You cannot get a prescription for retatrutide anywhere in the world right now. No doctor can prescribe it. No pharmacy can fill it. As of March 2026, it is still an experimental drug being tested in clinical trials run by Eli Lilly.
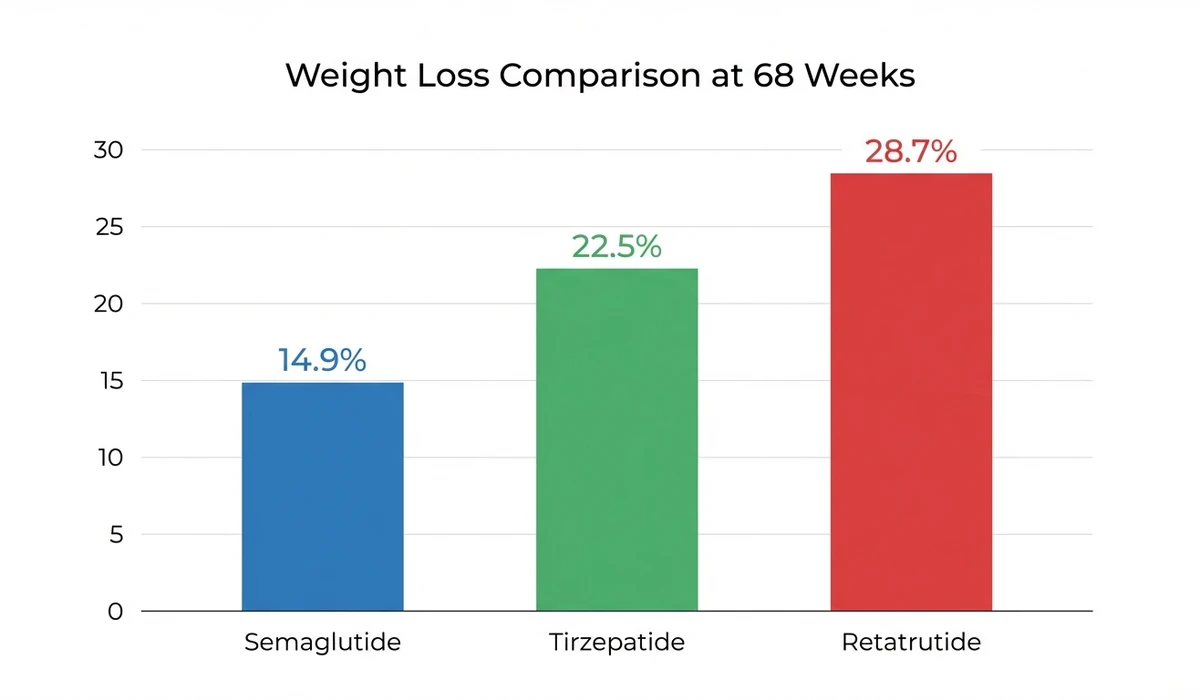
Retatrutide (also known by its lab name LY3437943) works by activating three gut hormone receptors at once: GLP-1, GIP, and glucagon. Think of it as a next-generation version of drugs like Ozempic (which hits one receptor) and Zepbound (which hits two). In the largest trial so far, people on retatrutide lost an average of 28.7% of their body weight over about 16 months. That is the highest weight loss ever recorded for any drug (Lilly Press Release, Dec 2025). The FDA is expected to approve it around mid-2027, with pharmacy availability in late 2027 or early 2028.
