Retatrutide (LY3437943) is an experimental weight loss injection made by Eli Lilly. In clinical trials, people taking the highest dose lost an average of 71 pounds over 16 months, a 28.7% reduction in body weight. That is the most weight loss ever recorded for any drug (Lilly Press Release, Dec 2025).
What makes retatrutide different from drugs like Ozempic or Zepbound: it activates three hormone receptors in your body instead of one or two. Ozempic targets one. Zepbound targets two. Retatrutide targets all three. More targets means more weight loss, but also more side effects.
Retatrutide is not FDA-approved and cannot be prescribed anywhere in the world as of March 2026. It is still in large-scale clinical trials (Phase 3). Approval is expected around mid-2027, with the drug reaching pharmacies in late 2027 or early 2028.
This guide explains how retatrutide works in plain language, what the clinical trial data actually shows, how it compares to existing drugs and bariatric surgery, and what is coming next in the obesity drug pipeline. Last updated: March 2026.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is Retatrutide?
Retatrutide is a once-weekly injection being developed by Eli Lilly as a treatment for obesity, type 2 diabetes, and fatty liver disease. Its lab name is LY3437943. Some media outlets call it "Triple G" because it targets three receptors that all start with G: GLP-1, GIP, and Glucagon.
It belongs to the same family of drugs as semaglutide (the active ingredient in Ozempic and Wegovy) and tirzepatide (the active ingredient in Mounjaro and Zepbound). Each generation of these drugs targets more receptors and produces more weight loss.
Think of it as evolution: Ozempic was the first generation. Zepbound was the second. Retatrutide is the third.
Retatrutide is a synthetic peptide, a small protein made of 39 amino acids. It has a half-life of about 6 days, which is why once-weekly dosing works. You inject it under the skin (like an insulin shot), same day each week.
How Does Retatrutide Work? The Triple Agonist Mechanism
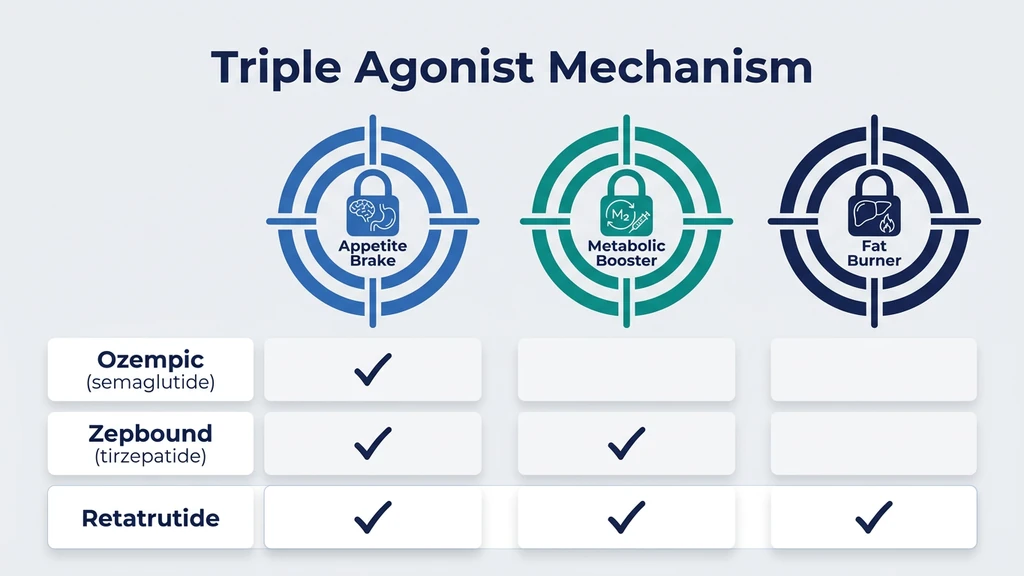
The word "agonist" just means something that activates a receptor. A receptor is like a lock on a cell. Retatrutide carries three keys that fit three different locks in your body. Each lock controls a different part of how your body handles food, fat, and energy.
GLP-1: The Appetite Brake
This is the same receptor targeted by Ozempic and Wegovy. When activated, it does two things. First, it slows down how fast your stomach empties. Food sits in your stomach longer, so you feel full faster and stay full longer. Second, it sends signals to your brain that reduce hunger and cravings.
