Your provider mentions retatrutide as the next-generation GLP-1 drug, and you want to know how it stacks up against the tirzepatide prescription already in your fridge. Retatrutide produced 28.7% body weight loss at 68 weeks in Phase 3 trials. Tirzepatide produced 22.5% at 72 weeks. Both are once-weekly injections from Eli Lilly, but retatrutide adds a third hormone receptor (glucagon) on top of the GLP-1 and GIP receptors that tirzepatide already targets.
Tirzepatide is FDA-approved and available now as Mounjaro (type 2 diabetes) and Zepbound (obesity). Retatrutide remains investigational, with Phase 3 trials ongoing and approval expected in late 2027. For a full overview of retatrutide mechanism, trials, and pipeline, see our complete retatrutide guide.
| Feature | Retatrutide | Tirzepatide |
|---|---|---|
| Mechanism | Triple agonist (GLP-1 + GIP + Glucagon) | Dual agonist (GLP-1 + GIP) |
| Brand names | None (investigational) | Mounjaro, Zepbound |
| Max weight loss | 28.7% at 68 weeks | 22.5% at 72 weeks |
| Max dose | 12 mg/week | 15 mg/week |
| FDA approved | No (Phase 3) | Yes (2022/2023) |
| Nausea rate | ~43% | ~31% |
| Liver fat reduction | Up to 82% | ~55% |
| Cost/month | Not available | $299-$1,086 |
Eli Lilly is running a direct head-to-head trial comparing the two drugs (NCT06662383, 800 patients). Results are expected in December 2026. Until then, all comparisons rely on cross-trial data.
This article covers mechanism, clinical trial results, side effects, dosing, liver benefits, cost, and practical guidance for choosing between them. All data comes from published trials and Eli Lilly press releases. Last updated: March 2026, including Phase 3 TRIUMPH-4 results and December 2025 Zepbound pricing.
This is educational content: consult a healthcare provider before starting any medication.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
How Do Retatrutide and Tirzepatide Work?
Your body produces hormones in the gut that control hunger, blood sugar, and how you burn energy. Both drugs are synthetic versions of those hormones, designed to activate specific receptors in the body. The critical difference is how many receptors each drug activates.
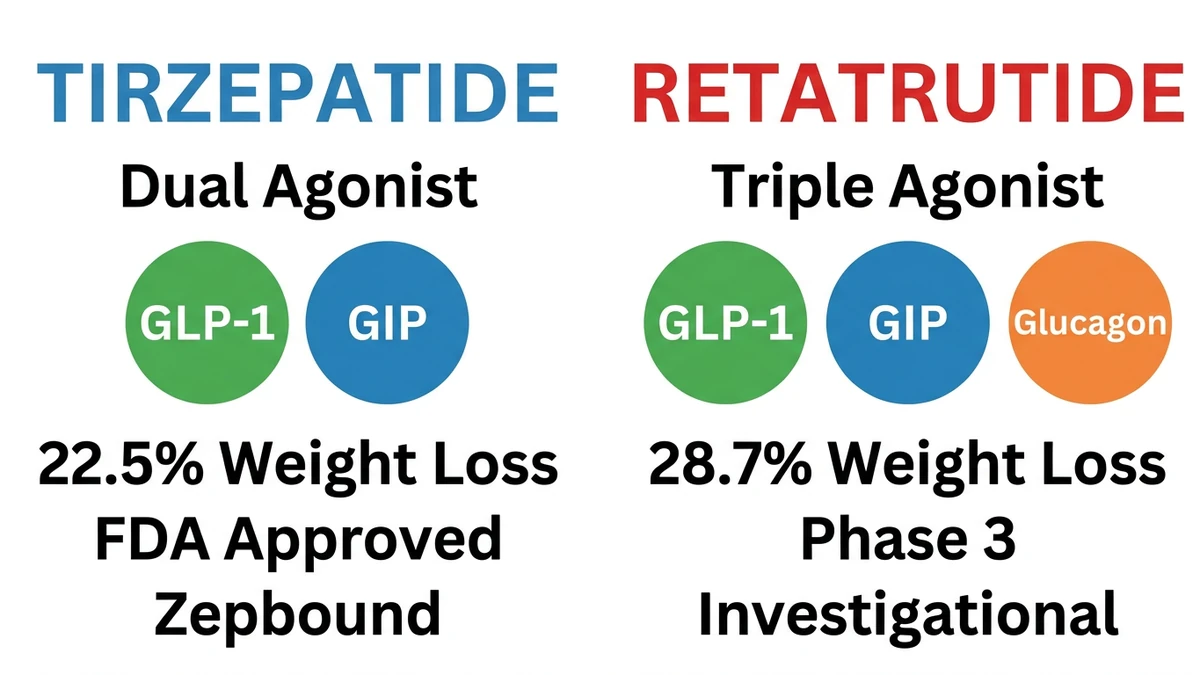
Tirzepatide: GLP-1 + GIP (Dual Agonist)
Tirzepatide activates two receptors. GLP-1 (glucagon-like peptide-1) suppresses appetite and slows digestion, so you feel full longer after eating. GIP (glucose-dependent insulinotropic polypeptide) improves how your body processes blood sugar and works with GLP-1 to make the "I'm full" signal stronger.
