You received compounded retatrutide. The vial contains a white lyophilized powder. The bacteriostatic water is beside it. Your protocol says "inject 2 mg weekly," but the vial has no mixing instructions and the syringe reads in units, not milligrams.
To reconstitute retatrutide, draw bacteriostatic water into a syringe, inject it slowly against the glass wall of the peptide vial, and swirl gently until the powder dissolves completely. For a 5 mg vial, add 1 mL of water to get 5 mg/mL. For a 10 mg vial, add 1 mL for 10 mg/mL or 2 mL for 5 mg/mL. The process takes under five minutes.
Retatrutide is a 39-amino-acid triple agonist peptide (GLP-1, GIP, and glucagon receptors) that produced 28.7% body weight loss in Phase 3 trials (Lilly Press Release, Dec 2025). Because it is still investigational and not available in prefilled pens, compounded retatrutide arrives as lyophilized powder that requires manual reconstitution before injection.
The rest of this guide covers every common vial size, concentration tables, syringe unit conversions, storage rules, shelf life, equipment, and the mistakes that lead to dosing errors. For instant math on any combination, use the Reconstitution Calculator.
*This article is for research and educational purposes only. It is not medical advice. Retatrutide is an investigational compound that has not received FDA approval. Consult a licensed healthcare provider before using any GLP-1 receptor agonist.*
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What You Need Before Starting
Gather every supply before opening the vial. Once the rubber stopper is punctured, you want to move quickly to minimize contamination risk.
Required materials:
- Lyophilized retatrutide vial (5 mg, 10 mg, or 20 mg)
- Bacteriostatic water (0.9% benzyl alcohol preserved)
- 3 mL or 5 mL syringe with an 18 to 21 gauge drawing needle
- Insulin syringes for dosing (U-100, 29 to 31 gauge, 0.3 mL or 0.5 mL)
- Alcohol swabs (70% isopropyl)
- Fine-point marker or label tape
- Clean, flat work surface
Optional but recommended:
- Sharps container for used needles
- Nitrile gloves
- 0.22 micron syringe filter (if concerned about particulate contamination)
The drawing syringe transfers water into the peptide vial. The insulin syringes measure and deliver individual doses afterward. These are two separate tools. Never use the same syringe for both tasks. For a full explanation of syringe types and gauge selection, see the peptide safety guide.
Bacteriostatic Water vs Sterile Water
Bacteriostatic water (BAC water) contains 0.9% benzyl alcohol as a preservative. This antimicrobial agent inhibits bacterial growth inside the vial after each needle puncture. BAC water extends the usable life of reconstituted retatrutide to 28 days when refrigerated at 2 to 8 degrees Celsius.
Sterile water contains no preservative. It works for single-use reconstitution where the entire vial is consumed within 24 hours. For multi-dose vials (anything you plan to draw from more than once), sterile water is a contamination risk. Each needle insertion introduces potential bacteria, and without benzyl alcohol, nothing stops them from multiplying.
For retatrutide reconstitution, bacteriostatic water is the standard and recommended solvent. The retatrutide dosage guide uses doses ranging from 2 mg to 12 mg per week. Unless you are using a small vial in a single injection, you will draw from the vial multiple times. That means BAC water.
For a deeper comparison of solvents across peptide types, see how much bacteriostatic water for semaglutide, which covers the same solvent principles in detail.
| Solvent | Preservative | Multi-Dose Safe? | Shelf Life After Mixing | Best For |
|---|---|---|---|---|
| Bacteriostatic water | 0.9% benzyl alcohol | Yes | 28 days at 2-8°C | All multi-dose vials |
| Sterile water | None | No | 24 hours | Single-use only |
| Normal saline (0.9% NaCl) | None | No | 24 hours | Not standard for retatrutide |
Step-by-Step Reconstitution Process
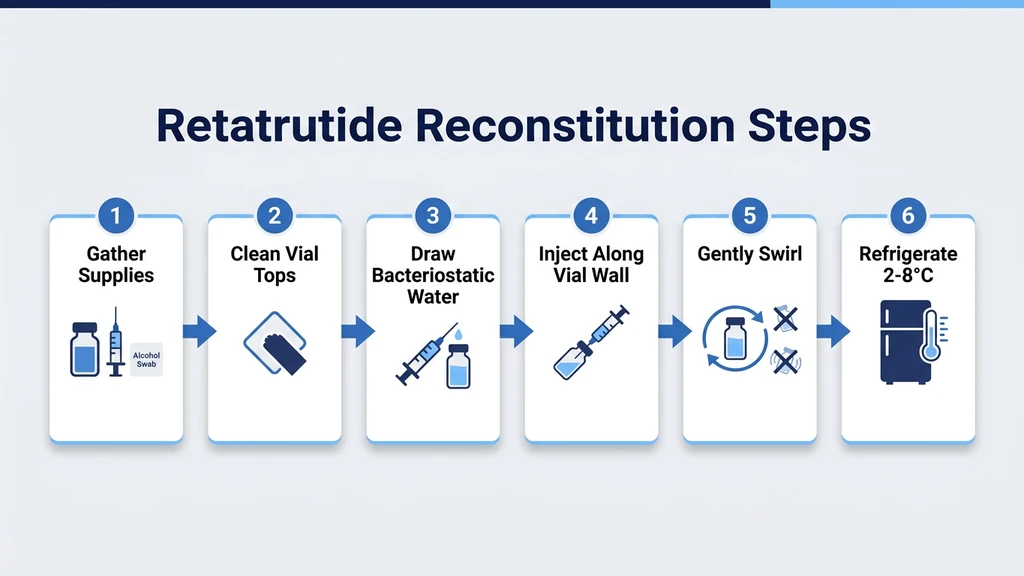
Follow these seven steps. The process takes two to five minutes from start to finish.