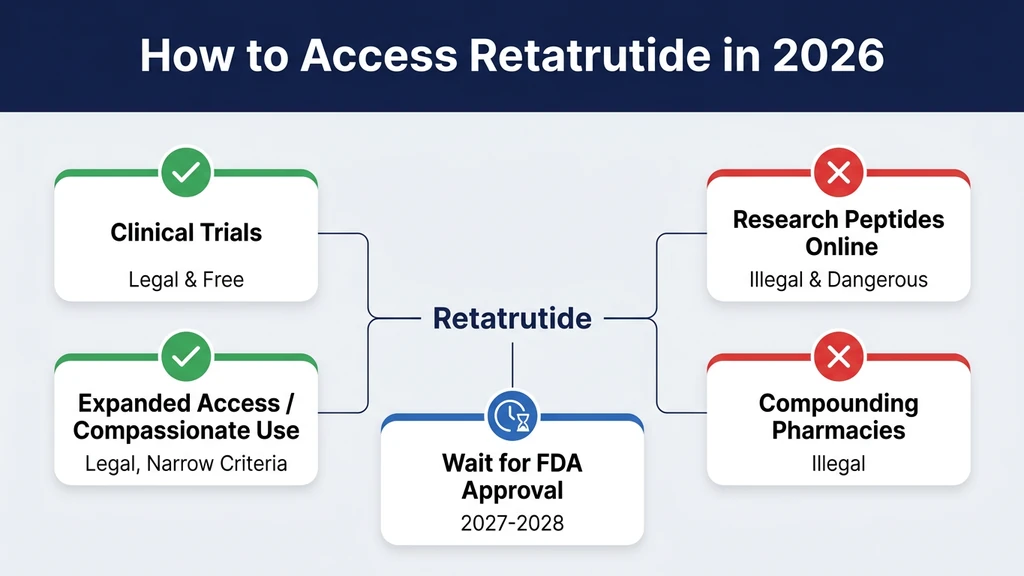
This guide walks through every way to access retatrutide today (clinical trials, expanded access, research peptides), explains the legal and safety landscape, and covers what you can do while waiting. Last updated: March 2026.
For a full overview of what retatrutide is, how it works, and what the trials show, see our complete retatrutide guide. This is educational content about an experimental drug. Talk to a doctor before making any medical decisions.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Is Retatrutide Available Yet?
No. Retatrutide is still in the testing phase. It has not been approved by the FDA (the US agency that decides which drugs are safe enough to sell), the European Medicines Agency, or any other health authority anywhere in the world. No doctor can legally prescribe it, and no pharmacy can dispense it.
Here is what we know as of March 2026:
The biggest trial so far (called TRIUMPH-4) released results in December 2025. People taking the highest dose (12 mg) lost an average of 71 pounds over 16 months, starting from an average weight of about 249 pounds. That works out to 28.7% of their body weight. People were still losing weight when the trial ended, meaning the final number could be even higher with longer use (Lilly Press Release, Dec 2025).
Seven more large-scale trials (the TRIUMPH program, over 5,800 people total) are still running and will release results throughout 2026. These cover obesity, type 2 diabetes, heart disease, knee arthritis, and liver disease (Giblin et al., DOM 2026).
For a detailed look at what retatrutide does and how it compares to existing drugs, see our retatrutide vs tirzepatide comparison.
When Will Retatrutide Be FDA-Approved?
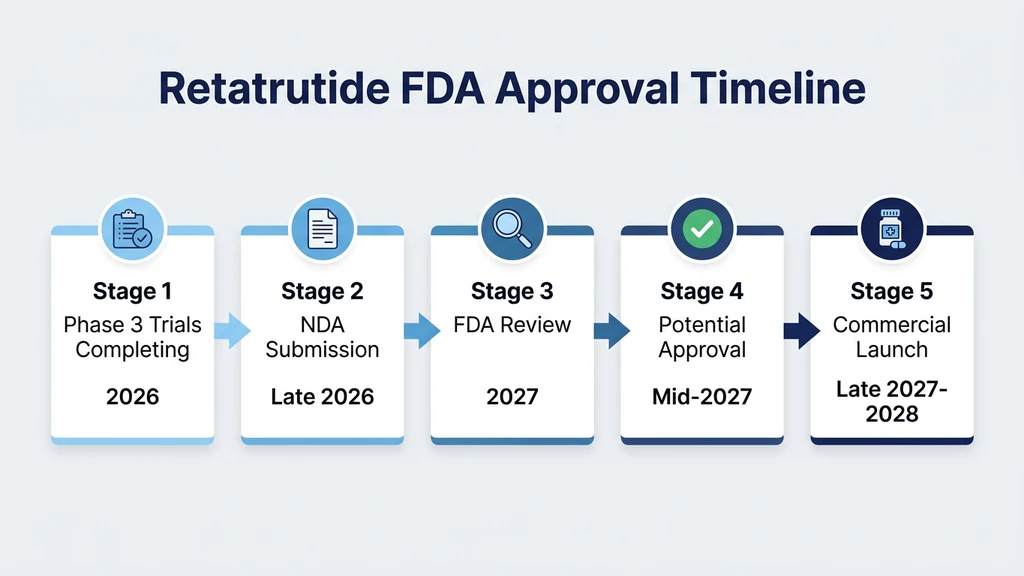
The approval process has several steps, each with its own timeline. No step can be skipped.
