You filled the prescription from a compounding pharmacy two weeks ago. The scale has not moved. You are wondering whether the vial in your refrigerator contains what the label claims. Yes, compounded tirzepatide can work when the pharmacy uses the correct base form of the molecule at verified potency, but the clinical evidence behind that "can" rests entirely on brand-name trials, not on compounded product testing. Every gram of efficacy data comes from Eli Lilly's SURMOUNT program using FDA-approved Mounjaro and Zepbound. No compounding pharmacy has published a single controlled trial on its own product.

That distinction matters more than most patients realize. The active molecule is the same 39-amino-acid dual GIP/GLP-1 receptor agonist regardless of source. But "same molecule" does not mean "same product." Potency can vary, salt forms may differ, sterility standards are inconsistent, and the regulatory framework governing compounded versions changed dramatically in late 2024 when the FDA resolved the tirzepatide shortage (FDA Drug Shortage Resolution, 2024).
| Quick Reference | Details |
|---|---|
| Active molecule | Tirzepatide (dual GIP/GLP-1 receptor agonist, 39 amino acids) |
| Brand names | Mounjaro (diabetes) / Zepbound (obesity) |
| SURMOUNT-1 weight loss | 16.0% (5 mg), 21.4% (10 mg), 22.5% (15 mg) at 72 weeks |
| Compounded versions tested in trials? | No. Zero published RCTs on compounded tirzepatide |
| Salt form concern | Only tirzepatide base was used in FDA trials; acetate and sodium forms lack bioequivalence data |
| Legal status (2026) | Highly restricted; FDA ended shortage exemption in late 2024 |
| Cost difference | Brand: $900-1,100/month; Compounded: $150-450/month |
For background on compounding safety standards, see our guide on whether compounded tirzepatide is safe. For dosing protocols, check the compound tirzepatide dosage chart.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What the SURMOUNT Trials Actually Proved
The entire case for tirzepatide's efficacy rests on Eli Lilly's SURMOUNT clinical trial program. Five large-scale Phase 3 trials enrolled over 10,000 participants and generated the data that led to FDA approval. Understanding what these trials tested, and what they did not, is essential for evaluating whether a compounded version will deliver similar results.
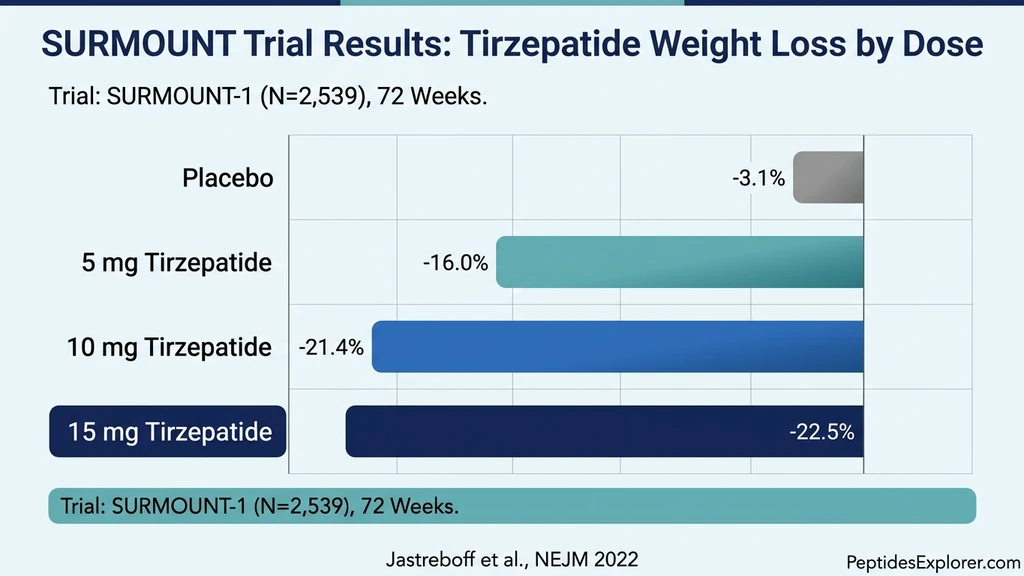
Every participant in these trials received tirzepatide manufactured by Eli Lilly under current good manufacturing practice (cGMP) standards. The drug product used tirzepatide in its base form, not a salt derivative, in a pre-filled autoinjector pen with verified potency, sterility, and stability at every batch (Jastreboff et al., NEJM, 2022).
