Retatrutide is dosed at 2 mg once weekly for the first four weeks, then escalated every four weeks to a maintenance dose of 8 mg or 12 mg. The full titration takes 12 weeks. All injections are subcutaneous, once per week, on the same day each week.
In clinical trials, the 12 mg maintenance dose produced 24.2% body weight loss at 48 weeks (Phase 2) and 28.7% at 68 weeks (Phase 3). No other peptide in development has matched these numbers.
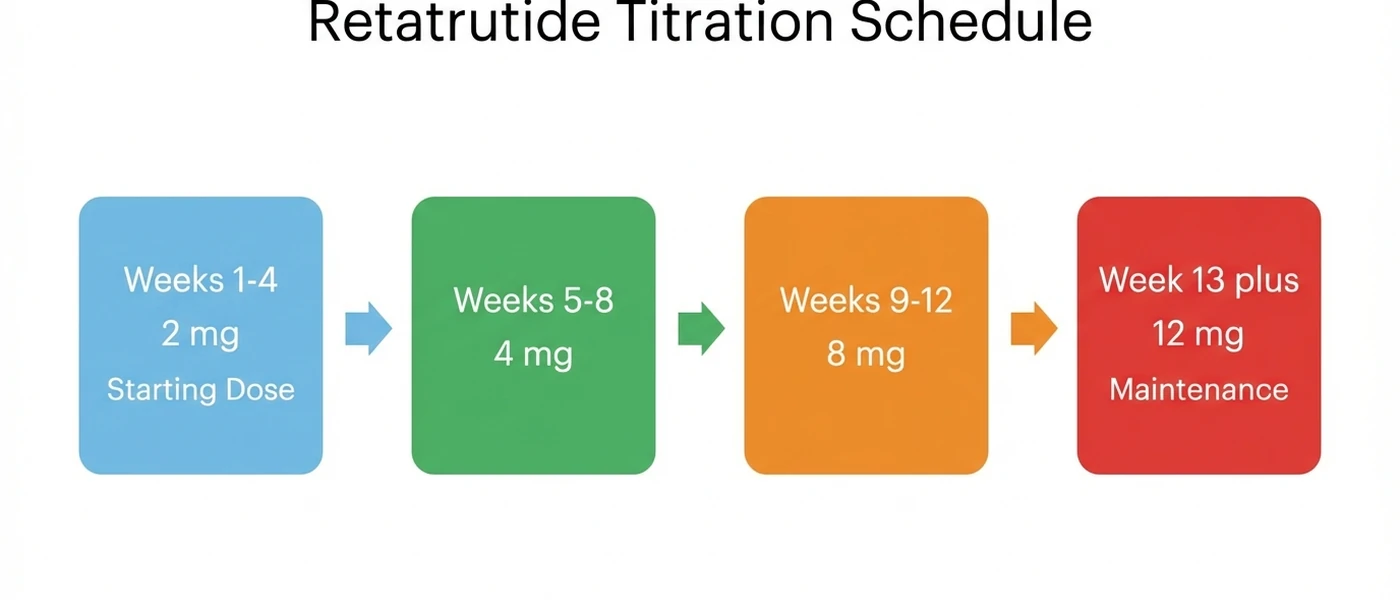
Quick reference: retatrutide titration schedule
| Weeks | Dose | Frequency | Notes |
|---|---|---|---|
| 1-4 | 2 mg | Once weekly | Starting dose for all patients |
| 5-8 | 4 mg | Once weekly | First escalation |
| 9-12 | 8 mg | Once weekly | Second escalation |
| 13+ | 12 mg | Once weekly | Maintenance (maximum dose) |
Use our retatrutide dosage calculator to plan your exact protocol. For dosage reference across all peptides, see the complete peptide dosage chart.
Retatrutide is investigational. It has not received FDA approval. All dosage information below comes from published clinical trial data. This is an educational resource: consult a qualified healthcare provider before acting on any information. For a broad overview of what retatrutide is and how it works, see our complete retatrutide guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is Retatrutide?