Phase 3 Trial Completion (2026)
Before Eli Lilly can ask for approval, it needs to finish its main trials. Drug trials happen in phases: Phase 1 tests safety in a small group, Phase 2 checks if it works, and Phase 3 tests it in thousands of people to confirm everything. Retatrutide is in Phase 3.
The two biggest ongoing trials are TRIUMPH-1 (2,300 people with obesity) and TRIUMPH-2 (1,000 people with type 2 diabetes and obesity). Both should finish around mid-2026.
There is also a trial specifically testing whether retatrutide prevents heart attacks and strokes (TRIUMPH-3, 1,800 people), and a head-to-head trial comparing retatrutide directly against tirzepatide (the active ingredient in Zepbound) under identical conditions (NCT06662383, 800 patients). That comparison should have results by December 2026.
Eli Lilly Files the Paperwork (Late 2026)
After the trials finish, Eli Lilly puts together a massive application (called a New Drug Application, or NDA) that includes all the safety and effectiveness data. Think of it as the company's homework proving the drug works and is safe enough to sell. This takes several months to prepare. Analysts expect Lilly to file in late 2026, though the company has not confirmed an exact date.
FDA Review (2027)
Once the FDA receives the application, it takes 10 months for a standard review. There is a faster track called Priority Review (6 months) for drugs that represent a major improvement over what is already available. Given that retatrutide produced nearly 29% weight loss, more than any existing drug, the faster review is possible. Earliest approval decision: mid-2027.
Available at Pharmacies (Late 2027 to Early 2028)
After approval, Eli Lilly needs a few more months to manufacture enough supply and negotiate insurance coverage. Based on Zepbound's launch (Lilly's other weight loss drug), expect a price around $1,000 or more per month before insurance. Lilly usually offers savings cards that can drop the cost significantly. Industry analysts project retatrutide will hit pharmacy shelves in 2028.
| Milestone | Expected Timeline |
|---|---|
| TRIUMPH-1 and TRIUMPH-2 results | Mid-2026 |
| Head-to-head vs tirzepatide results | December 2026 |
| NDA submission to FDA | Late 2026 |
| FDA review period | 6 to 10 months |
| Potential approval | Mid-2027 |
| Commercial availability | Late 2027 to early 2028 |
How Can You Get Retatrutide Through Clinical Trials?
If you want pharmaceutical-grade retatrutide (the real thing, made by Eli Lilly, under medical supervision), a clinical trial is the way to get it. Everything is free: the drug itself, doctor visits, blood work, and health monitoring.
The TRIUMPH Trial Program
Eli Lilly's Phase 3 program includes eight trials spanning multiple conditions. Several are still recruiting participants.
| Trial | Focus | NCT ID | Enrollment | Status |
|---|---|---|---|---|
| TRIUMPH-1 | Obesity | NCT05929066 | 2,300 | Active |
| TRIUMPH-2 | Type 2 diabetes + obesity | NCT05929079 | 1,000 | Active |
| TRIUMPH-3 | Cardiovascular + obesity | NCT05882045 | 1,800 | Active |
| TRIUMPH-4 | Knee osteoarthritis | NCT05931367 | 445 | Completed (Dec 2025) |
| TRIUMPH-Outcomes | CV/kidney long-term | NCT06383390 | TBD | Enrolling |
| Head-to-head | Retatrutide vs tirzepatide | NCT06662383 | 800 | Active |
| Maintenance | Dose reduction after weight loss | NCT06859268 | TBD | Recruiting |
| New enrollment | Obesity/overweight | NCT07232719 | 250 | Recruiting |
Step 1: Search for Active Trials Near You
Two places to search:
ClinicalTrials.gov: Go to clinicaltrials.gov and search for "retatrutide" or "LY3437943" (the research designation). Filter by location and recruiting status.