SURMOUNT-1: The Landmark Obesity Trial
SURMOUNT-1 randomized 2,539 adults with obesity (BMI of 30 or higher) or overweight (BMI of 27 or higher) with at least one weight-related comorbidity to tirzepatide 5 mg, 10 mg, or 15 mg versus placebo for 72 weeks. None of the participants had diabetes.
Results were striking. Mean weight reduction from baseline was 16.0% with 5 mg, 21.4% with 10 mg, and 22.5% with 15 mg, compared to 3.1% with placebo. More than one-third of participants receiving the 15 mg dose lost 25% or more of their body weight. No anti-obesity medication had previously achieved these numbers in a controlled trial (Jastreboff et al., NEJM, 2022).
These figures represent what happens when a patient receives verified-potency, cGMP-manufactured tirzepatide base in a controlled clinical setting. They do not represent what happens with a compounded product from an unknown pharmacy using an untested salt form. The difference is not academic. It is the difference between a guaranteed 22% weight loss and a hope that the product works similarly. For a full profile of the molecule, visit our tirzepatide overview.
SURMOUNT-3 and SURMOUNT-4: Maintenance and Durability
SURMOUNT-3 tested tirzepatide in participants who had already lost at least 5% body weight through intensive lifestyle intervention. The tirzepatide group lost an additional 18.4% of body weight over 72 weeks, compared to a 2.5% gain in the placebo group. This demonstrated that tirzepatide produces results beyond what diet and exercise alone can achieve (Wadden et al., Nature Medicine, 2023).
SURMOUNT-4 addressed durability. After 36 weeks of open-label tirzepatide treatment (mean weight reduction of 20.9%), participants were randomized to continue tirzepatide or switch to placebo for an additional 52 weeks. Those who continued lost an additional 5.5%, reaching a total reduction of 25.3% at 88 weeks. Those switched to placebo regained weight, ending at 9.9% total reduction (Aronne et al., JAMA, 2024).
The takeaway for compounded users: tirzepatide works when the drug is consistently potent and consistently administered. Any interruption in potency, whether from a sub-potent batch, degraded product, or inconsistent salt form, can trigger the same rebound seen in the placebo arm of SURMOUNT-4.
SURMOUNT-5: Tirzepatide Outperforms Semaglutide
The most recent trial, SURMOUNT-5, directly compared tirzepatide 15 mg to semaglutide 2.4 mg (Wegovy) in adults with obesity. At 72 weeks, tirzepatide produced superior weight reduction and greater waist circumference decrease compared to semaglutide (Lilly Investor Relations, 2024). This head-to-head result reinforced tirzepatide's position as the most effective GLP-1-class medication currently available.
However, both arms used manufacturer-produced, FDA-approved products. Neither arm used compounded versions of either drug. The superiority of tirzepatide over semaglutide has been demonstrated only with brand-name formulations. For patients comparing weight loss options, see our best peptides for weight loss guide.
Salt Forms: The Variable That Could Undermine Everything
This is where compounded tirzepatide diverges most sharply from brand-name product, and where the question of "does it work" gets complicated. Compounding pharmacies do not always use the same form of tirzepatide that Eli Lilly tested in clinical trials.

Eli Lilly's FDA-approved products (Mounjaro and Zepbound) use tirzepatide in its free base form. This is the exact molecular structure that underwent Phase 1, Phase 2, and Phase 3 clinical testing, pharmacokinetic profiling, stability testing, and post-market surveillance. Every data point about efficacy and safety applies specifically to tirzepatide base.
Tirzepatide Base: The FDA-Tested Standard
Tirzepatide base has a molecular weight of 4,813.45 Da. It is the uncharged, unmodified peptide molecule. All pharmacokinetic parameters published in the FDA approval package, including absorption rate, bioavailability, half-life (approximately 5 days), and receptor binding affinity, were measured using this form.
