
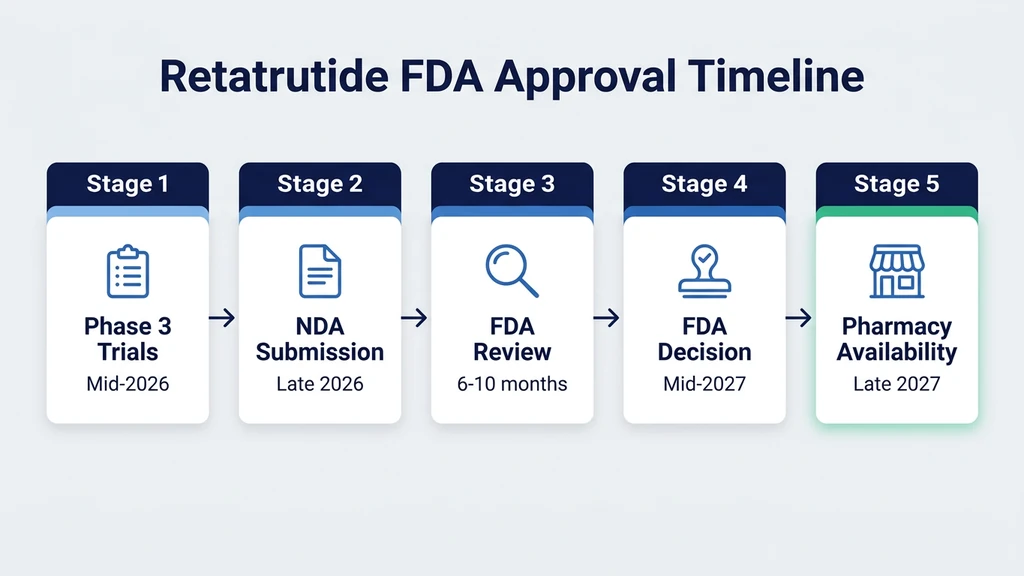
On December 19, 2025, Eli Lilly announced that retatrutide produced 28.7% body weight loss in the Phase 3 TRIUMPH-4 trial. That translates to roughly 71 pounds off a 249-pound starting weight. Retatrutide is not available yet. The earliest realistic FDA approval date is mid-2027, with pharmacy shelves stocked by late 2027 or early 2028.
Think of the drug approval process like building a house. You need architectural plans (Phase 3 data), a building permit (FDA submission), an inspection (FDA review), and finally a certificate of occupancy (approval) before anyone can move in. Retatrutide is still in the architectural plans phase. Seven large-scale trials are running. Two will finish mid-2026. Then Eli Lilly files the paperwork, the FDA reviews it for 6 to 10 months, and only then can your doctor write a prescription.
| Quick Reference | Details |
|---|---|
| Drug name | Retatrutide (LY3437943) |
| Manufacturer | Eli Lilly and Company |
| Mechanism | Triple agonist: GLP-1 + GIP + Glucagon |
| Max weight loss in trials | 28.7% body weight (71 lbs) at 68 weeks |
| Current status | Phase 3 clinical trials (not approved anywhere) |
| Expected NDA submission | Late 2026 |
| Expected FDA decision | Mid-2027 |
| Expected pharmacy availability | Late 2027 to early 2028 |
| Current access options | Clinical trials, expanded access (rare) |
This article walks through every milestone between now and the day you can fill a retatrutide prescription. We cover where each Phase 3 trial stands, what Eli Lilly has said publicly, how this timeline compares to semaglutide and tirzepatide approvals, and what you can do while waiting. For a complete overview of the drug itself, see our retatrutide guide. For side effect data, see retatrutide side effects.
This is educational content about an experimental drug. Consult a healthcare provider before making any medical decisions.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Where Does Retatrutide Stand Right Now?
As of March 2026, retatrutide is an investigational drug in Phase 3 clinical trials. No regulatory agency on Earth has approved it. You cannot get a prescription for it from any doctor, and no pharmacy can legally dispense it.
Here is what that means in practical terms. Phase 3 is the final and largest testing phase before a company can ask the FDA for approval. Eli Lilly is running eight trials under the umbrella name TRIUMPH, enrolling over 5,800 patients across multiple countries. These trials test retatrutide for obesity, type 2 diabetes, cardiovascular disease, knee osteoarthritis, and liver disease (Giblin et al., Diabetes, Obesity and Metabolism 2026).
