The most common retatrutide side effects are gastrointestinal: nausea (26 to 43% across Phase 3 trials), diarrhea (22 to 33%), vomiting (18 to 21%), and constipation (22 to 25%). A new safety signal, dysesthesia (abnormal skin sensations like tingling or burning), appeared at rates ranging from 4.4% to 20.9% depending on the trial population. Most side effects are dose-dependent, mild to moderate, and improve within 8 to 12 weeks as the body adjusts.
Key findings from 2 Phase 3 trials (1,200+ patients): - GI side effects are common but manageable and time-limited - Dysesthesia rates varied dramatically: 4.4% in TRANSCEND-T2D-1 (diabetes) vs 20.9% in TRIUMPH-4 (obesity + knee OA) - Heart rate increase of ~6.7 bpm, higher than tirzepatide (~2-3 bpm) or semaglutide (~3-4 bpm) - Liver fat reduction of 86% at 48 weeks, with no liver damage signals - Discontinuation rates: 5.1% (TRANSCEND-T2D-1) to 18.2% (TRIUMPH-4) at 12 mg
Retatrutide (LY3437943) is a triple hormone receptor agonist made by Eli Lilly. It activates three receptors: GLP-1, GIP, and glucagon. This triple mechanism produced 28.7% body weight loss in TRIUMPH-4, approaching bariatric surgery results. The drug is not FDA approved. All safety data comes from clinical trials: the Phase 2 obesity trial (Jastreboff et al., NEJM 2023), the Phase 3 TRIUMPH-4 trial (Lilly Press Release, Dec 2025), and the Phase 3 TRANSCEND-T2D-1 trial in type 2 diabetes (Lilly Press Release, 2026).
For dosing protocols and titration schedules, see our retatrutide dosage guide. For preparation instructions, see how to reconstitute retatrutide. For a broad overview of how retatrutide works, see our complete retatrutide guide. For the timeline to results, see how long does retatrutide take to work. Last updated: April 2026.
This is educational content for research purposes. Consult a healthcare provider before starting any medication.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
How Does Retatrutide's Triple Mechanism Cause Side Effects?
Retatrutide activates three hormone receptors in your body. Each one contributes a different set of side effects. Understanding which receptor causes what helps explain why retatrutide's safety profile differs from older drugs like semaglutide (one receptor) or tirzepatide (two receptors). For the full mechanism breakdown, see how does retatrutide work.
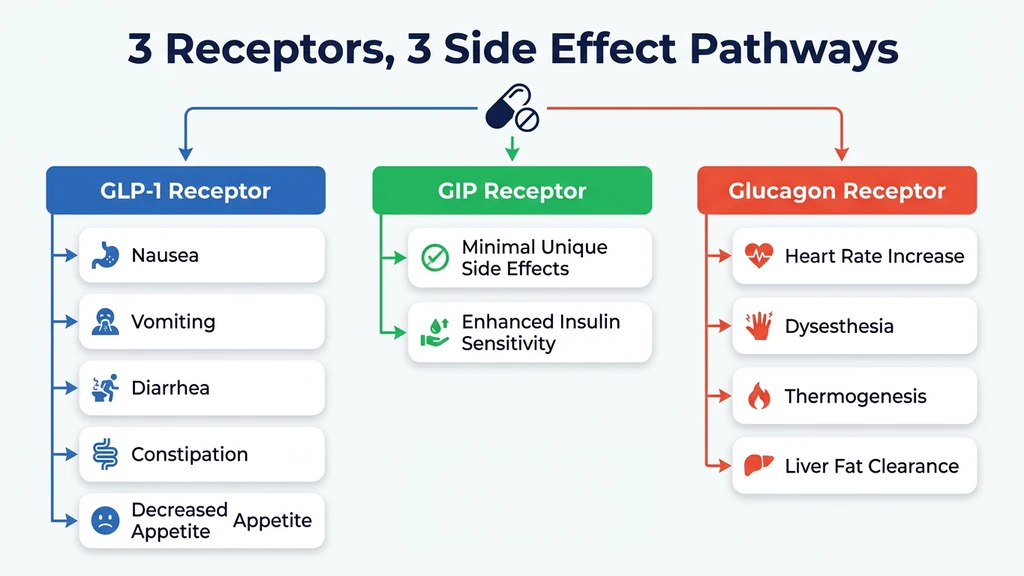
GLP-1: Appetite Suppression and Stomach Slowdown
The GLP-1 component slows down how fast your stomach empties food. This is why you feel full longer after eating. The downside: food sitting in the stomach longer causes nausea, bloating, and sometimes vomiting. This is the same mechanism behind the stomach issues people experience on Ozempic or Wegovy.
Every GLP-1 drug on the market causes these stomach side effects. They are an unavoidable part of how the drug works. The intensity fades as your body adapts, typically within 4 to 8 weeks at each dose level (Jastreboff et al., NEJM 2023).
GIP: Metabolic Fine-Tuning
The GIP (glucose-dependent insulinotropic polypeptide) component improves how your body handles blood sugar and enhances insulin sensitivity. GIP is generally the gentlest of the three receptors. It does not appear to add significant side effects on its own. Its main role is amplifying the weight loss and blood sugar benefits of GLP-1.
Glucagon: The Differentiator (and the Source of New Signals)
The glucagon receptor is what makes retatrutide unique. Glucagon tells your liver to burn stored fat for energy and increases your body's heat production, a process called thermogenesis. You burn more calories even at rest.
