
How Peptides Differ from Drugs and Steroids
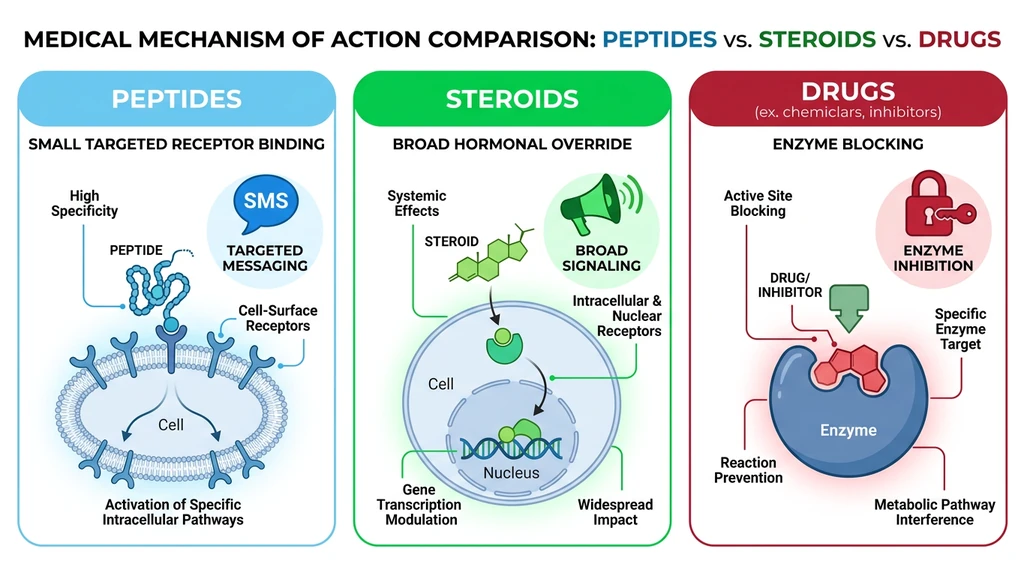
Peptides are short amino acid chains, typically 2 to 50 amino acids long, that act as signaling molecules. They bind to specific receptors on target cells and trigger defined biological responses. Think of a peptide as a text message sent to one person: precise, targeted, delivered to a specific recipient. Anabolic steroids are more like a loudspeaker in a crowded room. Every cell with a steroid receptor responds, whether you intended it to or not.
This distinction matters for safety. Testosterone, the most common anabolic steroid, binds androgen receptors throughout the body: muscle, prostate, liver, heart, brain, skin, hair follicles. That broad activation produces muscle growth alongside acne, hair loss, liver strain, cardiovascular stress, and hormonal shutdown (Basaria et al., NEJM 2010). BPC-157, by contrast, modulates growth factor pathways (VEGF, FGF, NO system) involved in tissue repair without directly activating hormone receptors (Sikiric et al., J Physiol Pharmacol 2018).
Receptor specificity also explains why peptide side effects tend to be predictable. Semaglutide activates GLP-1 receptors concentrated in the gut, pancreas, and brain. The nausea and appetite suppression it causes follow directly from the locations of those receptors (Wilding et al., NEJM 2021). There are few "off-target" surprises. Steroids, because they affect so many tissue types, generate a far wider and less predictable side effect profile.
None of this means peptides are harmless. Specificity is not the same as safety. A peptide that precisely activates growth hormone release still carries the risks associated with elevated growth hormone, including insulin resistance and potential tumor growth promotion. The advantage is that those risks are more predictable and more manageable than the systemic disruption caused by broad-acting compounds.
Side Effects by Peptide Class

Side effects cluster by mechanism of action. Peptides within the same class tend to produce similar adverse events because they target the same biological pathways. The sections below organize the 15 peptides from the reference table into five functional classes.
Healing Peptides: BPC-157 and TB-500
BPC-157 is a synthetic fragment of a protein found in human gastric juice. Animal studies spanning over 20 years show gastric ulcer healing, tendon repair, muscle regeneration, and neuroprotection with minimal toxicity, even at doses 10 to 100 times the therapeutic range (Sikiric et al., Curr Pharm Des 2018). No lethal dose has been established in animal models.
