
You open the refrigerator and spot a vial of tirzepatide you bought three weeks ago. The cap is still sealed, no water added. Is it still good? Unreconstituted tirzepatide lasts in the fridge until its printed expiration date, typically 18 to 24 months from manufacture, when stored at 2-8°C. Reconstituted tirzepatide (mixed with bacteriostatic water) lasts 28 days refrigerated. Pre-filled Mounjaro and Zepbound pens follow the manufacturer's printed date while unopened and refrigerated.
These timelines shift based on form, solvent, and handling. A single overnight lapse at room temperature does not ruin a vial, but repeated temperature excursions compound into measurable potency loss.
| Tirzepatide Form | Storage Temp | Fridge Duration | Key Notes |
|---|---|---|---|
| Lyophilized powder (sealed) | 2-8°C | 18-24 months | Keep in original packaging, away from light |
| Lyophilized powder (sealed) | -20°C (freezer) | 24-36 months | Maximum stability option |
| Reconstituted (bacteriostatic water) | 2-8°C | 28 days | Mark the reconstitution date on the vial |
| Reconstituted (sterile water) | 2-8°C | 24-48 hours | No preservative; draw all doses immediately |
| Mounjaro/Zepbound pen (unopened) | 2-8°C | Until printed expiry (18-24 months) | Store in original carton |
| Mounjaro/Zepbound pen (opened/used) | Room temp (up to 30°C) | 21 days | Do not return to fridge and restart the clock |
| Compounded vial (lyophilized, sealed) | 2-8°C | Per BUD label (typically 6 months) | Based on USP <797>, not product-specific testing |
For reconstitution volumes and technique, use the peptide reconstitution calculator. For dosing protocols, see the tirzepatide dosage chart in units.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Unreconstituted Tirzepatide: Fridge Shelf Life Explained
Unreconstituted tirzepatide arrives as a lyophilized (freeze-dried) powder or as a liquid in a pre-filled pen. In both cases, refrigeration at 2-8°C preserves the peptide's 39-amino-acid structure by slowing every chemical degradation pathway to near-zero velocity.
Think of it like frozen bread. A loaf left on the counter goes stale in two days. The same loaf in the freezer lasts months. The bread has not changed; the environment has. Tirzepatide in powder form behaves the same way: remove moisture and lower the temperature, and the molecule sits inert. The literal science: lyophilization halts deamidation and oxidation, the two primary degradation reactions, by eliminating the water they require (Manning et al., Pharm Res, 2010).
Lyophilized Powder at 2-8°C
Sealed lyophilized tirzepatide stored at standard refrigerator temperature retains greater than 95% potency for 18 to 24 months. The exact duration depends on the manufacturer's stability data and the excipients used in the formulation. Most compounding pharmacies assign a beyond-use date (BUD) of 6 months because USP <797> requires product-specific stability data to claim longer, and many pharmacies have not conducted those tests.
The practical implication: a compounded vial labeled with a 6-month BUD may retain potency longer than 6 months. But without stability data to confirm that, the labeled date is the only reliable guideline. Respect it.
Store the vial in the back of the refrigerator, never in the door. Door shelves experience temperature swings of 5-8°C with each opening. A vial stored in the door for three months accumulates more thermal stress than one stored on a back shelf for six months. For comprehensive storage principles, see the peptide storage guide.
Pre-Filled Pens (Mounjaro and Zepbound)
Eli Lilly's pre-filled pens contain tirzepatide in a proprietary liquid formulation engineered for stability. Unopened pens stored at 2-8°C remain potent until the printed expiration date, typically 18 to 24 months from production. The buffer system, excipients, and controlled manufacturing environment give these pens a longer verified shelf life than most compounded preparations.
Once you remove a pen from the refrigerator or use it for the first time, Eli Lilly specifies a 21-day window at room temperature (up to 30°C / 86°F). After 21 days at room temperature, discard the pen. Do not return it to the fridge and assume the clock resets. It does not. The 21-day limit accounts for cumulative degradation at higher temperatures.
Never freeze a pre-filled pen. The freeze-thaw cycle damages the protein structure and can alter the pen's delivery mechanism. If a pen has been frozen, discard it.