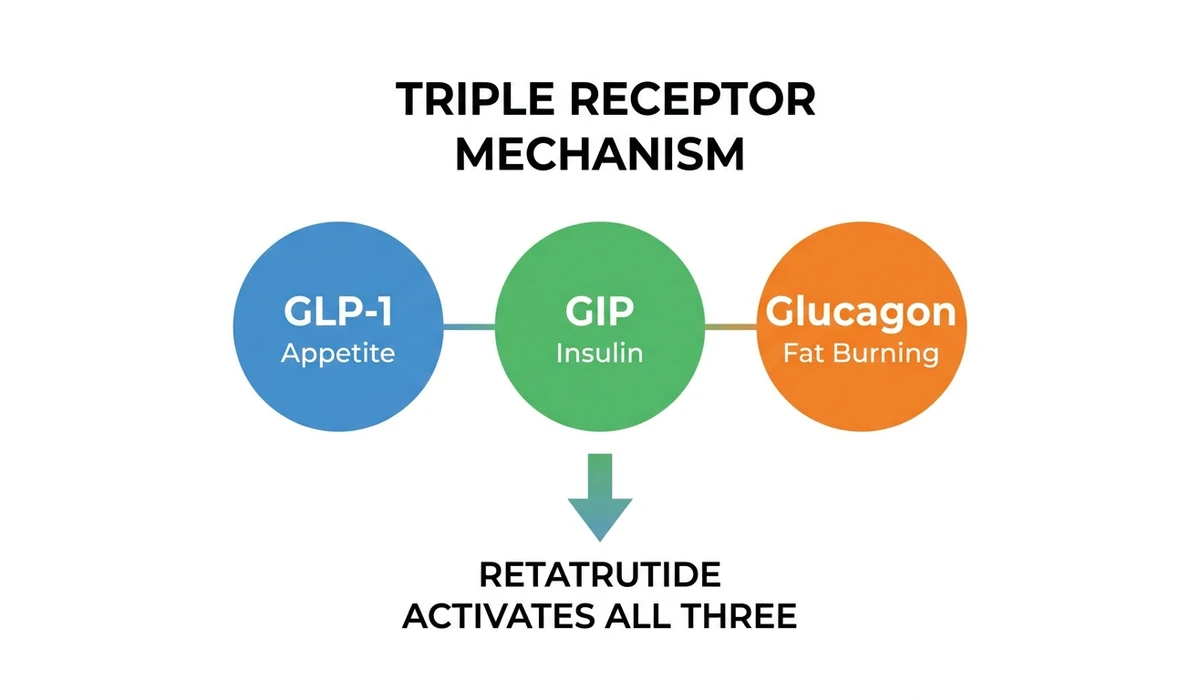
Retatrutide (LY3437943) is a triple hormone receptor agonist developed by Eli Lilly. It activates three receptors simultaneously: GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon.
This triple mechanism is what separates retatrutide from every other weight loss peptide on the market.
| Peptide | Receptors | Mechanism | Max Weight Loss (Trials) |
|---|---|---|---|
| Semaglutide | GLP-1 only | Single agonist | ~15% at 68 weeks |
| Tirzepatide | GLP-1 + GIP | Dual agonist | ~22.5% at 72 weeks |
| Retatrutide | GLP-1 + GIP + Glucagon | Triple agonist | 28.7% at 68 weeks |
The GLP-1 component suppresses appetite and slows gastric emptying. GIP enhances insulin sensitivity and works synergistically with GLP-1 to amplify satiety signals. Glucagon receptor activation is the differentiator: it increases energy expenditure by stimulating thermogenesis and fat oxidation in the liver.
In plain terms, semaglutide makes you eat less. Tirzepatide makes you eat less and improves how your body handles the food you do eat. Retatrutide does both of those things and actively burns more energy at rest.
Retatrutide Dosing Schedule (Week by Week)
The retatrutide titration schedule follows a 4-week escalation pattern. Every dose group in the Phase 2 trial started at 2 mg. Starting higher caused significantly more gastrointestinal side effects.