The catch: no published, peer-reviewed human randomized controlled trial exists for BPC-157 as of March 2026. All safety data comes from animal models (primarily rats and mice). User reports from thousands of self-experimenters describe mild nausea, occasional headache, and injection site redness as the most common complaints. Serious adverse events are rare in anecdotal reports, but anecdotal data cannot substitute for controlled trials.
TB-500 (Thymosin Beta-4) promotes angiogenesis, reduces inflammation, and accelerates wound healing. It has been studied in equine medicine and in limited human trials for cardiac repair post-myocardial infarction. Reported side effects include temporary lethargy, headache, and mild nausea. One theoretical concern: because TB-500 promotes new blood vessel growth, some researchers question whether it could support tumor vasculature. No published study has demonstrated this, but the question remains open (Crockford et al., Ann N Y Acad Sci 2010).
For both peptides, the primary safety concern is source quality, not the molecules themselves. Contaminated or mislabeled vials introduce risks that have nothing to do with BPC-157 or TB-500 pharmacology. Women considering BPC-157 should review our guide on BPC-157 benefits and considerations for women.
Weight Loss Peptides: Semaglutide, Tirzepatide, Retatrutide
These peptides have the strongest human safety data of any class because they have been through large-scale, FDA-regulated clinical trials.
Semaglutide (Ozempic, Wegovy): The STEP trial program enrolled over 4,500 participants. Nausea occurred in 44% of participants at the 2.4 mg dose, vomiting in 24%, diarrhea in 30%. Most GI symptoms were mild to moderate and diminished by week 12 to 16. Serious adverse events included gallbladder disorders (2.6% vs 1.2% placebo) and acute pancreatitis (rare, <1%). Discontinuation due to side effects was 7% (Wilding et al., NEJM 2021).
Tirzepatide (Mounjaro, Zepbound): The SURMOUNT-1 trial enrolled 2,539 participants. Nausea rates reached 31% at the 15 mg dose, diarrhea 23%, constipation 17%. Serious adverse events were similar to semaglutide. Cholelithiasis (gallstones) occurred in 0.4% to 1.6% depending on dose. The overall discontinuation rate was 4.3% to 7.1% (Jastreboff et al., NEJM 2022).
Retatrutide: This triple agonist (GLP-1 + GIP + glucagon) produced 28.7% weight loss in Phase 3 but introduced a new safety signal: dysesthesia (abnormal skin sensations) in 4.4 to 20.9% of participants at the 12 mg dose depending on trial population. Nausea ranged from 26.5% (TRANSCEND-T2D-1) to 43% (TRIUMPH-4). Higher resting heart rate (up to 6.7 bpm increase) was also noted. For detailed side effect rates and management strategies, see our retatrutide side effects guide.
The key pattern: GI side effects are dose-dependent and time-limited. Slow dose escalation reduces their severity. Gallbladder and pancreatic risks, while low in absolute terms, require monitoring, especially in patients with a history of gallstones or pancreatitis.
Growth Hormone Secretagogues: Ipamorelin, CJC-1295, Hexarelin
These peptides stimulate the pituitary gland to release growth hormone (GH). Their side effects mirror the effects of elevated GH itself.
Ipamorelin is considered the mildest GH secretagogue. It stimulates GH release without significantly elevating cortisol or prolactin (Raun et al., Eur J Endocrinol 1998). Common side effects include transient water retention, tingling in the extremities, and mild hunger after injection. These effects are dose-dependent and typically resolve within 30 to 60 minutes.
CJC-1295 (with DAC) provides sustained GH release over days rather than minutes. This extended action increases the risk of side effects seen with prolonged GH elevation: facial flushing, water retention, joint stiffness, and numbness or tingling in hands (carpal tunnel symptoms). A small clinical trial showed a 2 to 10-fold increase in GH levels lasting up to 6 days after a single injection (Teichman et al., J Clin Endocrinol Metab 2006).
Hexarelin is the most potent GH secretagogue but also the least selective. Unlike ipamorelin, hexarelin elevates cortisol and prolactin alongside growth hormone (Arvat et al., J Clin Endocrinol Metab 1997). Cortisol elevation can disrupt sleep, increase blood pressure, and promote fat storage, precisely the opposite of what most users intend. Hexarelin also shows rapid tachyphylaxis: the GH response diminishes significantly after 4 to 6 weeks of continuous use.