Freezer Storage for Maximum Longevity
If you plan to store lyophilized tirzepatide for longer than six months before use, a standard kitchen freezer at -20°C extends shelf life to 24-36 months. At this temperature, degradation reactions slow to the point where potency loss is negligible over two years.
Freezer storage is appropriate only for lyophilized (powder) tirzepatide. Never freeze reconstituted tirzepatide. Ice crystals physically shear peptide bonds and cause irreversible aggregation, destroying 30-50% of the active molecule in a single freeze-thaw cycle. This distinction matters: powder in the freezer is fine; liquid in the freezer is destroyed.
Wrap vials in aluminum foil or store them in the original packaging to block light. Even in a dark freezer, brief light exposure during door openings compounds over months.
Reconstituted Tirzepatide: The 28-Day Refrigerated Window
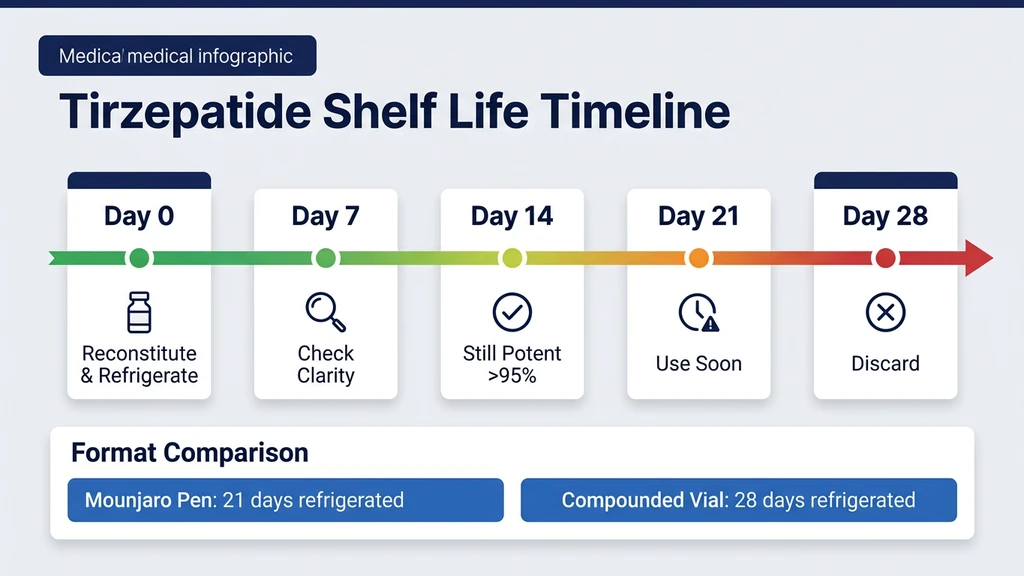
The moment you add bacteriostatic water to lyophilized tirzepatide, the shelf life drops from months to days. Every degradation pathway activates in aqueous solution. Deamidation of asparagine residues, oxidation of methionine, and microbial contamination risk all begin simultaneously.
With bacteriostatic water (containing 0.9% benzyl alcohol as a preservative), reconstituted tirzepatide lasts 28 days at 2-8°C. With plain sterile water (no preservative), the window collapses to 24-48 hours because nothing inhibits bacterial growth. Bacteriostatic water is the standard choice for multi-dose vials.
Why Exactly 28 Days?
The 28-day limit is not arbitrary. It reflects the point at which the peptide is expected to retain at least 90% of its labeled potency under refrigeration, the minimum threshold the pharmaceutical industry considers acceptable. Below 90%, dose-response relationships become unreliable.
At 2-8°C, deamidation proceeds slowly enough to keep potency above that line for four weeks. At room temperature (25°C), the same 10% loss occurs in roughly 7 to 10 days. The Arrhenius equation predicts that reaction rates approximately double for every 10°C increase in temperature. This is why refrigeration buys four times the shelf life of a countertop.
The SURMOUNT-1 trial demonstrated that each tirzepatide dose tier (5 mg, 10 mg, 15 mg) produced measurably different weight loss outcomes (Jastreboff et al., NEJM, 2022). A 10-15% potency loss effectively drops your dose to a lower tier without your knowledge. For dosing details, see how many units is 2.5 mg of tirzepatide.