This third receptor is also likely responsible for two side effects that are more pronounced with retatrutide than with older drugs: a higher resting heart rate (up to 6.7 beats per minute above normal) and dysesthesia (abnormal skin sensations). Neither tirzepatide nor semaglutide activate the glucagon receptor, which is why these signals are stronger with retatrutide (Nature Medicine, 2024).
What Are the Most Common Gastrointestinal Side Effects?
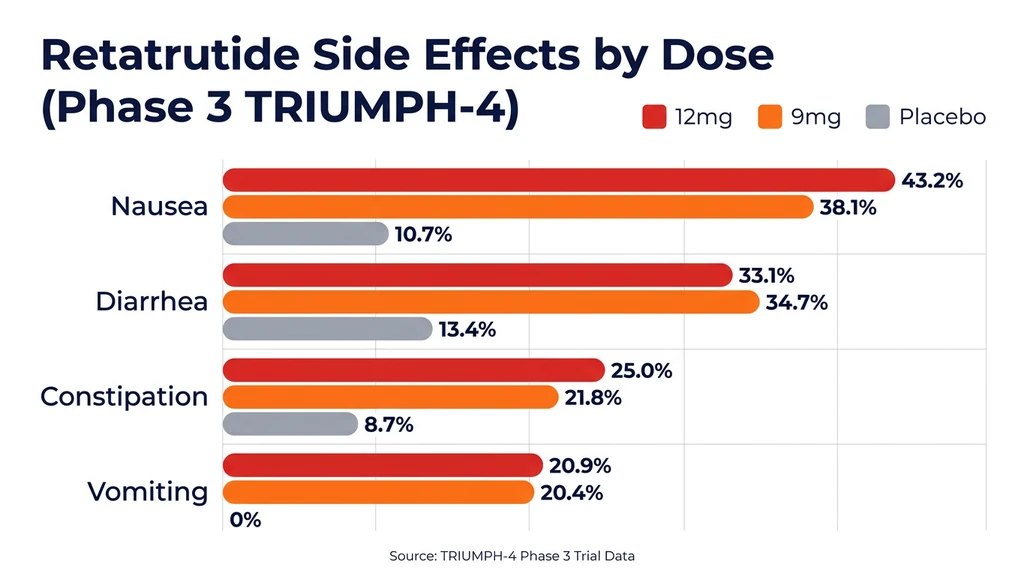
Stomach and digestive issues are the most frequent retatrutide side effects by a wide margin. The table below shows exact rates from both the Phase 2 trial (338 patients, 48 weeks) and the Phase 3 TRIUMPH-4 trial (445 patients, 68 weeks). The Phase 3 trial enrolled adults with obesity and knee osteoarthritis.
| Side Effect | Placebo | 12 mg TRIUMPH-4 | 12 mg TRANSCEND-T2D-1 | 12 mg Phase 2 |
|---|---|---|---|---|
| Nausea | 3.7-10.7% | 43.2% | 26.5% | ~60% |
| Diarrhea | 4.5-13.4% | 33.1% | 22.8% | ~22% |
| Vomiting | 0.0-2.2% | 20.9% | 17.6% | ~26% |
| Constipation | 8.7% | 25.0% | ~16% | |
| Decreased appetite | 9.4% | 18.2% | ||
| Fatigue | Reported | Reported | ||
| Headache | Reported | Reported |
The TRANSCEND-T2D-1 trial (type 2 diabetes population) showed consistently lower GI rates than TRIUMPH-4 (obesity + knee OA). Nausea at 12 mg was 26.5% vs 43.2%. This suggests that the patient population significantly influences side effect severity. The Phase 2 nausea rate (~60% at 12 mg) was the highest, likely reflecting less optimized dose escalation protocols.
Sources: NEJM 2023, Lilly TRIUMPH-4 press release, Lilly TRANSCEND-T2D-1 press release
Nausea: The Most Common Side Effect
Nausea affects roughly 4 in 10 people taking retatrutide at the 12 mg TRIUMPH-4 dose, though rates were lower in TRANSCEND-T2D-1 (26.5% at 12 mg). It typically appears during the first 1 to 2 weeks at each new dose level and feels similar to mild motion sickness or early pregnancy nausea. For most people, it subsides within 4 to 8 weeks. For a dedicated deep-dive, see our retatrutide nausea guide.
A 2025 meta-analysis (a study pooling data from multiple trials) found that at the 8 mg dose, you are about 4.3 times more likely to experience nausea than someone taking a sugar pill. At 12 mg, the odds climb higher still (PMC meta-analysis, 2025).
Severity matters: the vast majority of nausea cases were classified as mild to moderate. Severe nausea requiring medical attention was uncommon.
Diarrhea
About 1 in 3 people on 12 mg experienced diarrhea in TRIUMPH-4. TRANSCEND-T2D-1 showed a lower rate of 22.8%. Pooled data across trials shows roughly 2.5 times the risk compared to a sugar pill at the 8 mg dose (PMC meta-analysis, 2025).
Diarrhea tends to be intermittent rather than constant. It is worst during the first weeks at a new dose and improves with time. Staying well hydrated and replacing electrolytes (sodium, potassium, magnesium) are essential during these episodes. See our complete retatrutide diarrhea guide for detailed management strategies.
Vomiting
Vomiting is less common than nausea but still significant: about 1 in 5 people at the 12 mg dose. The Phase 2 trial showed a clear dose-response pattern: 3% at 1 mg rising to 26% at 12 mg.
Pooled data suggests that at 12 mg, vomiting is roughly 9 times more likely than on placebo. If vomiting prevents you from keeping fluids down for more than 48 hours, seek medical attention immediately.