For all GH secretagogues, the long-term concern is chronically elevated IGF-1 levels. IGF-1 is a growth factor that promotes cell proliferation. Persistently elevated IGF-1 has been associated with increased risk of certain cancers in epidemiological studies (Renehan et al., Lancet 2004). Monitoring IGF-1 through bloodwork every 8 to 12 weeks is essential during GH secretagogue protocols.
Cosmetic Peptides: GHK-Cu and Melanotan-2
GHK-Cu (copper peptide) has one of the strongest safety profiles of any research peptide. It occurs naturally in human plasma, saliva, and urine. Topical GHK-Cu has been used in wound healing and skincare for over three decades with a well-documented safety record. Skin irritation at the application site is the most reported adverse effect. Subcutaneous injection may cause temporary redness and swelling. Systemic toxicity is extremely low (Pickart et al., BioMed Res Int 2012). For specific applications, see our guides on GHK-Cu for hair growth and GHK-Cu microneedling protocols.
Melanotan-2 is a different story. This synthetic analog of alpha-melanocyte stimulating hormone (alpha-MSH) produces skin darkening, increased libido, and appetite suppression. Side effects are common and well-documented: nausea (85% of first-time users), facial flushing, fatigue, and injection site pain. The more concerning issue is mole darkening and new mole formation. Case reports have documented atypical moles and melanoma diagnoses in melanotan-2 users (Brennan et al., Br J Dermatol 2014). Whether MT-2 directly causes melanoma or simply accelerates the progression of pre-existing melanocytic lesions remains unresolved. Multiple dermatology societies have issued warnings.
The safety gap between GHK-Cu and melanotan-2 illustrates why "peptide" is not a meaningful safety category. Each compound has its own risk profile based on its mechanism and the tissues it targets.
Immune Peptides: Thymosin Alpha-1, LL-37, and KPV
Thymosin Alpha-1 has the deepest clinical track record of any peptide on this list. It is approved in over 30 countries for hepatitis B and C treatment and as an immune adjuvant. A pooled analysis of over 11,000 patients showed an adverse event profile comparable to placebo (Tuthill et al., Ann N Y Acad Sci 2010). Injection site reactions (redness, mild soreness) are the most common complaint. Thymosin Alpha-1 modulates rather than stimulates the immune system, reducing the risk of autoimmune overactivation.
LL-37 is an antimicrobial peptide that forms part of the innate immune system. Two small randomized controlled trials in humans have been published. The most common side effects are injection site pain, redness, and transient warmth at the injection area. At higher doses, some concern exists about pro-inflammatory effects: LL-37 can activate NF-kB pathways, potentially worsening certain inflammatory conditions. Dose-dependent safety monitoring is recommended. For a deeper analysis, see our LL-37 benefits guide.
KPV is a tripeptide fragment of alpha-MSH with potent anti-inflammatory activity. It inhibits NF-kB and reduces pro-inflammatory cytokine production. Published safety data comes entirely from animal studies, which show excellent tolerability with no reported serious adverse events. User reports describe occasional injection site reactions. KPV's small molecular weight (3 amino acids) and its derivation from a naturally occurring hormone fragment suggest a favorable safety profile, but human trial data is needed. More details are available in our immune system peptides guide.
Who Should NOT Use Peptides
Certain medical conditions create clear contraindications for specific peptides. The risks below are based on known mechanisms of action and published safety data. This is not an exhaustive list. Any pre-existing condition warrants a conversation with a physician before starting a peptide protocol.