In practical terms, people on tirzepatide report eating smaller portions without forcing it. Cravings for high-calorie foods decrease noticeably within the first few weeks.
The dual mechanism explains why tirzepatide outperforms semaglutide, which activates only GLP-1. Adding the second receptor improved weight loss from roughly 15% to 22.5% in clinical trials (Jastreboff et al., NEJM 2022).
Retatrutide: GLP-1 + GIP + Glucagon (Triple Agonist)
Retatrutide (LY3437943) keeps the same two receptors as tirzepatide, then adds a third: glucagon. This is the key difference.
Glucagon tells your liver to burn stored fat for energy and increases your body's heat production (a process called thermogenesis). The result: you burn more calories even while sitting still. Tirzepatide reduces how much you eat. Retatrutide does that and makes your body spend more energy at rest.
One natural concern: glucagon normally raises blood sugar. In retatrutide, the GLP-1 and GIP components offset this effect, keeping blood sugar stable while still capturing glucagon's fat-burning benefits (Jastreboff et al., NEJM 2023).
Why the Glucagon Component Matters
Think of weight loss drugs on a spectrum. Semaglutide (Wegovy) is one lever: eat less. Tirzepatide (Zepbound) is two levers: eat less and process food better. Retatrutide is three levers: eat less, process food better, and burn more energy at rest.
This three-lever approach has two practical consequences. First, greater total weight loss (28.7% vs 22.5%). Second, dramatic liver fat clearance. Your liver stores excess energy as fat, and in many people this buildup causes a condition called MASLD (metabolic dysfunction-associated steatotic liver disease, formerly known as "fatty liver"). An estimated 25% of US adults have it. Retatrutide reduced liver fat by 82% in trials, compared to roughly 55% for tirzepatide (Nature Medicine, 2024).
For someone whose doctor has flagged elevated liver enzymes or fatty liver on an ultrasound, this distinction matters more than the headline weight loss numbers.
How Much Weight Do You Lose on Each Drug?
The weight loss numbers below come from three separate clinical trials, each with different patients and different durations. Comparing results across trials is imperfect, because the participants were not identical. Still, the difference is large enough to be meaningful, and a direct head-to-head comparison is underway (see below).
Tirzepatide: SURMOUNT-1 (72 Weeks)
The SURMOUNT-1 trial enrolled 2,539 adults with obesity (BMI ≥30) or overweight with comorbidities. Participants received tirzepatide or placebo for 72 weeks (Jastreboff et al., NEJM 2022).
| Dose | Weight Loss | ≥20% Weight Loss |
|---|---|---|
| 5 mg | -16.0% | 27% |
| 10 mg | -21.4% | 46% |
| 15 mg | -22.5% | 57% |
| Placebo | -2.4% | 1.3% |
At the 15 mg dose, participants lost an average of 52 lbs (23.6 kg). An important nuance: about three-quarters of the weight lost was fat, while roughly one-quarter was lean mass (muscle, water, bone density). This ratio is typical of any major weight loss, whether from drugs, surgery, or dieting.