When a compounding pharmacy uses tirzepatide base as its starting material, the active ingredient is chemically identical to what Eli Lilly puts in Mounjaro pens. The remaining variables are potency (did the pharmacy get the concentration right?), sterility (is the solution free of contaminants?), and stability (will the molecule remain intact through the beyond-use date?). These are serious variables, but the molecule itself is not a question mark.
For patients preparing compounded tirzepatide from vials, our how to reconstitute tirzepatide guide covers proper technique. Use the peptide reconstitution calculator to verify dilution volumes.
Tirzepatide Acetate: Common but Untested
Tirzepatide acetate is a salt form where the peptide molecule is combined with acetic acid as a counterion. Many compounding pharmacies source tirzepatide acetate from raw material suppliers because it is more stable as a powder, easier to handle, and often cheaper than the free base form.
The problem: no clinical trial has tested tirzepatide acetate for efficacy or safety in humans. Salt form changes in peptide drugs can alter solubility, pH in solution, absorption kinetics, and injection site tolerability. For many small-molecule drugs, salt form differences are well-characterized and clinically irrelevant. For large peptides like tirzepatide (39 amino acids), the impact is less predictable (FDA Guidance on Compounding, 2023).
This does not mean tirzepatide acetate is ineffective. The tirzepatide molecule itself is likely intact and bioactive. But "likely" is not the same as "proven." The acetate counterion may change the pH of the reconstituted solution, which could affect subcutaneous absorption rates. It may alter the stability profile, leading to faster degradation in the vial. It may irritate injection sites differently than the base form. Without published pharmacokinetic studies comparing acetate to base, patients are relying on theoretical reasoning rather than clinical evidence.
Tirzepatide Sodium: The FDA's Flagged Concern
Tirzepatide sodium salt (CAS 2023788-19-2) is another common compounding starting material. The FDA has specifically flagged the use of tirzepatide salts as a safety concern, noting that compounded products using salt forms are not copies of the approved drug and may have different pharmacological properties (FDA Safety Communication, 2024).
The sodium counterion adds molecular weight and can change the molecule's behavior in solution. Different pH, different osmolality, potentially different absorption. A sodium salt of tirzepatide is not the same chemical entity as tirzepatide base, even though the active peptide sequence is identical. The FDA considers this distinction important enough to have issued public warnings about it.
For patients evaluating compounded products, the salt form used is one of the first questions to ask the pharmacy. If they cannot tell you whether they use base, acetate, or sodium, or if they do not understand why it matters, that is a red flag about their overall quality standards.
Potency Testing: Where Compounded Products Often Fail
Even when a compounding pharmacy uses tirzepatide base, the product is only as good as its potency testing. Potency testing determines whether the vial you inject actually contains the labeled amount of active ingredient. For brand-name Mounjaro, every batch undergoes validated high-performance liquid chromatography (HPLC) assay as part of Eli Lilly's cGMP manufacturing process. For compounded tirzepatide, potency testing ranges from rigorous to nonexistent, depending on the pharmacy.
What Proper Potency Testing Looks Like
A reputable compounding pharmacy will test each batch of tirzepatide for potency using an HPLC assay or equivalent validated method. The result should show that the actual concentration falls within 90-110% of the labeled concentration, the standard range for injectable drugs. The pharmacy should be able to provide a Certificate of Analysis (CoA) for each batch, showing the test date, method, result, and the identity of the testing laboratory.
503B outsourcing facilities are required to conduct potency testing on every batch before release. This is a federal requirement under cGMP rules. 503A pharmacies, which operate under state regulation, may or may not test each batch, depending on state law and the pharmacy's internal policies. Some 503A pharmacies send samples to third-party labs. Others rely on the potency certificate from their raw material supplier, which tests the powder before compounding but not the final product after reconstitution.
The difference matters. A raw material CoA confirms that the tirzepatide powder is potent. It does not confirm that the pharmacy measured it correctly, dissolved it properly, maintained sterility, or stored it at the right temperature. Final product testing is the only way to verify what the patient actually receives. To plan your dosing protocol around verified potency, use our tirzepatide dosage calculator.