| Condition | Contraindicated Peptides | Reason |
|---|---|---|
| Active cancer or history of cancer | GH secretagogues (ipamorelin, CJC-1295, hexarelin), follistatin-344, TB-500 | Elevated IGF-1 and growth factors may promote tumor growth and angiogenesis |
| Pregnancy or breastfeeding | All peptides | No safety data exists for any research peptide during pregnancy; unknown fetal effects |
| Under 18 years old | All peptides (especially GH secretagogues) | May interfere with natural growth and hormonal development during puberty |
| Chronic kidney disease (stage 3+) | BPC-157, semaglutide, tirzepatide | Altered clearance may increase drug levels; semaglutide carries acute kidney injury risk at higher doses |
| Autoimmune disease on immunosuppressants | Thymosin Alpha-1, LL-37 | Immune-stimulating peptides may counteract immunosuppressive therapy and trigger flares |
| History of pancreatitis | Semaglutide, tirzepatide, retatrutide | GLP-1 agonists carry a small but documented risk of acute pancreatitis |
| History of medullary thyroid carcinoma | Semaglutide, tirzepatide, retatrutide | GLP-1 agonists carry a black box warning for thyroid C-cell tumors (based on rodent data) |
| History of melanoma | Melanotan-2 | Stimulates melanocyte activity; may promote melanocytic lesion progression |
The single most important takeaway: if you have active cancer, are pregnant, or are under 18, no research peptide has sufficient safety data to justify use.
Peptide-Medication Interactions
Research on peptide-drug interactions is limited, but several mechanisms are well-established. The table below covers the most clinically relevant interactions.
| Peptide | Interacting Medication | Mechanism | Clinical Consequence |
|---|---|---|---|
| Semaglutide, tirzepatide, retatrutide | Oral contraceptives, levothyroxine, warfarin, oral antibiotics | GLP-1 agonists slow gastric emptying by 30 to 50% | Delayed absorption of oral medications; reduced peak drug levels; potential therapeutic failure |
| Semaglutide, tirzepatide | Insulin, sulfonylureas | Additive blood sugar lowering | Hypoglycemia risk; insulin doses may need 20 to 50% reduction |
| GH secretagogues (ipamorelin, CJC-1295, hexarelin) | Insulin | Growth hormone opposes insulin action | Blood sugar elevation; may require insulin dose adjustment |
| GH secretagogues | Corticosteroids (prednisone, dexamethasone) | Both elevate blood sugar; cortisol blunts GH effects | Compounded metabolic disruption; reduced GH secretagogue efficacy |
| BPC-157 | Antihypertensives (ACE inhibitors, ARBs) | BPC-157 modulates the NO system and may lower blood pressure independently | Potential additive blood pressure reduction; monitor for dizziness or lightheadedness |
| BPC-157 | NSAIDs (ibuprofen, naproxen) | BPC-157 counteracts NSAID-induced gastric damage in animal studies | Potentially protective interaction; may reduce NSAID gastrointestinal toxicity |
| Melanotan-2 | Blood pressure medications | MT-2 can transiently raise blood pressure | May counteract antihypertensive therapy |
If you take daily medications, use our peptide interaction checker to screen for known conflicts before starting a protocol. The most practically important interaction involves GLP-1 agonists and oral medications. If you take oral birth control, thyroid medication, or blood thinners while using semaglutide or tirzepatide, take those medications at least 1 hour before your peptide injection or discuss timing adjustments with your prescriber.
Source Quality: The Biggest Safety Variable
The peptide molecule matters less than where it came from. A perfectly safe compound becomes dangerous when it contains bacterial endotoxins, heavy metals, residual solvents, or the wrong peptide entirely.
A 2023 analysis published in the *Journal of the American Medical Association* (JAMA) examined compounded semaglutide products and found that approximately 40% contained impurities, incorrect concentrations, or failed sterility testing. Some vials contained less than 60% of the labeled dose. Others contained degradation products not present in pharmaceutical-grade semaglutide.
This problem extends beyond semaglutide. Research peptides purchased from unverified sources face no mandatory quality control. No regulatory body inspects the manufacturing facility. No third party verifies purity before sale. The buyer assumes all risk.
Three markers separate reliable sources from dangerous ones:
1. Third-party Certificate of Analysis (CoA). A legitimate CoA comes from an independent lab (not the vendor's in-house lab). It reports purity by HPLC (should be 98% or higher), mass spectrometry confirmation of molecular identity, and endotoxin/sterility testing. If a vendor cannot produce a CoA for each batch, do not buy from them.