One trial has already finished. TRIUMPH-4 (445 patients with knee osteoarthritis) reported results in December 2025, showing the 28.7% weight loss figure that made headlines worldwide (Lilly Press Release, Dec 2025). The remaining trials are still active and will report throughout 2026.
The TRIUMPH Trial Program: Trial-by-Trial Status
Each trial in the TRIUMPH program serves a specific purpose. The FDA needs data across different patient populations to feel confident that the drug is both safe and effective broadly, not just in one narrow group.
| Trial | Focus | Patients | NCT ID | Status (March 2026) |
|---|---|---|---|---|
| TRIUMPH-1 | Obesity (primary) | 2,300 | NCT05929066 | Active, results expected mid-2026 |
| TRIUMPH-2 | Type 2 diabetes + obesity | 1,000 | NCT05929079 | Active, results expected mid-2026 |
| TRIUMPH-3 | Cardiovascular outcomes | 1,800 | NCT05882045 | Active, long-term |
| TRIUMPH-4 | Knee osteoarthritis | 445 | NCT05931367 | Completed (Dec 2025) |
| TRIUMPH-Outcomes | CV/kidney long-term | TBD | NCT06383390 | Enrolling |
| Head-to-head | Retatrutide vs tirzepatide | 800 | NCT06662383 | Active, results expected Dec 2026 |
| Maintenance | Dose reduction | TBD | NCT06859268 | Recruiting |
| New enrollment | Obesity/overweight | 250 | NCT07232719 | Recruiting |
TRIUMPH-1 and TRIUMPH-2 are the pivotal trials. "Pivotal" means the FDA considers their results the primary evidence for an approval decision. Both are expected to report results by mid-2026. Without positive results from these two trials, the entire timeline shifts later. With positive results, Eli Lilly can begin assembling the application immediately.
The head-to-head trial against tirzepatide (NCT06662383) is particularly interesting. This is the first trial directly comparing retatrutide and tirzepatide in 800 patients under identical conditions. Results are expected in December 2026. For an existing comparison based on separate trials, see our retatrutide vs tirzepatide analysis.
What the Phase 2 Data Already Proved
The Phase 2 trial published in the New England Journal of Medicine in June 2023 was what put retatrutide on the map. In that trial, 338 adults with obesity received weekly injections for 48 weeks. People on the highest dose (12 mg) lost 24.2% of their body weight. Some participants lost over 30% (Jastreboff et al., NEJM 2023).
That was Phase 2, designed mainly to figure out the right dose and confirm the drug actually works. Phase 3 extends the testing to larger groups, longer durations, and more diverse patient populations. The TRIUMPH-4 result of 28.7% at 68 weeks exceeded the Phase 2 result, suggesting the effect strengthens with longer treatment.
A substudy within the Phase 2 trial also found that retatrutide reduced liver fat by up to 82% in patients with metabolic dysfunction-associated steatotic liver disease (MASLD), formerly called fatty liver disease. That reduction occurred within 48 weeks and exceeded the results of any other drug tested for that condition (Sanyal et al., NEJM 2024). For a deeper look at the mechanism behind these results, see how does retatrutide work.
The FDA Approval Timeline: Step by Step
Drug approval is not a single event. It is a sequence of regulatory steps, each with its own timeline. No step can be skipped, and each one must be completed before the next can begin.
Step 1: Complete Pivotal Trials (Mid-2026)
Eli Lilly needs final data from TRIUMPH-1 (2,300 obesity patients) and TRIUMPH-2 (1,000 type 2 diabetes patients). These are the trials the FDA will scrutinize most closely. Both are expected to deliver top-line results around the middle of 2026.
"Top-line results" means the key findings: did people lose significantly more weight than placebo, and was the drug safe enough? The detailed analysis, including subgroup breakdowns, long-term safety data, and secondary endpoints, takes additional months to compile.
Based on TRIUMPH-4 and Phase 2 data, analysts widely expect positive results. But biology can surprise. If a serious safety signal emerges in a larger population, the timeline could extend by a year or more. In the Phase 3 TRIUMPH-4 trial, a new safety signal called dysesthesia (abnormal skin sensations like tingling or burning) appeared in up to 20.9% of patients on the highest dose. The FDA will want to understand this signal fully before approving the drug. For details on known side effects, see our retatrutide side effects guide.