FDA Testing Reveals Widespread Quality Failures
In 2023, the FDA tested samples from multiple compounding pharmacies producing GLP-1 receptor agonists including tirzepatide. The results were alarming: 28% of tested sterile compounded preparations failed quality standards. Failures included sub-potent active ingredients (less drug than labeled), super-potent batches (more drug than labeled, increasing side effect risk), contamination with particulate matter, and incorrect pH values (FDA Compounding Survey, 2023).
Sub-potent tirzepatide means less weight loss, or no weight loss at all. A vial labeled as 10 mg/mL that actually contains 6 mg/mL will not produce SURMOUNT-level results, no matter how faithfully the patient follows the dosing schedule. The patient concludes that "compounded tirzepatide does not work" when the real problem is that they received 60% of the intended dose.
Super-potent batches carry the opposite risk. A patient titrating carefully from 2.5 mg to 5 mg who unknowingly receives a 7 mg dose may experience severe nausea, vomiting, or pancreatitis. The tirzepatide dosage chart in units assumes accurate potency. Inaccurate potency makes the entire dosing protocol unreliable.
How to Verify Your Pharmacy's Testing
Before filling a compounded tirzepatide prescription, ask the pharmacy these questions:
- 1.Do you test each batch for potency before dispensing? The only acceptable answer is yes.
- 2.What method do you use for potency testing? HPLC or a validated equivalent is the standard.
- 3.Can you provide a Certificate of Analysis for my specific batch? If they cannot, the product was not tested.
- 4.Is your testing done in-house or by a third-party laboratory? Third-party testing from an ISO-accredited lab is more reliable than in-house testing.
- 5.What is your acceptance range for potency? 90-110% of label claim is the industry standard for injectables.
If the pharmacy cannot answer these questions clearly, or if they become defensive, source your prescription elsewhere. The cost savings of compounded tirzepatide mean nothing if the product does not contain the right amount of drug. For cost comparisons between compounded and brand-name options, see our tirzepatide cost with insurance breakdown and use the peptide cost calculator.
Real-World Outcomes: What Compounded Users Report
No published clinical trials have evaluated compounded tirzepatide, but real-world reports from patients and clinicians provide a partial picture. These reports are not controlled evidence. They are subject to selection bias, recall bias, and the placebo effect. They are also the only outcome data available for compounded products.
Positive Outcomes from Quality Pharmacies
Patients receiving compounded tirzepatide from PCAB-accredited 503B outsourcing facilities frequently report weight loss trajectories consistent with SURMOUNT trial data. Weight loss of 10-15% over 12-16 weeks at maintenance doses of 10-15 mg per week is commonly reported in online patient communities and telehealth provider testimonials.
These outcomes make pharmacological sense. If the compounded product uses tirzepatide base at correct potency, the molecule binds the same GIP and GLP-1 receptors, activates the same intracellular signaling cascades, and produces the same appetite suppression, delayed gastric emptying, and improved insulin sensitivity. The chemistry does not care whether the vial has a Lilly logo or a compounding pharmacy label.
Several obesity medicine physicians have reported in conference presentations and medical forums that their patients on compounded tirzepatide from reputable 503B pharmacies show comparable weight loss, HbA1c reduction, and metabolic improvement to those on brand-name Mounjaro. These clinical observations, while not peer-reviewed data, are consistent with the expected pharmacology. For expected timelines, see our guide on how long tirzepatide takes to work.
Negative Outcomes and Red Flags
Not all reports are positive. Common complaints about compounded tirzepatide include:
No weight loss or minimal weight loss. The most likely explanation is sub-potent product. If the vial contains less tirzepatide than labeled, the patient receives a lower effective dose. A patient who thinks they are taking 10 mg but is actually receiving 5 mg will lose weight more slowly, if at all.
Excessive side effects at low doses. Severe nausea, persistent vomiting, or diarrhea at starting doses (2.5 mg) can indicate a super-potent batch. Brand-name tirzepatide at 2.5 mg typically causes mild nausea in 12-18% of patients. If a compounded 2.5 mg dose causes severe GI distress, the actual dose may be higher. Review tirzepatide before and after results to understand expected side effect patterns.
Injection site reactions disproportionate to dose. Excessive redness, swelling, or pain at the injection site may indicate incorrect pH, improper buffering, or contamination in the compounded solution rather than a reaction to tirzepatide itself.