- 1.Clean both vial stoppers. Wipe the retatrutide vial stopper and the bacteriostatic water vial stopper with separate alcohol swabs. Let each air dry for 10 seconds. This removes surface bacteria that could enter the vial during needle insertion.
- 1.Draw bacteriostatic water. Attach the drawing needle (18 to 21 gauge) to the 3 mL syringe. Insert it into the BAC water vial, invert the vial, and pull the plunger to your target volume. Refer to the concentration tables below to choose how much water to add. Tap the syringe barrel to dislodge air bubbles and push them out.
- 1.Insert the needle into the retatrutide vial. Push the needle through the center of the rubber stopper at a slight angle with the bevel facing up. This prevents coring (punching a rubber fragment into the solution). Once through the stopper, straighten the needle.
- 1.Release the water slowly against the glass wall. Aim the needle tip at the inside wall of the vial, not directly onto the powder cake. Push the plunger steadily over 30 to 60 seconds. Let the water trickle down the glass and pool at the bottom beneath the lyophilized powder.
- 1.Remove the syringe and swirl gently. Hold the vial between your thumb and forefinger and rotate it in slow circles. Never shake. Vigorous agitation creates foam and can denature the peptide through mechanical shearing. Retatrutide is a 39-amino-acid peptide with a fatty acid moiety similar to tirzepatide, making it sensitive to agitation-induced aggregation. Most powder dissolves within 1 to 3 minutes of gentle swirling.
- 1.Inspect the solution. Reconstituted retatrutide should be clear and colorless. Hold the vial up to a light source. If you see particles, cloudiness, or discoloration, do not inject. A few small bubbles on the surface are normal and will dissipate within minutes.
- 1.Label and refrigerate immediately. Write the peptide name, concentration (e.g., "Retatrutide 10 mg/mL"), and today's date on the vial. Store upright in the refrigerator at 2 to 8 degrees Celsius. The solution is now ready for dosing.
If undissolved powder remains after 5 minutes of swirling, place the vial upright in the refrigerator for 10 to 15 minutes, then swirl again. Do not apply heat. Do not shake. Patience dissolves what force cannot.
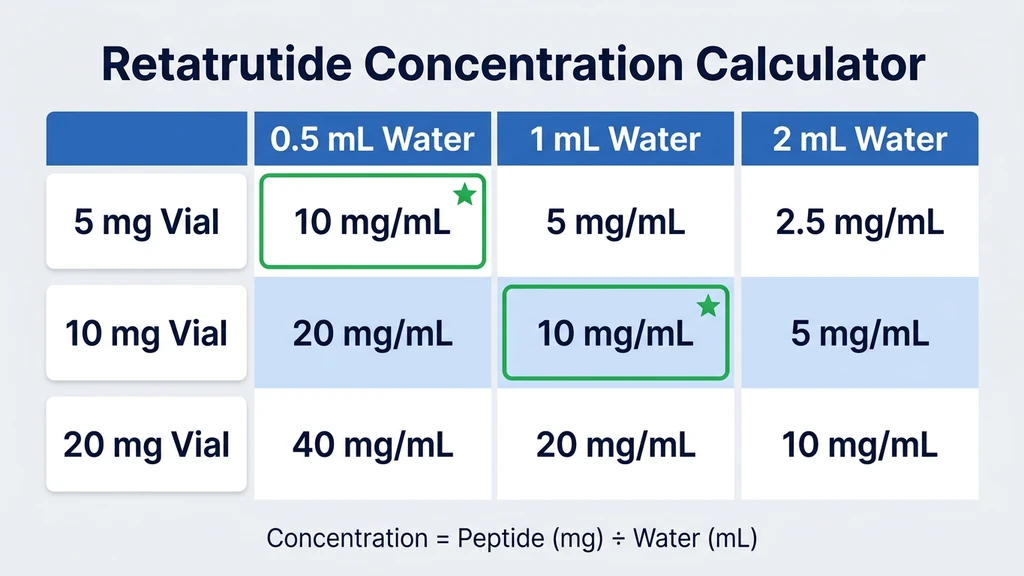
Concentration Tables by Vial Size
The reconstitution formula is: Concentration (mg/mL) = Peptide amount (mg) / Water added (mL).