Starting Dose (Weeks 1-4): 2 mg
Every patient begins at 2 mg once weekly, regardless of target maintenance dose. This is non-negotiable based on the clinical trial data.
In the Phase 2 trial, one group was assigned to start directly at 4 mg. Their rate of gastrointestinal side effects nearly doubled compared to the group that started at 2 mg and escalated (Jastreboff et al., NEJM 2023). Nausea, diarrhea, and vomiting were all significantly higher in the direct-start group.
The 2 mg starting dose allows the GI tract to adapt to GLP-1 receptor activation. Most side effects that do occur at this dose are mild: slight nausea in the first few days, reduced appetite, and occasionally loose stools. These typically resolve within the first week.
First Escalation (Weeks 5-8): 4 mg
At week 5, the dose doubles to 4 mg once weekly. Appetite suppression becomes noticeably stronger at this level. Most users report a significant reduction in food cravings and portion sizes.
Weight loss accelerates during this phase. In the Phase 2 trial, the 4 mg group had already lost 12.9% of body weight by week 24, compared to 1.6% for placebo.
GI symptoms may briefly return when the dose increases. If nausea is severe, hold at 4 mg for an additional 2-4 weeks before escalating. The clinical trial protocol allowed for dose delays when side effects were intolerable.
Second Escalation (Weeks 9-12): 8 mg
The 8 mg dose is where the glucagon receptor activation becomes more pronounced. Energy expenditure increases measurably. The 8 mg group in the Phase 2 trial achieved 22.8% body weight loss at 48 weeks.
For some patients, 8 mg is the optimal maintenance dose. If side effects are manageable and weight loss is progressing well, there is no strict requirement to escalate to 12 mg. The difference between 8 mg and 12 mg at 48 weeks was 1.4 percentage points (22.8% vs 24.2%).
Maintenance Dose (Week 13+): 12 mg
The 12 mg dose produced the highest weight loss in every trial: 24.2% at 48 weeks (Phase 2) and 28.7% at 68 weeks (Phase 3 TRIUMPH-4). This is the maximum studied dose.
At 12 mg, response rates were remarkable. In the Phase 2 trial at 48 weeks (NEJM 2023):
| Threshold | 12 mg Group | Placebo |
|---|---|---|
| Lost ≥5% body weight | 100% | 27% |
| Lost ≥10% body weight | 93% | 9% |
| Lost ≥15% body weight | 83% | 2% |
Every single participant on 12 mg lost at least 5% of their body weight. No other obesity medication has achieved a 100% response rate at this threshold.
Continue the 12 mg maintenance dose for as long as treatment is needed. The Phase 3 TRIUMPH-4 trial ran for 68 weeks with continued weight loss throughout, suggesting the plateau has not yet been reached at this timepoint (Lilly TRIUMPH-4 Press Release).
Slower Titration Option
Some practitioners use a more gradual escalation with 2 mg increments every four weeks. This approach reduces GI side effects further and is useful for patients with a history of GLP-1 intolerance.
| Weeks | Dose |
|---|---|
| 1-4 | 2 mg |
| 5-8 | 4 mg |
| 9-12 | 6 mg |
| 13-16 | 8 mg |
| 17-20 | 10 mg |
| 21+ | 12 mg |
This schedule takes 20 weeks to reach the maximum dose instead of 12. The tradeoff: slower initial weight loss in exchange for better tolerability. For patients who struggled with nausea on tirzepatide or semaglutide, the slower schedule is often the better choice.
Retatrutide Weight Loss Results by Dose
Two major clinical trials provide dosage-specific weight loss data. The Phase 2 trial (338 adults, 48 weeks) was published in the New England Journal of Medicine. The Phase 3 TRIUMPH-4 trial (68 weeks) reported topline results in December 2025.
Phase 2 Trial Results (48 Weeks)
The Phase 2 trial randomized 338 adults with obesity (BMI ≥30) or overweight with at least one weight-related condition to retatrutide or placebo for 48 weeks (Jastreboff et al., NEJM 2023).