Product appearance changes. Cloudiness, particulate matter, color changes, or unusual viscosity in the vial indicate potential degradation, contamination, or formulation errors. Brand-name Mounjaro is a clear, colorless to slightly yellow solution. Any compounded product that looks different should not be injected.
If you experience unexpected results at any point, consult your prescriber. For help with proper injection technique, see where to buy tirzepatide for sourcing guidance and review the full tirzepatide profile.
FDA Regulatory Status in 2026: The Legal Landscape
Whether compounded tirzepatide "works" is partly a regulatory question. A product that cannot be legally obtained, or that is produced under compromised conditions due to regulatory pressure, may not perform as expected regardless of the molecule's inherent efficacy.
The Shortage Exemption That Opened the Floodgates
When the FDA placed tirzepatide on its Drug Shortage List in December 2022, it activated a legal pathway under federal law that allows compounding pharmacies to produce copies of commercially available drugs during confirmed shortages. Demand for Mounjaro had outstripped Eli Lilly's manufacturing capacity within months of the drug's June 2022 launch.
This shortage exemption led to explosive growth in compounded tirzepatide. Hundreds of pharmacies began producing the peptide. Telehealth platforms built entire business models around prescribing compounded GLP-1 agonists at a fraction of brand-name cost. By mid-2024, an estimated 50,000 to 100,000 patients were receiving compounded tirzepatide monthly.
Eli Lilly invested over $5 billion in new manufacturing facilities. By late 2024, the FDA determined that the tirzepatide shortage had been resolved. This resolution ended the legal basis for most compounding under the shortage exemption. For a broader view of how this affects peptide access, see our FDA peptide crackdown 2026 analysis.
Post-Shortage Legal Battles (2025-2026)
The end of the shortage exemption triggered immediate legal challenges. Multiple compounding pharmacies and industry organizations filed lawsuits against the FDA, arguing that the shortage resolution was premature and that patients would lose access to affordable medication.
Several courts issued temporary restraining orders allowing continued compounding during litigation. As of April 2026, the legal landscape remains fragmented. Some 503A pharmacies continue to compound tirzepatide under patient-specific prescriptions, arguing that the right to compound is independent of shortage status when a physician determines medical necessity. Many 503B outsourcing facilities have scaled back or ceased tirzepatide production due to increased FDA scrutiny.
The practical effect for patients: compounded tirzepatide is harder to obtain in 2026 than it was in 2023-2024. Pharmacies that continue to produce it face uncertain legal footing. Some have shifted to tirzepatide salt forms (acetate, sodium) rather than copies of the exact base form, arguing that salt forms are different chemical entities not subject to the same compounding restrictions. This legal argument is contested, and it introduces the potency and bioequivalence concerns discussed earlier.
What This Means for Patients Today
If you are currently using compounded tirzepatide or considering it, here is the regulatory reality:
503B outsourcing facilities that continue to produce tirzepatide do so under increasing FDA scrutiny. Some have received warning letters. Others operate under court orders allowing continued production during litigation. The quality of product from these facilities may be affected by the uncertainty, as some have reduced quality control investment or shifted to cheaper salt form starting materials.
503A pharmacies may still compound tirzepatide with a valid patient-specific prescription from a licensed prescriber who documents medical necessity. The legal basis is strongest when the prescriber can demonstrate that the patient has a legitimate clinical need that brand-name products cannot meet, such as an allergy to an inactive ingredient in Mounjaro.
Online and international sources operate entirely outside the US regulatory framework. Products from these sources carry the highest risk of sub-potency, contamination, and counterfeit ingredients. The FDA has seized shipments of unapproved tirzepatide at the border and issued import alerts.
Regardless of the source, anyone using compounded tirzepatide should track their results carefully. Weight loss of 3-5% per month during the titration phase (first 16-20 weeks) is consistent with the SURMOUNT data. If you are not seeing results on compounded product, the molecule may not be the problem. The product may be. For more details on sourcing, see where to buy tirzepatide.