This is also the receptor responsible for the nausea that many people experience. Your stomach is not used to holding food for that long, especially in the first weeks. The nausea usually fades as your body adjusts.
GLP-1 also improves how your body releases insulin (the hormone that controls blood sugar), which is why these drugs work for type 2 diabetes too.
GIP: The Metabolic Booster
This is the receptor that Zepbound and Mounjaro added to the mix. GIP (glucose-dependent insulinotropic polypeptide) improves how your body processes insulin and appears to change where your body stores fat, shifting it away from dangerous visceral fat (the fat packed around your organs) toward less harmful subcutaneous fat (the fat under your skin).
When GIP is combined with GLP-1, weight loss jumps from about 15% (GLP-1 alone) to about 22.5% (GLP-1 plus GIP). The side effect profile stays relatively similar.
Glucagon: The Fat Burner (What Makes Retatrutide Different)
This is retatrutide's secret weapon, the third receptor that no approved drug targets yet.
Glucagon increases your resting energy expenditure. In plain language: you burn more calories even when you are doing nothing. If GLP-1 is the brake on hunger, glucagon is the accelerator on fat burning. It tells your body to break down stored fat for energy, especially in the liver.
This is why retatrutide produced an 82% reduction in liver fat in trials (Nature Medicine, 2024). Semaglutide reduces liver fat by about 40 to 50%. Tirzepatide by about 55%. Retatrutide nearly doubles them because of this glucagon component.
The trade-off: glucagon normally raises blood sugar (it is the body's emergency "release stored energy" signal). In retatrutide, the GLP-1 component counterbalances this, keeping blood sugar stable. Glucagon is also the suspected source of dysesthesia, a new side effect (tingling or burning skin sensations) that appeared in Phase 3 trials.
Why Three Receptors Produce More Weight Loss Than Two
Each generation of these drugs added a receptor and gained roughly 6 to 8 percentage points of additional weight loss:
| Drug | Receptors | What Each Does | Max Weight Loss |
|---|---|---|---|
| Semaglutide (Wegovy) | GLP-1 | Reduces appetite | ~15% (68 weeks) |
| Tirzepatide (Zepbound) | GLP-1 + GIP | Reduces appetite + improves fat metabolism | ~22.5% (72 weeks) |
| Retatrutide | GLP-1 + GIP + Glucagon | Reduces appetite + improves fat metabolism + burns more energy | 28.7% (68 weeks) |
Semaglutide reduced how much you eat. Tirzepatide improved how your body processes what you eat. Retatrutide added burning more energy on top of both (Jastreboff et al., NEJM 2023).
How Much Weight Do You Lose on Retatrutide?
Two major trials provide the data. The Phase 2 trial (338 people, 48 weeks) was published in the New England Journal of Medicine, one of the most prestigious medical journals in the world. The Phase 3 TRIUMPH-4 trial (445 people, 68 weeks) reported results in December 2025.
Phase 2 Results (48 Weeks)
This trial, led by researcher Anja Jastreboff and published in the NEJM in 2023, tested five different doses of retatrutide against a placebo (fake injection) in 338 adults with obesity (Jastreboff et al., NEJM 2023).