This determines how much liquid you draw per dose. More water means a lower concentration, which means a larger draw volume per dose. Larger draw volumes are easier to measure accurately on an insulin syringe. Less water means a higher concentration and smaller, harder-to-read draw volumes.
Retatrutide clinical trial doses range from 2 mg (starting) to 12 mg (maximum maintenance), administered once weekly by subcutaneous injection (Jastreboff et al., NEJM 2023). All concentration tables below map to this dosing range. For any combination not listed, the Reconstitution Calculator gives instant results.
5 mg Vial
The 5 mg vial is practical for the starting dose phase (2 mg per week for weeks 1 through 4).
| BAC Water Added | Concentration | 2 mg Dose = | Syringe Units (U-100) |
|---|---|---|---|
| 0.5 mL | 10 mg/mL | 0.20 mL | 20 units |
| 1 mL | 5 mg/mL | 0.40 mL | 40 units |
| 2 mL | 2.5 mg/mL | 0.80 mL | 80 units |
Recommended for 5 mg vials: Add 1 mL for a 5 mg/mL concentration. At this concentration, the starting dose of 2 mg requires 40 units on a U-100 insulin syringe, which is easy to read on a 0.5 mL (50-unit) syringe. Adding 0.5 mL gives 10 mg/mL, which works but produces draw volumes of only 20 units, slightly harder to measure precisely.
A single 5 mg vial provides two doses at 2 mg per week (two weeks of supply at the starting dose) or one dose at 4 mg. Either fits comfortably within the 28-day stability window.
10 mg Vial
The 10 mg vial is the most versatile size. It covers the starting dose (2 mg), the first escalation (4 mg), and the second escalation (8 mg).
| BAC Water Added | Concentration | 2 mg = | 4 mg = | 8 mg = | Syringe Units per 2 mg |
|---|---|---|---|---|---|
| 0.5 mL | 20 mg/mL | 0.10 mL | 0.20 mL | 0.40 mL | 10 units |
| 1 mL | 10 mg/mL | 0.20 mL | 0.40 mL | 0.80 mL | 20 units |
| 2 mL | 5 mg/mL | 0.40 mL | 0.80 mL | 1.60 mL* | 40 units |
*Exceeds a 1 mL (100-unit) insulin syringe. Use a higher concentration or split into two draws.*
Recommended for 10 mg vials: Add 1 mL for 10 mg/mL. At this concentration, the starting dose of 2 mg requires 20 units, 4 mg requires 40 units, and 8 mg requires 80 units. All three fit within a standard 1 mL insulin syringe. For additional dose calculations, use the Retatrutide Dosage Calculator.
A 10 mg vial at 2 mg per week lasts 5 weeks. At 4 mg per week, it lasts 2.5 weeks. Both fit within the 28-day stability window. For the complete titration schedule, see the retatrutide dosage guide.
20 mg Vial
The 20 mg vial suits users on the higher maintenance doses of 8 mg or 12 mg per week.
| BAC Water Added | Concentration | 4 mg = | 8 mg = | 12 mg = | Syringe Units per 4 mg |
|---|---|---|---|---|---|
| 1 mL | 20 mg/mL | 0.20 mL | 0.40 mL | 0.60 mL | 20 units |
| 2 mL | 10 mg/mL | 0.40 mL | 0.80 mL | 1.20 mL* | 40 units |
| 4 mL | 5 mg/mL | 0.80 mL | 1.60 mL* | 2.40 mL* | 80 units |
*Exceeds a 1 mL (100-unit) insulin syringe. Use a higher concentration or split into two draws.*
Recommended for 20 mg vials: Add 1 mL for 20 mg/mL. This keeps draw volumes manageable even at the maximum 12 mg dose (0.60 mL = 60 units). At 8 mg per week, the vial provides 2.5 weeks of doses. At 12 mg per week, one vial lasts just under 2 weeks.
If you are unsure which vial size matches your protocol, check the retatrutide dosage guide for the full titration schedule and dose recommendations from the Phase 2 and Phase 3 trials.