Potency Decline Timeline After Reconstitution
Reconstituted tirzepatide does not degrade linearly. Potency drops gradually at first, then accelerates as degradation products catalyze further breakdown.
| Days After Reconstitution | Estimated Potency at 2-8°C | Estimated Potency at 25°C |
|---|---|---|
| Day 0 | 100% | 100% |
| Day 7 | 97-99% | 90-95% |
| Day 14 | 95-98% | 80-88% |
| Day 21 | 92-96% | 70-80% |
| Day 28 | 90-95% | 60-72% |
| Day 42 | 82-90% | 45-55% |
By day 28 at 2-8°C, potency remains within the pharmaceutical standard. By day 42, it has dropped below 90% for many vials. By day 28 at room temperature, more than a third of the active peptide may be gone. This is why reconstituted vials belong in the fridge at all times except the 30-60 seconds you need to draw a dose.
For broader context on reconstituted peptide timelines, see how long do reconstituted peptides last.
Bacteriostatic Water vs. Sterile Water
The solvent you choose determines whether your vial lasts 28 days or 48 hours. Bacteriostatic water contains 0.9% benzyl alcohol, a preservative that suppresses bacterial growth. Sterile water for injection contains no preservative.
Use bacteriostatic water for any vial you plan to draw from more than once. Use sterile water only if you intend to draw the entire contents in a single session and discard the vial immediately.
Each needle puncture through the rubber stopper introduces a contamination pathway. After 15 to 20 punctures, the stopper may no longer seal properly. Keep puncture counts low by drawing accurate volumes each time. Use the tirzepatide dosage calculator to determine the exact volume before you uncap a needle. For mixing ratios, see how much bacteriostatic water for semaglutide (the principles apply across all GLP-1 agonists).
Mounjaro Pen vs. Compounded Vial: Fridge Life Differences
The container changes the rules. A Mounjaro pen and a compounded vial both contain tirzepatide, but their refrigeration timelines differ because of formulation, quality control, and regulatory standards.
Mounjaro and Zepbound Pens
Eli Lilly conducts 12 to 24 months of accelerated stability testing under International Council for Harmonisation (ICH) Q1A guidelines before assigning expiration dates. The pens benefit from a proprietary buffer system, precise pH control, and excipients selected for long-term stability (ICH Q1A(R2), 2003).
The result: unopened pens last until the printed date (18-24 months from manufacture) in the fridge. After first use or removal from refrigeration, 21 days at room temperature. This window is clinically validated through stability testing. Pens should not be stored above 30°C or exposed to direct sunlight.
If you travel frequently, the 21-day room-temperature tolerance makes pens more practical than reconstituted vials. A two-week vacation does not require a cold pack if the pen stays below 30°C.
Compounded Vials
Compounding pharmacies operate under USP <797> and USP <795> standards, which are less rigorous than ICH guidelines. Many pharmacies assign a beyond-use date of 6 months for lyophilized preparations and 28 to 45 days for reconstituted solutions. These dates are conservative estimates, not the result of product-specific stability studies.
A compounded vial labeled with a 6-month BUD stored at 2-8°C may retain potency for 12 months or more. But without testing data, that remains speculation. The quality of excipients, the purity of the starting material, and the sterility of the compounding environment all introduce variables that Eli Lilly's manufacturing process controls more tightly.
The cost calculation matters here. A compounded vial that expires before you finish it wastes both product and money. Plan your tirzepatide dosing schedule against the labeled BUD. If the math does not work, order smaller vials or split orders with your prescriber's guidance.
What Happens If Tirzepatide Is Left Out of the Fridge
You forgot to put the vial back after your morning injection. Eight hours later, you find it on the bathroom counter. Is it ruined?
Probably not. But the damage depends on temperature, duration, and whether the vial was reconstituted or sealed.
Left Out Overnight (8-12 Hours)
A reconstituted vial left at room temperature (20-25°C) overnight loses approximately 1-3% potency. That loss is measurable in a laboratory but unlikely to produce a noticeable change in clinical effect. Return the vial to the fridge and continue using it, but note the lapse. Multiple overnight lapses compound.
