Tirzepatide produces an average of 22.5% body weight loss at the 15 mg dose over 72 weeks. For a person starting at 250 pounds, that is roughly 56 pounds. These numbers come from SURMOUNT-1, a randomized trial of 2,539 adults published in the New England Journal of Medicine (Jastreboff et al., NEJM 2022).
Real-world results are lower. An Optum study of 239 patients found 11.9% average weight loss at 6 months, roughly 28 pounds from a typical starting weight. The gap exists because real-world patients titrate more slowly, stay on lower doses longer, and face cost-driven interruptions that clinical trial participants do not experience.
| Quick Reference | Details |
|---|---|
| Drug | Tirzepatide (Mounjaro, Zepbound) |
| Mechanism | Dual agonist: GLP-1 + GIP |
| Trial weight loss (15 mg, 72 wks) | 22.5% (52 lbs from 231 lb baseline) |
| Real-world weight loss (6 mo) | 11.9% (28 lbs) |
| Time to visible changes | 8-12 weeks |
| Plateau timing | 24-36 weeks (varies by BMI) |
| Weight regained after stopping | 82.5% regain 25%+ within 1 year |
| Side effects (15 mg) | Nausea 31%, diarrhea 23%, constipation 12% |
| FDA approved | Yes (Mounjaro 2022, Zepbound 2023) |
This article presents week-by-week timelines, dose-by-dose results, real-world versus clinical trial comparisons, and the factors that predict how much weight you will lose. All data comes from published SURMOUNT trials and real-world studies.
This is educational content. Consult a healthcare provider before starting any medication.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Clinical Trial Results by Dose
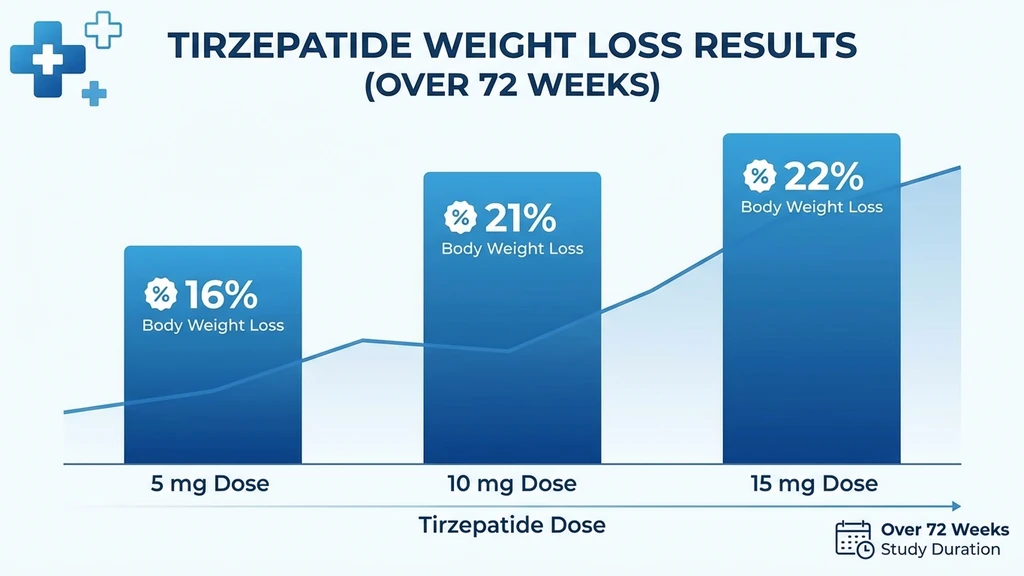
The SURMOUNT-1 trial tested three tirzepatide doses against placebo in 2,539 adults with obesity (BMI 30+) or overweight with comorbidities (BMI 27+) and no type 2 diabetes. Results at 72 weeks:
| Metric | 5 mg | 10 mg | 15 mg | Placebo |
|---|---|---|---|---|
| Body weight loss | 16.0% | 21.4% | 22.5% | 2.4% |
| Pounds lost (from ~231 lbs) | 35 lbs | 49 lbs | 52 lbs | 5 lbs |
| Achieved 5%+ loss | 89% | 96% | 96% | 28% |
| Achieved 10%+ loss | 73% | 86% | 90% | 14% |
| Achieved 15%+ loss | 50% | 74% | 78% | 6% |
| Achieved 20%+ loss | 32% | 55% | 63% | 1.3% |
| Achieved 25%+ loss | 16.5% | 35% | 39.7% | 0.3% |
Source: Jastreboff et al., NEJM 2022
Two patterns stand out. First, the jump from 5 mg to 10 mg is larger than the jump from 10 mg to 15 mg (5.4 percentage points versus 1.1). This means most of the additional weight loss benefit comes from reaching 10 mg. Second, even the lowest dose (5 mg) outperforms semaglutide's maximum approved dose in head-to-head data.