Weight loss at 24 weeks (primary endpoint):
| Dose | Mean Weight Loss | vs Placebo |
|---|---|---|
| Placebo | -1.6% | |
| 1 mg | -7.2% | +5.6% |
| 4 mg | -12.9% | +11.3% |
| 8 mg | -17.3% | +15.7% |
| 12 mg | -17.5% (41.2 lbs) | +15.9% |
Weight loss at 48 weeks (secondary endpoint):
| Dose | Mean Weight Loss | vs Placebo |
|---|---|---|
| Placebo | -2.1% | |
| 1 mg | -8.7% | +6.6% |
| 4 mg | -17.1% | +15.0% |
| 8 mg | -22.8% | +20.7% |
| 12 mg | -24.2% (57.8 lbs) | +22.1% |
The weight loss curves had not plateaued at 48 weeks. The 8 mg and 12 mg groups were still losing weight when the study ended, suggesting longer treatment durations would produce even greater reductions.
Phase 3 TRIUMPH-4 Results (68 Weeks)
The TRIUMPH-4 trial enrolled adults with obesity and knee osteoarthritis. Results announced in December 2025 confirmed the Phase 2 findings at a longer timepoint (Lilly Press Release, Dec 2025):
| Metric | 12 mg Retatrutide |
|---|---|
| Mean body weight loss | 28.7% |
| Absolute weight loss | 71.2 lbs (from 248.5 lbs baseline) |
| WOMAC pain score reduction | 75.8% (4.5 points) |
| Patients completely pain-free | 1 in 8 |
| Duration | 68 weeks |
The 28.7% weight loss at 68 weeks exceeds both semaglutide (~15% in STEP trials) and tirzepatide (~22.5% in SURMOUNT trials) at similar timepoints.
Seven additional Phase 3 TRIUMPH trials are expected to report results throughout 2026, covering obesity, type 2 diabetes, sleep apnea, chronic low back pain, and metabolic liver disease (ClinicalTrials.gov).
Weight Loss Comparison: Retatrutide vs Tirzepatide vs Semaglutide
How does retatrutide compare to the current standard of care? The data comes from separate trials with different populations, so direct comparison has limitations. Still, the magnitude of difference is striking.
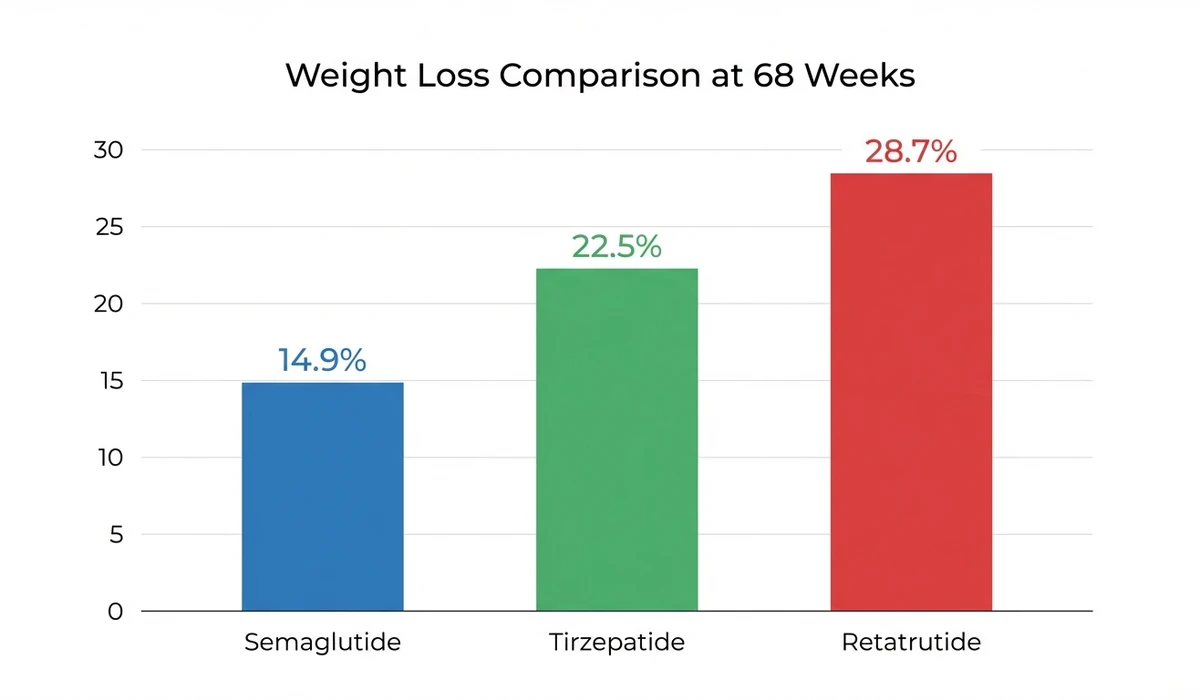
| Peptide | Trial | Duration | Max Dose | Weight Loss | Mechanism |
|---|---|---|---|---|---|
| Semaglutide 2.4 mg | STEP 1 | 68 weeks | 2.4 mg/week | -14.9% | GLP-1 |