2. USP-grade or pharmaceutical-grade ingredients. Bacteriostatic water, mannitol (a common filler in lyophilized peptides), and the peptide itself should meet United States Pharmacopeia standards. "Research grade" without further specification is a red flag.
3. Proper cold-chain shipping and storage. Peptides degrade at room temperature. Any vendor shipping lyophilized peptides without cold packs in warm weather is selling compromised product. After reconstitution, vials must be stored at 2 to 8 degrees Celsius and used within the timeframe specified for the bacteriostatic water used. For detailed shelf life guidance, see our guide on how long reconstituted peptides last.
For a curated list of vendors that meet these criteria, see our where to buy peptides in 2026 guide.
How to Minimize Risks: 6 Practical Steps
Peptide safety is partly about the compound and mostly about your protocol. These six steps address the controllable variables.
Step 1: Start at the lowest effective dose. Every peptide-class section above shows dose-dependent side effect rates. Semaglutide nausea at 0.25 mg is roughly 20%; at 2.4 mg it reaches 44%. Beginning low and titrating upward over weeks lets your body adapt and reveals your personal tolerance threshold before reaching high doses.
Step 2: Use bacteriostatic water, not sterile water. Bacteriostatic water contains 0.9% benzyl alcohol, which inhibits microbial growth. A vial reconstituted with bacteriostatic water remains usable for up to 28 days when refrigerated. Sterile water contains no preservative. Once the stopper is punctured, bacteria can colonize the vial within 24 hours. This is the single most preventable source of infection in peptide use. Calculate your exact reconstitution volumes with our reconstitution calculator.
Step 3: Follow proper injection technique. Clean the vial stopper and injection site with alcohol swabs. Use a new, sterile insulin syringe (29 to 31 gauge) for every injection. Inject subcutaneously at a 45-degree angle into abdominal fat, thigh, or upper arm. Rotate injection sites to prevent lipodystrophy (localized fat loss or hardening). If you are new to peptide injection, our beginner's guide walks through each step with photographs.
Step 4: Monitor bloodwork every 8 to 12 weeks. At minimum, test IGF-1 (for GH secretagogues), fasting glucose and HbA1c (for GLP-1 agonists), and a complete metabolic panel. Changes in these markers can signal problems before symptoms appear. IGF-1 levels can triple without producing noticeable symptoms, silently increasing cancer risk.
Step 5: Cycle appropriately. Most peptides are not designed for indefinite use. BPC-157 and TB-500 are typically run for 4 to 12 weeks, then discontinued once the injury has healed. GH secretagogues benefit from cycling (8 to 12 weeks on, 4 weeks off) to prevent receptor desensitization, especially hexarelin. FDA-approved GLP-1 agonists are the exception: they are designed for continuous use, and weight regain occurs rapidly after discontinuation.
Step 6: Verify your source with a batch-specific CoA. Request the Certificate of Analysis before purchasing. Confirm that the lot number on the CoA matches the lot number on your vial. Cross-reference the testing lab's name to verify it is a real, independent laboratory. A 30-second verification step eliminates the largest single risk factor in peptide use.
Common Mistakes That Increase Risk
Mistake 1: Using sterile water instead of bacteriostatic water. A vial reconstituted with sterile water has no antimicrobial protection. Every time you puncture the stopper with a needle, you introduce bacteria. Within 24 hours at refrigerator temperature, bacterial colonies can reach levels sufficient to cause injection site infections, abscesses, or systemic infection. Bacteriostatic water extends safe multi-use to 28 days. One bottle of bacteriostatic water costs $5 to $10. An abscess requiring medical drainage costs $500 to $2,000 and leaves a scar.
Mistake 2: Skipping bloodwork during GH secretagogue protocols. Ipamorelin and CJC-1295 can elevate IGF-1 levels 2 to 3 times above baseline without producing any perceptible symptoms. You feel fine. Meanwhile, sustained IGF-1 elevation above 300 ng/mL has been associated with increased colorectal and breast cancer risk in epidemiological data (Renehan et al., Lancet 2004). A basic IGF-1 blood test costs $30 to $60 through direct-to-consumer lab services. It takes 5 minutes and can reveal a problem that has no symptoms.