For detailed dosing information, see our tirzepatide dosage chart and use our tirzepatide dosage calculator. For compound formulations, refer to the compound tirzepatide dosage chart.
Week-by-Week Timeline
The following timeline is based on combined data from SURMOUNT trials and real-world patient reports. Individual results vary based on dose, diet, exercise, starting weight, and metabolic health.
| Timeframe | Expected Loss | Dose Phase | What to Expect |
|---|---|---|---|
| Week 1-4 | 1-3% (2-7 lbs) | 2.5 mg starter | Appetite reduction begins. Nausea common. Food noise decreases. |
| Week 5-8 | 3-6% (7-14 lbs) | 5 mg | First clinically meaningful loss. Cravings decrease noticeably. |
| Week 9-12 | 6-10% (14-23 lbs) | 7.5 mg | Visible changes: face thinning, clothes loosening. |
| Week 13-16 | 8-12% (18-28 lbs) | 10 mg | Consistent 1-3 lbs/week losses. Others start noticing. |
| Week 17-20 | 10-15% (23-35 lbs) | 12.5 mg | Significant visible transformation. |
| Week 21-24 | 12-16% (28-37 lbs) | 15 mg max | Rate of loss begins decelerating. |
| Week 24-36 | 14-19% (32-44 lbs) | 15 mg | Most patients hit their first plateau. |
| Week 36-52 | 16-21% (37-49 lbs) | 15 mg | Gradual additional loss. Body recomposition begins. |
| Week 52-72 | 16-22.5% (37-52 lbs) | 15 mg | Near maximum. Maintenance phase for most. |
All percentages and pound figures assume a starting weight of approximately 230 pounds. Scale these proportionally for your starting weight.
Timing your dose can influence tolerability. Our guide on the best time to take tirzepatide covers morning versus evening dosing and meal timing strategies. For injection technique, see how to inject tirzepatide.
When Plateaus Hit
A 2025 study by Horn et al. analyzed plateau timing by starting BMI:
| Starting BMI | Plateau Timing |
|---|---|
| BMI 27-30 (overweight) | ~24.3 weeks |
| BMI 30-35 (class I obesity) | ~26.0 weeks |
| BMI 35-40 (class II obesity) | ~36.1 weeks |
| BMI 40+ (class III obesity) | ~36.1 weeks |
Higher starting BMI correlates with later plateaus. This makes sense because there is more excess weight to lose. The plateau does not mean the medication stopped working. It means your body has reached a new energy equilibrium at the current dose. Options include increasing the dose (if not yet at 15 mg), adding structured exercise, or adjusting dietary habits.
For strategies when weight loss stalls, see our guide on why you may not be losing weight on tirzepatide.
What to Expect by Starting Weight
The table below projects expected weight loss at the 15 mg dose over 72 weeks, based on the 22.5% average from SURMOUNT-1. Individual results will vary.
| Starting Weight | Projected Loss (22.5%) | Projected End Weight |
|---|---|---|
| 180 lbs | 40-41 lbs | ~139-140 lbs |
| 200 lbs | 45 lbs | ~155 lbs |
| 220 lbs | 49-50 lbs | ~170-171 lbs |
| 250 lbs | 56-57 lbs | ~193-194 lbs |
| 275 lbs | 62 lbs | ~213 lbs |
| 300 lbs | 67-68 lbs | ~232-233 lbs |
| 350 lbs | 79 lbs | ~271 lbs |
These projections assume 72 weeks at the maximum dose with consistent compliance. Real-world results run 10 to 30% lower than clinical trial numbers. A conservative estimate uses 15 to 18% loss rather than 22.5%.