| Tirzepatide 15 mg | SURMOUNT-1 | 72 weeks | 15 mg/week | -22.5% | GLP-1 + GIP |
| Retatrutide 12 mg | TRIUMPH-4 | 68 weeks | 12 mg/week | -28.7% | GLP-1 + GIP + Glucagon |
The glucagon receptor component accounts for much of the difference. Glucagon stimulates hepatic fat oxidation and thermogenesis, burning calories that GLP-1 alone cannot access. This is why retatrutide produces greater weight loss even at a lower milligram dose than tirzepatide.
For a broader comparison of weight loss peptides, see our best peptides for weight loss guide.
How to Inject Retatrutide
Retatrutide is administered as a subcutaneous injection once per week. The injection technique is identical to other peptide injections. If you have experience with semaglutide, tirzepatide, or BPC-157, the process is the same.
For a complete injection tutorial, see our peptide injections guide.
Injection Sites
Three subcutaneous injection sites are standard:
- Abdomen: The most common site. Pinch a fold of skin at least 2 inches from the navel. Inject at a 45-degree angle.
- Front of thigh: Middle third of the upper leg. Good alternative if the abdomen is sore from repeated injections.
- Back of upper arm: Harder to self-inject but viable with practice.
Rotate injection sites each week. Do not inject into the same spot two weeks in a row. Rotation prevents lipodystrophy (localized fat changes) and reduces injection site irritation.
Injection Frequency and Timing
Inject once per week on the same day each week. The specific day does not matter. Choose a day that is easy to remember: many users pick Sunday or Monday.
The time of day is also flexible. Morning or evening, with or without food. Consistency matters more than timing. If you miss your scheduled day, inject as soon as you remember within 3 days. If more than 3 days have passed, skip the missed dose and resume on the next scheduled day.
Retatrutide has a half-life of approximately 6 days, which supports the once-weekly dosing schedule. Steady-state blood levels are typically reached by week 4-5 of treatment.