Mistake 3: Buying from unverified sources. The 40% contamination rate found in compounded semaglutide products applies broadly to the unregulated peptide market. Users who select vendors based on price alone expose themselves to underdosed, overdosed, contaminated, or entirely mislabeled products. Saving $20 on a vial while injecting unknown substances is not a reasonable tradeoff. Use our vendor guide to identify sources that provide batch-specific third-party testing.
Mistake 4: Ignoring dose-response curves. Doubling your BPC-157 dose from 250 mcg to 500 mcg does not double the healing speed. Most peptides follow a logarithmic dose-response curve: initial dose increases produce large effects, but further increases yield diminishing returns and increasing side effects. In the STEP trials, semaglutide at 1.0 mg produced 13.2% weight loss; the 2.4 mg dose produced 16.9%, only 3.7% more despite more than doubling the dose and significantly increasing GI side effects (Wilding et al., NEJM 2021). More is not better. More is more side effects.
Peptides and the Regulatory Landscape
The FDA's 2024-2026 enforcement actions significantly reshaped the peptide market. Several compounds, including BPC-157 and certain GH secretagogues, were removed from compounding pharmacies. This did not make these peptides illegal to possess for research purposes, but it did eliminate the most quality-controlled supply channel for many users.
The regulatory picture varies by compound:
FDA-approved peptides (semaglutide, tirzepatide) are available by prescription with full manufacturing oversight, adverse event reporting, and post-market surveillance. Their safety profiles are the most thoroughly documented. For compounded alternatives, see is compound tirzepatide safe and compound semaglutide with B12.
Compounding-eligible peptides (thymosin alpha-1, certain others) can be prepared by licensed compounding pharmacies under physician supervision in some jurisdictions. Quality control exists but is less rigorous than for FDA-approved drugs.
Research-only peptides (BPC-157, TB-500, melanotan-2, most GH secretagogues) are sold labeled "for research purposes only" and "not for human consumption." Quality control depends entirely on the vendor. This is where source verification matters most.
The safety implication: an FDA-approved peptide manufactured by Eli Lilly or Novo Nordisk carries fundamentally different purity and dosing assurances than a research peptide from an overseas synthesis lab. Factor this reality into your risk assessment.
Frequently Asked Questions
Are peptides safe for long-term use?
It depends on the peptide. FDA-approved GLP-1 agonists like semaglutide have 2+ year safety data from clinical trials and are designed for continuous use. Research peptides like BPC-157 and TB-500 lack long-term human studies; most protocols recommend 4 to 12 week cycles. GH secretagogues require cycling to prevent receptor desensitization and should include IGF-1 monitoring every 8 to 12 weeks. Thymosin Alpha-1 has been used continuously in clinical settings for months with minimal adverse events across 11,000+ patients.
Are peptides safer than steroids?
In general, yes. Anabolic steroids activate receptors throughout the body, causing systemic effects: liver toxicity, cardiovascular strain, hormonal shutdown, acne, hair loss. Peptides target specific receptors with narrower effects. BPC-157 affects growth factor pathways without touching androgen receptors. Ipamorelin stimulates GH release without elevating cortisol. The key difference is receptor specificity. However, some peptides (hexarelin, follistatin-344) carry risks comparable to mild steroid use, so blanket statements are misleading.
Can peptides cause cancer?
No peptide has been proven to cause cancer in humans. The concern centers on growth-promoting peptides (GH secretagogues, follistatin-344, TB-500) that elevate IGF-1 or stimulate angiogenesis. Epidemiological data links chronically elevated IGF-1 levels above 300 ng/mL to increased colorectal and breast cancer risk (Renehan et al., Lancet 2004). GLP-1 agonists carry a rodent-based thyroid tumor warning, but human data over 7+ years has not confirmed this risk. If you have active cancer or a cancer history, avoid growth-promoting peptides entirely.
Are peptides safe for women?
Most peptides studied show similar safety profiles in men and women. Semaglutide and tirzepatide trials enrolled roughly equal numbers of both sexes with comparable adverse event rates. BPC-157 has no known sex-specific risks in animal studies. GH secretagogues affect women and men differently because baseline GH levels vary; women typically need lower doses. The main contraindication is pregnancy and breastfeeding, where no safety data exists for any research peptide. See our BPC-157 guide for women for female-specific considerations.