Master Dosing Conversion Table
This single table covers every standard retatrutide dose at the four most common concentrations. All values are for a U-100 insulin syringe, where 100 units = 1 mL.
| Dose | 2.5 mg/mL | 5 mg/mL | 10 mg/mL | 20 mg/mL |
|---|---|---|---|---|
| 2 mg | 80 units (0.80 mL) | 40 units (0.40 mL) | 20 units (0.20 mL) | 10 units (0.10 mL) |
| 4 mg | 160 units* | 80 units (0.80 mL) | 40 units (0.40 mL) | 20 units (0.20 mL) |
| 8 mg | 320 units* | 160 units* | 80 units (0.80 mL) | 40 units (0.40 mL) |
| 12 mg | 480 units* | 240 units* | 120 units* | 60 units (0.60 mL) |
*Exceeds a 100-unit (1 mL) syringe. Use a higher concentration or split into two injections.*
The formula behind every number in this table: Units = (Dose in mg / Concentration in mg/mL) x 100. For example, 8 mg at 10 mg/mL: (8 / 10) x 100 = 80 units. At 20 mg/mL: (8 / 20) x 100 = 40 units.
Notice the pattern: the 12 mg maintenance dose only fits in a single syringe draw at 20 mg/mL (60 units). At 10 mg/mL, 12 mg requires 120 units, which exceeds a standard 1 mL insulin syringe. This is why 20 mg/mL is the preferred concentration for users on the maximum maintenance dose. For any combination, verify with the Peptide Unit Converter.
Understanding Concentration: Why the Water Volume Matters
Think of it like coffee. A shot of espresso and a large mug of drip coffee can both contain the same amount of caffeine. The espresso is more concentrated: more caffeine in less liquid. A 10 mg vial of retatrutide works the same way. Whether you add 0.5 mL or 2 mL of water, the vial still contains 10 mg of peptide. The concentration changes. The total drug does not.
More water means lower concentration and larger draw volumes per dose. Larger volumes are easier to read on a syringe. Less water means higher concentration and smaller draw volumes, which are harder to measure precisely but allow higher doses to fit in a single syringe pull.
For retatrutide specifically, concentration choice matters more than it does for tirzepatide or semaglutide. The reason: retatrutide doses reach 12 mg per week at maximum maintenance, compared to 15 mg for tirzepatide and 2.4 mg for semaglutide. At 12 mg, any concentration below 12 mg/mL requires more than 1 mL per injection. That forces you to either split the dose into two syringe pulls or use a non-insulin syringe, both of which increase measurement error.
The practical rule: if your maintenance dose will be 8 mg or 12 mg, start with 20 mg/mL from the beginning. This keeps all dose levels within the range of a single syringe draw. If you are in the early titration phase (2 mg or 4 mg), 5 mg/mL or 10 mg/mL both work well and produce easier-to-read volumes.
How to Draw Your Dose After Reconstitution
Reconstitution gets the solution into the vial. Drawing gets the right amount into your syringe. This second step is where most dosing errors happen.
Choosing the right insulin syringe:
| Syringe Size | Total Capacity | Best For | Smallest Graduation |
|---|---|---|---|
| 0.3 mL (30 units) | 30 units | Doses under 25 units | 0.5 units |
| 0.5 mL (50 units) | 50 units | Doses of 10 to 50 units | 1 unit |
| 1.0 mL (100 units) | 100 units | Doses of 25 to 100 units | 2 units |
Match the syringe to your draw volume. At 10 mg/mL, the 2 mg starting dose requires 20 units. A 0.3 mL or 0.5 mL syringe gives the clearest markings for that volume. At 20 mg/mL, the 12 mg maintenance dose requires 60 units. A 1.0 mL syringe handles that comfortably.
Drawing step by step:
- 1.Roll the reconstituted vial gently between your palms 5 to 10 times. This ensures uniform concentration throughout the solution.
- 2.Wipe the vial stopper with a fresh alcohol swab.
- 3.Pull air into the insulin syringe equal to your target draw volume. For 40 units, pull the plunger to 40 units of air.
- 4.Insert the needle through the stopper and push the air into the vial. This equalizes pressure and prevents vacuum buildup over multiple draws.
- 5.Invert the vial so the stopper faces down. Pull the plunger past your target by 5 to 10 units.
- 6.Tap the syringe barrel to move air bubbles toward the needle. Push the plunger slowly until it sits exactly at your target mark and the air is expelled.
- 7.Withdraw the needle from the vial.
This air-replacement technique prevents negative pressure from accumulating inside the vial. Without it, each successive draw becomes harder and pulls less liquid than intended. For complete injection technique, see the peptide injection guide.
Storage After Reconstitution
Proper storage determines whether your retatrutide remains potent for the full 28-day window or degrades within a week. For detailed storage protocols covering all peptide types, see how to store peptides.