Step 2: Assemble and Submit the NDA (Late 2026)
After the pivotal trials finish, Eli Lilly assembles a New Drug Application (NDA). This is a massive document, sometimes exceeding 100,000 pages, that contains every piece of data the FDA needs to evaluate the drug: efficacy results, safety data, manufacturing details, proposed labeling, and the plan for post-approval monitoring.
Preparing an NDA typically takes 4 to 6 months after the last pivotal trial reports. Analysts expect Lilly to submit in late 2026, possibly Q4. The company has not publicly confirmed a submission date, but its quarterly investor calls have consistently described retatrutide as a "late-stage pipeline priority."
Lilly could also pursue a pre-NDA meeting with the FDA, where the agency provides informal guidance on what the application should contain. This meeting helps avoid last-minute surprises that could delay the review. Given the scale and importance of the TRIUMPH program, a pre-NDA meeting is almost certain.
Step 3: FDA Review Period (6 to 10 Months)
Once the FDA accepts the NDA for review, it has 10 months for a Standard Review or 6 months for a Priority Review. Priority Review is granted when a drug represents a "significant improvement" over existing treatments.
Does retatrutide qualify for Priority Review? There is a strong argument that it does. No approved drug produces 28.7% weight loss. The closest, tirzepatide, peaks around 22.5%. That 6-percentage-point gap is clinically meaningful, representing an additional 15 to 20 pounds of weight loss for the average patient. The liver fat reduction data and potential cardiovascular benefits add further justification.
However, the FDA has been cautious with weight loss drugs historically. The agency may want the full 10-month Standard Review to thoroughly examine the dysesthesia signal and the cardiovascular safety data from TRIUMPH-3. If TRIUMPH-3 is not complete by the time of submission, the FDA may require Lilly to submit that data as a post-marketing commitment, which would allow approval to proceed but with conditions.
| Review Type | Duration | Likelihood for Retatrutide |
|---|---|---|
| Priority Review | 6 months | Possible (unprecedented weight loss) |
| Standard Review | 10 months | More likely (cautious FDA approach) |
If Lilly submits in Q4 2026 and receives Standard Review, the FDA decision would come around August to October 2027. With Priority Review, the decision could arrive as early as April to June 2027.
Step 4: Advisory Committee Meeting (If Needed)
The FDA sometimes convenes an independent panel of outside experts, called an Advisory Committee, to publicly debate whether a drug should be approved. These meetings are not mandatory. The FDA skipped the advisory committee for tirzepatide's obesity indication (Zepbound) in 2023, approving it directly.
For retatrutide, an advisory committee meeting is possible but not certain. The dysesthesia safety signal and the novel triple-agonist mechanism might prompt the FDA to seek external input. If scheduled, the meeting typically occurs 1 to 2 months before the approval decision date (called the PDUFA date). The committee's recommendation is non-binding but almost always followed.
Advisory committees rarely derail drugs that have strong efficacy data. The bigger risk is a "complete response letter," where the FDA asks for additional data or changes before granting approval. This would push the timeline back by 6 to 12 months. Complete response letters have become uncommon for obesity drugs with large, well-run trial programs (FDA CDER).
Step 5: Commercial Launch (Late 2027 to Early 2028)
FDA approval is not the same as pharmacy availability. After approval, Eli Lilly needs to manufacture enough supply, negotiate insurance coverage, set up distribution, and train healthcare providers on the new drug. Based on how Lilly launched Zepbound (tirzepatide for weight loss), this ramp-up takes 2 to 4 months.
Zepbound was approved on November 8, 2023, and was available at pharmacies within a few weeks. But it took months for insurance coverage to expand, and supply shortages persisted into 2024. Lilly has learned from that experience and is likely investing in manufacturing capacity for retatrutide well ahead of approval.
| Milestone | Best Case | Most Likely |
|---|---|---|
| Pivotal trial results | Mid-2026 | Mid-2026 |
| NDA submission | Q3 2026 | Q4 2026 |
| FDA decision | Q2 2027 | Q3-Q4 2027 |
| First prescriptions filled | Q3 2027 | Q4 2027 to Q1 2028 |
| Broad insurance coverage | Q4 2027 | Mid-2028 |
The cost at launch will likely mirror Zepbound's pricing: approximately $1,000 to $1,100 per month before insurance. Eli Lilly typically offers manufacturer savings cards that can reduce the out-of-pocket cost to $25 per month for eligible patients. Use our peptide cost calculator to compare current options. For details on insurance dynamics, see tirzepatide cost with insurance.