A sealed lyophilized vial left out overnight experiences negligible degradation. Powder tolerates room temperature for months. A single night is inconsequential.
A Mounjaro pen left out overnight begins its 21-day room-temperature clock if it was previously refrigerated. Eli Lilly's guidance does not distinguish between "accidentally left out for 8 hours" and "intentionally stored at room temperature." Once the pen warms, the countdown starts.
Left Out for 24-48 Hours
At this point, a reconstituted vial has lost 3-7% potency at 20-25°C. If the room was warmer (near 30°C in summer), losses can reach 8-10%. The vial is still usable if it passes visual inspection (clear, colorless, no particles), but you have effectively shortened its 28-day window by several days.
Pre-filled pens are within their 21-day tolerance. No action needed beyond noting the start of the countdown. Lyophilized powder remains unaffected.
Left Out for Several Days or Longer
A reconstituted vial left at room temperature for 5 or more days has likely lost 10-20% potency. By day 7 at 25°C, potency drops to 90-95%, approaching the pharmaceutical threshold. If the room was warm and the vial was exposed to light, discard it.
A reconstituted vial left in a hot car (40°C+) for even a few hours should be discarded. At 40°C, degradation accelerates roughly 8-fold compared to 2-8°C. A GLP-1 receptor agonist stability study found that oxidation of a single methionine residue at elevated temperatures reduced receptor binding affinity by 40-60% (Houen & Jakobsen, J Pharm Sci, 2019).
For signs that your vial has crossed the line, see our guide on whether tirzepatide expires.
Signs Your Refrigerated Tirzepatide Has Gone Bad
Cold storage slows degradation. It does not prevent it forever. Even a properly refrigerated vial will eventually degrade. Here is how to identify a vial that has passed its useful life.
Visual Indicators
Cloudiness. Fresh tirzepatide solution is clear and colorless. Any haziness or opalescence indicates protein aggregation. Aggregated peptides are less potent and potentially immunogenic. The immune system can recognize peptide aggregates as foreign and mount an antibody response. Discard cloudy solutions.
Particles. Hold the vial against a light source and rotate it slowly. Floating specks, flakes, or fibers mean contamination or advanced aggregation. A single small fiber may be a stopper fragment. Multiple particles mean the vial is done.
Color shift. The solution should be colorless to very faintly yellow. Any amber, brown, or unusual tint indicates oxidation. Discard immediately.
Gel or film. In advanced degradation, peptide aggregates form a gel-like coating on the vial walls or a film at the solution surface. This represents extensive structural damage beyond recovery.
Functional Warning Signs
The most telling sign of degraded tirzepatide is reduced efficacy. If your appetite suppression, weight loss, or glucose control has diminished on a previously stable dose without any change in diet, exercise, or medication, suspect the vial.
A 15-20% potency loss turns a 10 mg dose into an effective 8 to 8.5 mg dose. The SURMOUNT trials showed meaningful differences between dose tiers. Dropping from your intended dose to a lower one because of degradation will show in your results within 1-2 weeks.
Increased injection site discomfort can also signal degradation. Breakdown products lower the solution's pH, increasing tissue irritation. If a previously comfortable injection starts burning or stinging, that vial warrants scrutiny.
For a full breakdown of efficacy concerns, see does tirzepatide burn fat. If you experience side effects, consult does tirzepatide cause headaches.
Travel and Temperature Excursion Guidelines
Maintaining the cold chain during travel is the most common challenge tirzepatide users face. The approach differs based on whether you are carrying a pre-filled pen or a reconstituted vial.
Pre-Filled Pens During Travel
Mounjaro and Zepbound pens tolerate 21 days at room temperature, making them the most travel-friendly option. For trips under three weeks in moderate climates, no cold storage is required. Keep the pen in your carry-on bag, away from direct sunlight, and below 30°C.
For trips in hot climates or exceeding 21 days, pack the pen in an insulated pouch with a cold gel pack. Avoid direct contact between the ice pack and the pen. A sock or small towel between them prevents accidental freezing. Frio wallets (evaporative cooling pouches) maintain 18-26°C for 24-48 hours without ice and are widely used by insulin-dependent travelers.