| Dose | Weight Loss at 48 Weeks | Weight Loss at 24 Weeks |
|---|---|---|
| Placebo | -2.1% | -1.6% |
| 1 mg | -8.7% | -7.2% |
| 4 mg | -17.1% | -12.9% |
| 8 mg | -22.8% | -17.3% |
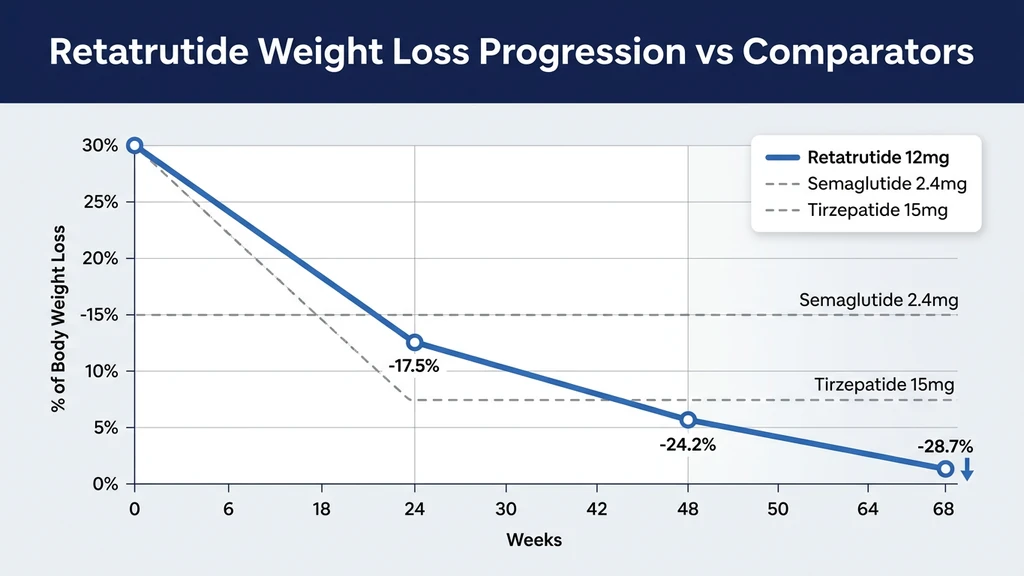
| 12 mg | -24.2% | -17.5% |
At 12 mg, every single participant lost at least 5% of their body weight. 83 out of 100 lost at least 15%. Weight loss was still accelerating when the trial ended at 48 weeks, meaning longer treatment would produce even more loss.
Phase 3 TRIUMPH-4 Results (68 Weeks)
The larger Phase 3 trial enrolled 445 adults with obesity and knee arthritis. It ran for 68 weeks, about 16 months (Lilly Press Release, Dec 2025).
| Metric | 9 mg Dose | 12 mg Dose | Placebo |
|---|---|---|---|
| Average weight loss | 26.4% (64.2 lbs) | 28.7% (71.2 lbs) | 2.1% |
| Knee pain improvement | Significant | 75.8% reduction | Moderate |
| Blood pressure drop | Significant | 14 mmHg reduction | Modest |
For perspective: someone starting at 250 pounds would lose about 72 pounds on the highest dose. That brings them from 250 to 178 pounds over 16 months. The weight loss curve was still going down when the trial ended, meaning the final number could be even higher with continued treatment.
How Does That Compare to Weight Loss Surgery?
Retatrutide's 28.7% weight loss puts it in the same range as bariatric surgery for the first time.
| Factor | Retatrutide (12 mg) | Gastric Sleeve | Gastric Bypass |
|---|---|---|---|
| Weight loss | 28.7% (16 months) | 25 to 30% (1 year) | 30 to 35% (1 year) |
| Reversible | Yes (stop the injection) | No (permanent surgery) | No (permanent surgery) |
| How it works | Weekly injection | Surgery removes part of stomach | Surgery reroutes digestive system |
| Recovery time | None | 2 to 4 weeks | 4 to 6 weeks |
| Weight regain risk | High if you stop the drug | 10 to 20% regain common | 10 to 15% regain |
| Approximate cost (US) | Est. $1,100 to $1,500/month ongoing | $15,000 to $25,000 one-time | $20,000 to $35,000 one-time |
| Liver fat reduction | 82% | Variable | Variable |
One major difference: surgery is a one-time procedure. Retatrutide requires weekly injections for as long as you want to maintain the weight loss. If you stop, the weight is likely to return, based on what we have seen with semaglutide and tirzepatide. Long-term data beyond 68 weeks does not yet exist for retatrutide.
The Liver Fat Breakthrough Most Articles Skip
Retatrutide's weight loss numbers get all the headlines. But the liver fat data may be equally important for millions of people.
A substudy of 98 participants with fatty liver disease (technically called MASLD, or metabolic dysfunction-associated steatotic liver disease) was published in Nature Medicine in 2024 (Nature Medicine, 2024). Fatty liver is extremely common: it affects the majority of people with obesity and can progress to liver scarring, liver failure, and liver cancer if untreated.
| Dose | Liver Fat Reduction at 24 Weeks | Liver Returned to Normal (<5% fat) |
|---|---|---|
| Placebo | +0.3% (got worse) | 0% |
| 1 mg | 42.9% | 27% |
| 4 mg | 57.0% | 52% |
| 8 mg | 81.4% | 79% |
| 12 mg | 82.4% | 86% |
At the highest dose, 86 out of 100 people with fatty liver had their liver fat return to normal levels. No liver damage was caused by the drug itself.
