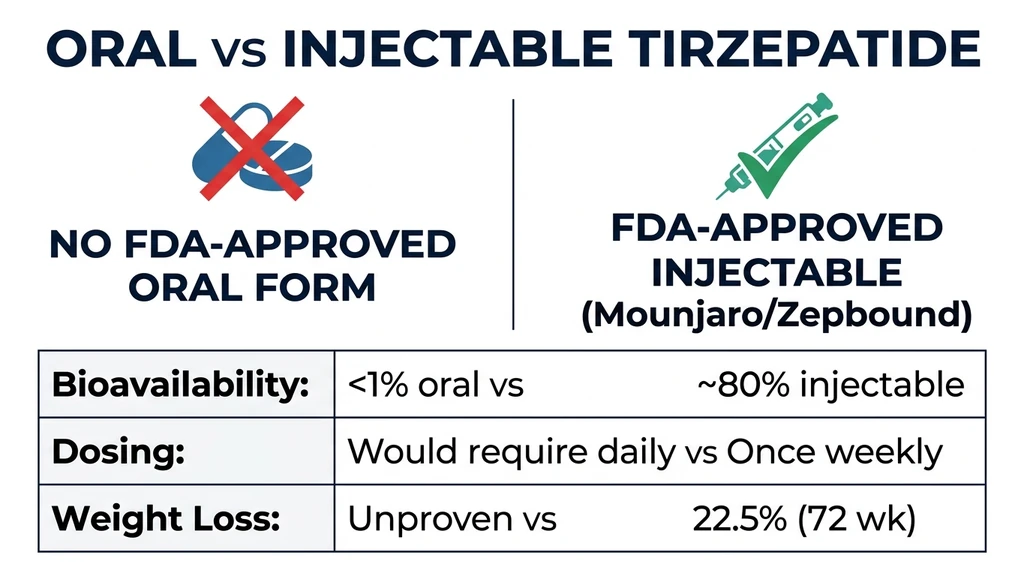
You found a website selling "oral tirzepatide drops" and want to know if swallowing or placing this peptide under your tongue actually produces the same weight loss as the injection. No FDA-approved oral tirzepatide exists as of April 2026. Compounded sublingual formulations have unknown bioavailability, and no published clinical trial supports oral tirzepatide at therapeutic levels. The only proven route remains a once-weekly subcutaneous injection (Mounjaro for type 2 diabetes, Zepbound for obesity).
| Quick Reference | Details |
|---|---|
| Oral tirzepatide FDA-approved? | No. Injection only (April 2026) |
| Sublingual drops available? | Previously from compounding pharmacies; legality ended March 2025 |
| Sublingual bioavailability | Estimated <2-3%, highly variable between patients |
| Injectable bioavailability | ~80% (subcutaneous) |
| Injectable weight loss (15 mg) | 22.5% at 72 weeks (SURMOUNT-1) |
| Oral GLP-1 alternatives | Wegovy pill (25 mg semaglutide, approved Dec 2025), orforglipron (Phase 3) |
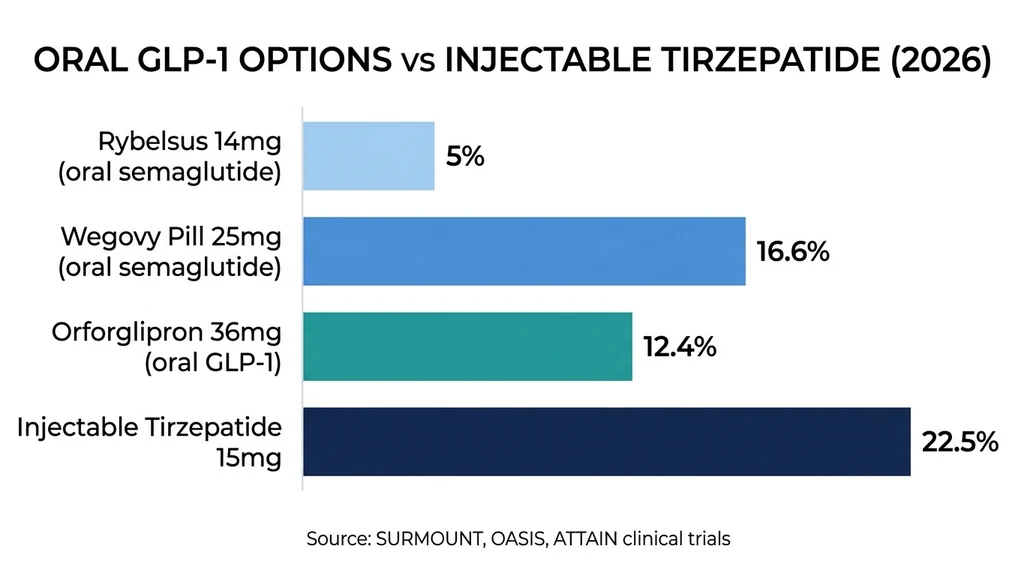
| Best oral weight loss result | Wegovy pill 25 mg: 16.6% at 64 weeks |
| Injectable tirzepatide advantage | Dual GLP-1/GIP activation; no oral drug replicates this |
That gap between 2-3% sublingual bioavailability and 80% injectable bioavailability is the core problem. You would need to absorb roughly 40 times more drug orally to match a single weekly injection. This article walks through the science behind that barrier, examines what compounded oral products actually deliver, compares every oral GLP-1 alternative available in 2026, and helps you decide whether to start injectable tirzepatide now or wait for a pill that may never fully replicate this drug's dual-receptor power. For current dosing protocols, see our compound tirzepatide dosage chart.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
The Short Answer: Oral Tirzepatide Does Not Work Like the Injection
Tirzepatide is a 39-amino-acid peptide weighing 4,810 daltons. Every property of that molecule works against oral delivery. Stomach acid unfolds its three-dimensional structure within minutes. Pepsin and intestinal proteases chop the linearized chain into fragments too small to activate GLP-1 or GIP receptors. Even if a fraction survives enzymatic attack, the molecule is nearly ten times too large to cross the intestinal wall through normal absorption pathways.
The result is oral bioavailability well below 1% without specialized delivery technology. For a 15 mg injectable dose to produce the same blood levels orally, you would need to swallow approximately 1,500 mg or more. No compounding pharmacy formulates at that scale, and no absorption enhancer has been validated for tirzepatide specifically (Drucker, 2020).