For air travel, carry tirzepatide in your cabin bag. Checked luggage endures temperature extremes in cargo holds that can reach -30°C or 50°C depending on the aircraft and season. TSA permits injectable medications with proper documentation. A letter from your prescriber or a pharmacy label is sufficient.
Reconstituted Vials During Travel
Reconstituted vials require unbroken refrigeration. Pack them in a medical-grade insulated cooler bag with gel packs pre-chilled to 2-8°C. Do not use loose ice cubes; they melt into water that can soak labels and compromise stopper seals.
Most hotel rooms have minibars or small refrigerators. Request one at booking. If unavailable, ask the hotel concierge to store your medication in the kitchen refrigerator. Medical storage requests are routine in the hospitality industry.
For road trips, a powered cooler (thermoelectric or compressor) that plugs into your car's 12V outlet maintains a consistent 2-8°C indefinitely. Standard coolers with ice packs hold temperature for 12-18 hours depending on ambient heat.
For more on storage fundamentals, see how to store peptides. For reconstitution instructions that start the 28-day clock, see how to reconstitute tirzepatide.
Temperature Excursion Tolerance
Pharmaceutical stability testing builds in a margin of safety. A single brief temperature excursion (2-4 hours above 8°C) does not typically push the peptide below the 90% potency threshold. The concern is cumulative exposure.
A vial that spends 30 minutes at room temperature during each of 28 daily dose draws accumulates 14 hours of warm exposure over its life. That is equivalent to roughly one full day at room temperature, enough to shave 1-3% off total potency. Minimize handling time. Draw the dose, return the vial, close the door. Sixty seconds or less.
If your home refrigerator runs warmer than expected (above 8°C), every vial in it degrades faster than the labeled timeline assumes. A $5 refrigerator thermometer eliminates this risk. Consumer refrigerators frequently run 2-3°C warmer than their dial setting, especially older models. Use the reconstitution calculator to plan dose volumes that minimize the number of draws per vial.
Common Mistakes That Shorten Fridge Life
Most premature tirzepatide degradation is avoidable. These are the errors that cost people days or weeks of shelf life from otherwise good vials.
Storing in the refrigerator door. The door is the warmest zone. Each opening sends a burst of room-temperature air across everything on the shelf. Temperature data loggers placed in refrigerator doors show swings of 5-8°C per opening cycle. Vials belong on a back shelf or in a dedicated drawer.
Forgetting to label the reconstitution date. Without a date written on the vial, you are guessing at the 28-day window. Write the date with a permanent marker the moment you add water. A vial without a date is a vial you will use too long.
Shaking the vial. Agitation creates air-liquid interfaces that promote protein aggregation. Aggregation reduces potency and can trigger immune responses. Swirl gently to mix. Never shake.
Reconstituting with sterile water and storing as if it were bacteriostatic. Sterile water contains no preservative. A vial mixed with sterile water that sits in the fridge for a week may harbor dangerous levels of bacteria even if the solution looks clear. Always use bacteriostatic water for multi-dose vials.
Taking the vial out to "warm it up" before injecting. Some users leave the vial at room temperature for 15-30 minutes to reduce injection discomfort. This practice is unnecessary. Cold injections cause brief stinging; warm degradation is permanent. If cold injections bother you, roll the syringe (not the vial) between your palms for 10 seconds after drawing. The small volume warms quickly.
Using the same needle for multiple draws. Each puncture widens the stopper hole slightly. After multiple reuses, the stopper leaks, admitting air and bacteria. Use a fresh needle every time.
Freezing Tirzepatide: When It Helps and When It Destroys
Freezing is the best preservation method for one form of tirzepatide and the worst for the other. Getting this distinction wrong is irreversible.
Lyophilized Powder: Freeze It
Moving lyophilized tirzepatide from the refrigerator (2-8°C) to the freezer (-20°C) extends shelf life from 18-24 months to 24-36 months. The powder contains virtually no moisture, so ice crystal formation is not a concern. A study on peptide pharmaceutical stability found that lyophilized formulations stored at -20°C retained greater than 95% potency for three years under ideal conditions (Manning et al., Pharm Res, 2010).
Standard kitchen freezers maintain approximately -18°C to -20°C, which is adequate. Deep freezers at -40°C offer marginal additional benefit. The meaningful jump is from 2-8°C to -20°C, not from -20°C to -40°C.