Retatrutide: Phase 2 (48 Weeks) and Phase 3 TRIUMPH-4 (68 Weeks)
Retatrutide has two major datasets. The Phase 2 trial enrolled 338 adults for 48 weeks. The Phase 3 TRIUMPH-4 trial enrolled 445 adults with obesity and knee osteoarthritis for 68 weeks.
Phase 2 results at 48 weeks (Jastreboff et al., NEJM 2023):
| Dose | Weight Loss |
|---|---|
| 4 mg | -17.1% |
| 8 mg | -22.8% |
| 12 mg | -24.2% |
Phase 3 TRIUMPH-4 at 68 weeks (Lilly Press Release, Dec 2025):
The 12 mg dose produced 28.7% body weight loss, an average of 71.2 lbs from a 248.5 lb baseline. Weight loss curves had not plateaued, suggesting longer treatment would produce further reduction.
To put 28.7% in perspective: for someone weighing 250 lbs, that means losing about 72 lbs over 16 months. This approaches the results of bariatric surgery, which typically produces 25-35% weight loss.
At 12 mg in Phase 2, every single participant (100%) lost at least 5% of body weight. 83% lost at least 15%. No other obesity medication has achieved a 100% response rate at the 5% threshold.
Three-Way Comparison: Retatrutide vs Tirzepatide vs Semaglutide
Adding semaglutide provides context for the full generational progression of GLP-1 based therapies.
| Metric | Semaglutide 2.4 mg | Tirzepatide 15 mg | Retatrutide 12 mg |
|---|---|---|---|
| Mechanism | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Trial | STEP 1 | SURMOUNT-1 | TRIUMPH-4 |
| Duration | 68 weeks | 72 weeks | 68 weeks |
| Weight loss | -14.9% | -22.5% | -28.7% |
| Absolute loss | ~34 lbs | ~52 lbs | ~71 lbs |
| FDA approved | Yes (Wegovy) | Yes (Zepbound) | No (Phase 3) |
| Brand | Ozempic/Wegovy | Mounjaro/Zepbound | Investigational |
Each generation added a receptor and gained roughly 6-8 percentage points of additional weight loss. A 2025 meta-analysis (a study that pools data from multiple trials to find patterns) confirmed this ranking, with retatrutide showing a mean difference of -16.34 kg vs tirzepatide's -11.82 kg vs placebo (J Endocrine Soc, 2025).
These are cross-trial comparisons with different populations and designs. For definitive answers, the head-to-head trial (see below) will be essential. For a broader overview of weight loss peptides, see our best peptides for weight loss guide.
The Head-to-Head Trial: Retatrutide vs Tirzepatide Direct
Everything above relies on comparing results from separate trials with different patients. That is useful but imperfect. The gold standard in medicine is a head-to-head trial: give one group retatrutide, another group tirzepatide, and see who loses more weight under identical conditions.
Eli Lilly is running exactly that trial.
| Parameter | Detail |
|---|---|
| Trial ID | NCT06662383 |
| Design | Phase 3, randomized, double-blind (neither patients nor doctors know who gets which drug) |
| Enrollment | ~800 patients |
| Duration | 89 weeks (about 20 months) |
| Start date | November 2024 |
| Expected results | December 2026 |
This is the trial that will settle the debate. Same patients, same clinics, same duration, random assignment. The 89-week duration is longer than either drug's previous pivotal trials, which will also show whether retatrutide keeps pulling ahead or whether the gap narrows over time.
The results will shape how doctors prescribe these drugs and how insurance companies decide which to cover. We will update this article when data is published. For detailed retatrutide dosing protocols in the meantime, see our retatrutide dosage guide.
What Are the Side Effects of Each Drug?
Both drugs cause stomach-related side effects, mostly nausea, diarrhea, and vomiting. This happens because GLP-1 slows down digestion, and your stomach needs time to adjust. Retatrutide has higher rates across the board, plus a new safety signal not seen with tirzepatide. For the full Phase 3 safety profile, see our complete retatrutide side effects guide.