Eli Lilly Trial Finder: Visit trials.lilly.com for a more patient-friendly interface. You can enter your zip code and medical conditions to see matching trials.
Look for trials with "Recruiting" or "Enrolling by invitation" status. "Active, not recruiting" means the trial has filled its enrollment.
Step 2: Check If You Qualify
Most TRIUMPH trials require:
You need a BMI of 30 or higher. BMI is a number calculated from your height and weight. A 5'9" person weighing 203 lbs has a BMI of 30. You can also qualify with a BMI of 27 or higher if you have a weight-related health problem like high blood pressure, high cholesterol, sleep apnea, or heart disease. You must be at least 18 years old, and you must have tried to lose weight through diet or exercise before without lasting success.
You will likely be turned away if you have a history of pancreatitis (pancreas inflammation), a thyroid cancer called medullary thyroid carcinoma in your family, if you are pregnant, if your weight has changed by more than 11 lbs in the past 3 months, or if you are already taking a weight loss drug.
Step 3: Apply and Go Through Screening
Contact the study coordinator listed on the trial page. They will walk you through a screening process: your medical history, blood tests, a physical exam, and a consent form.
One important thing to understand: you might not get the actual drug. In these trials, some participants receive a placebo (a fake injection with no active ingredient). This is how researchers prove the drug actually works, by comparing it against nothing. The trial is "double-blind," which means neither you nor your doctor knows whether you got the real drug or the placebo until the trial is over.
Benefits and Risks of Joining a Trial
The upside: Free access to pharmaceutical-grade retatrutide (if you get the real drug and not the placebo), regular checkups, blood work, and health monitoring at no cost. You also help advance research for future patients.
The downside: You might get the placebo. Side effects are common: about 43% of people felt nauseous, 33% had diarrhea, and about 1 in 5 experienced tingling or burning skin sensations (called dysesthesia) at the highest dose. You will need to show up for clinic visits regularly, sometimes for over a year. You cannot pick your dose, and you need to live close enough to a trial site to attend in person.
For a full breakdown of what side effects to expect, see our retatrutide side effects guide.
Can You Get Retatrutide Through Expanded Access (Compassionate Use)?
The FDA has a special program called "expanded access" (sometimes called compassionate use) for people who are seriously ill and have run out of options. It lets doctors request experimental drugs for individual patients outside of clinical trials.
Eli Lilly accepts these requests for retatrutide. Your doctor (not you directly) can call Lilly at 1-800-545-5979 (1-800-Lilly-Rx). The company promises to respond within 5 business days (Lilly Expanded Access).
The catch: being overweight alone probably will not qualify. This pathway is designed for people in immediate medical danger, for example someone whose obesity is causing severe heart failure or who cannot breathe properly because of their weight. You also need to have tried everything else and be unable to join a clinical trial. It is a very narrow door, but worth knowing about if you are in a serious medical situation.
Can You Buy Retatrutide as a Research Peptide?
Yes, retatrutide is sold online by research peptide companies. If you search for it, you will find dozens of vendors selling vials of retatrutide powder. These products are labeled "for research use only" and are not intended for human use. Here is what you need to understand about this market.
What Are Research Peptides?
Research peptide companies sell synthetic peptides as raw chemicals for laboratory use. The products typically arrive as a freeze-dried (lyophilized) powder in small vials, usually 5 mg or 10 mg. You would need to add sterile water to dissolve the powder before it could be used. Prices range from about $150 to $500 per month depending on the vendor and dose.