Reconstituted solution (mixed with BAC water): - Refrigerate at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit) - Store the vial upright with the stopper facing up - Protect from direct light - Stable for up to 28 days - Do not freeze reconstituted retatrutide. Ice crystal formation denatures the peptide irreversibly
Unreconstituted powder (lyophilized): - Store in the freezer at minus 20 degrees Celsius for maximum shelf life (months to years) - Refrigerated storage at 2 to 8 degrees Celsius is acceptable for several months - Room temperature tolerance: 2 to 4 weeks without significant degradation - Keep the desiccant packet in the container to absorb moisture
Practical storage tips: - Store vials on a middle shelf of the refrigerator, not in the door. Door shelves experience temperature fluctuations of 5 to 10 degrees with each opening. - Use a dedicated container or small box to prevent the vial from tipping over. - If you travel with reconstituted retatrutide, use an insulated cooler bag with an ice pack. Keep the vial between 2 and 8 degrees Celsius. A car glove compartment in summer can exceed 60 degrees Celsius and will destroy the peptide in hours.
For shelf life data broken down by peptide type, see how long do reconstituted peptides last.
How Many Doses Per Vial?
This table shows how many weekly doses each vial size provides at standard retatrutide titration doses. Use it to plan how many vials you need per month and to ensure you use the reconstituted solution within the 28-day stability window.
| Vial Size | Weekly Dose | Doses per Vial | Weeks of Supply | Within 28-Day Window? |
|---|---|---|---|---|
| 5 mg | 2 mg | 2.5 | ~2.5 weeks | Yes |
| 10 mg | 2 mg | 5 | 5 weeks* | Borderline |
| 10 mg | 4 mg | 2.5 | 2.5 weeks | Yes |
| 10 mg | 8 mg | 1.25 | ~1 week | Yes |
| 20 mg | 4 mg | 5 | 5 weeks* | Borderline |
| 20 mg | 8 mg | 2.5 | 2.5 weeks | Yes |
| 20 mg | 12 mg | 1.67 | ~1.5 weeks | Yes |
*A 10 mg vial at 2 mg per week or a 20 mg vial at 4 mg per week stretches slightly beyond 28 days. Consider using a smaller vial or plan to discard any remaining solution after day 28.*
Retatrutide is administered as a once-weekly subcutaneous injection. The titration schedule starts at 2 mg per week for 4 weeks, escalates to 4 mg for weeks 5 through 8, then to 8 mg for weeks 9 through 12, and reaches the 12 mg maintenance dose at week 13 (Jastreboff et al., NEJM 2023). Your provider may adjust this schedule based on tolerability and response. For the full week-by-week protocol, see the retatrutide dosage guide.
Common Mistakes and How to Avoid Them
Five errors account for the vast majority of reconstitution problems with compounded retatrutide. Each is preventable.
Mistake 1: Spraying water directly onto the powder cake. The force of the stream can partially denature the peptide by disrupting hydrogen bonds in the folded structure. The solution may look normal but deliver reduced potency. Fix: aim the needle at the glass wall and let the water trickle down slowly over 30 to 60 seconds.
Mistake 2: Shaking the vial to dissolve the powder. Shaking creates foam, introduces air, and subjects the peptide to mechanical stress. Retatrutide, like tirzepatide, contains a fatty acid sidechain that makes it vulnerable to agitation-induced aggregation. Fix: swirl gently. If the powder is stubborn, refrigerate the vial for 10 to 15 minutes and swirl again.
Mistake 3: Using the wrong water volume for the vial size. Adding 1 mL to a 20 mg vial gives 20 mg/mL. Adding 1 mL to a 5 mg vial gives 5 mg/mL. Same volume of water, four-fold difference in concentration. If you follow instructions meant for a different vial size, every injection will be wrong. Fix: always confirm the vial label for the peptide mass before calculating your water volume.
Mistake 4: Forgetting to label the vial. Reconstituted retatrutide is a clear, colorless liquid. So is reconstituted tirzepatide, semaglutide, and bacteriostatic water itself. An unlabeled vial is an accident waiting to happen. Fix: write the peptide name, concentration, and reconstitution date on the vial immediately after mixing.
Mistake 5: Using sterile water for a multi-dose vial. Sterile water has no antimicrobial preservative. After the first needle puncture, bacteria can enter and multiply unchecked. By the third or fourth draw, the solution may contain a clinically relevant bacterial load. Fix: always use bacteriostatic water for any vial you plan to draw from more than once. If you already reconstituted with sterile water, use the vial within 24 hours and discard the remainder.
Danger Scenarios: What Goes Wrong with Bad Math
Reconstitution errors produce silent dosing errors. You inject what appears to be the correct number of units, but the concentration is wrong and the actual dose is higher or lower than intended. With retatrutide, which activates three hormone receptors and has a steep dose-response curve for gastrointestinal side effects, these errors carry real consequences.