This does not mean all oral GLP-1 therapy is impossible. Oral semaglutide exists (Rybelsus, and now the Wegovy pill), and a non-peptide oral GLP-1 agonist called orforglipron is advancing through Phase 3 trials. But neither of these is tirzepatide, and neither replicates its dual GLP-1/GIP mechanism. The distinction matters because that dual activation is what gives tirzepatide its edge in weight loss trials.
Why Tirzepatide Cannot Survive the Digestive Tract
Understanding the biology explains why "oral tirzepatide" is not simply a matter of putting the drug into a capsule. Three sequential barriers destroy the molecule before it can reach your bloodstream.
Barrier 1: Gastric Acid and Pepsin
Your stomach maintains a pH between 1.5 and 3.5, acidic enough to denature most proteins on contact. Tirzepatide's folded conformation, the precise shape that lets it dock into both GLP-1 and GIP receptors, unravels within minutes at that pH. Pepsin, a protease enzyme activated by stomach acid, then cleaves the linearized peptide at multiple sites along its 39-amino-acid backbone.
Enteric coatings can shield a tablet through the stomach, but the small intestine deploys its own arsenal: trypsin, chymotrypsin, and carboxypeptidases. These enzymes attack different amino acid bonds from different angles. Surviving one protease means nothing if three others are waiting downstream (Drucker, 2020).
Barrier 2: Intestinal Permeability
Even if tirzepatide survived enzymatic destruction, its size blocks absorption. Small molecules under 500 daltons cross the intestinal wall through gaps between cells or directly through cell membranes. Tirzepatide, at 4,810 daltons, is roughly ten times too large for either route.
Think of the intestinal lining as a security checkpoint that only allows carry-on luggage. Tirzepatide is an oversized suitcase. Without a permeation enhancer that temporarily widens the gaps between cells, the molecule simply sits in the intestinal lumen and is excreted. Oral semaglutide uses a chemical called SNAC to widen those gaps, but SNAC was engineered specifically for semaglutide's physicochemical properties. No equivalent enhancer has been developed or tested for tirzepatide (Buckley et al., 2018).
Barrier 3: First-Pass Liver Metabolism
Any tirzepatide that somehow crossed the intestinal wall would enter the portal vein and flow directly to the liver before reaching general circulation. The liver metabolizes many peptides on first pass, further reducing the amount available to target tissues. Injectable delivery bypasses this entirely. The drug enters the bloodstream through subcutaneous capillaries, skipping the liver's initial filtration.