These companies source their peptides from chemical manufacturers, often based in China. The vials are labeled "for research purposes only, not for human consumption." This labeling is what allows them to sell the product without FDA approval, since technically they are selling a lab chemical, not a medicine.
In practice, many buyers purchase these peptides with the intention of self-administering them. This is an open secret in the peptide community. However, the "research use only" label means the product is not manufactured, tested, or regulated to pharmaceutical standards.
What Are the Risks?
The core problem is quality control. Pharmaceutical-grade retatrutide (the version used in Eli Lilly's clinical trials) is made under strict manufacturing standards with rigorous purity testing. Research peptides are not held to those standards.
Purity is not guaranteed. The FDA has found heavy metals, bacterial toxins (endotoxins), wrong peptide sequences, and contamination in seized samples of GLP-1 peptides sold online. Some vials contained something completely different from what the label said (FDA, 2025).
There is no medical supervision. Even the real, pharmaceutical-grade retatrutide causes significant side effects: nausea in 43% of people, diarrhea in 33%, and unusual skin sensations (tingling, burning) in about 1 in 5 people at the highest dose. These numbers come from trials where patients were monitored by doctors. Self-administering an unverified version without medical oversight adds a layer of risk.
Dosing is guesswork. Without knowing the actual purity of what is in the vial, you cannot accurately dose. Underdosing means it will not work. Overdosing increases side effect risk.
Some research peptide vendors provide third-party certificates of analysis (COA) showing purity testing. These can be helpful, but they vary in reliability: some come from reputable independent labs, others are easy to fabricate.
The Legal Landscape
Buying research peptides for personal research is a legal gray area. Selling them labeled "for research use only" is currently permitted. What is clearly illegal: marketing them for human use, making medical claims, or selling them as medicine.
The FDA is tightening enforcement. In September 2025, the agency issued over 50 warning letters to companies selling GLP-1 products (including retatrutide) with marketing that implied human use. In December 2025, Pinnacle Peptides received a warning letter for selling unapproved products (FDA Warning Letter). GenLabMeds got a similar letter in September 2025.
Congress is pushing to close loopholes. The SAFE Drugs Act, introduced in December 2025, would expand the FDA's power over compounded and unregulated GLP-1 products and the telehealth companies that prescribe them (Dykema analysis). US Customs can also seize peptide shipments at the border under Import Alert 66-80.
The Alliance for Pharmacy Compounding (the compounding industry's own trade group) has told its members twice: do not compound retatrutide.
What About Compounding Pharmacies?
Compounding pharmacies are specialty pharmacies that can custom-make certain medications. You may have heard of compounded semaglutide or compounded tirzepatide being available at lower prices. Retatrutide is different: compounding pharmacies cannot legally make it.
Why Compounding Pharmacies Cannot Make Retatrutide
US law only allows pharmacies to compound a drug if it meets certain conditions. Retatrutide fails all of them:
| Rule | What It Means in Plain English | Retatrutide |
|---|---|---|
| Published formula exists | There has to be an official recipe | No official recipe exists |
| Part of an approved drug | The ingredient must already be in an approved medication | Retatrutide has never been approved |
| Drug is in shortage | The approved version must be hard to get | There is no approved version, so a "shortage" is impossible |
The FDA has said this directly: retatrutide "cannot be used in compounding under federal law." Any pharmacy making it is breaking federal law, no matter what their state laws say (Ohio Board of Pharmacy / FDA Letter).
Why Could Semaglutide and Tirzepatide Be Compounded?
Those drugs followed a different path. Semaglutide (Ozempic/Wegovy) got FDA approval, then demand outstripped supply, so it went on the "drug shortage list." That shortage list temporarily opened a legal door for compounding pharmacies to make their own versions. Tirzepatide (Mounjaro/Zepbound) followed the same path.
As of early 2026, the FDA has shut down tirzepatide compounding because the shortage is over. Semaglutide compounding is in a gray area.
Retatrutide has never been through any of these steps. It has never been approved, so it cannot be in "shortage," so there is no legal basis for compounding. Period.
Could Compounding Become Legal Later?