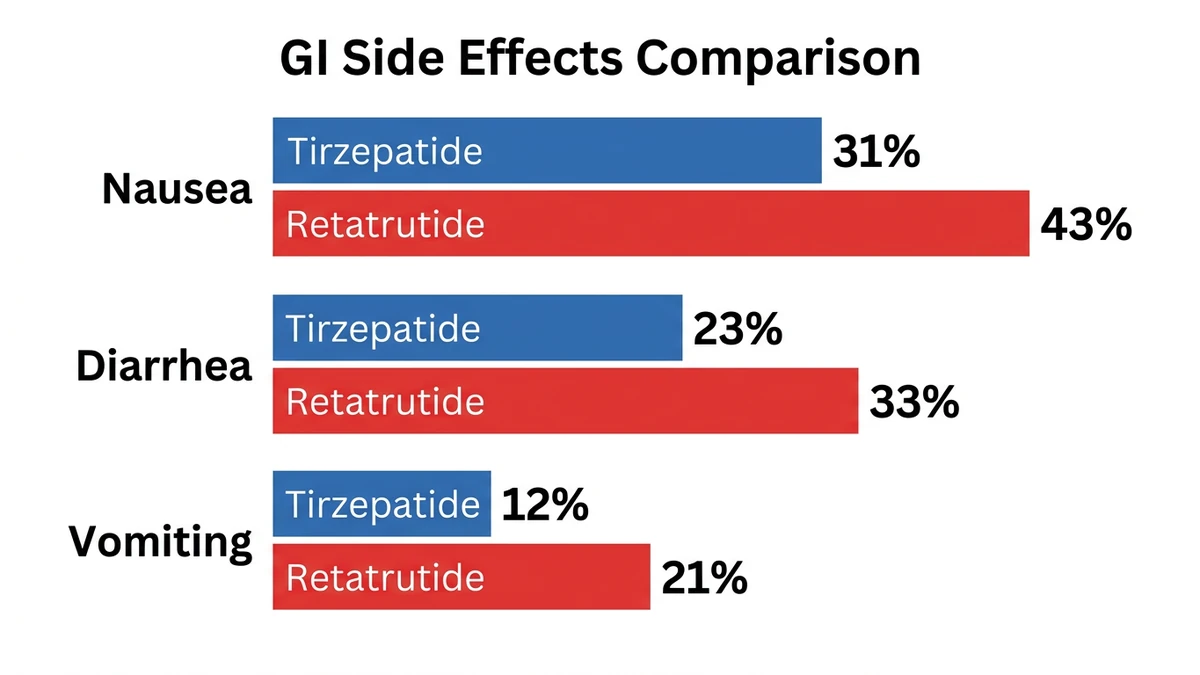
Gastrointestinal Side Effects
| Side Effect | Retatrutide 12 mg (TRIUMPH-4) | Tirzepatide 15 mg (SURMOUNT-1) |
|---|---|---|
| Nausea | 43% | 31% |
| Diarrhea | 33% | 23% |
| Vomiting | 21% | 12% |
| Constipation | ~15% | ~13% |
| Discontinuation due to AE | 18.2% | ~7% |
The pattern is consistent: retatrutide causes more stomach issues. The glucagon component likely adds to the disruption. In practice, this means more nausea during the first weeks of each dose increase.
The good news: for both drugs, side effects are worst during dose escalation and fade after 8-12 weeks at a stable dose. Most people describe the nausea as manageable, similar to mild motion sickness. Eating smaller, lower-fat meals and staying hydrated help significantly. Slow titration (increasing the dose gradually) reduces severity substantially (GI Tolerability Across SURMOUNT 1-4, DOM 2025).
Dysesthesia: A New Signal for Retatrutide
The TRIUMPH-4 Phase 3 trial revealed an unexpected finding: dysesthesia (abnormal touch sensations where normal sensations feel unusual or painful) in 20.9% of participants on 12 mg and 8.8% on 9 mg, compared to 0.7% on placebo (BioSpace, Dec 2025).
A second Phase 3 trial, TRANSCEND-T2D-1 (type 2 diabetes population), showed dramatically lower rates: 4.4% at 12 mg. The TRIUMPH-4 population (obesity + knee osteoarthritis) may have elevated baseline susceptibility due to existing nerve compression from joint inflammation.
Dysesthesia did not lead to significant treatment discontinuations in either trial, and Lilly characterized events as mild. The mechanism is under investigation but likely related to glucagon receptor activation. Tirzepatide has no comparable signal. This is a genuine difference in the safety profiles and warrants monitoring as more TRIUMPH trial data becomes available.