The combined effect of all three barriers means oral tirzepatide bioavailability would be far below 1%. At that level, achieving therapeutic blood concentrations requires impractical megadoses. This is not a manufacturing problem that a better pill can solve. It is a fundamental conflict between the molecule's structure and the human digestive system.
What About Sublingual Tirzepatide Drops and Troches?
Sublingual delivery (under the tongue) bypasses stomach acid, which is why some compounding pharmacies offered tirzepatide in this form during the medication shortage period. The sublingual mucosa is thin and vascularized, allowing some molecules to absorb directly into the bloodstream. The question is whether tirzepatide absorbs in meaningful quantities through this route.
Sublingual Bioavailability: The Data Gap
No peer-reviewed clinical trial has measured sublingual tirzepatide bioavailability in humans. The estimated absorption rate for large peptides via the sublingual route is below 2-3% without enhancers. Individual results with compounded sublingual tirzepatide ranged from significant weight loss to no detectable effect, likely reflecting wildly variable absorption between patients and between batches.
The sublingual mucosa has a surface area of approximately 26 square centimeters, compared to 32 square meters for the small intestine. Even with better permeability per unit area, the sublingual route simply does not offer enough absorptive surface for a large peptide. The molecule must also remain under the tongue long enough to absorb, typically 2-5 minutes, without being swallowed or diluted by saliva.
Compounding pharmacies sometimes add absorption enhancers or permeation modifiers to sublingual formulations. The effectiveness of these additives varies between formulations and between individual patients. Without standardized bioequivalence testing, there is no way to know whether a specific sublingual product delivers 0.5%, 2%, or 5% of its labeled dose into the bloodstream.
Legal Status: The Compounding Exemption Ended
During the tirzepatide shortage, the FDA granted temporary leniency to compounding pharmacies under section 503A and 503B of the Federal Food, Drug, and Cosmetic Act. This allowed pharmacies to compound tirzepatide products, including sublingual formulations, while the shortage persisted. That leniency ended for 503A pharmacies on February 11, 2025, and for 503B outsourcing facilities on March 19, 2025.
As of April 2026, compounding tirzepatide is no longer legally permitted under the shortage exemption. Some pharmacies may still offer these products, but they do so outside the regulatory framework that previously authorized their activity. For patients who used compounded sublingual tirzepatide, switching to FDA-approved injectable Mounjaro or Zepbound ensures consistent dosing and known bioavailability. See our guide on where to buy tirzepatide for current sourcing options.
Compounding Quality Concerns
Beyond bioavailability, compounded sublingual formulations carry quality risks. Troches and drops are mixed without the standardized manufacturing processes required of FDA-approved drugs. Concentration can vary between batches, between individual troches in the same batch, and even within a single troche if the active ingredient distributes unevenly.
A patient receiving 5 mg of tirzepatide from one troche might receive 3 mg from the next and 7 mg from a third, even if all are labeled identically. With injectable tirzepatide, each prefilled pen delivers a precise, factory-calibrated dose. The difference is the difference between a hand-poured candle and a machine-tooled engine part. Both can "work," but only one delivers repeatable precision.
For patients considering any compounded tirzepatide product, our guide on compound tirzepatide safety provides a detailed risk-benefit framework. For reconstitution of injectable vials, see our tirzepatide reconstitution guide.
How Oral Semaglutide Actually Works (And Why Tirzepatide Cannot Copy It)
Oral semaglutide is the proof that oral peptide delivery is possible, but the specific technology behind it reveals why the same approach cannot simply be applied to tirzepatide.
The SNAC Absorption Enhancer System
Novo Nordisk developed a proprietary enhancer called SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate) specifically for semaglutide. SNAC raises the local pH around the tablet in the stomach, creating a protective buffer that shields semaglutide from pepsin. It also promotes transcellular absorption through the gastric epithelium, allowing intact semaglutide to cross into the bloodstream directly from the stomach wall (Buckley et al., 2018).
Even with SNAC, oral semaglutide achieves only about 1% bioavailability. That means 99% of each dose is destroyed in the gut. The system works because semaglutide has specific physicochemical properties (albumin binding, relative protease resistance due to amino acid modifications) that make it compatible with SNAC. Tirzepatide has different structural characteristics. Its larger size, different amino acid modifications, and dual-receptor binding domain would require a fundamentally different enhancer system that does not exist.