Body Composition: Fat vs. Muscle Loss
Weight loss medications reduce both fat and lean mass. The critical question is the ratio. A DXA substudy of tirzepatide patients measured body composition changes directly (Look et al., Diabetes Obes Metab 2025).
Tirzepatide patients lost 15.9 kg of fat mass and 5.6 kg of lean mass. Placebo patients lost 3.6 kg of fat mass and 1.2 kg of lean mass. The ratio in both groups was approximately 75% fat and 25% lean mass, which matches the composition of natural weight loss through caloric restriction alone.
This matters because lean mass loss reduces resting metabolic rate. Every pound of muscle burns roughly 6 calories per day at rest. Losing 12 pounds of lean mass (5.6 kg) reduces daily calorie burn by approximately 72 calories per day. Over a year, that deficit compounds.
Two strategies preserve lean mass during tirzepatide treatment. First, resistance training at least two to three sessions per week. Second, adequate protein intake of 0.7 to 1 gram per pound of lean body mass daily. A 200-pound person with 140 pounds of lean mass should target 98 to 140 grams of protein per day. For more on this mechanism, see our article on does tirzepatide burn fat.
SURMOUNT Trial Comparisons
Eli Lilly has run six major tirzepatide obesity trials. Each adds important context for understanding before-and-after results.
| Trial | Population | Duration | Key Result | Source |
|---|---|---|---|---|
| SURMOUNT-1 | 2,539 adults, no T2D | 72 weeks | 22.5% loss at 15 mg | NEJM 2022 |
| SURMOUNT-2 | 938 adults with T2D | 72 weeks | 15.7% loss at 15 mg | Lilly PR |
| SURMOUNT-3 | Lifestyle + drug | 84 weeks | 26.6% total loss | Nature Medicine 2023 |
| SURMOUNT-4 | Discontinuation study | 88 weeks | Regained to -9.9% after stopping | JAMA 2024 |
| SURMOUNT-5 | Head-to-head vs semaglutide | 72 weeks | 20.2% tirz vs 13.7% sema | NEJM 2025 |
| SURMOUNT-OSA | Obstructive sleep apnea | 52 weeks | AHI reduced ~25-30 events/hr; 51.5% disease resolution | NEJM 2024 |
Type 2 Diabetes Reduces Results by ~30%
SURMOUNT-2 tested tirzepatide in patients with type 2 diabetes. The 15 mg dose produced 15.7% weight loss, compared to 22.5% in the non-diabetic SURMOUNT-1 population. The approximately 30% reduction in weight loss for diabetic patients is consistent across all GLP-1 drug trials.
The reason: insulin resistance and diabetes medications (especially insulin and sulfonylureas) promote weight retention. Patients with T2D should set expectations accordingly. A realistic goal is 12 to 16% weight loss rather than 20%+.
Lifestyle Intervention Amplifies Results
SURMOUNT-3 combined tirzepatide with an intensive lifestyle intervention (calorie-restricted diet and exercise program) during a 12-week lead-in period. Total weight loss reached 26.6%, the highest of any SURMOUNT trial.
The implication: tirzepatide is not a replacement for diet and exercise. It is a multiplier. Patients who combine medication with structured dietary changes and regular physical activity consistently achieve better results than medication alone.
Stopping Means Regaining
SURMOUNT-4 tested what happens when patients stop tirzepatide. After 36 weeks on drug (achieving significant weight loss), participants were randomized to either continue or switch to placebo for another 52 weeks.
Results: patients who continued lost a total of 25.3% body weight. Patients who stopped regained weight, ending at just 9.9% below their original weight. That means they regained roughly 60% of the weight they had lost. 82.5% of patients who stopped treatment regained at least 25% of their lost weight within one year.
This is the most important before-and-after data point. Tirzepatide is not a short-term treatment. Stopping the drug leads to significant weight regain for most patients. Long-term treatment appears necessary to maintain results. See our guide on tirzepatide maintenance dosing after weight loss.
47% More Weight Loss Than Semaglutide
SURMOUNT-5 was the first head-to-head trial comparing tirzepatide to semaglutide. At 72 weeks, tirzepatide produced 20.2% weight loss versus 13.7% for semaglutide. That is 47% more weight loss with tirzepatide.