Why is retatrutide so much better for the liver than other drugs? The glucagon receptor. Glucagon directly tells the liver to burn its stored fat. Semaglutide and tirzepatide reduce liver fat indirectly through weight loss. Retatrutide does both: indirect reduction from weight loss plus direct fat burning in the liver itself.
For anyone whose primary concern is liver health alongside weight loss, this is retatrutide's strongest differentiator. A dedicated MASLD trial (NCT07035093) is part of the broader TRIUMPH program.
What Are the Side Effects?
Retatrutide causes more side effects than older drugs in its class. This is the trade-off for more weight loss. Most side effects are stomach-related and improve over time, but one new signal deserves attention.
The Common Side Effects
The most frequent side effects from the Phase 3 TRIUMPH-4 trial (BioSpace, Dec 2025):
| Side Effect | 12 mg Dose | 9 mg Dose | Placebo |
|---|---|---|---|
| Nausea | 43% | 38% | 11% |
| Diarrhea | 33% | 35% | 13% |
| Constipation | 25% | 22% | 7% |
| Vomiting | 21% | 20% | 3% |
| Decreased appetite | 18% | 19% | 9% |
These numbers are higher than semaglutide (~30% nausea) and tirzepatide (~31% nausea). The symptoms are worst during the first weeks on a new dose and improve as your body adjusts. Slow dose escalation (starting low and increasing gradually every 4 weeks) reduces these significantly.
Dysesthesia: The New Side Effect Nobody Expected
TRIUMPH-4 revealed something that did not show up in the smaller Phase 2 trial: dysesthesia. This is a medical term for abnormal skin sensations. People described it as tingling, burning, or heightened sensitivity, similar to the feeling when your foot falls asleep, but in random body parts.
At the highest dose, about 1 in 5 people (20.9%) experienced this. At the lower 9 mg dose, it was about 1 in 11 (8.8%). On placebo, almost nobody reported it (0.7%).
The good news: it was generally mild. It did not cause significant numbers of people to quit the trial. There is no evidence of long-term nerve damage.
The likely cause is the glucagon receptor. Semaglutide and tirzepatide do not cause this because they do not activate glucagon. This is genuinely new territory for weight loss drugs.
How Many People Stopped Because of Side Effects?
In TRIUMPH-4, 18.2% of people on 12 mg dropped out due to side effects, compared to 4% on placebo. That means roughly 4 out of 5 people completed the trial at the highest dose.
An interesting pattern: people with higher starting weight had lower dropout rates. At 12 mg, only 12.1% of people with a BMI above 35 dropped out (versus 18.2% overall). The likely reason: when the drug is producing dramatic weight loss, people are more willing to tolerate the side effects.
For the complete side effect breakdown with management strategies, see our retatrutide side effects guide.
How Is Retatrutide Dosed?
Retatrutide is injected once per week under the skin, similar to Ozempic or Zepbound. You pick the same day each week (for example, every Monday). The dose starts low and increases gradually over 12 weeks.
| Phase | Weeks | Dose | Purpose |
|---|---|---|---|
| Starting | 1 to 4 | 2 mg | Let your stomach adjust |
| First increase | 5 to 8 | 4 mg | Gradual ramp up |
| Second increase | 9 to 12 | 8 mg | Approaching full dose |
| Maintenance | 13 onward | 12 mg | Full dose for maximum results |
The gradual increase matters. In the Phase 2 trial, one group skipped the low starting dose and went straight to 4 mg. They had nearly double the rate of nausea and vomiting. Every Phase 3 trial uses the slow ramp-up.
Eli Lilly is also testing a 4 mg maintenance dose for people who lose enough weight at lower doses or cannot tolerate the higher ones (NCT06859268).
For the complete dosing protocol including how to prepare the injection, see our retatrutide dosage guide. Plan your doses with our retatrutide dosage calculator.
When Will Retatrutide Be Available?