How Retatrutide's Timeline Compares to Semaglutide and Tirzepatide

Historical comparisons help calibrate expectations. Both semaglutide and tirzepatide followed similar paths to the one retatrutide is on now. Looking at their timelines reveals how long each step actually took.
Semaglutide: From Phase 3 to Pharmacy in 3 Years
Novo Nordisk began the STEP clinical trial program for semaglutide (Wegovy) in 2018. The STEP 1 trial published results in the New England Journal of Medicine in February 2021, showing 14.9% weight loss at 68 weeks (Wilding et al., NEJM 2021).
The NDA for Wegovy was submitted in December 2020, before the pivotal trial was even published. The FDA approved Wegovy on June 4, 2021, using Priority Review. Total time from first Phase 3 enrollment to approval: roughly 3 years. However, supply shortages delayed widespread availability until late 2023.
| Semaglutide (Wegovy) | Date |
|---|---|
| STEP 1 trial start | June 2018 |
| NDA submission | December 2020 |
| FDA approval | June 2021 |
| Widespread availability | Late 2023 |
For a look at semaglutide results, see semaglutide before and after.
Tirzepatide: From Phase 3 to Pharmacy in 2.5 Years
Eli Lilly started the SURMOUNT trial program for tirzepatide in late 2020. SURMOUNT-1 published in the New England Journal of Medicine in July 2022, showing 22.5% weight loss at 72 weeks (Jastreboff et al., NEJM 2022).
Lilly submitted the NDA for Zepbound in mid-2023 and received FDA approval on November 8, 2023. Total time from first Phase 3 enrollment to approval: roughly 3 years. Unlike semaglutide, Zepbound was available at pharmacies within weeks of approval, though supply constraints persisted for months.
| Tirzepatide (Zepbound) | Date |
|---|---|
| SURMOUNT-1 trial start | Late 2020 |
| SURMOUNT-1 results | July 2022 |
| NDA submission | Mid-2023 |
| FDA approval | November 2023 |
| Pharmacy availability | December 2023 |
For dosing guidance on tirzepatide, see how long does tirzepatide take to work.
What This Tells Us About Retatrutide
Both semaglutide and tirzepatide took approximately 3 years from first Phase 3 enrollment to FDA approval. The TRIUMPH program began enrolling in mid-2023. Applying the same 3-year window points to mid-2026 at the earliest and mid-2027 as the more realistic target.
Lilly has one advantage this time: institutional experience. The company navigated the tirzepatide approval process just two years ago. The regulatory team, manufacturing infrastructure, and commercial playbook are already in place. This could shave a few months off the timeline, particularly in the NDA preparation and launch phases.
The potential delay factor is the triple-agonist novelty. Semaglutide and tirzepatide had predecessor drugs in their same class that the FDA had already reviewed (liraglutide and native GIP, respectively). Retatrutide's glucagon component is new territory for the agency. The FDA may take extra time to evaluate the glucagon-related safety signals, particularly the heart rate increase (up to 6.7 bpm above baseline) and dysesthesia (Nature Medicine, 2024).
Why Is the Approval Taking So Long?
If retatrutide works so well, why not fast-track it immediately? The answer comes down to safety, scale, and regulatory precedent.
The FDA Requires Large-Scale Safety Data
Phase 2 tested retatrutide in 338 people. That is enough to confirm the drug works but not enough to catch rare side effects that might affect 1 in 500 or 1 in 1,000 patients. The Phase 3 TRIUMPH program enrolls over 5,800 people specifically to find those rare events.
Consider the math. A side effect that occurs in 0.1% of patients (1 in 1,000) has only a 28% chance of appearing in a 338-person trial. In a 5,800-person program, that same side effect has a 99.7% chance of being detected. The FDA wants that 99.7% confidence level before 50 million Americans start taking a new weight loss drug (FDA Guidance on Clinical Trial Size).
The dysesthesia signal illustrates why this matters. It was barely noticeable in Phase 2 but appeared in up to 20.9% of patients on the highest dose in Phase 3. Without the larger trial, this side effect might not have been characterized until after millions of people were already taking the drug.