For a detailed comparison, see our semaglutide before and after guide. If you are considering switching between the two drugs, our semaglutide to tirzepatide conversion guide covers the transition process.
First Drug Approved for Obstructive Sleep Apnea
SURMOUNT-OSA tested tirzepatide in adults with moderate-to-severe obstructive sleep apnea and obesity. Published in the New England Journal of Medicine in 2024, this trial demonstrated that weight loss from tirzepatide produces measurable improvements in sleep-disordered breathing.
Participants treated with tirzepatide experienced a reduction in the apnea-hypopnea index (AHI) of approximately 25 to 30 events per hour compared to placebo. 51.5% of participants achieved complete disease resolution, defined as AHI below 5 events per hour. Based on these results, Zepbound became the first medication ever FDA-approved for moderate-to-severe obstructive sleep apnea in 2024.
This trial matters because it proves weight loss from tirzepatide produces health outcomes beyond the scale. Sleep apnea contributes to cardiovascular disease, daytime fatigue, and metabolic dysfunction. Resolving it through pharmacologic weight loss represents a new treatment paradigm.
Tirzepatide vs. Semaglutide: Side-by-Side Results
Two data sources now allow direct comparison between tirzepatide and semaglutide: the SURMOUNT-5 clinical trial and the SHAPE real-world study.
| Metric | Tirzepatide 15 mg | Semaglutide 2.4 mg | Source |
|---|---|---|---|
| Weight loss (72 weeks) | 20.2% | 13.7% | SURMOUNT-5, NEJM 2025 (PMID: 40353578) |
| Waist circumference | -18.4 cm | -13.0 cm | SURMOUNT-5 |
| GI discontinuation rate | 2.7% | 5.6% | SURMOUNT-5 |
| Achieved 25%+ loss | 31.6% | 16.1% | SURMOUNT-5 |
| Real-world weight loss (1 year) | -16.5% | -14.1% | SHAPE 2025 (PMID: 40875186) |
SURMOUNT-5 randomized 751 adults to tirzepatide 15 mg or semaglutide 2.4 mg for 72 weeks (PMID: 40353578). Tirzepatide produced 47% more weight loss, 41% greater waist circumference reduction, and nearly double the proportion of patients achieving 25%+ weight loss. Notably, tirzepatide had a lower GI-related discontinuation rate (2.7% vs. 5.6%), suggesting better tolerability at maximum doses.
The SHAPE study (PMID: 40875186) confirmed these findings in a real-world setting. At one year, tirzepatide patients lost 16.5% of body weight versus 14.1% for semaglutide patients. The gap narrows in real-world practice, likely because many patients do not reach the maximum dose of either drug.
The mechanism behind tirzepatide's advantage is its dual receptor activity. Tirzepatide activates both GIP and GLP-1 receptors, while semaglutide targets GLP-1 alone. GIP receptor activation enhances fat oxidation, improves insulin sensitivity, and may reduce nausea relative to pure GLP-1 agonism. For detailed guidance on switching between these medications, see our semaglutide to tirzepatide conversion guide.
Real-World vs. Clinical Trial Results
Clinical trials represent best-case scenarios. Participants receive free medication, regular monitoring, and consistent dosing. Real-world patients face cost barriers, insurance complications, medication shortages, and slower titration schedules.
An Optum real-world study of 239 tirzepatide patients found:
| Metric | Real-World (6 months) | SURMOUNT-1 (72 weeks) |
|---|---|---|
| Average weight loss | 11.9% (27.7 lbs) | 22.5% (52 lbs) |
| Achieved 5%+ loss | 85.8% | 96% |
| Achieved 10%+ loss | 61.5% | 90% |
| Achieved 20%+ loss | 10.5% | 63% |
| Typical dose at assessment | <10 mg (74.2% of patients) | 15 mg |
The 10-percentage-point gap between real-world and clinical trial results has three main explanations. First, 74.2% of real-world patients were still on doses below 10 mg at the 6-month mark. Clinical trial participants follow strict titration schedules that reach 15 mg faster. Second, real-world patients experience medication gaps due to cost, shortage, and insurance issues. Third, the Optum study measured at 6 months, while SURMOUNT-1 measured at 72 weeks (16.5 months).