Serious Adverse Events and Contraindications
Serious side effects (hospitalization-level events) were rare with both drugs. Retatrutide Phase 2 reported 4%, matching placebo. Tirzepatide has years of real-world use confirming its overall safety.
Both drugs carry the same FDA boxed warning (the most serious type of warning) about thyroid C-cell tumors found in rodent studies. This has not been confirmed in humans, but as a precaution, neither drug should be used by people with a personal or family history of medullary thyroid cancer (MTC) or a rare genetic condition called MEN2.
Other shared contraindications: history of pancreatitis (inflamed pancreas), pregnancy or breastfeeding, and severe gastroparesis (a condition where the stomach empties too slowly). Tirzepatide's longer track record in real patients provides greater confidence in its safety profile. Retatrutide's profile will sharpen as the remaining seven TRIUMPH trials report over 2026.
Which Drug Is Better for Fatty Liver Disease?
MASLD (metabolic dysfunction-associated steatotic liver disease) is what most people know as "fatty liver." It means excess fat has built up in the liver, which over time can cause inflammation, scarring, and serious liver damage. An estimated 25% of US adults have it, often without knowing. If your doctor has mentioned elevated liver enzymes on a blood test or fatty liver on an ultrasound, this section is particularly relevant.
The glucagon receptor gives retatrutide a pronounced advantage here. A substudy published in Nature Medicine enrolled 98 participants with MASLD for 48 weeks (Nature Medicine, 2024):
| Dose | Liver Fat Reduction | Achieved Normal (<5%) |
|---|---|---|
| 1 mg | -42.9% | Not reported |
| 4 mg | -57.0% | Not reported |
| 8 mg | -81.4% | 89% |
| 12 mg | -82.4% | 93% |
| Placebo | +0.3% | Not reported |
At 12 mg, 93% of participants with fatty liver achieved normal liver fat levels. The drug did not cause liver damage (no hepatotoxicity signals).
Tirzepatide also reduces liver fat, with estimates around 50-60% from available data. Semaglutide achieves roughly 40-50%. Retatrutide's 82% reduction is in a different category entirely. The glucagon component directly tells the liver to burn its stored fat, which is why the gap is so large.
For someone whose primary concern is liver health alongside weight loss, this is the most important distinction between the two drugs. A dedicated MASLD trial is part of the broader TRIUMPH program.
How Are the Dosing Schedules Different?
Both drugs use once-weekly subcutaneous injection with gradual dose escalation. The titration patterns differ.
| Parameter | Tirzepatide | Retatrutide |
|---|---|---|
| Starting dose | 2.5 mg | 2 mg |
| Escalation pattern | +2.5 mg every 4 weeks | Non-uniform (2→4→8→12 mg) |
| Dose steps | 2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg | 2 → 4 → 8 → 12 mg |
| Time to max dose | ~20 weeks | ~12 weeks |
| Max dose | 15 mg | 12 mg |
| Injection frequency | Once weekly | Once weekly |
| Half-life | ~5 days | ~6 days |
Tirzepatide increases by the same amount each time (2.5 mg steps), which makes escalation predictable. Retatrutide doubles at each step (2→4→8) before the final jump to 12 mg. The larger jumps mean more stomach discomfort during dose changes, but the total ramp-up is shorter: 12 weeks vs 20 weeks to reach the full dose.
What does this feel like in practice? Each time the dose increases, expect 3-7 days of reduced appetite and possible nausea, similar to mild food poisoning. This settles as your body adjusts. For people who struggled with nausea on tirzepatide, retatrutide offers a slower alternative protocol (2 mg increments over 20 weeks, detailed in our retatrutide dosage guide).
Use our tirzepatide dosage calculator or retatrutide dosage calculator to plan specific protocols. For a complete dosing reference, see the peptide dosage chart. If you are new to self-injection, our peptide injections complete guide covers technique step by step.
How Much Does Each Drug Cost?
This is the most practical difference right now, and for many people, the only one that matters today. Tirzepatide is available at your pharmacy. Retatrutide is not.
Tirzepatide (Available Now)
Tirzepatide is sold under two brand names. Mounjaro is indicated for type 2 diabetes (FDA approved May 2022). Zepbound is indicated for chronic weight management (FDA approved November 2023).