Retatrutide cannot be prescribed or bought at a pharmacy anywhere in the world right now. Here is the realistic timeline.
| Scenario | Lilly Files Application | FDA Decision | At Your Pharmacy |
|---|---|---|---|
| Best case | Late 2026 | Mid-2027 | Late 2027 |
| Most likely | Early 2027 | Late 2027 | Early 2028 |
| Delayed | Mid-2027 | Mid-2028 | Late 2028 |
Seven Phase 3 trials are still running (the TRIUMPH program, 5,800+ people total). The most important remaining data: TRIUMPH-1 (the main obesity trial, 2,300 people) and TRIUMPH-2 (obesity with type 2 diabetes, 1,000 people) should report results in mid-2026. A head-to-head trial comparing retatrutide directly against tirzepatide (NCT06662383, 800 patients) should report in late 2026.
Your options today: clinical trials (free, pharmaceutical-grade drug, medical supervision), expanded access (for severe cases), or research peptides (unregulated, sold for research use only). FDA-approved alternatives like tirzepatide and semaglutide are available by prescription right now.
For the full breakdown of every access pathway, trial enrollment steps, and what to do while waiting, see our guide to getting retatrutide.
Retatrutide vs Other Weight Loss Drugs
Here is how retatrutide stacks up against the two FDA-approved options:
| Feature | Semaglutide (Wegovy/Ozempic) | Tirzepatide (Zepbound/Mounjaro) | Retatrutide |
|---|---|---|---|
| Maker | Novo Nordisk | Eli Lilly | Eli Lilly |
| Receptors | GLP-1 (1 target) | GLP-1 + GIP (2 targets) | GLP-1 + GIP + Glucagon (3 targets) |
| Max weight loss | ~15% | ~22.5% | 28.7% |
| Liver fat reduction | ~40 to 50% | ~55% | 82% |
| Nausea rate | ~30% | ~31% | ~43% |
| Dysesthesia | Rare | Rare | 20.9% at 12 mg |
| FDA status | Approved (2021) | Approved (2023) | Phase 3 (est. 2027) |
| How you take it | Weekly injection | Weekly injection | Weekly injection |
| Monthly cost | ~$1,300 | ~$1,000 | Not available (est. $1,100 to $1,500) |
A direct head-to-head trial comparing retatrutide against tirzepatide under identical conditions (NCT06662383, 800 patients) will report results around December 2026. That will be the first apples-to-apples comparison. Until then, all the numbers above come from separate trials with different patients.
For the full comparison including mechanism breakdown and practical guidance on choosing, see our retatrutide vs tirzepatide comparison. Use our tirzepatide dosage calculator or semaglutide dosage calculator if you are considering those options now.
The Obesity Drug Pipeline: What Else Is Coming?
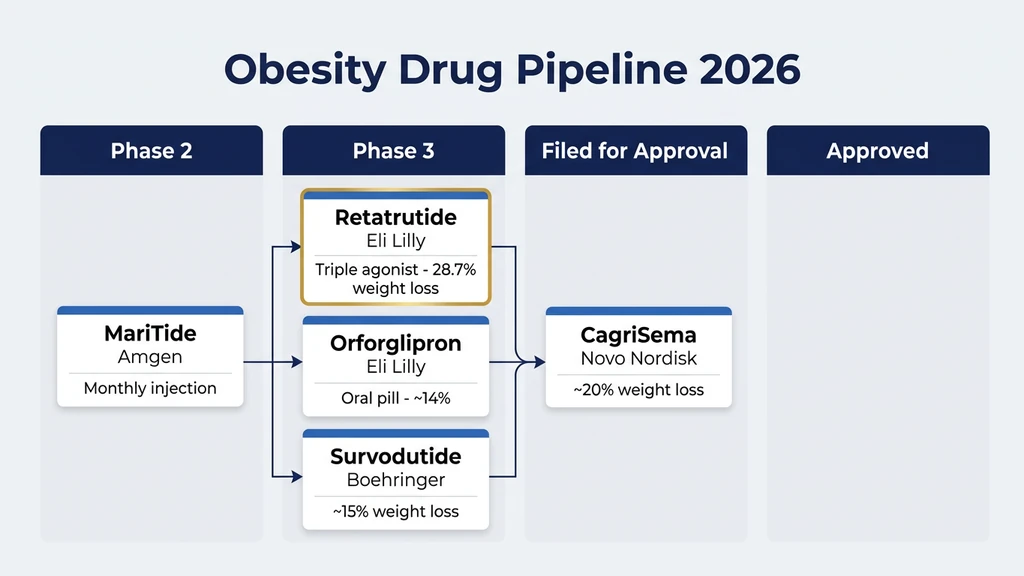
Retatrutide is not the only next-generation weight loss drug in development. The field is moving fast. Here is where everything stands:
| Drug | Company | How It Works | Stage | Weight Loss | What Makes It Different |
|---|---|---|---|---|---|
| Retatrutide | Eli Lilly | GLP-1 + GIP + Glucagon | Phase 3 | 28.7% | Triple agonist, best liver fat data |
| CagriSema | Novo Nordisk | GLP-1 + Amylin | Filed for approval (2026) | ~20% | May preserve more muscle mass |
| Orforglipron | Eli Lilly | GLP-1 (oral pill) | Phase 3 | ~14 to 15% | First effective weight loss pill, no injections |
| Survodutide | Boehringer Ingelheim | GLP-1 + Glucagon | Phase 3 | ~15% (46 weeks) | Strong fatty liver data |