Scenario 1: Half the water, double the dose. You have a 10 mg vial. You intend to add 2 mL for 5 mg/mL but accidentally add only 1 mL. The actual concentration is 10 mg/mL. You draw 40 units thinking you are getting 2 mg. You actually inject 4 mg. That is double the starting dose and equivalent to jumping straight to week 5 of the titration. In the Phase 2 trial, participants who started directly at 4 mg (without the 2 mg titration period) experienced nearly double the rate of gastrointestinal adverse events compared to those who started at 2 mg (Jastreboff et al., NEJM 2023). Expect nausea, vomiting, and possibly diarrhea lasting 48 to 72 hours.
Scenario 2: Wrong vial assumption. You have a 20 mg vial but follow instructions written for a 5 mg vial. You add 1 mL of water thinking you are making 5 mg/mL. The actual concentration is 20 mg/mL. When you draw 40 units for what you believe is 2 mg, you inject 8 mg. That is four times your intended starting dose and matches the second escalation level. At 8 mg without prior titration, the risk of severe gastrointestinal distress is high. The glucagon receptor activation at this dose level also increases resting heart rate by up to 6.7 beats per minute and may trigger dysesthesia (tingling or burning skin sensations) reported in 8.8% to 20.9% of participants at higher doses in Phase 3 trials.
Scenario 3: Contaminated solution from skipping sterile technique. You reconstitute with bacteriostatic water but skip the alcohol swab step. Over 3 weeks of use, bacteria introduced during the first needle puncture multiply to a level that causes injection-site infection. Symptoms include localized redness, swelling, warmth, and pain at the injection site. In severe cases, an abscess can form requiring incision and drainage.
The safeguard for all three: Always verify the vial label before adding water. Calculate or confirm using the Reconstitution Calculator. Write the concentration on the vial. Use clean technique every time.
Retatrutide Reconstitution vs Tirzepatide Reconstitution
If you have reconstituted tirzepatide before, the process for retatrutide is nearly identical. The mechanics (add water, aim at glass, swirl, refrigerate) do not change. The differences are practical, not procedural.
| Factor | Retatrutide | Tirzepatide |
|---|---|---|
| Mechanism | Triple agonist (GLP-1 + GIP + Glucagon) | Dual agonist (GLP-1 + GIP) |
| Common vial sizes | 5, 10, 20 mg | 5, 10, 15, 30 mg |
| Dose range (weekly) | 2 to 12 mg | 2.5 to 15 mg |
| Starting dose | 2 mg | 2.5 mg |
| Titration schedule | 4-week escalation steps | 4-week escalation steps |
| Solution appearance | Clear, colorless | Clear, colorless to slightly yellow |
| Shelf life (reconstituted, BAC water) | 28 days at 2-8°C | 28 days at 2-8°C |
| Shake tolerance | Low (swirl only) | Low (swirl only) |
The most important practical difference is dose magnitude relative to vial size. Retatrutide maxes out at 12 mg per week. Tirzepatide maxes out at 15 mg per week. Both require careful concentration planning to keep draw volumes within a single syringe pull.
The reconstitution math is the same for both: Concentration = mg / mL. The syringe reading technique is the same. The storage conditions are the same. If you can reconstitute one, you can reconstitute the other.
For a full comparison of these two peptides beyond reconstitution, including mechanism, weight loss data, and side effect profiles, see retatrutide vs tirzepatide. For information on how the triple mechanism produces additional weight loss, see how does retatrutide work.
Troubleshooting Common Problems
The powder will not dissolve. Set the vial upright in the refrigerator for 10 to 15 minutes. Then swirl again for 2 to 3 minutes. Lyophilized retatrutide occasionally takes longer to dissolve, especially in vials that were exposed to temperature fluctuations during shipping. If clumps remain after 30 minutes of gentle swirling and cold rest, the peptide may have degraded. Contact the supplier before using it.
Foam on the surface of the solution. A thin layer of small bubbles is normal and dissipates within 5 to 10 minutes of undisturbed rest. A thick, persistent foam indicates the vial was shaken or the water was injected too forcefully. Let the vial sit undisturbed in the refrigerator for 15 minutes. Draw from beneath the foam layer. The peptide in the foam is likely partially denatured and should not be injected.
The solution looks cloudy or has visible particles. Discard the vial. Cloudiness in a reconstituted peptide solution can indicate bacterial contamination, peptide aggregation from improper handling, or a degraded product. Do not attempt to filter and use a cloudy solution. A fresh vial reconstituted with proper technique will always be clear.