The Wegovy Pill: What FDA Approval Means for Oral GLP-1
In December 2025, the FDA approved oral Wegovy (semaglutide 25 mg tablets) for chronic weight management, making it the first oral GLP-1 approved for obesity. The OASIS clinical trial program demonstrated that the 25 mg oral dose achieved 16.6% weight loss at 64 weeks in adherent patients. One-third of participants lost at least 20% of their body weight.
This approval is a milestone for oral peptide delivery, but it does not change the outlook for oral tirzepatide. Wegovy pills use the same SNAC technology as Rybelsus, optimized for semaglutide. The pill requires the same fasting protocol: take on an empty stomach with no more than 4 ounces of water, wait 30 minutes before eating. Novo Nordisk launched the Wegovy pill in January 2026 at approximately $149/month, substantially less than injectable Wegovy.
For patients currently on injectable semaglutide considering the switch to the pill form, the weight loss results are comparable. But for patients comparing oral Wegovy to injectable tirzepatide, the 16.6% vs. 22.5% gap remains significant. Tirzepatide's dual GLP-1/GIP activation produces roughly 6 percentage points more weight loss. See our guide on best peptides for weight loss for a comprehensive comparison.
Oral Semaglutide vs Injectable Tirzepatide: Head-to-Head Data
No trial has directly compared oral semaglutide to injectable tirzepatide. However, cross-trial comparisons using matched populations and endpoints provide a reasonable estimate.
| Metric | Oral Wegovy (25 mg) | Injectable Tirzepatide (15 mg) |
|---|---|---|
| Route | Oral tablet, daily | Subcutaneous injection, weekly |
| Weight loss (adherent) | 16.6% (64 wk) | 22.5% (72 wk) |
| Patients losing 20%+ | ~33% | ~55% |
| A1C reduction (T2D) | ~1.5% | ~2.4% |
| Mechanism | GLP-1 only | GLP-1 + GIP dual agonist |
| Fasting required | Yes, 30 min | No |
| Cost (list) | ~$149/month | ~$1,000/month |
| FDA approved for obesity | Yes (Dec 2025) | Yes (Nov 2023) |
The Wegovy pill offers meaningful weight loss at a lower price point and without injections. But it does not replicate tirzepatide's dual mechanism or match its peak efficacy. Patients seeking maximum weight loss still benefit most from injectable tirzepatide. For those considering a switch between these drug classes, see our semaglutide to tirzepatide switching guide.
Orforglipron: Eli Lilly's Oral Alternative to Tirzepatide
Eli Lilly is not developing an oral version of tirzepatide. Instead, the company is pursuing orforglipron, a completely different molecule engineered specifically for oral delivery. Understanding the distinction is critical for anyone hoping Lilly will eventually put tirzepatide in a pill.
Small Molecule vs Peptide: A Fundamental Difference
Orforglipron is not a peptide. It is a non-peptide, small-molecule GLP-1 receptor agonist with a molecular weight under 600 daltons. That size allows it to survive stomach acid, cross the intestinal wall through standard absorption pathways, and achieve oral bioavailability that peptides cannot match. No specialized enhancer like SNAC is needed.
Orforglipron activates only the GLP-1 receptor. It does not activate the GIP receptor. This is the key limitation: tirzepatide's dual GLP-1/GIP activation is what drives its superior weight loss over semaglutide and other GLP-1-only drugs. Orforglipron is pharmacologically closer to oral semaglutide than to injectable tirzepatide (Wharton et al., NEJM 2023).
Phase 3 Results: The ATTAIN and ACHIEVE Programs
Lilly's Phase 3 data for orforglipron paints a picture of a capable but not dominant oral option.
In the ATTAIN-1 trial (obesity), orforglipron 36 mg daily produced approximately 12.4% weight loss (average of 27.3 lbs) at 72 weeks. The ATTAIN-2 trial in adults with type 2 diabetes and obesity showed 10.5% weight loss (22.9 lbs) at 72 weeks with the 36 mg dose. In ATTAIN-MAINTAIN, patients switching from injectable incretins to oral orforglipron maintained their previous weight loss.