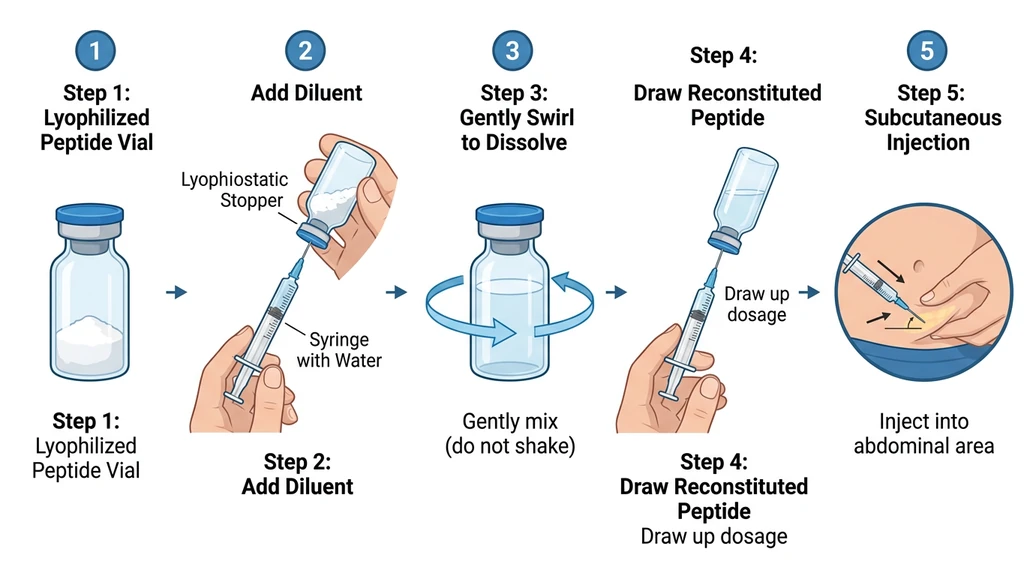
How to Reconstitute Retatrutide
Research-grade retatrutide typically arrives as a lyophilized (freeze-dried) powder that must be reconstituted before injection. Pre-filled pens are used in clinical trials but are not available outside the trial setting.
What You Need
| Supply | Specification |
|---|---|
| Retatrutide vial | Lyophilized powder (typically 5 mg or 10 mg vials) |
| Bacteriostatic water (BAC water) | 0.9% benzyl alcohol preserved |
| Insulin syringes | 29-31 gauge, 0.5 mL or 1 mL |
| Alcohol swabs | For cleaning vial tops and injection sites |
Use bacteriostatic water, not sterile water. The benzyl alcohol preservative allows the reconstituted solution to last 28-30 days refrigerated. Sterile water has no preservative and must be used within 5-7 days. For the complete step-by-step process, see how to reconstitute retatrutide. For storage guidelines, see our peptide storage guide.
Step-by-Step Reconstitution
- 1.Clean the top of the retatrutide vial and the BAC water vial with alcohol swabs.
- 2.Draw your chosen volume of BAC water into the syringe. For a 10 mg vial, 2 mL of BAC water gives a concentration of 5 mg/mL (convenient for dosing).
- 3.Insert the needle into the retatrutide vial at an angle. Let the water run slowly down the inside wall of the vial. Do not spray directly onto the powder.
- 4.Swirl gently. Never shake. Shaking creates foam and can damage the peptide.
- 5.Wait until the powder is fully dissolved. The solution should be clear and colorless. If cloudy, let it sit in the fridge for 15-30 minutes and swirl again.
- 6.Label the vial with the date and concentration.
- 7.Refrigerate immediately.
Use our reconstitution calculator to determine exact volumes for your vial size and target dose. For general reconstitution guidance, see how to reconstitute peptides.
Dosing After Reconstitution
With a 10 mg vial reconstituted in 2 mL BAC water (5 mg/mL concentration):
| Target Dose | Volume to Draw |
|---|---|
| 2 mg | 0.40 mL (40 units on insulin syringe) |
| 4 mg | 0.80 mL (80 units) |
| 8 mg | 1.60 mL (use two draws from a 1 mL syringe) |
| 12 mg | 2.40 mL (use three draws, or reconstitute at higher concentration) |
For the 8 mg and 12 mg doses, a higher concentration is more practical. Reconstituting a 10 mg vial in 1 mL BAC water gives 10 mg/mL:
| Target Dose | Volume to Draw |
|---|---|
| 2 mg | 0.20 mL (20 units) |
| 4 mg | 0.40 mL (40 units) |
| 8 mg | 0.80 mL (80 units) |
| 12 mg | 1.20 mL (use two 0.5 mL syringes or one 1 mL + one 0.5 mL) |
Store the reconstituted vial in the refrigerator at 2-8°C and use within 30 days.