The SHAPE study (2025) provides additional real-world context at the one-year mark. In a large retrospective cohort, tirzepatide patients achieved 16.5% weight loss versus 14.1% for semaglutide patients (PMID: 40875186). This narrows the gap between clinical trial and real-world results, suggesting that longer treatment duration in practice brings outcomes closer to trial benchmarks.
Three-year extension data from SURMOUNT-1 further supports durability. Most patients who remained on tirzepatide regained only 5% or less from their lowest weight (nadir). This suggests that continued treatment maintains the vast majority of weight loss over multiple years.
The practical takeaway: expect 10 to 18% weight loss in your first year of real-world use, not the 22.5% headline number from trials. With continued treatment and adequate titration, results improve toward the 16 to 20% range by year two.
Side Effects During the Weight Loss Journey
Side effects follow a predictable pattern tied to dose escalation.
| Phase | Common Side Effects | Severity |
|---|---|---|
| Weeks 1-4 (2.5 mg) | Mild nausea, reduced appetite, mild GI discomfort | Mild |
| Weeks 5-12 (5-7.5 mg) | Nausea peaks during dose increases, diarrhea, constipation | Mild to Moderate |
| Weeks 13-20 (10-12.5 mg) | GI often improves, fatigue from caloric deficit | Moderate |
| Weeks 21+ (15 mg) | Most GI resolved, potential hair thinning | Mild |
SURMOUNT-1 side effect rates at 15 mg: nausea 31%, diarrhea 23%, constipation 11.7%, vomiting 12.2%. Only 4 to 7% of participants discontinued due to side effects. The key pattern: GI side effects occur primarily during dose escalation, not at stable doses.
Hair thinning (telogen effluvium) can occur at any point during rapid weight loss. It is caused by the caloric deficit, not the medication directly. Hair typically regrows after weight stabilizes. For more on this topic, see our guide on does tirzepatide cause hair loss.
For managing specific side effects, see our guides on tirzepatide and diarrhea, tirzepatide constipation, tirzepatide headaches, and tirzepatide fatigue.
Factors That Predict Your Results
Not everyone loses the same amount of weight. Research identifies several factors that influence tirzepatide outcomes.
Factors That Improve Results
- Higher dose: 15 mg produces 22.5% loss versus 16% at 5 mg. Reaching the maximum tolerated dose matters.
- No type 2 diabetes: Non-diabetic patients lose roughly 30% more weight than diabetic patients.
- Combining with lifestyle changes: Diet and exercise alongside medication produced 26.6% total loss (SURMOUNT-3).
- Higher starting BMI: More absolute weight is lost at higher starting weights.
- Consistent weekly dosing: Gaps in treatment reduce cumulative results.
- Adequate protein intake: 0.7 to 1 gram per pound of lean body mass helps preserve muscle.
- Resistance training: Maintains lean mass during weight loss, improving body composition.
Factors That Reduce Results
- Type 2 diabetes: Insulin resistance limits weight loss to approximately 15-16% at the maximum dose.
- Slower titration: Staying on lower doses longer means less total loss during the same timeframe.
- Medication gaps: Cost, shortages, or insurance issues that interrupt dosing.
- Liquid calories: Alcohol and sugary beverages bypass the appetite suppression mechanism.
- Concurrent medications: Insulin, sulfonylureas, and some antidepressants promote weight retention.
- Inadequate protein: Accelerates lean mass loss, reducing metabolic rate.
For guidance on what happens if weight loss stalls, see our detailed guide on why you may not be losing weight on tirzepatide and our tirzepatide maintenance dose guide.
Frequently Asked Questions
How much weight will I lose on tirzepatide?
Clinical trials show an average of 22.5% body weight loss at the 15 mg dose over 72 weeks. For a 250-pound person, that is about 56 pounds. Real-world results average 10 to 18% in the first year due to slower dose titration, medication gaps, and cost barriers. Type 2 diabetes reduces results by roughly 30%.
How long does it take to see results on tirzepatide?
Most patients notice appetite suppression within the first 1-2 weeks. Measurable weight loss (2-7 pounds) typically occurs in the first month on the starter 2.5 mg dose. Visible changes that others notice usually appear around weeks 9-12, when cumulative loss reaches 6-10% of body weight.