The ACHIEVE-3 trial compared orforglipron head-to-head against oral semaglutide in type 2 diabetes. Orforglipron outperformed oral semaglutide on A1C reduction and weight loss across all endpoints. This positions orforglipron as the strongest oral GLP-1 agonist in development.
| Orforglipron Trial | Population | Weight Loss | Duration |
|---|---|---|---|
| ATTAIN-1 (Phase 3) | Obesity | 12.4% | 72 weeks |
| ATTAIN-2 (Phase 3) | T2D + obesity | 10.5% | 72 weeks |
| ACHIEVE-3 (Phase 3) | T2D (vs oral sema) | Superior to oral semaglutide | Published Feb 2026 |
| Phase 2 (48 mg) | Obesity | 14.7% | 36 weeks |
Lilly has filed for global regulatory approval. A US FDA decision for obesity is anticipated in Q2 2026. If approved, orforglipron would be the first non-peptide oral GLP-1 on the market. But even at its best (14.7% in Phase 2 at 48 mg), orforglipron trails injectable tirzepatide by nearly 8 percentage points. For a deeper comparison, see our article on orforglipron vs tirzepatide.
Injectable Tirzepatide: What the Clinical Trials Actually Show
To understand what oral alternatives are competing against, consider the clinical evidence behind injectable tirzepatide. The SURMOUNT trial program represents the most extensive obesity pharmacotherapy dataset ever collected.
SURMOUNT-1: The Landmark Obesity Trial
SURMOUNT-1 enrolled 2,539 adults with obesity (BMI 30+) or overweight (BMI 27+) with at least one weight-related condition. Participants received tirzepatide 5 mg, 10 mg, or 15 mg once weekly for 72 weeks. The results reset expectations for what pharmacotherapy could achieve.
At 72 weeks, the 15 mg dose produced 22.5% mean weight loss. Among patients who completed treatment, 63% lost at least 20% of their body weight, and 36% lost at least 25%. The placebo group lost 3.1%. These numbers rival bariatric surgery outcomes for the first time in pharmaceutical history (Jastreboff et al., NEJM 2022).
For patients tracking their own progress against these benchmarks, our tirzepatide before and after guide provides realistic timeline expectations. Results typically become visible by weeks 8-12, with peak effects at months 9-12.
SURMOUNT-5: Tirzepatide vs Semaglutide Head-to-Head
The SURMOUNT-5 trial directly compared tirzepatide to injectable semaglutide in adults with obesity. At 72 weeks, tirzepatide 15 mg produced 20.2% weight loss compared to 13.7% for semaglutide 2.4 mg. Patients on tirzepatide were significantly more likely to achieve weight loss thresholds of 10%, 15%, 20%, and 25%.
This 6.5 percentage point gap between the two most potent injectable options puts the oral alternatives in perspective. No oral GLP-1, whether semaglutide-based or small-molecule, has come within 3 percentage points of injectable tirzepatide. The dual GLP-1/GIP mechanism appears to confer an advantage that single-receptor oral drugs cannot replicate.
For patients comparing all available incretin options, our guide on retatrutide vs tirzepatide covers the emerging triple-agonist class (GLP-1/GIP/glucagon), which produced even greater weight loss (24.2%) in Phase 2.
Complete Comparison: Every Oral and Injectable GLP-1 Option in 2026

The landscape of incretin-based therapies has expanded rapidly. This table captures every major option available or in late-stage development as of April 2026, sorted by weight loss efficacy.
| Drug | Route | Dose | Weight Loss | Mechanism | Status (Apr 2026) |
|---|---|---|---|---|---|
| Retatrutide | Injection (weekly) | 12 mg | 24.2% (48 wk) | GLP-1/GIP/Glucagon | Phase 3 |
| Tirzepatide (Zepbound) | Injection (weekly) | 15 mg | 22.5% (72 wk) | GLP-1/GIP | FDA approved |
| Semaglutide (Wegovy inj.) | Injection (weekly) | 2.4 mg | 14.9% (68 wk) | GLP-1 | FDA approved |
| Semaglutide (Wegovy pill) | Oral (daily) | 25 mg | 16.6% (64 wk) | GLP-1 | FDA approved |
| Orforglipron | Oral (daily) | 36 mg | 12.4% (72 wk) | GLP-1 | Phase 3 / Filing |
| Semaglutide (Rybelsus) | Oral (daily) | 14 mg | ~5% (26 wk) | GLP-1 | FDA approved |
| Oral tirzepatide | Sublingual | Varies | Unproven | GLP-1/GIP | Not FDA-approved |
Three patterns emerge from this data. First, injectable formulations consistently outperform oral ones because of superior bioavailability. Second, dual- and triple-agonist drugs (tirzepatide, retatrutide) outperform single-receptor drugs regardless of route. Third, the most effective oral option (Wegovy pill at 16.6%) still falls short of injectable tirzepatide by nearly 6 percentage points.