Constipation and Decreased Appetite
Constipation affected about 25% of participants at 12 mg, roughly triple the placebo rate. GLP-1 slows the entire digestive tract, not just the stomach.
Decreased appetite (18 to 19%) is worth noting because it occupies an unusual space: appetite reduction is the intended effect of the drug, but when it becomes excessive, it crosses into side effect territory. Some trial participants described eating less than 1,000 calories per day without feeling hungry. If you find yourself unable to eat enough to maintain basic nutrition, discuss a dose reduction with your provider.
Fatigue and Headache
Fatigue and headache are commonly reported with retatrutide, though they received less attention in trial publications than GI symptoms. Fatigue is a predictable consequence of sharply reduced caloric intake combined with metabolic shifts from the glucagon receptor. Your body is burning stored fat at an accelerated rate while receiving less food energy.
Headaches tend to cluster in the first 2 to 4 weeks and often correlate with dehydration. When diarrhea or reduced fluid intake accompanies the start of treatment, headaches follow. The fix is straightforward: maintain 2 to 3 liters of water daily, keep electrolytes balanced, and ensure adequate protein intake even when appetite is suppressed.
What Is Dysesthesia, and Why Did It Appear in Phase 3?
Dysesthesia is the safety signal that got the most attention when TRIUMPH-4 results were announced. It was not detected in the earlier Phase 2 trial. This section explains what it is, how common it was, and what researchers think causes it.
Dysesthesia Explained in Plain Language
Dysesthesia means your sense of touch is altered. Normal sensations, like clothing brushing against skin or water from a shower, feel unusual, uncomfortable, or even mildly painful. People describe it as tingling, burning, numbness, or heightened skin sensitivity.
It is not the same as neuropathy (nerve damage). Dysesthesia is a change in how your brain processes sensory input, not a sign that nerves are being destroyed. Think of it as the volume knob on your skin's sensitivity being turned up too high.
How Common Is Dysesthesia With Retatrutide?
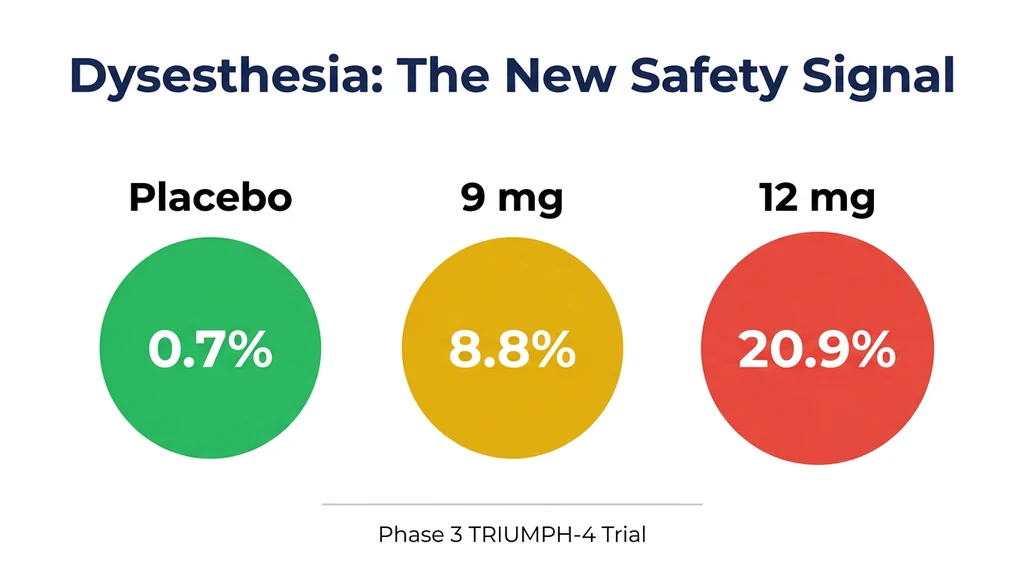
Two Phase 3 trials now provide dysesthesia data, and the rates differ dramatically:
| Trial | Population | 4 mg | 9 mg | 12 mg | Placebo |
|---|---|---|---|---|---|
| TRIUMPH-4 | Obesity + knee OA | 8.8% | 20.9% | 0.7% | |
| TRANSCEND-T2D-1 | Type 2 diabetes | 4.5% | 2.3% | 4.4% | 0.0% |
In TRIUMPH-4, roughly 1 in 5 people at 12 mg experienced altered skin sensation. In TRANSCEND-T2D-1, the rate dropped to 1 in 23. The events were classified as generally mild in both trials and did not cause significant numbers of people to stop treatment (BioSpace, Dec 2025, Lilly TRANSCEND-T2D-1 Press Release).
The gap between trials matters. The TRIUMPH-4 population (obesity with knee osteoarthritis) may have elevated baseline susceptibility to nerve-related side effects due to existing joint inflammation and nerve compression. The TRANSCEND-T2D-1 population (type 2 diabetes) showed rates much closer to what earlier GLP-1 drugs produce. The 20.9% figure from TRIUMPH-4 may not represent the general population. The pivotal TRIUMPH-1 and TRIUMPH-2 trials (obesity-only, no OA requirement) will clarify the true rate when they report later in 2026.
Why was dysesthesia not seen in Phase 2? The most likely explanation is sample size. Phase 2 enrolled 338 people. TRIUMPH-4 enrolled 445. Larger trials detect rarer side effects that smaller studies miss. The longer duration (68 weeks vs 48 weeks) may also have allowed the signal to emerge.
What Causes Dysesthesia With Retatrutide?