In December 2025, Eli Lilly reduced Zepbound single-dose vial prices (Lilly Press Release):
| Format | Monthly Cost |
|---|---|
| Zepbound vials (2.5 mg) | $299/month |
| Zepbound vials (5 mg) | $399/month |
| Zepbound pens (list price) | ~$1,086/month |
| With insurance + savings card | As low as $25/month |
| Lilly Direct (without insurance) | $499/month (pens) |
Insurance coverage varies widely. A practical tip: Mounjaro (the diabetes version) often has better insurance coverage than Zepbound (the weight loss version), because insurance companies are more likely to cover medications prescribed for diabetes. Some doctors prescribe Mounjaro off-label for weight loss when patients also have blood sugar issues. Use our peptide cost calculator for estimates.
Retatrutide (Not Yet Available)
Retatrutide has no commercial pricing. It is accessible only through clinical trials. For the complete FDA timeline, trial enrollment steps, and legal access pathways, see our how to get retatrutide guide.
The timeline for availability:
| Milestone | Expected Date |
|---|---|
| TRIUMPH program completion | Throughout 2026 |
| Head-to-head trial results | December 2026 |
| NDA submission to FDA | Late 2026 or early 2027 |
| FDA review period | 10-12 months |
| Potential approval | Late 2027 |
| Commercial availability | Late 2027 to early 2028 |
Lilly will likely price retatrutide at or above Zepbound levels, given the superior efficacy data. The company has not disclosed pricing plans.
Who Should Choose Which?
The choice depends on timeline, medical history, and priorities.
Consider tirzepatide (Zepbound/Mounjaro) if:
- You want a proven, FDA-approved medication available today
- You prefer a drug with years of real-world safety data
- You have insurance coverage for weight management medications
- You have type 2 diabetes (Mounjaro is indicated for T2D)
- You are sensitive to GI side effects (lower nausea rate: 31% vs 43%)
- You want the option of pre-filled pens for easier injection
Consider waiting for retatrutide if:
- You have plateaued on tirzepatide or semaglutide and want the next-generation option
- You have MASLD or fatty liver disease (glucagon component reduces liver fat by 82%)
- Maximum possible weight loss is your primary goal
- You are eligible for and enrolled in a clinical trial
- You are comfortable waiting until 2027-2028 for availability
On switching from tirzepatide to retatrutide:
Once retatrutide is approved, switching will likely be straightforward since both are Lilly products. The standard approach: start retatrutide at the lowest dose and titrate up regardless of your tirzepatide dose. The glucagon receptor component introduces new pharmacology that requires its own titration period. Clinical guidance on switching protocols will emerge post-approval.
The TRIUMPH Trial Program: What to Expect in 2026
Retatrutide's Phase 3 program consists of eight trials spanning multiple indications. TRIUMPH-4 reported in December 2025. Seven more are expected in 2026.