Rubber stopper fragments in the vial. This happens when the needle cores the stopper. Prevention: insert at a 45-degree angle with the bevel facing up before straightening to 90 degrees. If you spot fragments, draw the solution through a 0.22 micron syringe filter before each injection, or discard the vial and reconstitute a fresh one.
You accidentally added too much water. Adding extra water does not destroy the peptide. It lowers the concentration. Recalculate: new concentration equals vial mass divided by total water added. For a 10 mg vial with 3 mL of water, the concentration is 3.33 mg/mL instead of 10 mg/mL. Adjust your draw volume accordingly and write the corrected concentration on the vial. You cannot remove excess water once it has been added.
Equipment Guide: Choosing the Right Syringe
The syringe you use matters as much as the reconstitution itself. A syringe that is too large for your draw volume introduces measurement error. A syringe that is too small cannot hold the required volume.
Every insulin syringe is calibrated in "units." On a U-100 syringe, 100 units equals 1 mL. Each unit equals 0.01 mL. The markings printed on the barrel translate directly to volume. When your calculation says "draw 40 units," you are drawing 0.40 mL.
Syringe selection guide by dose and concentration:
| Your Dose | At 5 mg/mL | At 10 mg/mL | At 20 mg/mL | Recommended Syringe |
|---|---|---|---|---|
| 2 mg | 40 units | 20 units | 10 units | 0.5 mL (50-unit) |
| 4 mg | 80 units | 40 units | 20 units | 0.5 mL or 1.0 mL |
| 8 mg | 160 units* | 80 units | 40 units | 1.0 mL (100-unit) |
| 12 mg | 240 units* | 120 units* | 60 units | 1.0 mL (100-unit) |
*Exceeds syringe capacity. Use a higher concentration or split into two injections.*
At 20 mg/mL, even the maximum 12 mg dose fits within a 1 mL syringe (60 units). This is why 20 mg/mL is the practical choice for anyone on a higher maintenance dose. At 10 mg/mL, doses of 8 mg (80 units) still fit, but 12 mg (120 units) does not.
If your calculated draw volume falls below 10 units on any syringe, switch to a 0.3 mL (30-unit) syringe with half-unit markings. At volumes this small, each unit of error represents a proportionally large dosing error. The Peptide Unit Converter shows exact unit counts for any concentration and dose combination.
Tips for First-Time Reconstitution
If this is your first time reconstituting any peptide, three habits will prevent virtually every common error.
Habit 1: Read the vial label twice. Before drawing bacteriostatic water, confirm the peptide name and the mass printed on the label. A 5 mg vial and a 20 mg vial of retatrutide can look identical from the outside. The only difference is the number on the label. Reading it twice takes 5 seconds and eliminates the most dangerous reconstitution mistake.
Habit 2: Write on the vial before you put it in the refrigerator. The moment you finish swirling, pick up the marker and write the concentration and date. Do not tell yourself you will remember. In a refrigerator with multiple vials, an unlabeled clear solution is indistinguishable from every other unlabeled clear solution.
Habit 3: Use a calculator for every new vial. Even if you have reconstituted the same vial size 10 times, confirm the math. Complacency is the root of dosing errors. The Reconstitution Calculator takes less time than second-guessing yourself.
For a general reconstitution guide that covers all peptide types, see how to reconstitute peptides. For tirzepatide specifically (same process, different dose range), see how to reconstitute tirzepatide. For GHK-Cu (which has a unique solvent restriction), see how to reconstitute GHK-Cu. The same fundamental technique applies to every lyophilized peptide: clean, draw, aim at the wall, swirl, label, refrigerate.
For comprehensive peptide stacking protocols that include retatrutide alongside other compounds, see the peptide stacking guide. For safe injection practices including site rotation and angle, see the peptide safety guide.
Frequently Asked Questions
How much bacteriostatic water do I add to a 10 mg retatrutide vial?
Add 1 mL for a 10 mg/mL concentration, which is the most practical standard for the starting and mid-range doses. At 10 mg/mL, the 2 mg starting dose equals 20 units and the 4 mg escalation dose equals 40 units on a U-100 insulin syringe. For higher maintenance doses (8 mg or 12 mg), consider adding only 0.5 mL for 20 mg/mL to keep draw volumes within a single syringe pull.
How long does reconstituted retatrutide last in the fridge?
Reconstituted retatrutide mixed with bacteriostatic water remains stable for 28 days when stored at 2 to 8 degrees Celsius. Do not freeze it after reconstitution. If the solution becomes cloudy or develops particles before 28 days, discard it. Unreconstituted lyophilized powder lasts months to years when stored at minus 20 degrees Celsius.
Can I use sterile water instead of bacteriostatic water for retatrutide?