Reconstituted Solution: Never Freeze It
Freezing reconstituted tirzepatide creates ice crystals that physically tear peptide bonds. The freeze-thaw cycle causes irreversible aggregation, destroying 30-50% of the active peptide in a single event. A vial that was accidentally frozen should be discarded even if the solution looks clear after thawing. Aggregation damage is not always visible immediately; it manifests as reduced receptor binding and diminished clinical effect.
This rule applies to all reconstituted peptides. For a broader overview of reconstituted peptide handling, see how long do reconstituted peptides last.
Pre-Filled Pens: Never Freeze Them
Mounjaro and Zepbound pens contain liquid tirzepatide. Freezing follows the same physics as reconstituted vials: ice crystals damage the peptide structure. Additionally, freezing can damage the pen's spring mechanism and needle assembly, making it mechanically unreliable.
If a pen was left in a checked bag that reached sub-zero temperatures in a cargo hold, or stored near the back of a freezer by accident, discard it. Do not attempt to use it even if the solution appears normal.
Important Safety Warnings
Never use reconstituted tirzepatide beyond 28 days regardless of appearance. Microbial contamination is not always visible. A clear solution can harbor dangerous bacterial loads.
Do not use any tirzepatide that shows cloudiness, visible particles, color change, or unusual odor. These signs indicate structural degradation and potential immunogenicity. Injecting aggregated peptides can trigger antibody formation against tirzepatide, potentially reducing the efficacy of future doses. Clinical trials report anti-tirzepatide antibodies in approximately 2.1% of subjects receiving standard doses. Degraded formulations with higher aggregate content could increase that rate.
If you suspect your tirzepatide has lost potency (diminished appetite suppression, reduced weight loss, worsening glucose control on a stable dose), contact your prescriber. Do not compensate by increasing the dose on your own. Unpredictable potency in a degraded vial means one draw could deliver 70% of intended dose while the next delivers 110%. For diabetic patients, this inconsistency creates dangerous blood sugar swings.
Do not share vials between individuals. Multi-dose vials from compounding pharmacies are for single-patient use. Sharing introduces cross-contamination risk and is both medically and legally inappropriate.
For a complete overview of safety considerations, see the peptide safety guide. For information on tirzepatide side effects, see does tirzepatide cause headaches and does tirzepatide cause diarrhea.
Frequently Asked Questions
How long does tirzepatide last in the fridge after reconstitution?
Reconstituted tirzepatide lasts 28 days refrigerated at 2-8°C when mixed with bacteriostatic water (0.9% benzyl alcohol). With plain sterile water, use within 24-48 hours. After 28 days, potency drops below 90%, the pharmaceutical acceptability threshold. Write the mixing date on the vial with a permanent marker to track this window accurately.
Can tirzepatide go bad in the fridge?
Yes. Reconstituted tirzepatide degrades even under refrigeration, reaching unacceptable potency levels after approximately 28 days. Sealed lyophilized powder lasts 18-24 months in the fridge. Signs of a degraded vial include cloudiness, visible particles, color shift from clear to amber, or diminished clinical effect on a previously stable dose.
What happens if tirzepatide is left out of the fridge overnight?
A reconstituted vial left at room temperature (20-25°C) for 8-12 hours loses approximately 1-3% potency. Return it to the fridge and continue using it, but note the lapse. An unopened lyophilized vial is unaffected. A Mounjaro pen begins its 21-day room-temperature clock once it leaves refrigeration, regardless of whether the removal was intentional.
Can you freeze tirzepatide to make it last longer?
Freeze lyophilized (powder) tirzepatide only. Storage at -20°C extends shelf life from 18-24 months to 24-36 months. Never freeze reconstituted tirzepatide or pre-filled pens. Ice crystals physically shear peptide bonds, destroying 30-50% of the active molecule in one freeze-thaw cycle. The damage is irreversible.
How long does a Mounjaro pen last in the fridge?
An unopened Mounjaro pen lasts until its printed expiration date (18-24 months from manufacture) when stored at 2-8°C in the original carton. After first use or removal from the fridge, the pen is stable for 21 days at room temperature (up to 30°C). Do not store above 30°C or expose to direct sunlight.