Compounded vs. Brand: A Head-to-Head Comparison
The table below summarizes the practical differences between compounded and brand-name tirzepatide across every dimension that affects whether the product works.
| Factor | Brand (Mounjaro/Zepbound) | Compounded (503B) | Compounded (503A) |
|---|---|---|---|
| Active ingredient | Tirzepatide base | Base, acetate, or sodium | Base, acetate, or sodium |
| Clinical trial data | SURMOUNT 1-5 (10,000+ patients) | None | None |
| FDA approval | Yes (2022/2023) | No | No |
| cGMP manufacturing | Yes (required) | Yes (required) | No (state-regulated) |
| Batch potency testing | Every batch, HPLC-validated | Every batch (required) | Varies by state/pharmacy |
| Sterility testing | Every batch | Every batch (required) | Varies |
| Stability data | Published, validated | Limited or none | Limited or none |
| Delivery format | Pre-filled pen | Multi-dose vial | Multi-dose vial |
| Monthly cost | $900-1,100 (without insurance) | $250-450 | $150-350 |
| Insurance coverage | Variable; improving in 2026 | Rarely covered | Rarely covered |
| Legal status (2026) | Fully legal | Contested (ongoing litigation) | Contested (requires medical necessity) |
The efficacy gap is not inherent to compounding. A properly manufactured compounded tirzepatide base at correct potency will activate the same receptors as Mounjaro. The gap is in quality assurance. Brand products guarantee potency and sterility. Compounded products depend on the specific pharmacy's standards. Compare costs in detail using our peptide cost calculator.
How to Maximize Effectiveness of Compounded Tirzepatide
If you choose compounded tirzepatide after weighing the risks and benefits, these steps will maximize the probability that your product works as intended.
Source from a PCAB-Accredited 503B Facility
The Pharmacy Compounding Accreditation Board (PCAB) provides voluntary accreditation for compounding pharmacies that meet rigorous quality standards, including environmental monitoring, personnel competency, potency and sterility testing, and documented standard operating procedures. PCAB accreditation does not guarantee perfection, but it represents the highest quality tier available for compounded products.
A PCAB-accredited 503B facility is the closest you can get to pharmaceutical-grade manufacturing outside of a drug company. These facilities undergo regular inspections, maintain detailed batch records, and test every lot before release. If your pharmacy is not PCAB-accredited, ask why.
Confirm the Salt Form
Ask your pharmacy whether they use tirzepatide base, tirzepatide acetate, or tirzepatide sodium. If they use a salt form, ask for the rationale and any bioequivalence data they rely on. The strongest evidence supports tirzepatide base, as it is the form tested in all SURMOUNT trials.
Some pharmacies may use acetate or sodium because the base form is harder to source or more expensive. That is a business decision, not a clinical one. If your pharmacy uses a salt form, you should understand that you are injecting a chemical entity that has never been tested in a published clinical trial. The risk may be low. It is not zero.
Track Your Response Objectively
Weigh yourself at the same time each week, under the same conditions. Track your food intake. Note side effects. Compare your trajectory to the SURMOUNT data.
Expected weight loss on properly dosed tirzepatide: - Weeks 1-4 (2.5 mg): 1-3% body weight loss - Weeks 5-8 (5 mg): Additional 2-4% loss - Weeks 9-12 (7.5 mg): Additional 2-3% loss - Weeks 13-20 (10-15 mg): Additional 3-6% loss - Total at 20 weeks: 8-16% body weight loss
If your results fall significantly below these benchmarks despite adherence to dosing and dietary recommendations, the product potency is suspect. Discuss switching to brand-name product or a different pharmacy with your prescriber. For detailed titration protocols, review the compound tirzepatide dosage chart and the tirzepatide dosage chart in units.
Handle and Store Correctly
Compounded tirzepatide in multi-dose vials is more sensitive to storage conditions than brand-name pre-filled pens. Improper handling can degrade the peptide, reducing potency even if the pharmacy produced it correctly.
Storage rules: - Refrigerate at 2-8 degrees Celsius (36-46 degrees Fahrenheit) at all times after receiving - Never freeze tirzepatide - Protect from light (keep in original packaging or wrap vial in foil) - Use within the beyond-use date assigned by the pharmacy (typically 28-42 days after compounding) - Do not use if the solution appears cloudy, discolored, or contains particles - After reconstitution (if applicable), keep refrigerated and use within 28 days
For step-by-step preparation instructions, see our how to reconstitute tirzepatide guide. Our peptide reconstitution calculator will help you calculate exact volumes for your prescribed dose.