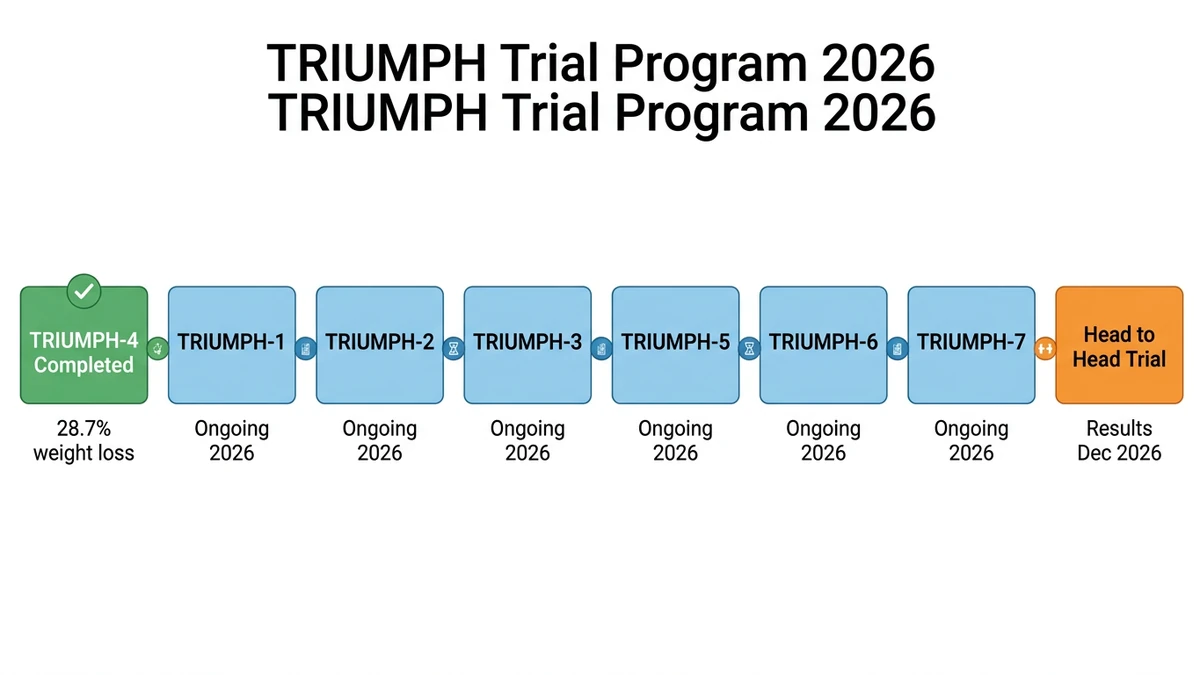
| Trial | Population | Status | Expected |
|---|---|---|---|
| TRIUMPH-1 | Pivotal obesity (registrational) | Ongoing | 2026 |
| TRIUMPH-2 | Pivotal obesity (registrational) | Ongoing | 2026 |
| TRIUMPH-3 | Obesity + cardiovascular disease | Ongoing | 2026 |
| TRIUMPH-4 | Obesity + knee OA | Completed | Dec 2025: 28.7% loss |
| TRIUMPH-5 | Type 2 diabetes | Ongoing | 2026 |
| TRIUMPH-6 | Obstructive sleep apnea | Ongoing | 2026 |
| TRIUMPH-7/8 | Additional indications (likely MASLD, maintenance) | Ongoing | 2026-2027 |
| Head-to-head | Retatrutide vs tirzepatide | Ongoing | Dec 2026 |
TRIUMPH-1 and TRIUMPH-2 are the pivotal trials, the ones Lilly will submit to the FDA to get retatrutide approved. Their results are the most important for the approval timeline. The full program design was published in Diabetes Obesity and Metabolism in early 2026 (Giblin et al., DOM 2026).
A 4 mg maintenance dose is also being studied alongside 9 mg and 12 mg. If it works well enough, this would give doctors a lower-dose option for patients who respond without needing the maximum, similar to how many tirzepatide patients do well on 10 mg without going to 15 mg. For the complete program registry, see ClinicalTrials.gov.
Retatrutide vs Tirzepatide FAQ
Frequently Asked Questions
Is retatrutide better than tirzepatide for weight loss?
In cross-trial comparisons, retatrutide shows greater weight loss: 28.7% vs 22.5% at similar timepoints. A direct head-to-head trial (NCT06662383, 800 patients) is underway with results in December 2026. Until then, retatrutide appears more potent but tirzepatide is proven, approved, and available now via the tirzepatide dosage chart in units.
When will retatrutide be FDA approved?
Retatrutide is expected to complete its Phase 3 TRIUMPH program in 2026. If results are positive, Eli Lilly will likely submit a New Drug Application in late 2026 or early 2027. Standard FDA review takes 10 to 12 months. Potential approval lands in late 2027. For trial enrollment paths today, see how to get retatrutide in 2026.
Can I switch from tirzepatide to retatrutide?
Not yet. Retatrutide is not approved or commercially available. Once approved, switching will likely be possible since both drugs are made by Eli Lilly. You would start retatrutide at the lowest dose and titrate regardless of your current tirzepatide dose. The principle mirrors the semaglutide-to-tirzepatide switching protocol of restarting titration.
What is the main difference between retatrutide and tirzepatide?