You can, but the vial must be used within 24 hours because sterile water contains no antimicrobial preservative. Bacteriostatic water (preserved with 0.9% benzyl alcohol) allows multi-dose use over 28 days. Since retatrutide is dosed once weekly and most vials contain multiple doses, bacteriostatic water is the correct choice for nearly all situations.
How many units is 2 mg of retatrutide at 10 mg/mL?
At 10 mg/mL, 2 mg of retatrutide equals 20 units on a U-100 insulin syringe. The formula is (2 mg / 10 mg per mL) times 100 = 20 units. At 5 mg/mL, the same dose is 40 units. At 20 mg/mL, it is 10 units. Always confirm the concentration written on your vial label before drawing.
What happens if I add too much water to my retatrutide vial?
Adding extra water does not destroy the peptide. It lowers the concentration, which means you need to draw a larger volume per dose. Recalculate using the formula: new concentration equals vial mass divided by total water added. For a 10 mg vial with 3 mL of water, the concentration is 3.33 mg/mL. Adjust your draw volume and write the corrected concentration on the vial.
Do I need to shake the vial to dissolve retatrutide?
Never shake a retatrutide vial. Shaking causes foaming and can denature the peptide through mechanical stress. Retatrutide contains a fatty acid sidechain similar to tirzepatide, making it sensitive to agitation. Swirl the vial gently in a circular motion for 1 to 3 minutes. If powder remains, refrigerate for 10 to 15 minutes and swirl again.
What is the best concentration for compounded retatrutide?
For the starting dose phase (2 mg per week), 5 mg/mL or 10 mg/mL both work well. For the maintenance dose of 8 mg or 12 mg per week, 20 mg/mL is the most practical concentration. At 20 mg/mL, the maximum 12 mg dose requires only 60 units on a U-100 syringe, fitting comfortably within a single draw.
Is reconstituting retatrutide different from reconstituting tirzepatide?
The process is identical: add bacteriostatic water, aim at the glass wall, swirl gently, label, refrigerate. The only practical difference is dose range. Retatrutide doses go from 2 mg to 12 mg weekly, while tirzepatide ranges from 2.5 mg to 15 mg. Both are 39-amino-acid peptides with fatty acid sidechains. Same technique, same storage conditions, same 28-day stability window.
The Bottom Line
Reconstituting retatrutide follows the same five steps as any lyophilized peptide: clean the stoppers, draw bacteriostatic water, inject slowly against the glass wall, swirl gently, and refrigerate. The entire process takes under five minutes.
The critical variable is getting the concentration right. Confirm the vial size on the label, choose your water volume from the tables above, and write the resulting concentration on the vial immediately after mixing. For the maximum 12 mg maintenance dose, use 20 mg/mL to keep the draw volume within a single syringe pull (60 units). When in doubt, verify with the Reconstitution Calculator.
For the full titration schedule and week-by-week dosing protocol, see the retatrutide dosage guide. For unit conversions at your specific concentration, use the Retatrutide Dosage Calculator. For the pharmacology and mechanism of action, visit how does retatrutide work. For the full drug overview including availability timeline, see the complete retatrutide guide.
Related articles: - Retatrutide Side Effects: Phase 3 safety data including dysesthesia and GI rates by dose - Retatrutide vs Tirzepatide: full comparison of triple vs dual agonist - How to Get Retatrutide: availability, sourcing, and legal status in 2026 - How to Reconstitute Tirzepatide: same process for the dual agonist - How to Store Peptides: storage conditions for all peptide types - How Long Do Reconstituted Peptides Last?: shelf life data by peptide
More from this series: - Is Reconstitution Solution the Same as Bacteriostatic Water? — comparing diluent types for peptide mixing - Where to Buy Bacteriostatic Water for Injection — sourcing guide with price comparison and quality checks - Does Bacteriostatic Water Need to Be Refrigerated? — storage rules for opened and unopened vials - How Long Does Tirzepatide Last in the Fridge? — refrigeration timelines for reconstituted and lyophilized forms - How Many mg Is 20 Units of Tirzepatide? — unit-to-mg conversion for compounded tirzepatide
Related Articles
How to Reconstitute Tirzepatide
How to reconstitute tirzepatide step by step for 5mg, 10mg, 15mg, and 30mg vials. Concentration tables, BAC water volumes, syringe units, and storage guide.
How to Take Retatrutide (2026)
How to take retatrutide: once-weekly subcutaneous injection, 2mg to 12mg titration over 12 weeks. Injection sites, meal timing, and storage.
How to Reconstitute GHK-Cu: Guide
How to reconstitute GHK-Cu step by step for 50mg and 100mg vials. Concentration table, bacteriostatic water volumes, syringe units, and storage instructions.
How Long Does Retatrutide Take to Work?
Retatrutide suppresses appetite within days, produces weight loss by week 4, and delivers up to 24.2% loss at 48 weeks in trials.