How should I store tirzepatide when traveling?
Pre-filled pens tolerate 21 days at room temperature below 30°C, requiring no cold storage for short trips. Reconstituted vials need insulated cooler bags with gel packs at 2-8°C. Use Frio evaporative cooling pouches for 24-48 hours without ice. Carry tirzepatide in cabin luggage; cargo holds reach extreme temperatures. TSA allows injectables with a prescriber letter or pharmacy label.
Is tirzepatide still good after 30 days in the fridge?
Reconstituted tirzepatide at 30 days has likely dropped below 90% potency, the pharmaceutical standard. Some vials retain 88-92% at day 30, but without lab testing you cannot confirm yours is above the threshold. At day 28, discard the vial and reconstitute a fresh one. The cost of a new vial is lower than the cost of subtherapeutic dosing.
Does the type of water used to mix tirzepatide affect how long it lasts?
Yes. Bacteriostatic water (containing 0.9% benzyl alcohol preservative) allows 28 days of refrigerated storage. Sterile water for injection contains no preservative, limiting safe use to 24-48 hours. Always use bacteriostatic water for multi-dose vials. Draw all doses immediately if using sterile water, and discard the vial within 48 hours.
The Bottom Line
Unreconstituted tirzepatide lasts in the fridge until its printed expiration date, typically 18-24 months at 2-8°C. Reconstituted tirzepatide lasts 28 days refrigerated with bacteriostatic water. Pre-filled Mounjaro and Zepbound pens follow the manufacturer's date when unopened and cold, then 21 days at room temperature after first use.
The difference between a vial that reaches day 28 at full potency and one that degrades early comes down to handling: store in the back of the fridge (not the door), label the reconstitution date, minimize time at room temperature during draws, and use bacteriostatic water for multi-dose vials. These are small habits with measurable results.
Use the tirzepatide dosage calculator to plan exact doses and the peptide reconstitution calculator to mix accurately. For the complete picture of tirzepatide degradation chemistry, read does tirzepatide expire.
Related articles: - How to Reconstitute Tirzepatide — step-by-step guide that starts the 28-day clock - How to Store Peptides — comprehensive storage principles for all peptides - How Long Do Reconstituted Peptides Last? — shelf life data across compounds - Does Tirzepatide Expire? — degradation chemistry and potency loss curves - Tirzepatide Dosage Chart in Units — dosing protocols to plan against your vial's shelf life - Does Tirzepatide Burn Fat? — what proper storage preserves in clinical effect - Does Tirzepatide Cause Headaches? — side effects to monitor during treatment - Can You Drink Alcohol on Tirzepatide? — safety considerations during treatment
More from this series: - Does Bacteriostatic Water Need to Be Refrigerated? — storage rules for opened and unopened bacteriostatic water vials - Where to Buy Bacteriostatic Water for Injection — sourcing guide with price comparison and red flags - How to Reconstitute Retatrutide — step-by-step mixing guide for the triple agonist - Does Tirzepatide Make You Tired? — fatigue data and 8 management strategies - How Long Does Tirzepatide Stay in Your System? — half-life, clearance timeline, and what to expect after stopping - How Many mg Is 20 Units of Tirzepatide? — unit-to-mg conversion for compounded tirzepatide
Related Articles
Does Tirzepatide Expire? Shelf Life
Tirzepatide expires in 24-36 months as powder and 28 days once reconstituted. Learn storage temps, degradation signs, and potency loss timelines.
Semaglutide Refrigeration Guide
Yes, semaglutide must be refrigerated at 2-8°C before first use. Ozempic pens last 56 days at room temp after opening. Compounded vials: 28 days.
Does Tirzepatide Help With Inflammation? Clinical Evidence
Tirzepatide reduces CRP by 32.9% and IL-6 by 17.8% in clinical trials. Review the SURMOUNT data, adipose tissue mechanisms, and practical implications for inflammation.
Does Compounded Tirzepatide Work? Efficacy Evidence, Salt Forms, and What the Trials Actually Show
Does compounded tirzepatide work? Review SURMOUNT trial data, salt form differences (base vs acetate vs sodium), potency testing gaps, and FDA regulatory status in 2026.