For patients prioritizing maximum weight loss, injectable tirzepatide remains the standard. For those who cannot or will not inject, the Wegovy pill now offers a legitimate oral alternative with meaningful efficacy. Use our tirzepatide dosage calculator to plan an injectable protocol, or explore our semaglutide dosage chart for the oral semaglutide pathway.
Could an Oral Tirzepatide Equivalent Ever Exist?
The barriers are real but not necessarily permanent. Several technologies could theoretically enable an oral drug with tirzepatide-like dual GLP-1/GIP activation. None are close to market.
Small-Molecule Dual Agonists
The most plausible path to an "oral tirzepatide" is not reformulating the peptide but discovering an entirely new small molecule that activates both GLP-1 and GIP receptors. Orforglipron proved that small molecules can potently activate GLP-1 receptors. Extending that approach to GIP would require a molecule shaped to fit both receptor binding pockets, a significant medicinal chemistry challenge.
No oral small-molecule GIP agonist has reached clinical trials as of 2026. Several pharmaceutical companies are working on this problem, but the timeline from discovery to approval typically spans 10-15 years. A realistic estimate for an oral dual agonist matching tirzepatide's efficacy is 2030 at the earliest, and that assumes no setbacks in development.
Ingestible Injection Devices
Devices like SOMA (Self-Orienting Millimeter-Scale Applicator) blur the line between pills and injections. You swallow a capsule, but inside your stomach, a spring-loaded needle made of compressed drug deploys against the stomach wall and delivers the peptide locally. A study in swine showed SOMA-delivered semaglutide achieved plasma levels comparable to subcutaneous injection (Abramson et al., Science 2019).
Adapting SOMA or similar platforms for tirzepatide is theoretically feasible. The device bypasses acid degradation, the permeability barrier, and first-pass metabolism simultaneously. However, these devices remain in early-stage research. The engineering challenges of reliably orienting inside the stomach, deploying without causing perforation, and delivering precise doses across varied gastric environments are substantial. Clinical availability is years away.
Next-Generation Permeation Enhancers
Researchers are developing enhancers beyond SNAC that temporarily open intestinal tight junctions more effectively. Cell-penetrating peptide conjugates and sodium caprate (C10) have shown promise in preclinical models, potentially increasing bioavailability of large peptides from below 1% to 3-5%. Even that improvement would still require extremely large oral doses, but it narrows the gap (Maher et al., 2019).
The bottom line: an oral tirzepatide equivalent will eventually arrive, but not soon. Patients delaying treatment while waiting for that breakthrough sacrifice years of weight loss and metabolic improvement available through injectable tirzepatide right now.
Should You Start Injectable Tirzepatide Now or Wait for a Pill?
This is the practical question beneath the science. The answer depends on why you prefer oral dosing and how much weight loss you are willing to forgo.
If Needle Fear Is the Barrier
Needle phobia affects 20-25% of adults and is the most common reason patients avoid injectable medications (McLenon & Rogers, 2019). Modern autoinjectors like the Mounjaro KwikPen use a hidden needle. You press the pen against your skin and press a button. The injection takes 10 seconds. Pain is typically less than a blood glucose finger stick.
Our guide on how to inject tirzepatide covers every step. Most patients report their anxiety before the first injection was far worse than the experience itself. For optimal injection site rotation, see our guide on best injection sites for tirzepatide. Starting now ensures you do not lose years of benefit while waiting for oral alternatives.
If Convenience Is the Priority
A weekly injection takes 2 minutes. The Wegovy pill requires daily dosing on an empty stomach with a 30-minute fast every morning. Adherence data consistently shows once-weekly injectables achieve 15-20% higher compliance than daily oral medications.
For most patients, a weekly injection is actually more convenient than a daily pill with dietary restrictions. The practical inconvenience of injectable tirzepatide is lower than expected. See best time to take tirzepatide for scheduling strategies, and our guide on what to eat on tirzepatide for dietary optimization during treatment.
If Cost Is the Primary Concern
Injectable tirzepatide costs approximately $1,000/month without insurance. The Wegovy pill launched at $149/month. That price difference is substantial and may make oral semaglutide the better economic choice for patients paying out of pocket, even though it produces less weight loss.
For cost strategies with injectable tirzepatide, see our guide on tirzepatide cost with insurance. Our peptide cost calculator helps compare total treatment costs across different peptides and formulations. For patients after reaching their goal, see our guide on tirzepatide maintenance dose after weight loss.