Only if retatrutide gets FDA approval and then ends up on the drug shortage list, like semaglutide and tirzepatide did. But Eli Lilly has already shown it will fight compounders in court (the company has active lawsuits against pharmacies that compounded its other drugs). Congress is also moving to tighten the rules with the SAFE Drugs Act, which would cap compounding of copycat drugs at 20 units per month.
What FDA-Approved Alternatives Are Available Right Now?
If you want a weight loss medication today, two FDA-approved options exist. Both are available by prescription.
Tirzepatide (Zepbound / Mounjaro)
Tirzepatide is the closest available drug to retatrutide. It shares two of the three receptors (GLP-1 and GIP) and is made by the same company, Eli Lilly. FDA-approved for type 2 diabetes (Mounjaro, 2022) and chronic weight management (Zepbound, 2023).
Weight loss: approximately 22.5% at 72 weeks in the SURMOUNT-1 trial. Cost: $299 to $1,086 per month depending on format. Eli Lilly's savings card can bring the price down to $25 per month for eligible patients. Plan your dosing with our tirzepatide dosage calculator. For a detailed comparison, see retatrutide vs tirzepatide.
Semaglutide (Wegovy / Ozempic)
Semaglutide is the original "new generation" weight loss drug. It activates one receptor (GLP-1) instead of two or three. Made by Novo Nordisk. You may know it as Ozempic (for diabetes) or Wegovy (for weight loss). It produces about 15 to 17% body weight loss over 16 months.
Semaglutide is the most widely prescribed option and has the longest safety track record. It also has a proven heart benefit: in a large trial, it reduced heart attacks and strokes by 20%. Use our semaglutide dosage calculator for dosing guidance.
How Do They Compare?
| Feature | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Receptors | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| FDA status | Approved | Approved | Phase 3 |
| Max weight loss | ~17% (68 weeks) | ~22.5% (72 weeks) | ~28.7% (68 weeks) |
| Availability | Now | Now | Est. late 2027 |
| Brand names | Wegovy / Ozempic | Zepbound / Mounjaro | None yet |
| Nausea rate | ~20% | ~31% | ~43% |
| Liver fat reduction | ~40-50% | ~55% | ~82% |
| Injection | Once weekly | Once weekly | Once weekly |
Each generation of these drugs targets more receptors and produces more weight loss. The tradeoff is that more receptors also means more side effects, especially nausea and (for retatrutide) unusual skin sensations. For a broader view of peptide options, see our best peptides for weight loss guide.
Other New Drugs Coming Soon
Retatrutide is not the only next-generation weight loss drug in development. Several others are in late-stage testing:
Orforglipron (Eli Lilly): This one is a pill, not an injection. It works on the same GLP-1 receptor as Ozempic, but you swallow it daily instead of injecting weekly. Results are expected in 2026. If approved, this could be a game-changer for people who do not want to use needles.
Survodutide (Boehringer Ingelheim): Targets two receptors (GLP-1 and glucagon), like a simpler version of retatrutide. It showed about 15% weight loss in trials, with very promising results for reducing liver fat in people with fatty liver disease.
CagriSema (Novo Nordisk): A combination of two drugs (cagrilintide and semaglutide) that work through different pathways than retatrutide. Still in testing, results pending.
What Should You Do While Waiting for Retatrutide?
Retatrutide is at least 18 to 24 months away from being available at your pharmacy. Here is what you can do in the meantime.
Talk to Your Doctor About Available Options
Tirzepatide and semaglutide are available today and produce meaningful weight loss (15 to 22.5%). If your insurance covers weight management medications, a prescription is a straightforward first step. If cost is a barrier, manufacturer savings cards can reduce Zepbound to $25 per month and Wegovy has similar programs.
Starting one of these medications now does not lock you in. Once retatrutide is approved, you could switch. Since both retatrutide and tirzepatide are made by Eli Lilly, the transition should be straightforward. You would start retatrutide at the lowest dose and gradually increase it (called titration), regardless of what dose of tirzepatide you were on. See our retatrutide dosage guide for how the dose increase works.