Researchers have two leading hypotheses:
GLP-1 receptor effects on peripheral nerves. GLP-1 receptors exist on nerve cells. At high enough activation intensity, they may alter nerve signaling. Supporting evidence: dysesthesia has been reported at lower rates with high-dose oral semaglutide and tirzepatide (PMC, 2025).
The glucagon receptor as amplifier. Retatrutide's substantially higher dysesthesia rates compared to semaglutide and tirzepatide point to the glucagon component as a contributing factor. Glucagon affects small blood vessels. Reduced blood flow to sensory nerves could alter how they transmit signals. This hypothesis would explain why the third receptor produces a side effect that the first two receptors only hint at.
Neither hypothesis is confirmed. Eli Lilly has stated that dysesthesia warrants monitoring in upcoming TRIUMPH readouts expected throughout 2026.
Should You Be Concerned About Dysesthesia?
Based on current data, dysesthesia with retatrutide appears manageable. The events were generally mild and self-limiting. They did not lead to significant treatment discontinuation. There is no evidence of long-term nerve damage from clinical trials so far.
That said, 20.9% is a high rate for a side effect that essentially did not exist with older GLP-1 drugs. This is new territory. Anyone considering retatrutide should be aware of it and discuss it with their provider. If tingling or burning sensations develop and become bothersome, a dose reduction is the first-line response.
Does Retatrutide Affect Your Heart?
Retatrutide has measurable cardiovascular effects, some concerning and some beneficial. The net picture from clinical trials is mixed but not alarming.
Heart Rate Increase
Retatrutide raises resting heart rate in a dose-dependent pattern. At the 12 mg dose, the increase peaked at approximately 6.7 beats per minute above baseline around week 24, then declined at weeks 36 and 48. Both the GLP-1 and glucagon components naturally speed up the heart.
For context, tirzepatide raises heart rate by about 2 to 3 bpm and semaglutide by about 3 to 4 bpm. Retatrutide's ~7 bpm increase is notably higher, likely because of the glucagon component.
No serious cardiac events were attributed to retatrutide in clinical trials. People with existing heart conditions should discuss this with their cardiologist before considering the drug (NEJM 2023).
Blood Pressure and Cholesterol Improvements
The cardiovascular news is not all concerning. Retatrutide significantly improved several heart disease risk markers:
Blood pressure dropped enough that 41% of participants on 8 mg and 30% on 12 mg were able to stop their blood pressure medications entirely. Blood fat levels, the numbers your doctor checks to assess heart disease risk, improved substantially over 48 weeks:
| What it measures | Improvement at 48 weeks |
|---|---|
| "Bad" cholesterol (non-HDL) | -26.9% |
| Particles that clog arteries (ApoB) | -24.2% |
| Blood fats / triglycerides | -40.6% |
One exception: HDL cholesterol ("good cholesterol") did not improve.
For anyone whose doctor has flagged high cholesterol or elevated triglycerides, these are significant improvements. The full cardiovascular outcomes trial (TRIUMPH-3) will tell us whether these better numbers actually translate into fewer heart attacks and strokes, which is what really matters (ESC 2024 / European Heart Journal).
Cardiac Arrhythmias
Irregular heartbeat patterns were reported in 4 to 14% of retatrutide groups compared to 2 to 3% on placebo in Phase 2 data. None were classified as serious adverse events. This warrants monitoring but has not raised safety alarms so far.
Is Retatrutide Safe for Your Liver?
The liver story is one of retatrutide's strongest safety points. Not only did the drug show no signs of liver damage, it dramatically reversed fatty liver disease.
Liver Fat Reduction
A substudy of the Phase 2 trial enrolled 98 participants with MASLD (metabolic dysfunction-associated steatotic liver disease, the medical term for what most people call "fatty liver"). Fatty liver affects an estimated 25% of US adults, often without symptoms.
| Dose | Liver Fat Reduction (48 weeks) | Normal Liver Fat Achieved |
|---|---|---|
| 1 mg | -42.9% | |
| 4 mg | -57.0% | |
| 8 mg | -81.4% | 89% |
| 12 mg | -86.0% | 93% |
| Placebo | +0.3% |
At 12 mg, 93% of participants who started with fatty liver reached normal liver fat levels after 48 weeks. The liver fat reduction at that dose was 86% over 48 weeks. Tirzepatide reduces liver fat by roughly 50 to 60%. Semaglutide achieves 40 to 50%. Retatrutide's 86% reduction, driven by the glucagon receptor telling the liver to burn its stored fat, is in a different category.
Sources: Nature Medicine, 2024, AASLD Liver Meeting
No Liver Damage Signals
Liver enzymes (ALT and AST, the blood markers your doctor checks to monitor liver inflammation) were tracked throughout both trials using standardized safety analysis. No signs of liver damage were detected through 48 weeks in either the general obesity population or the fatty liver subset.
This is reassuring. When a drug reduces liver fat by 82%, there is a theoretical concern that the process of clearing that fat could stress the liver. The data show that it does not.
What About Injection Site Reactions?
Injection site reactions, including redness, mild itching, and small nodules, occurred in 5 to 15% of participants across trials. These rates were similar to placebo, meaning the act of injecting itself (not the drug) causes most of them.
Reactions were mild and self-resolving. Management is straightforward: rotate injection sites between abdomen, thigh, and upper arm. Inject at least 2 inches from the navel. Let the alcohol swab dry before injecting. A cool compress resolves redness within 30 minutes to a few hours. Over-the-counter hydrocortisone cream helps persistent itching.
For detailed injection technique, see our peptide injections complete guide.