Retatrutide Side Effects by Dose
Side effects are dose-dependent and concentrated in the gastrointestinal system. The Phase 2 trial provides the most detailed safety data by dose level. For a comprehensive safety guide including Phase 3 TRIUMPH-4 data, dysesthesia, cardiovascular effects, and management strategies, see our complete retatrutide side effects guide.
Common Side Effects
| Side Effect | 1 mg | 4 mg | 8 mg | 12 mg | Placebo |
|---|---|---|---|---|---|
| Nausea | Mild | Moderate | Moderate | Moderate-Severe | Rare |
| Diarrhea | Rare | Mild | Moderate | Moderate | Rare |
| Vomiting | Rare | Mild | Mild-Moderate | Moderate | Rare |
| Decreased appetite | Mild | Moderate | Strong | Strong | None |
| Constipation | Rare | Mild | Mild | Mild | Rare |
Most GI side effects occur during the first 1-2 weeks of each dose escalation and resolve without intervention. The severity was described as "mostly mild to moderate" in the NEJM publication, with events partially mitigated by the lower starting dose protocol (Jastreboff et al., NEJM 2023).
Why Starting at 2 mg Matters
The Phase 2 trial included a direct comparison: one 4 mg group started at 2 mg and escalated, while another started directly at 4 mg. The direct-start group experienced nearly double the rate of GI adverse events.
This finding changed the protocol for all subsequent trials. Every Phase 3 TRIUMPH trial uses the 2 mg starting dose. Skipping the initial titration is the single most common mistake in retatrutide dosing, and it is entirely avoidable.
If you have used GLP-1 agonists before and tolerated them well, you still start at 2 mg. Prior GLP-1 experience does not eliminate the need for titration with retatrutide, because the glucagon receptor component introduces a new variable.
Managing Side Effects
Practical strategies that reduce GI symptoms during dose escalation:
- Eat smaller meals. Large meals overwhelm a slowed gastric emptying system. Five small meals outperforms three large ones during titration.
- Avoid fatty foods. Fat takes the longest to digest and is most likely to trigger nausea when gastric emptying is delayed.
- Stay hydrated. Diarrhea and vomiting deplete fluids. Aim for 2-3 liters of water daily.
- Time the injection after your last meal. Injecting before bed, after dinner, means any nausea peaks during sleep.
- Delay escalation if needed. If side effects at 4 mg are intolerable, stay at 4 mg for 6-8 weeks instead of 4. The clinical protocol allowed dose delays.
- Ginger or peppermint tea. Anecdotally helpful for mild nausea. No clinical data, but no risk.
Retatrutide vs Tirzepatide vs Semaglutide: Dosage Comparison
All three peptides use once-weekly subcutaneous dosing, but the dose ranges, titration schedules, and maintenance doses differ significantly.
| Parameter | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Brand name | Ozempic / Wegovy | Mounjaro / Zepbound | Investigational (LY3437943) |
| Mechanism | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Starting dose | 0.25 mg | 2.5 mg | 2 mg |
| Max dose | 2.4 mg | 15 mg | 12 mg |
| Titration duration | 16-20 weeks | 16-20 weeks | 12 weeks |
| Injection frequency | Weekly | Weekly | Weekly |
| Max weight loss (trials) | ~15% | ~22.5% | ~28.7% |
| FDA approved | Yes (2021) | Yes (2022) | No (expected 2027) |
Retatrutide reaches its maximum dose faster (12 weeks vs 16-20 weeks) and produces greater weight loss despite a lower milligram dose than tirzepatide. The glucagon receptor activation drives energy expenditure in a way the other two cannot match.
For patients switching from semaglutide or tirzepatide to retatrutide, there is no established crossover protocol. The standard recommendation: start retatrutide at 2 mg regardless of prior dose, then titrate normally. Do not attempt to "match" your prior dose from another peptide.
Retatrutide Dosage FAQ
Frequently Asked Questions
What is the maximum dose of retatrutide?