Retatrutide activates three hormone receptors (GLP-1, GIP, and glucagon) while tirzepatide activates two (GLP-1 and GIP). The added glucagon receptor increases resting energy expenditure, promotes liver fat clearance (82% reduction vs ~55%), and contributes to greater overall weight loss (28.7% vs 22.5%). The tradeoff is a new dysesthesia signal (4.4 to 20.9% at 12 mg).
Is retatrutide safer than tirzepatide?
Tirzepatide currently has a better-established safety profile due to years of real-world use and multiple large trials. In TRIUMPH-4, retatrutide showed higher GI rates (nausea 43% vs 31%, vomiting 21% vs 12%). Dysesthesia (4.4 to 20.9% at 12 mg) has not been reported with tirzepatide. See retatrutide side effects for the full safety profile.
How does semaglutide compare to retatrutide and tirzepatide?
Semaglutide (Wegovy/Ozempic) activates only GLP-1 and produces roughly 15% weight loss at 68 weeks. Tirzepatide adds GIP for 22.5%. Retatrutide adds glucagon for 28.7%. Each generation built on the last. Semaglutide has the longest safety record and the SELECT trial proving cardiovascular benefits. See best peptides for weight loss for a broader ranking.
Does retatrutide cause muscle loss?
All weight loss interventions cause some lean mass loss alongside fat loss. In tirzepatide trials, fat mass decreased 33.9% while lean mass decreased 10.9%. Retatrutide-specific body composition data from Phase 3 has not been fully published. To preserve muscle on any GLP-1, stack resistance training with adequate protein. See muscle loss on tirzepatide for prevention strategies.
What is the TRIUMPH trial program?
TRIUMPH is retatrutide's Phase 3 program: eight trials across obesity, type 2 diabetes, knee osteoarthritis, sleep apnea, cardiovascular disease, and liver disease populations. TRIUMPH-4 reported positive results in December 2025 (28.7% weight loss). Seven additional trials will report throughout 2026. The program enrolls over 5,800 participants total. For the retatrutide dosage guide covering all trial doses, see the full protocol.
The Bottom Line
Retatrutide and tirzepatide use the same basic approach: mimicking gut hormones to reduce appetite and improve metabolism. Tirzepatide activates two receptors and produces 22.5% weight loss. Retatrutide activates three and reaches 28.7%. The added glucagon receptor is responsible for the gap, delivering both higher weight loss and dramatically better liver fat clearance.
The practical bottom line: if you need a weight loss medication today, tirzepatide is the proven, available option with years of safety data. If you are already on tirzepatide and doing well, there is no medical reason to wait for something else. Keep going.
If you have plateaued on current medications, have fatty liver disease, or simply want to understand what is coming next, retatrutide is the most promising drug in the pipeline. The head-to-head trial results in December 2026 will clarify the picture. We will update this article when those results are published.
Explore peptide profiles, dosage calculators, and research tools at PeptidesExplorer. For retatrutide-specific dosing, see our retatrutide dosage guide. For storage best practices, see how to store peptides. For the full mechanism explanation, see how does retatrutide work. If you are struggling with current GLP-1 therapies, see not losing weight on semaglutide or why am I not losing weight on tirzepatide. For the regulatory landscape, see FDA peptide crackdown 2026. For overall safety considerations, see our peptide safety guide.
Related Articles: - Retatrutide: Complete Guide - Retatrutide Side Effects - How to Get Retatrutide - Best Peptides for Weight Loss - Getting Started with Peptides
Helpful Tools
Related Articles
Retatrutide Availability & Timeline
Retatrutide is expected to receive FDA approval in mid-2027 with pharmacy availability in late 2027. Full timeline and Phase 3 trial status.
Retatrutide: Mechanism & Trial Results
Retatrutide (LY3437943) is a triple agonist weight loss drug by Eli Lilly. 28.7% weight loss in trials, 82% liver fat reduction. Not yet FDA-approved.
How to Get Retatrutide in 2026
How to access retatrutide in 2026: clinical trial enrollment, compounding pharmacy status, FDA approval timeline (est. 2027). Updated March 2026.
Retatrutide Dosage: Titration Schedule
Retatrutide dosage chart with weekly titration schedule. Phase 2 & 3 trial data: up to 28.7% weight loss at 12 mg. Dose escalation and side effects.