When Compounded Tirzepatide Is the Wrong Choice
Not every situation warrants the risks of compounding. Compounded tirzepatide is the wrong choice when:
You can access brand-name product. If your insurance covers Mounjaro or Zepbound, or if you qualify for Eli Lilly's savings programs, brand-name product eliminates every quality concern associated with compounding. FDA-approved products have verified potency, proven sterility, and clinical trial data behind every dose.
Your pharmacy cannot provide documentation. If the compounding pharmacy cannot produce a current 503B registration, a CoA for your batch, or clear answers about salt form and potency testing, the risk-to-savings ratio is unacceptable.
You have had adverse reactions to compounded product. Unusual injection site reactions, unexpected GI side effects at low doses, or no response at therapeutic doses all suggest a product quality issue. Switch to brand-name or a different pharmacy immediately.
You are using it without a prescription. Compounded tirzepatide without a valid prescription from a licensed prescriber is illegal, unmonitored, and dangerous. Tirzepatide requires medical oversight for dose titration, metabolic monitoring, and management of potential complications including pancreatitis, gallbladder disease, and thyroid tumors.
You are sourcing internationally or online without verification. Unregulated international sources carry the highest risk of counterfeit product, contamination, and mislabeling. The lowest price is not a bargain if the vial contains the wrong drug or no drug at all.
For patients who cannot access tirzepatide in any form, alternative GLP-1 receptor agonists may be available. See our comparison of semaglutide and the broader best peptides for weight loss guide.
Frequently Asked Questions
Is compounded tirzepatide the same as Mounjaro?
Compounded tirzepatide uses the same active molecule as Mounjaro (tirzepatide, a 39-amino-acid dual GIP/GLP-1 receptor agonist), but it is not the same product. Mounjaro is manufactured by Eli Lilly under FDA-approved cGMP conditions using tirzepatide base. Compounded versions may use different salt forms (acetate or sodium), lack batch-level potency verification, and have no clinical trial data supporting their specific formulation. The molecule is the same; the quality assurance is not. For a detailed safety analysis, see our guide on whether compounded tirzepatide is safe.
How much weight can I lose on compounded tirzepatide?
If your compounded tirzepatide contains verified-potency tirzepatide base, weight loss should approximate the SURMOUNT trial results: 16-22.5% of body weight over 72 weeks, depending on dose (5-15 mg). However, compounded products with unknown potency or untested salt forms may produce less weight loss or none at all. Track your progress against the tirzepatide dosage chart and consult your prescriber if results fall below expectations. Expected early-phase loss is 1-3% body weight in the first four weeks at 2.5 mg.
Why am I not losing weight on compounded tirzepatide?
The most common reasons are sub-potent product (the vial contains less tirzepatide than labeled), incorrect salt form (acetate or sodium instead of the tested base form), degraded peptide due to improper storage, insufficient dose (still in the titration phase), or dietary factors. Request a Certificate of Analysis from your pharmacy to verify batch potency. If the pharmacy cannot provide one, switch providers. Also review our why am I not losing weight on tirzepatide guide for non-product-related causes.
Is compounded tirzepatide legal in 2026?
The legal status is contested. The FDA ended the tirzepatide shortage exemption in late 2024, removing the primary legal basis for most compounding. Multiple lawsuits are ongoing, and some courts have allowed continued compounding during litigation. 503A pharmacies may compound with a valid patient-specific prescription when a prescriber documents medical necessity. 503B facilities face increasing scrutiny. The legal landscape varies by jurisdiction and changes frequently. For the latest regulatory updates, see our FDA peptide crackdown 2026 article.
What is the difference between tirzepatide base and tirzepatide acetate?
Tirzepatide base is the free, uncharged form of the molecule used in all FDA-approved products and clinical trials. Tirzepatide acetate is a salt form where the peptide is combined with acetic acid as a counterion. The active peptide sequence is identical, but the salt form can alter solubility, pH, absorption kinetics, and injection site tolerability. No clinical trial has tested tirzepatide acetate in humans for weight loss or diabetes. Use the peptide reconstitution calculator to ensure proper dilution regardless of form.