Consider Clinical Trial Enrollment
If you meet the eligibility criteria and live near a trial site, clinical trial enrollment gives you free access to pharmaceutical-grade retatrutide under close medical supervision. You may receive placebo, but even that comes with regular medical monitoring that many people do not otherwise get. See the step-by-step enrollment guide above.
Explore Expanded Access If You Have Severe Complications
If you have life-threatening obesity-related complications, are ineligible for clinical trials, and have exhausted all approved treatments, ask your physician to contact Eli Lilly at 1-800-545-5979 about expanded access. This is a narrow pathway, but it exists for the most severe cases.
Build Good Habits Now
Weight loss drugs work better when combined with healthier eating and more movement. Building that foundation now will amplify whatever medication you eventually take. Clinical trials also require that you have already tried to lose weight through diet and exercise, so documenting those efforts now helps if you apply later.
Start tracking a few basic numbers: your weight, waist measurement, and blood pressure. If you can, get bloodwork done (blood sugar and cholesterol levels). This gives you a starting point so you can actually measure how much a future treatment helps.
Stay Informed
Key dates to watch in 2026: TRIUMPH-1 and TRIUMPH-2 results (mid-2026), head-to-head retatrutide vs tirzepatide results (December 2026), and any NDA filing announcements from Eli Lilly.
Set up free email alerts on ClinicalTrials.gov for "retatrutide" to be notified of new trial openings. Follow Eli Lilly's investor relations page for press releases. We will update this article as new data emerges. For dosing reference across all peptides, see the peptide dosage chart. For injection fundamentals, see the peptide injections complete guide.
Frequently Asked Questions
Is retatrutide available for purchase anywhere in 2026?
Not as a prescription drug. No doctor can prescribe it and no pharmacy can sell it as of March 2026. You can find it sold online as a research peptide (labeled 'for research use only, not for human consumption'), but these products are unregulated and not manufactured to pharmaceutical standards. The only way to get pharmaceutical-grade retatrutide is through a clinical trial.
When will retatrutide be FDA-approved?
Eli Lilly will likely submit its application in late 2026. The FDA then takes 6 to 10 months to review it. Best case: approval around mid-2027. More realistic estimate: late 2027, with the drug available at pharmacies in early 2028. Eli Lilly has not confirmed an exact date.
How do I sign up for a retatrutide clinical trial?
Go to ClinicalTrials.gov and search for 'retatrutide,' or visit trials.lilly.com and enter your zip code. Look for trials that say 'Recruiting.' You typically need a BMI of 30 or higher (or 27+ with a health condition like high blood pressure). Contact the study coordinator listed on the page to start the screening process. Everything is free: the drug, doctor visits, and lab work.
What is the difference between retatrutide and tirzepatide (Zepbound)?
Retatrutide targets three gut hormone receptors, tirzepatide targets two. More receptors means more weight loss: 28.7% for retatrutide vs 22.5% for tirzepatide in trials, but also more side effects. The key practical difference: tirzepatide (sold as Zepbound and Mounjaro) is approved and available now. Retatrutide is still being tested. A head-to-head comparison trial with 800 patients will give direct results in late 2026.
Can a compounding pharmacy make retatrutide?
No. Unlike semaglutide and tirzepatide (which could be compounded during shortages), retatrutide has never been FDA-approved. US law only allows compounding of drugs that already have approval, so there is no legal basis for any pharmacy to make it. The FDA sent over 50 warning letters in September 2025 to companies doing it anyway. Even the compounding industry's own trade group has told its members: do not compound retatrutide.
Is it safe to buy retatrutide peptides online?
Research peptide vendors sell retatrutide labeled 'for research use only.' These products are not manufactured to pharmaceutical standards and are not tested or regulated by the FDA. Some vendors provide third-party purity testing (certificates of analysis), which can offer some reassurance, but quality varies widely between suppliers. The FDA has found contamination in seized GLP-1 peptide samples, including heavy metals and bacterial toxins. If you choose to purchase research peptides, understand that you are taking on risk that does not exist with pharmaceutical-grade products.
What is the best weight loss drug you can actually get right now?