Managing Injectable Tirzepatide Treatment
If you decide to start injectable tirzepatide rather than wait, several practical resources help you manage treatment effectively.
The standard titration begins at 2.5 mg weekly for 4 weeks, increasing by 2.5 mg every 4 weeks to a target of 10-15 mg. Our compound tirzepatide dosage chart provides the complete schedule with unit conversions for different vial concentrations. Use the tirzepatide dosage calculator to plan your specific titration.
Common side effects during titration include nausea, diarrhea, constipation, headaches, and fatigue. Most resolve within 4-6 weeks at a stable dose. For specific side effect management, see our guides on tirzepatide and constipation, headaches, and fatigue.
Understanding how long tirzepatide takes to work sets realistic expectations: meaningful weight loss typically appears by weeks 8-12, with peak effects at months 9-12. If results stall, our troubleshooting guide covers why you may not be losing weight on tirzepatide.
Important Warnings
Tirzepatide is a prescription medication. Do not use it without medical supervision. It is approved for type 2 diabetes (Mounjaro) and chronic weight management in adults with obesity or overweight with at least one weight-related condition (Zepbound). It is not approved for cosmetic weight loss in normal-weight individuals.
Do not combine tirzepatide with other GLP-1 receptor agonists (semaglutide, liraglutide, dulaglutide). The combination increases the risk of severe gastrointestinal events including gastroparesis and bowel obstruction.
Individuals with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 should not use tirzepatide. The drug carries a boxed warning for thyroid C-cell tumors based on rodent studies.
Unregulated "oral tirzepatide" products sold online without a prescription are not FDA-evaluated. Their composition, potency, and safety are unknown. Purchasing prescription peptide products from unverified sources carries risks of contamination, misdosing, and counterfeit ingredients. For verified sourcing information, see our guide on where to buy tirzepatide.
This article is educational content for research purposes. Consult a healthcare provider before starting, adjusting, or discontinuing any medication.
Frequently Asked Questions
Does oral tirzepatide actually produce weight loss?
No FDA-approved oral tirzepatide exists. Compounded sublingual formulations have estimated bioavailability below 2-3%, meaning the vast majority of each dose is destroyed before reaching the bloodstream. Some patients reported weight loss with sublingual drops, but results were highly variable and no clinical trial has validated this route. Injectable tirzepatide at 15 mg produces 22.5% weight loss at 72 weeks with ~80% bioavailability. The gap between these delivery methods is enormous.
Is the Wegovy pill a good alternative to injectable tirzepatide?
The Wegovy pill (oral semaglutide 25 mg) was FDA-approved in December 2025 and achieves 16.6% weight loss at 64 weeks. That is meaningful but falls short of injectable tirzepatide's 22.5% at 72 weeks. The Wegovy pill costs approximately $149/month versus $1,000/month for tirzepatide. For patients prioritizing affordability and needle avoidance over maximum weight loss, the Wegovy pill is a legitimate option. See our best peptides for weight loss guide for a full comparison.
What is orforglipron and when will it be available?
Orforglipron is Eli Lilly's oral small-molecule GLP-1 receptor agonist. It is not tirzepatide in a pill. Unlike tirzepatide, it activates only the GLP-1 receptor, not GIP. Phase 3 data showed 12.4% weight loss at 36 mg daily over 72 weeks. Lilly filed for FDA approval and a US decision is expected in Q2 2026. See our detailed comparison at orforglipron vs tirzepatide.
Why can't Eli Lilly just put tirzepatide into a pill?
Tirzepatide is a 39-amino-acid peptide weighing 4,810 daltons. Stomach acid denatures it within minutes, intestinal enzymes chop it apart, and its size prevents absorption across the intestinal wall. Oral bioavailability would be below 1%, requiring a 1,500+ mg oral dose to match a 15 mg injection. No absorption enhancer has been developed for tirzepatide. Lilly's oral strategy instead focuses on orforglipron, an entirely different molecule. For injection guidance, see how to inject tirzepatide.
Are sublingual tirzepatide drops safe?
Sublingual tirzepatide drops from compounding pharmacies lack FDA evaluation for safety or efficacy. Quality varies between batches, and no standardized bioavailability testing exists for these products. The FDA's compounding exemption for tirzepatide ended in early 2025, making current sublingual products legally questionable. For safety information on compounded products, see our guide on compound tirzepatide safety.