| Amycretin | Novo Nordisk | GLP-1 + Amylin (oral pill) | Phase 2 | ~13% (36 weeks) | Oral, next-gen amylin pathway |
| MariTide | Amgen | Anti-GIPR antibody + GLP-1 | Phase 2 | ~14% (52 weeks) | Monthly injection instead of weekly |
Retatrutide leads on weight loss numbers. CagriSema is closest to FDA approval. Orforglipron and Amycretin represent the future of oral pills (no needles). The obesity drug landscape has shifted from "does anything work?" to "which one fits your situation?" (Medscape, 2026).
Who Should Consider Retatrutide?
This is not medical advice. But different situations call for different approaches.
If you are already on semaglutide or tirzepatide and your weight loss has stalled: The third receptor (glucagon) may unlock further loss. You cannot switch to retatrutide yet since it is not approved, but you could discuss clinical trial enrollment with your doctor, or simply wait for approval. Starting an approved drug now does not prevent you from switching later.
If fatty liver disease is a major concern: The 82% liver fat reduction is retatrutide's strongest selling point versus competitors. If your doctor has flagged fatty liver as a health risk, retatrutide may be particularly relevant once approved. A dedicated liver disease trial is underway (NCT07035093).
If you have never tried a weight loss medication and are considering starting: There is no reason to wait for retatrutide. Semaglutide and tirzepatide are available today, produce meaningful weight loss (15 to 22.5%), and have years of real-world safety data. Starting now is better than waiting 18+ months for a drug that may or may not work better for your body.
If you need the maximum possible weight loss: Retatrutide produced the highest numbers of any drug. If your BMI is very high and you want to avoid surgery, this drug may eventually be your best pharmaceutical option. Clinical trial enrollment is the only way to access it today.
Explore peptide profiles, dosage calculators, and research tools at PeptidesExplorer. For the full peptide dosage chart or to check for peptide interactions, use our free tools.
Frequently Asked Questions
What is retatrutide?
Retatrutide (LY3437943) is an experimental weight loss injection made by Eli Lilly. It activates three hormone receptors (GLP-1, GIP, and glucagon) simultaneously, which is why it is called a triple agonist. In clinical trials, the highest dose produced 28.7% average body weight loss over 68 weeks, the most ever recorded for any obesity drug. It is not FDA-approved yet.
How does retatrutide work?
Retatrutide works by activating three receptors at once. GLP-1 slows your stomach and reduces hunger (like Ozempic). GIP improves how your body handles insulin and fat storage (like Zepbound). Glucagon increases how many calories you burn at rest and directly tells your liver to burn stored fat. The combination of eating less, processing food better, and burning more energy produces greater weight loss than drugs targeting one or two receptors.
When will retatrutide be FDA-approved?
The most likely timeline: Eli Lilly submits its application in late 2026 or early 2027, the FDA takes 6 to 10 months to review it, and retatrutide reaches pharmacies in late 2027 or early 2028. Seven Phase 3 trials are still running and will report results throughout 2026. Eli Lilly has not confirmed an exact filing date.