Does Retatrutide Cause Thyroid Cancer? (The Boxed Warning)
Every GLP-1 drug, including semaglutide (Ozempic, Wegovy), tirzepatide (Mounjaro, Zepbound), and retatrutide, carries the same FDA boxed warning about thyroid cancer. This is the most serious type of warning a drug can carry. Here is what it means and why the human risk appears different from what the warning suggests.
The Rodent Data
In animal studies, GLP-1 receptor agonists caused thyroid C-cell tumors (a specific type of thyroid cancer called medullary thyroid carcinoma, or MTC). The effect was dose-dependent and time-dependent: higher doses for longer periods produced more tumors. This applies to the entire drug class, not just retatrutide.
Why Humans Are Different
The critical difference: GLP-1 receptors are densely expressed on thyroid C-cells in rodents but detected in only about 27% of human C-cell tumors (neoplasms, not normal tissue). Rodent thyroid cells are essentially covered in targets for the drug to hit. Human thyroid cells are not.
Additional reassurance:
No similar effect has been observed in non-human primates (which are closer to humans than rodents). No confirmed increase in human MTC rates has appeared in clinical trials or post-marketing surveillance of approved GLP-1 drugs. A Mayo Clinic study published in 2025 suggests that the slight association seen in some human data reflects detection bias (people on GLP-1 drugs get more medical monitoring, so more thyroid issues get found) rather than causation (Mayo Clinic, 2025). A 2025 Diabetes Care study found that GLP-1 receptor agonists carry similar thyroid cancer risk as DPP-IV inhibitors, a completely different drug class (Diabetes Care, 2025).
Who Should Not Take Retatrutide
Two groups have absolute contraindications:
People with a personal or family history of medullary thyroid carcinoma (MTC). People with Multiple Endocrine Neoplasia syndrome type 2 (MEN2), a rare genetic condition.
These are standard contraindications for every GLP-1 drug. If you have a family history of thyroid cancer, discuss the specific type with your doctor before considering any drug in this class.
What Rare but Serious Side Effects Should You Watch For?
Beyond the common GI issues, retatrutide carries risks shared by all GLP-1 receptor agonists. These are uncommon but important to know.
Acute Pancreatitis
One case of pancreatitis (inflammation of the pancreas) was reported in the Phase 2 trial out of 338 participants. This is a class effect for all GLP-1 drugs.
Warning signs: severe abdominal pain that radiates to your back, persistent vomiting, and inability to find a comfortable position. If these develop, stop the medication and seek emergency care immediately. Do not wait to see if it resolves.
Gallbladder Events
Rapid weight loss, from any cause (drugs, surgery, or dieting), increases gallstone risk. This is not unique to retatrutide. Any therapy producing more than 10% weight loss carries this risk. Retatrutide produces 28.7% weight loss, so gallbladder monitoring is prudent.
Gallbladder event rates in clinical trials were approximately 0.5 to 1%, consistent with other GLP-1 drugs at similar weight loss magnitudes. Gallstones form when bile composition shifts during rapid fat mobilization. Warning signs: sudden right upper abdominal pain after eating fatty foods, pain radiating to the right shoulder, nausea with fever. If these develop, seek evaluation promptly.
Hypoglycemia (Low Blood Sugar)
When used alone, retatrutide carries low risk of hypoglycemia. Both the GLP-1 and GIP components are glucose-dependent: they stimulate insulin only when blood sugar is elevated and shut off when glucose is normal.
The risk increases significantly if retatrutide is combined with insulin or sulfonylureas (another class of diabetes medications). Dose adjustments of those drugs are necessary. Use our peptide interaction checker to evaluate potential drug interactions.
Severe Allergic Reactions
Anaphylaxis (a life-threatening allergic reaction) is rare with any injectable medication. Standard first-dose monitoring applies. Signs include swelling of the face or throat, difficulty breathing, and widespread rash. Discontinue and seek emergency care if these occur.
How Many People Stop Treatment Because of Side Effects?
Discontinuation rates tell you how tolerable a drug actually is in practice. If side effects are truly mild, people stay on the drug. If they are unbearable, people quit. Here are the numbers.
| Trial | Dose | Stopped Due to Side Effects | Placebo |
|---|---|---|---|
| Phase 2 (48 weeks) | All doses | 6-16% | 0% |
| Phase 3 TRIUMPH-4 (68 weeks) | 9 mg | 12.2% | 4.0% |
| Phase 3 TRIUMPH-4 (68 weeks) | 12 mg | 18.2% | 4.0% |
| Phase 3 TRIUMPH-4 (BMI >=35 subgroup) | 9 mg | 8.8% | 4.8% |
| Phase 3 TRIUMPH-4 (BMI >=35 subgroup) | 12 mg | 12.1% | 4.8% |
| Phase 3 TRANSCEND-T2D-1 | 9 mg | 4.5% | 0.0% |
| Phase 3 TRANSCEND-T2D-1 | 12 mg | 5.1% | 0.0% |
The contrast between trials is striking. TRANSCEND-T2D-1 showed 5.1% discontinuation at 12 mg, compared to 18.2% in TRIUMPH-4. The diabetes trial population tolerated retatrutide at rates comparable to tirzepatide and semaglutide (~7%).
Within TRIUMPH-4, people with higher starting BMI had lower discontinuation rates. At 12 mg, 18.2% of all participants stopped, but only 12.1% of those with BMI above 35. The likely explanation is that when the drug is working dramatically (greater weight loss in heavier individuals), people tolerate the side effects better because the benefit feels worth it. Some discontinuations in the trial were actually due to "perceived excessive weight loss" in lower-BMI participants.