How does injectable tirzepatide compare to all oral GLP-1 options?
Injectable tirzepatide 15 mg produces 22.5% weight loss at 72 weeks. The best oral option, the Wegovy pill (25 mg oral semaglutide), achieves 16.6% at 64 weeks. Orforglipron achieves 12.4% at 72 weeks. Rybelsus (14 mg oral semaglutide) achieves about 5% at 26 weeks. No oral drug replicates tirzepatide's dual GLP-1/GIP mechanism. Use our tirzepatide dosage calculator to plan an injectable protocol.
Will there ever be an oral drug as effective as injectable tirzepatide?
Possibly, but not soon. An oral tirzepatide equivalent would require either a small-molecule dual GLP-1/GIP agonist (none in clinical trials as of 2026) or an ingestible injection device (early research stage). Realistic timeline: 2030 at the earliest. Waiting means forgoing years of weight loss available now through injectable tirzepatide. For emerging alternatives, see our comparison of retatrutide vs tirzepatide.
Is once-weekly injection really better than a daily pill?
For most patients, yes. A weekly tirzepatide injection takes 2 minutes. Daily oral GLP-1 medications require empty-stomach dosing with a 30-minute fast each morning. Adherence studies show once-weekly injectables achieve 15-20% higher compliance than daily pills. The autoinjector pen uses a hidden needle and causes minimal pain. See our guide on best time to take tirzepatide for scheduling strategies.
The Bottom Line
Oral tirzepatide does not work as a proven, reliable treatment. No FDA-approved oral form exists. Compounded sublingual drops delivered inconsistent and unvalidated results, and the legal basis for compounding tirzepatide ended in early 2025. The molecule's size, susceptibility to digestive enzymes, and poor intestinal absorption make oral delivery a fundamental chemistry problem, not a manufacturing shortcut away from being solved.
The oral GLP-1 landscape offers legitimate alternatives. The Wegovy pill (oral semaglutide 25 mg, approved December 2025) achieves 16.6% weight loss at a fraction of tirzepatide's cost. Orforglipron may reach FDA approval in 2026 with 12.4% weight loss. But neither replicates tirzepatide's dual GLP-1/GIP mechanism, and neither matches its 22.5% weight loss. An oral drug with equivalent dual-agonist efficacy is realistically a 2030+ prospect.
The practical takeaway: do not delay effective treatment while chasing unproven oral formulations. Injectable tirzepatide is the most potent weight loss medication available. The autoinjector is simple, nearly painless, and takes 2 minutes per week. Use our tirzepatide dosage calculator to plan your titration. For injection guidance, see how to inject tirzepatide. For real-world results, see tirzepatide before and after.
Not sure which peptide is right for your goals? Take our peptide quiz for a personalized recommendation based on your health objectives, experience level, and preferences.
Related articles: - Does Tirzepatide Come in Pill Form? - oral delivery barriers and pipeline analysis - How to Inject Tirzepatide - step-by-step injection guide - Tirzepatide Before and After - real results and timeline expectations - Compound Tirzepatide Dosage Chart - complete titration schedule - Tirzepatide Cost With Insurance - coverage strategies and savings - Semaglutide to Tirzepatide Switching - dose equivalence guide - Retatrutide vs Tirzepatide - triple vs dual agonist comparison - Best Peptides for Weight Loss - comprehensive weight loss peptide guide - Where to Buy Tirzepatide - verified sourcing options - Orforglipron vs Tirzepatide - oral vs injectable GLP-1 comparison
Explore all peptide profiles and tools at PeptidesExplorer.
Related Articles
Tirzepatide Pill Form: Status 2026
Tirzepatide is injection-only in 2026. Learn about Eli Lilly's oral GLP-1 pipeline, orforglipron, oral semaglutide comparison, and the timeline.
Does Compounded Tirzepatide Work? Real Data
Does compounded tirzepatide work? Review SURMOUNT data, salt form differences (base, acetate, sodium), potency gaps, and FDA status in 2026.
Does Tirzepatide Help With Inflammation?
Does tirzepatide help with inflammation? Yes: CRP drops 32.9%, IL-6 drops 17.8% in trials. SURMOUNT data and adipose tissue mechanisms.
Where to Buy Tirzepatide: 5 Legal Sources (2026)
Where to buy tirzepatide legally in 2026: Zepbound via LillyDirect from $299/mo, telehealth options, insurance paths, and red flags to avoid.