How does retatrutide compare to Ozempic and Zepbound?
Ozempic (semaglutide) targets one receptor and produces about 15% weight loss. Zepbound (tirzepatide) targets two receptors and produces about 22.5%. Retatrutide targets three receptors and produced 28.7%. Each generation adds a target and pushes weight loss further. The trade-off: retatrutide has higher nausea rates (43% vs about 30%) and a new side effect called dysesthesia (tingling or burning skin sensations in about 1 in 5 people at the highest dose).
What does triple agonist mean?
An agonist is a substance that activates a receptor in your body. A triple agonist activates three different receptors at the same time. Retatrutide activates GLP-1 (reduces appetite), GIP (improves metabolism), and glucagon (burns more energy). Older drugs like Ozempic are single agonists (one receptor). Zepbound is a dual agonist (two receptors). Retatrutide is the first triple agonist in late-stage clinical trials.
Is retatrutide better than weight loss surgery?
Retatrutide's 28.7% weight loss is in the same range as gastric sleeve surgery (25 to 30%) but below gastric bypass (30 to 35%). The key differences: retatrutide is a weekly injection with no recovery time, while surgery is permanent and requires weeks of recovery. However, retatrutide must be taken indefinitely to maintain results, while surgery is a one-time procedure. Long-term data beyond 16 months does not yet exist for retatrutide.
Can you buy retatrutide online?
Research peptide vendors sell retatrutide labeled 'for research use only.' These products are not manufactured to pharmaceutical standards, not FDA-regulated, and carry quality risks. The only way to get pharmaceutical-grade retatrutide is through a clinical trial. No pharmacy in the world can legally dispense it as a prescription. See our guide to getting retatrutide for all access options.
What is the retatrutide dosing schedule?
Retatrutide starts at 2 mg per week for the first 4 weeks, then increases to 4 mg (weeks 5 to 8), then 8 mg (weeks 9 to 12), then 12 mg from week 13 onward. All doses are once-weekly injections under the skin. The gradual increase over 12 weeks helps minimize nausea and other stomach-related side effects. See our retatrutide dosage guide for the full protocol.
The Bottom Line
Retatrutide is the most potent weight loss drug in clinical trials. 28.7% body weight reduction. 82% liver fat clearance. Three receptor targets instead of one or two. The numbers speak.
It is not available yet. Phase 3 trials are running, FDA approval is estimated for 2027, and pharmacy shelves will not stock it until late 2027 or 2028 at the earliest. The head-to-head trial against tirzepatide (December 2026) will be the most important data point of the year.
If you need treatment now, tirzepatide (Zepbound) and semaglutide (Wegovy) are proven, approved, and available today. Starting one does not prevent switching to retatrutide later.
We will update this guide as each TRIUMPH trial reports results throughout 2026. For how to reconstitute retatrutide, see our preparation guide. For the regulatory landscape, see our FDA peptide crackdown 2026 overview. Explore peptide profiles, dosage calculators, and research tools at PeptidesExplorer. For broader weight loss comparisons, see best peptides for weight loss. If you are struggling with current GLP-1 therapies, see not losing weight on semaglutide or why am I not losing weight on tirzepatide. New to peptides? Start with our getting started with peptides guide.
Related Articles: - Retatrutide Dosage Guide - Retatrutide Side Effects - How Does Retatrutide Work? - How to Get Retatrutide - Retatrutide vs Tirzepatide
Helpful Tools
Related Articles
Retatrutide Availability & Timeline
Retatrutide is expected to receive FDA approval in mid-2027 with pharmacy availability in late 2027. Full timeline and Phase 3 trial status.
How to Get Retatrutide in 2026
How to access retatrutide in 2026: clinical trial enrollment, compounding pharmacy status, FDA approval timeline (est. 2027). Updated March 2026.
How Does Retatrutide Work?
Retatrutide activates 3 receptors (GLP-1, GIP, glucagon) to reduce appetite, improve metabolism, and burn fat. 28.7% weight loss in trials.
GLP-3 Peptide: What It Really Is
GLP-3 does not exist. What clinics call 'GLP-3' is retatrutide (LY3437943), Eli Lilly's triple agonist targeting GLP-1, GIP, and glucagon receptors.