Pooled data across trials confirms the pattern: at 4 mg, dropout rates were only slightly above placebo. At 8 mg, the difference became statistically meaningful. At 12 mg, people were roughly 6.7 times more likely to quit than those on placebo (PMC meta-analysis, 2025).
For comparison, tirzepatide and semaglutide have discontinuation rates around 7%. Retatrutide's 18.2% at 12 mg is higher, though the TRIUMPH-4 population (patients with both obesity and knee osteoarthritis) may have broader health issues contributing to dropouts.
How Do Retatrutide Side Effects Compare to Tirzepatide and Semaglutide?
The table below compares side effect rates across the three most studied GLP-1-based weight loss medications, using the highest tested dose for each. Numbers come from different trials with different patients, durations, and endpoints. They are useful for relative tolerability, not head-to-head comparisons.
| Side Effect | Retatrutide 12 mg (TRIUMPH-4) | Retatrutide 12 mg (TRANSCEND-T2D-1) | Tirzepatide 15 mg | Semaglutide 2.4 mg |
|---|---|---|---|---|
| Nausea | 43% | 26.5% | ~31% | ~44% |
| Diarrhea | 33% | 22.8% | ~21% | ~30% |
| Vomiting | 21% | 17.6% | ~12% | ~24% |
| Constipation | 25% | ~11% | ~24% | |
| Dysesthesia | 20.9% | 4.4% | Not reported | Not reported* |
| Heart rate increase | ~6.7 bpm peak | ~2-3 bpm | ~3-4 bpm | |
| Max weight loss | 28.7% | 22.5% | 14.9% | |
| Stopped due to side effects | 18.2% | 5.1% | ~7% | ~7% |
*Dysesthesia has been reported anecdotally with semaglutide and tirzepatide at much lower rates per a 2025 case series in Cureus.
GI side effects are broadly similar across all three drugs because they share the GLP-1 receptor. The TRANSCEND-T2D-1 column is revealing: when retatrutide is tested in a different population, its GI rates drop below semaglutide's. The differences that persist: retatrutide's higher heart rate increase is likely due to the glucagon component. Dysesthesia at meaningful rates appears unique to retatrutide, though the true rate may be closer to 4 to 5% (TRANSCEND) than 21% (TRIUMPH-4). The higher discontinuation rate in TRIUMPH-4 likely reflects that population (obesity + knee OA) rather than the drug itself. CagriSema (cagrilintide + semaglutide), another next-generation combination, is also in Phase 3 trials but uses amylin rather than glucagon, producing a different side effect profile.
Hair loss has not been reported as a side effect in retatrutide clinical trials. For context on this common concern, see does retatrutide cause hair loss.
Eli Lilly is running a direct head-to-head trial of retatrutide vs tirzepatide (NCT06662383, 800 patients). Results are expected in December 2026. Until then, all comparisons are indirect. For a deep-dive into the comparison, see our retatrutide vs tirzepatide article.
How Can You Manage Retatrutide Side Effects?
Most retatrutide side effects can be managed without stopping the drug. The strategies below are evidence-based and used by clinicians managing patients on GLP-1 therapies.
Dose Escalation: The Single Most Important Factor
Always start at 2 mg. Never skip the starting dose. Phase 2 data showed that GI symptoms nearly doubled when patients started at 4 mg instead of titrating from 2 mg.
The standard schedule: 2 mg (weeks 1 to 4), then 4 mg (weeks 5 to 8), then 8 mg (weeks 9 to 12), then 12 mg (week 13 onward). For complete administration guidance, see how to take retatrutide. If side effects are severe at any step, hold the current dose for an additional 2 to 4 weeks before moving up. There is no penalty for going slowly. The drug still works at lower doses; you simply reach the maximum effect later.
Plan your titration with our retatrutide dosage calculator. For a visual reference, see the peptide dosage chart.
Dietary Strategies for GI Side Effects
What you eat during dose escalation matters significantly.
Eat smaller, more frequent meals: 5 to 6 per day instead of 3 large ones. Smaller portions put less strain on a stomach that is emptying slowly. Prioritize protein: Greek yogurt, cottage cheese, eggs, lean chicken, protein shakes. When solid food is poorly tolerated, liquid protein is easier on the stomach. Avoid fried, greasy, and high-sugar foods, especially during the first weeks at a new dose. These are the hardest for a slow-emptying stomach to process.
Evidence-based dietary protocols for GLP-1 patients recommend moderate carbs and low fat as the foundation (PMC dietary recommendations, 2025). Similar dietary principles apply across GLP-1 drugs; see what to eat on tirzepatide for detailed meal planning guidance.
Hydration and Electrolytes
Diarrhea and vomiting both cause fluid loss. Minimum intake: 2 to 3 liters of water daily. If diarrhea persists for more than a few days, add electrolyte supplementation (sodium, potassium, magnesium). An electrolyte drink mix or oral rehydration solution is more effective than water alone.
Anti-Nausea Options
Ginger is a well-studied natural antiemetic. Fresh ginger tea, ginger supplements (250 mg four times daily), or real ginger ale (check the label for actual ginger, not just flavoring) can reduce nausea by 20 to 40%. Many of these strategies overlap with other GLP-1 drugs; see how to relieve nausea from semaglutide for additional techniques.
Ondansetron (Zofran), a prescription anti-nausea medication, is commonly prescribed off-label for GLP-1 nausea. It works by blocking serotonin receptors in the gut. Use it short-term during dose escalation periods, not chronically to mask symptoms while escalating too fast.
Vitamin B6 (10 to 25 mg) is a safe over-the-counter option with mild antiemetic properties.