What is the tirzepatide weight loss timeline?
Weeks 1-4: 1-3% loss (2.5 mg dose). Weeks 5-8: 3-6% (5 mg). Weeks 9-12: 6-10% (7.5 mg, visible changes begin). Weeks 13-20: 10-15% (10-12.5 mg). Weeks 21-36: 12-19% (15 mg). Weeks 36-72: 16-22.5% (near maximum). Most plateaus occur between weeks 24 and 36.
Is tirzepatide better than semaglutide for weight loss?
Yes. SURMOUNT-5, the first head-to-head trial, showed tirzepatide produces 20.2% weight loss versus 13.7% for semaglutide at 72 weeks. That is 47% more weight loss. Tirzepatide targets two receptors (GLP-1 and GIP) versus semaglutide's one (GLP-1). Both are FDA-approved.
Do you gain weight back after stopping tirzepatide?
Yes. SURMOUNT-4 found that 82.5% of patients who stopped tirzepatide regained at least 25% of their lost weight within one year. Patients who stopped ended at only 9.9% below their original weight, versus 25.3% for those who continued. Long-term treatment appears necessary to maintain results.
What are the side effects of tirzepatide?
The most common side effects at the 15 mg dose are nausea (31%), diarrhea (23%), constipation (12%), and vomiting (12%). These primarily occur during dose escalation and improve at stable doses. Only 4-7% of trial participants stopped treatment due to side effects. Hair thinning can occur during rapid weight loss but typically reverses.
When do tirzepatide side effects go away?
GI side effects (nausea, diarrhea, constipation) typically improve within 2-4 weeks at each dose level. They peak during dose escalation and significantly decrease once you stabilize at your target dose. Most patients report manageable or no GI symptoms after the first 12-16 weeks of titration.
What is the maximum weight loss on tirzepatide?
SURMOUNT-3 recorded 26.6% total body weight loss when tirzepatide was combined with an intensive lifestyle intervention. In standard treatment (SURMOUNT-1), 39.7% of patients on 15 mg lost more than 25% of their body weight. Individual results vary, with some patients exceeding 30% loss.
Does tirzepatide help with sleep apnea?
Yes. In SURMOUNT-OSA, tirzepatide reduced sleep apnea severity (AHI) by 25 to 30 events per hour compared to placebo, and 51.5% of participants achieved disease resolution. Zepbound became the first medication FDA-approved for moderate-to-severe obstructive sleep apnea in 2024. Weight loss is the primary mechanism driving these improvements.
The Bottom Line
Tirzepatide produces transformative weight loss in clinical trials: 22.5% at the 15 mg dose, 47% more than semaglutide in head-to-head data. Real-world results run lower (10 to 18% in the first year) but remain clinically significant. Visible changes typically appear by weeks 9 to 12, with maximum results reached between weeks 52 and 72.
The critical caveat: stopping tirzepatide leads to significant weight regain. SURMOUNT-4 showed 82.5% of patients regained substantial weight within a year of discontinuation. This is not a short-term treatment.
Use our tirzepatide dosage calculator to understand your prescribed dose, and see our tirzepatide dosage chart for the complete titration schedule. For cost planning, check our tirzepatide cost with insurance guide and peptide cost calculator.
This is educational content based on published clinical trial data. It is not medical advice. Consult a healthcare provider before starting any weight loss medication.
Related Articles
Tirzepatide Men Before & After: Data for Males
Tirzepatide before and after for men: SURMOUNT-1 male subgroup data, testosterone impact, muscle loss risk, body composition timeline. Men-specific protocols.
Peptides Before and After: Real Results by Category
Before and after peptides: realistic timelines by class. GLP-1s, BPC-157, GHK-Cu, CJC-1295, MK-677, hair and fat-loss peptides, with trial data and photo tips.
BPC-157 Dosage Per Body Weight (Chart)
BPC-157 dosage by body weight: 1.6-10 mcg/kg. Full chart for 120-280 lbs, injection vs oral doses, and why most use flat 250-500 mcg.
Hims Weight Loss Reviews 2026: Pricing & Verdict
Hims weight loss reviews 2026: GLP-1s from $149/mo plus $149 membership. Branded Wegovy and Ozempic, Trustpilot 3.5/5. Pricing and red flags.