Tirzepatide (sold as Zepbound for weight loss, Mounjaro for diabetes) produces the most weight loss of any approved drug: about 22.5%. Semaglutide (Wegovy/Ozempic) is the other major option at about 15 to 17%. Both require a prescription and are covered by many insurance plans. Your doctor can help you decide which one makes sense for you.
Will insurance cover retatrutide when it is approved?
Nobody knows yet. Based on how it went with Zepbound and Wegovy, expect limited insurance coverage at first, especially for weight loss (diabetes coverage tends to be better). Eli Lilly usually offers savings cards that can dramatically reduce the cost. Right now, the Zepbound savings card brings the monthly cost down to $25 for people who qualify.
What are retatrutide's side effects?
Side effect rates vary by trial population. In TRIUMPH-4 (obesity + knee OA): nausea 43%, diarrhea 33%, vomiting 21%, constipation 25%, dysesthesia 20.9%. In TRANSCEND-T2D-1 (type 2 diabetes): nausea 26.5%, diarrhea 22.8%, vomiting 17.6%, dysesthesia 4.4%. Most side effects were mild to moderate and concentrated during dose escalation. See our retatrutide side effects guide for the full breakdown.
Can my doctor request retatrutide through compassionate use?
Possibly, but only in extreme cases. This pathway is for people who are seriously ill, have tried everything else, and cannot join a clinical trial. Your doctor can call Eli Lilly at 1-800-545-5979, and the company responds within 5 business days. Being overweight alone probably will not qualify. You would need life-threatening complications from your weight, like severe heart failure or the inability to breathe properly.
How much weight can you lose on retatrutide?
In the biggest trial, people on the highest dose lost an average of 71 pounds (28.7% of their body weight) over about 16 months. The lower dose produced 26.4% loss. For perspective, weight loss surgery typically produces 25 to 30% loss. Retatrutide is approaching surgical results without an operation.
The Bottom Line
Retatrutide is the most powerful weight loss drug in development, but it is not something you can get a prescription for yet. The FDA approval timeline points to late 2027 at the earliest, with pharmacy availability likely in early 2028.
Your options today: clinical trials give you access to the real, pharmaceutical-grade drug under medical supervision. Research peptides are sold online, but they are unregulated, not made to pharmaceutical standards, and carry quality risks. Compounding pharmacies cannot legally make retatrutide.
If you want a proven weight loss medication right now, tirzepatide (Zepbound) and semaglutide (Wegovy) are FDA-approved, available by prescription, and produce meaningful results. Starting one now does not prevent you from switching to retatrutide once it becomes available.
We will update this article as trial results come in throughout 2026. For the regulatory landscape, see FDA peptide crackdown 2026. For reconstitution, see how to reconstitute retatrutide. For the full mechanism overview, see how does retatrutide work. For sourcing other peptides, see where to buy peptides in 2026. For overall safety, see our peptide safety guide and getting started with peptides. Explore peptide profiles, dosage calculators, and research tools at PeptidesExplorer.
Related Articles: - Retatrutide: Complete Guide - Retatrutide Dosage Guide - Retatrutide Side Effects - Retatrutide vs Tirzepatide - FDA Peptide Crackdown 2026
Helpful Tools
Related Articles
Retatrutide Availability & Timeline
Retatrutide is expected to receive FDA approval in mid-2027 with pharmacy availability in late 2027. Full timeline and Phase 3 trial status.
Retatrutide: Mechanism & Trial Results
Retatrutide (LY3437943) is a triple agonist weight loss drug by Eli Lilly. 28.7% weight loss in trials, 82% liver fat reduction. Not yet FDA-approved.
Retatrutide vs Tirzepatide Compared
Retatrutide vs tirzepatide compared: 28.7% vs 22.5% weight loss, triple vs dual agonist mechanism, side effects, dosing, cost, and FDA status.
Retatrutide Side Effects: Phase 3 Trial Data
Retatrutide side effects from 2 Phase 3 trials (1,200+ patients): nausea 26-43%, dysesthesia 4-21%, heart rate +6.7 bpm. Tirzepatide compared.