When to call your doctor: persistent vomiting for more than 48 hours, inability to keep fluids down, signs of dehydration (dark urine, dizziness, dry mouth, rapid heartbeat).
Managing Dysesthesia
There is no established treatment protocol for retatrutide-associated dysesthesia. Current clinical guidance:
If mild (tingling that is noticeable but not distressing): continue treatment and monitor. If bothersome (affecting daily activities or sleep): discuss dose reduction with your provider. Symptoms have not been shown to cause long-term nerve damage based on available data.
Updated guidance will emerge as more TRIUMPH trials report results throughout 2026.
Injection Site Care
Rotate sites between abdomen, upper thigh, and upper arm. Inject at least 2 inches from the navel. Let the alcohol swab dry completely before injecting (injecting through wet alcohol stings). A cool compress resolves redness within 30 minutes. OTC hydrocortisone cream handles persistent itching.
For complete injection technique, see our peptide injections complete guide. For storage instructions, see how to store peptides.
When Should You Seek Medical Attention?
Most retatrutide side effects are manageable at home. The following situations require prompt medical care:
Severe abdominal pain radiating to the back. This could indicate pancreatitis. Stop the medication and go to an emergency room.
Persistent vomiting preventing fluid intake for more than 48 hours. Dehydration from GLP-1 drugs can become serious.
Signs of allergic reaction: swelling of the face or throat, difficulty breathing, widespread severe rash.
A lump or swelling in the neck. While the thyroid cancer risk in humans appears low, any thyroid changes should be evaluated.
Sudden right upper abdominal pain with fever or yellowing of the skin (jaundice). This could indicate a gallbladder attack.
Signs of hypoglycemia if you take insulin or sulfonylureas: shakiness, confusion, sweating, rapid heartbeat.
Chest pain, palpitations, or fainting. While the heart rate increase is expected, new cardiac symptoms always warrant evaluation.
What Do We Still Not Know About Long-Term Safety?
Retatrutide has been studied for a maximum of 68 weeks in Phase 3. That is about 16 months. For a drug that many people would take for years or even lifelong, this is a limited window.
Key unknowns:
Dysesthesia with extended use. Does it stabilize, worsen, or resolve with time? No one knows yet.
Cardiovascular outcomes. The TRIUMPH-3 trial is specifically designed to answer whether retatrutide prevents heart attacks and strokes. Results are pending.
Cancer risk with multi-year use. The thyroid C-cell concern requires years of surveillance data. Other cancer risks have not been evaluated beyond trial duration.
Weight regain after stopping. What happens to side effects and weight when the drug is discontinued? Data from semaglutide and tirzepatide shows significant weight regain after stopping. Retatrutide data on this does not exist.
Safety in special populations. Renal impairment, elderly patients, adolescents. These have not been thoroughly studied.
TRANSCEND-T2D-1 provided a second data point, and its substantially lower side effect rates suggest that the TRIUMPH-4 population may not be representative. Additional Phase 3 trials are reporting results throughout 2026, which will significantly expand the safety dataset. The TRIUMPH-1 and TRIUMPH-2 trials (obesity-only, without the knee OA comorbidity) will be particularly important for establishing the true side effect profile. The full TRIUMPH program design was published in Diabetes Obesity and Metabolism (Giblin et al., DOM 2026).
For an overview of where retatrutide fits among weight loss peptides, see our best peptides for weight loss guide. For beginners, start with getting started with peptides.
Frequently Asked Questions
What are the most common side effects of retatrutide?
Nausea (38 to 43%), diarrhea (33 to 35%), constipation (22 to 25%), and vomiting (20 to 21%) are the most common side effects based on Phase 3 TRIUMPH-4 data. These are primarily gastrointestinal and dose-dependent. They occur more frequently at higher doses and during dose escalation periods. Most cases are mild to moderate and improve within 8 to 12 weeks.
Does retatrutide cause dysesthesia (tingling or burning sensations)?
Yes. Dysesthesia was reported in 8.8% of patients on 9 mg and 20.9% on 12 mg in the Phase 3 TRIUMPH-4 trial, compared to 0.7% on placebo. The events were generally mild and did not lead to treatment discontinuation. This is a new safety signal not seen in the earlier Phase 2 trial. The glucagon receptor component is suspected as a contributing factor.
How long do retatrutide side effects last?
Most GI side effects peak during the first 4 to 8 weeks of treatment or during dose escalation. In clinical trials, symptoms diminished significantly after 8 to 12 weeks, particularly with gradual dose escalation starting at 2 mg. Heart rate increases peak at week 24 and decline thereafter. Dysesthesia duration data is limited, but events in TRIUMPH-4 were generally transient.
Is retatrutide safer than semaglutide or tirzepatide?
The GI side effect profiles are broadly similar across all three drugs. Retatrutide has a unique dysesthesia signal (4.4 to 20.9% at 12 mg depending on trial population) not seen with the other two drugs, and a higher heart rate increase (~6.7 bpm vs 2 to 4 bpm). TRANSCEND-T2D-1 showed GI rates (nausea 26.5%) comparable to or lower than semaglutide (44%) and tirzepatide (31%). Retatrutide produces substantially greater weight loss (28.7% vs 22.5% vs 14.9%) and liver fat reduction (86% vs ~55% vs ~45%). No head-to-head safety trials exist yet. Tirzepatide has the longest real-world safety track record.
Can retatrutide cause thyroid cancer?