Are peptides safe for people over 50?
Age alone is not a contraindication. Thymosin Alpha-1 trials included elderly patients and showed excellent tolerability. GLP-1 agonists have been studied in adults up to 75 years old. However, kidney function declines with age, which affects clearance of some peptides. GH secretagogues require more cautious dosing because cancer screening becomes more important as baseline cancer risk rises with age. Bloodwork monitoring every 8 to 12 weeks is more critical in the over-50 population than in younger users.
Do peptides affect hormones?
Some do, by design. GH secretagogues (ipamorelin, CJC-1295, hexarelin) stimulate growth hormone and IGF-1 release. Hexarelin also elevates cortisol and prolactin. GLP-1 agonists (semaglutide, tirzepatide) modulate insulin and glucagon secretion. Melanotan-2 affects melanocyte-stimulating hormone pathways. Healing peptides (BPC-157, TB-500, GHK-Cu) and immune peptides (thymosin alpha-1, KPV) do not directly alter hormone levels based on published data. If you take hormone-related medications, check interactions before starting any peptide.
Are peptides legal?
In the United States, semaglutide and tirzepatide are legal by prescription. Most other research peptides are legal to purchase labeled "for research purposes only." The FDA's 2024 to 2026 crackdown removed BPC-157 and several other compounds from compounding pharmacy availability, but did not criminalize possession. Legality varies by country. In Australia, most peptides require a prescription. In the UK, they are legal to possess but not to sell for human use. Check your local regulations before purchasing. See our FDA crackdown overview for the latest enforcement actions.
Can you take multiple peptides at once?
Yes, but with caveats. Common stacks like BPC-157 + TB-500 for injury healing have extensive anecdotal support and no known negative interactions. Stacking GH secretagogues (ipamorelin + CJC-1295) is standard practice and supported by pharmacological rationale. However, combining peptides from different classes (e.g., a GH secretagogue + a GLP-1 agonist + an immune peptide) creates unpredictable interaction potential. Screen any planned stack with our peptide interaction checker and introduce one peptide at a time to isolate side effects.
The Bottom Line
Most research peptides have favorable safety profiles when sourced from verified suppliers, dosed conservatively, and monitored with regular bloodwork. The compound matters, but the protocol matters more.
One principle governs peptide safety above all others: verify before you inject. Verify the source with a batch-specific CoA. Verify the reconstitution with bacteriostatic water and correct volumes. Verify your body's response with bloodwork at 8 to 12 week intervals. The 40% contamination rate in unregulated products means that skipping verification is the single largest risk factor, larger than any side effect profile of any peptide on this list.
Before starting or combining peptides, screen your planned protocol with our peptide interaction checker. It cross-references known interactions and contraindications in seconds.
Related articles: - Is Compound Tirzepatide Safe? — safety evaluation for compounded tirzepatide products - Compound Semaglutide With B12 — safety and efficacy of B12-enhanced semaglutide - Tirzepatide Cost With Insurance — brand-name vs compounded pricing comparison
Helpful Tools
Related Articles
How to Travel With Semaglutide: TSA Rules, Cold Chain, Packing & International Tips
Complete guide to traveling with semaglutide. TSA rules for Ozempic pens, cold chain packing, international customs regulations, hotel storage, and time zone dosing adjustments.
CJC-1295 With DAC: Complete Guide to the Long-Acting GHRH Analog
CJC-1295 with DAC guide: 6-8 day half-life, 2-10x GH increase for 6+ days, 1-2 mg weekly. GH bleed effect explained, pituitary desensitization risk, insulin sensitivity data, stacking protocols, and week-by-week results timeline.
BPC-157 Oral vs Injection: Which Route Is Right for Your Goals?
BPC-157 oral vs injection: 80-95% injectable bioavailability vs 3-90% oral. He et al. PK data, condition-by-condition matrix, combination protocol, and cost breakdown.
BPC-157 TB-500 Capsules: The Complete Oral Blend Guide
BPC-157 TB-500 capsules: oral bioavailability comparison, dosage protocols, side effects, and what to look for when buying. TB-500 oral evidence gap explained.