The maximum studied dose is 12 mg once weekly. This was the highest dose in the Phase 2 trial (NEJM 2023) and the Phase 3 TRIUMPH-4 trial. Higher doses have not been tested in humans.
How often do you inject retatrutide?
Once per week, subcutaneously, on the same day each week. The half-life of approximately 6 days supports weekly dosing. The time of day and relation to meals do not significantly affect efficacy.
How long does retatrutide take to work?
Appetite suppression begins within the first week at the 2 mg starting dose. Measurable weight loss is typically visible by weeks 4-8. In the Phase 2 trial, participants lost an average of 17.5% body weight by week 24 on the 12 mg dose.
Is retatrutide FDA approved?
No. As of March 2026, retatrutide is investigational. Phase 3 TRIUMPH trials are ongoing, with results expected throughout 2026. FDA approval is projected for 2027 at the earliest. See our how to get retatrutide guide for the full FDA timeline, clinical trial enrollment steps, and what alternatives are available now.
How much weight can you lose on retatrutide?
In clinical trials, the 12 mg dose produced 24.2% body weight loss at 48 weeks (Phase 2) and 28.7% at 68 weeks (Phase 3 TRIUMPH-4). At 12 mg, 100% of participants lost at least 5% of body weight and 83% lost at least 15%.
Can you take retatrutide orally?
Not currently. All clinical trials use subcutaneous injection. Eli Lilly has an oral formulation (orforglipron) in development, but it targets different receptors. An oral retatrutide formulation has not been announced.
What happens if you miss a retatrutide dose?
If fewer than 3 days have passed since the missed dose, inject as soon as you remember. If more than 3 days have passed, skip the missed dose and resume on the next scheduled injection day. Do not double up.
What is the difference between retatrutide and tirzepatide?
Tirzepatide activates two receptors (GLP-1 and GIP). Retatrutide activates three (GLP-1, GIP, and glucagon). The additional glucagon receptor activation increases energy expenditure and fat burning, producing approximately 6 percentage points more weight loss in clinical trials (28.7% vs 22.5%).
The Bottom Line
Retatrutide dosing follows a straightforward 12-week titration: 2 mg for four weeks, 4 mg for four weeks, 8 mg for four weeks, then 12 mg maintenance. Once weekly, subcutaneous, same day each week.
The clinical data speaks clearly. Phase 2 showed 24.2% weight loss at 48 weeks. Phase 3 confirmed 28.7% at 68 weeks. No other peptide in development has matched these results. The 100% response rate at the ≥5% threshold is unprecedented.
Start at 2 mg. Do not skip the titration. The Phase 2 data proved that starting higher doubles GI side effects with no benefit to long-term outcomes.
For help planning your protocol, use our retatrutide dosage calculator. For reconstitution volumes, use the reconstitution calculator. For storage, see how to store peptides and how long do reconstituted peptides last. For the full mechanism explanation, see how does retatrutide work. For safety, see the peptide safety guide. For the regulatory landscape, see FDA peptide crackdown 2026. Explore all peptide profiles, calculators, and research tools at PeptidesExplorer.
Related Articles: - Retatrutide: Complete Guide - Retatrutide Side Effects - Retatrutide vs Tirzepatide - How to Get Retatrutide - Best Peptides for Weight Loss
Helpful Tools
Related Articles
Retatrutide vs Tirzepatide Compared
Retatrutide vs tirzepatide compared: 28.7% vs 22.5% weight loss, triple vs dual agonist mechanism, side effects, dosing, cost, and FDA status.
Retatrutide Availability & Timeline
Retatrutide is expected to receive FDA approval in mid-2027 with pharmacy availability in late 2027. Full timeline and Phase 3 trial status.
Retatrutide: Mechanism & Trial Results
Retatrutide (LY3437943) is a triple agonist weight loss drug by Eli Lilly. 28.7% weight loss in trials, 82% liver fat reduction. Not yet FDA-approved.
How to Get Retatrutide in 2026
How to access retatrutide in 2026: clinical trial enrollment, compounding pharmacy status, FDA approval timeline (est. 2027). Updated March 2026.