GLP-1 receptor agonists caused thyroid C-cell tumors in rodent studies, but this has not been confirmed in humans. The key difference: GLP-1 receptors are heavily expressed on rodent thyroid C-cells but barely present on human C-cells. A 2025 Mayo Clinic study suggests detection bias, not causation. Retatrutide carries the same class-wide boxed warning. People with a personal or family history of medullary thyroid carcinoma (MTC) or MEN2 syndrome should not take it.
What is the discontinuation rate for retatrutide?
In Phase 3 TRIUMPH-4, 12.2% on 9 mg and 18.2% on 12 mg discontinued due to adverse events, compared to 4.0% on placebo. Patients with higher baseline BMI had lower discontinuation rates (12.1% at 12 mg for BMI above 35), suggesting better tolerability when the weight loss benefit is greater. For comparison, tirzepatide and semaglutide discontinuation rates are approximately 7%.
Does retatrutide affect your heart?
Retatrutide increases resting heart rate by approximately 5 to 7 bpm in a dose-dependent manner, peaking at 24 weeks then declining. It also significantly improves blood pressure (41% of patients on 8 mg discontinued antihypertensives) and lipid profiles (triglycerides down 40.6%, non-HDL-C down 26.9%). No serious cardiac events were attributed to retatrutide in clinical trials. The TRIUMPH-3 cardiovascular outcomes trial will provide definitive data.
Is retatrutide FDA approved?
No. As of April 2026, retatrutide is in Phase 3 clinical trials (the TRIUMPH and TRANSCEND programs). Eli Lilly reported positive results from TRIUMPH-4 in December 2025 and TRANSCEND-T2D-1 in early 2026. Additional Phase 3 trials are expected to report throughout 2026. FDA submission is anticipated for late 2026 or early 2027, with potential approval in late 2027. See our how to get retatrutide guide for clinical trial enrollment, expanded access options, and FDA-approved alternatives available now. For the regulatory landscape, see retatrutide availability timeline.
Can women take retatrutide during pregnancy?
No. Retatrutide is contraindicated during pregnancy and breastfeeding, consistent with all GLP-1 receptor agonists. Weight loss during pregnancy poses risks to the fetus. Women of childbearing potential should use effective contraception during treatment and for at least 4 weeks after the last dose. GLP-1 drugs slow gastric emptying, which can reduce absorption of oral contraceptives. Non-oral contraception (IUD, implant, injection) is recommended during treatment. Fertility itself does not appear to be affected, and some women may experience improved fertility as PCOS symptoms resolve with weight loss.
Does retatrutide affect hormones?
Retatrutide acts on three hormone receptors (GLP-1, GIP, glucagon) but does not directly alter sex hormones like testosterone or estrogen. Indirect hormonal changes occur through weight loss: testosterone levels in men often normalize, PCOS symptoms in women may improve, and insulin sensitivity increases substantially. These are consequences of weight loss itself, not direct drug effects. Thyroid hormone levels are monitored as a precaution due to the GLP-1 class warning, but no disruption has been observed in clinical trials.
The Bottom Line
Retatrutide's side effect profile reflects the power of its triple mechanism. Activating three receptors produces more weight loss than any previous drug in its class. It also produces more side effects, though the severity varies significantly by population. TRIUMPH-4 (obesity + knee OA) showed higher rates across the board. TRANSCEND-T2D-1 (type 2 diabetes) showed rates comparable to existing GLP-1 drugs. The truth likely sits somewhere between these two trials.
Nausea, diarrhea, and vomiting are common but manageable and time-limited. Dysesthesia requires monitoring, though its true prevalence may be closer to 4 to 5% than the 21% headline from TRIUMPH-4. The heart rate increase is higher than competitors. Liver safety is excellent.
For most people, the side effects are worst during the first months and during each dose increase. Slow titration, dietary adjustments, and hydration handle the majority of issues. Discontinuation rates ranged from 5.1% (TRANSCEND-T2D-1) to 18.2% (TRIUMPH-4) at 12 mg, meaning the vast majority of participants completed treatment.
Retatrutide is not approved, and long-term safety data beyond 68 weeks does not exist. Additional Phase 3 trials reporting in 2026, particularly the pivotal TRIUMPH-1 and TRIUMPH-2 obesity-only trials, will sharpen the picture considerably. Anyone considering this drug should discuss the specific risks and benefits with their healthcare provider.
For the full mechanism overview, see how does retatrutide work. For broader peptide safety considerations, see the peptide safety guide and our peptide therapy side effects overview. For alternatives if you struggle with GLP-1 side effects, see not losing weight on semaglutide or why am I not losing weight on tirzepatide. For the regulatory landscape, see our FDA peptide crackdown 2026 overview and peptide statistics 2026. Explore peptide profiles, dosage calculators, and research tools at PeptidesExplorer.
Related Articles: - Retatrutide: Complete Guide - Retatrutide Dosage Guide - Retatrutide vs Tirzepatide - How to Get Retatrutide - Best Peptides for Weight Loss
Related Articles
Retatrutide Availability & Timeline
Retatrutide is expected to receive FDA approval in mid-2027 with pharmacy availability in late 2027. Full timeline and Phase 3 trial status.
Retatrutide: Mechanism & Trial Results
Retatrutide (LY3437943) is a triple agonist weight loss drug by Eli Lilly. 28.7% weight loss in trials, 82% liver fat reduction. Not yet FDA-approved.
How to Get Retatrutide in 2026
How to access retatrutide in 2026: clinical trial enrollment, compounding pharmacy status, FDA approval timeline (est. 2027). Updated March 2026.
How Long Does Retatrutide Take to Work?
Retatrutide suppresses appetite within days, produces weight loss by week 4, and delivers up to 24.2% loss at 48 weeks in trials.