How do I know if my compounded tirzepatide is working?
Track weekly weight loss, appetite changes, and side effects. On properly dosed tirzepatide at verified potency, you should see 1-3% body weight loss in the first four weeks (2.5 mg dose), appetite suppression within 1-2 weeks, and mild GI side effects (nausea, reduced appetite) that indicate receptor activation. If you experience zero appetite change and zero weight loss after four weeks, the product potency is likely the issue. Compare your trajectory to our tirzepatide before and after data and discuss results with your prescriber.
Should I switch from compounded to brand-name tirzepatide?
Switch to brand-name if you can afford it or your insurance covers it, if your compounded product is not producing expected results, if your pharmacy cannot provide batch testing documentation, or if the legal status of compounded tirzepatide in your state becomes restrictive. Brand-name Mounjaro and Zepbound eliminate quality concerns entirely. Check our tirzepatide cost with insurance guide to explore coverage options and manufacturer savings programs.
Can a compounding pharmacy make tirzepatide as strong as Mounjaro?
Yes, a properly equipped compounding pharmacy can formulate tirzepatide at the same concentrations as Mounjaro (2.5 mg to 15 mg per dose). The question is whether they consistently do so. Brand-name Mounjaro undergoes HPLC-validated potency testing on every batch under cGMP. Compounded products vary. A 503B facility with PCAB accreditation and batch-level testing is the most reliable compounded option. Request the Certificate of Analysis and compare the reported potency to label claim. Anything below 90% or above 110% of label is substandard. Plan your protocol with the tirzepatide dosage calculator.
The Bottom Line
Compounded tirzepatide can work. The molecule is the same dual GIP/GLP-1 receptor agonist that produced 22.5% body weight reduction in the SURMOUNT-1 trial. When compounded correctly, using tirzepatide base at verified potency in a sterile formulation, it activates the same receptors and should produce similar weight loss outcomes.
The word "should" carries all the weight in that sentence. No clinical trial has tested any compounded tirzepatide product. Every efficacy claim relies on extrapolation from brand-name data. The variables that separate a working compounded product from a failing one are potency accuracy, salt form selection, sterility, storage, and the regulatory compliance of the pharmacy that produced it. None of these variables affect brand-name Mounjaro.
For patients who cannot access or afford brand-name tirzepatide, a compounded product from a PCAB-accredited 503B outsourcing facility using tirzepatide base, with batch-level CoA documentation, represents the most evidence-supported alternative. Track results against SURMOUNT benchmarks. If weight loss stalls or side effects are unusual, question the product before questioning the molecule.
Use our tirzepatide dosage calculator to plan titration schedules, the peptide reconstitution calculator for dilution math, and the peptide cost calculator to compare long-term costs between compounded and brand-name options.
Related articles: - Is Compounded Tirzepatide Safe? — safety analysis of compounding practices and pharmacy verification - Compound Tirzepatide Dosage Chart — complete dosing protocol for compounded formulations - Tirzepatide Before and After — real-world results and timeline expectations - How to Reconstitute Tirzepatide — step-by-step preparation for vial-based product - FDA Peptide Crackdown 2026 — regulatory updates affecting compounded peptide access - Best Peptides for Weight Loss — comparing all GLP-1 and metabolic peptide options - Where to Buy Tirzepatide — sourcing guide for brand and compounded tirzepatide
Explore all peptide profiles and tools at PeptidesExplorer.
Related Articles
Is Compounded Tirzepatide Safe?
Is compounded tirzepatide safe? Learn FDA 503A/503B rules, quality risks, how to verify a compounding pharmacy, and brand vs compounded differences.
Does Oral Tirzepatide Work? The Facts
Oral tirzepatide is not FDA-approved. Why peptide pills face absorption barriers, sublingual drops vs injections, and oral GLP-1 alternatives in 2026.
Tirzepatide Cost With Insurance (2026)
Tirzepatide costs $550-$1,060/month at list price. With commercial insurance, copays range from $25-$150/month. Mounjaro, Zepbound, Medicare breakdown.
Tirzepatide: How Long to Work?
Tirzepatide reduces appetite within the first week, produces measurable weight loss by week 4, and delivers significant results by weeks 12-16.