The Glucagon Component Is Uncharted Territory
Every currently approved weight loss drug works through GLP-1 alone (semaglutide) or GLP-1 plus GIP (tirzepatide). Retatrutide adds glucagon receptor activation, something no approved drug does. The FDA has no direct precedent for reviewing a glucagon agonist for chronic weight management.
Glucagon's primary natural role is raising blood sugar when it drops too low. A drug that activates glucagon receptors carries theoretical risks: it could raise fasting blood sugar, accelerate muscle protein breakdown, or overstimulate the heart. In the clinical trials so far, blood sugar effects have been manageable (retatrutide actually improved blood sugar in diabetic patients), and muscle loss has been proportional to weight loss. The heart rate increase of 4 to 7 beats per minute is within acceptable range but requires monitoring (Coskun et al., Nature 2022).
The FDA will want long-term cardiovascular data to confirm that the heart rate increase does not translate into higher rates of heart attacks or strokes. TRIUMPH-3 (the cardiovascular outcomes trial with 1,800 patients) is designed to answer exactly this question. For a comparison of retatrutide vs tirzepatide including safety profiles, see our dedicated analysis.
Manufacturing a Novel Peptide at Scale
Retatrutide is a 39-amino-acid synthetic peptide with a fatty acid modification that extends its half-life. Manufacturing peptides at pharmaceutical scale is technically challenging. Each amino acid must be added in precise sequence, the fatty acid must be attached at the correct position, and the final product must be purified to greater than 99% purity (Lau & Dunn, Bioorganic & Medicinal Chemistry 2018).
Eli Lilly invested $4 billion in expanding its manufacturing facilities in 2024 and 2025, partly in anticipation of retatrutide demand. The company opened a new facility in Lebanon, Indiana, and expanded its Research Triangle Park site in North Carolina. These investments suggest Lilly is confident in approval and is building capacity ahead of it.
Manufacturing bottlenecks delayed semaglutide (Wegovy) availability for over two years after approval. Lilly appears determined to avoid repeating that mistake with retatrutide. For context on current peptide sourcing, see where to buy peptides in 2026 and peptide sciences alternatives.
Can You Access Retatrutide Before FDA Approval?
Yes, but only through narrow channels. There is no legal way to get a retatrutide prescription today. There are, however, two legitimate pathways and one gray-market option. For a comprehensive walkthrough, see our retatrutide: how to get it guide.
Clinical Trials: The Primary Access Route
Enrolling in a TRIUMPH trial is the only way to receive pharmaceutical-grade retatrutide under medical supervision. Everything is free: the drug, doctor visits, lab work, and monitoring. You may receive placebo (an inactive injection), but even placebo participants get extensive medical care throughout the study.
To find active trials near you, go to ClinicalTrials.gov and search for "retatrutide," or visit Eli Lilly's trial finder at trials.lilly.com. General eligibility requirements include a BMI of 30 or higher (or 27+ with a weight-related health condition), age 18 or older, and no history of pancreatitis or medullary thyroid carcinoma.
As of March 2026, two trials are actively recruiting new participants: the Maintenance trial (NCT06859268, testing dose reduction after initial weight loss) and a new enrollment trial (NCT07232719, 250 participants with obesity or overweight). Competition for spots is intense. Apply early and be prepared for a screening process that typically takes 2 to 4 weeks.
Expanded Access (Compassionate Use)
If you have a life-threatening obesity-related condition, have exhausted all approved treatments, and cannot enroll in a clinical trial, your doctor can request expanded access directly from Eli Lilly. Call 1-800-545-5979. The company responds within 5 business days.
This pathway is extremely narrow. Being overweight, even severely overweight, typically does not qualify. You would need complications like severe heart failure, respiratory failure, or organ damage directly attributable to obesity. Fewer than 1% of requests are granted for experimental weight loss drugs.
Research Peptides: The Gray Market (Risks Apply)
Some online vendors sell retatrutide labeled "for research use only, not for human consumption." These products are not manufactured to pharmaceutical standards, are not inspected by the FDA, and their purity varies widely between suppliers.
The FDA has taken aggressive enforcement action against peptide vendors since 2024. Over 50 warning letters were issued to GLP-1 compounders in September 2025 alone. Peptide Sciences, one of the largest research vendors, shut down voluntarily in early 2026. The legal and safety landscape for research peptides is increasingly hostile. For the full regulatory picture, see FDA peptide crackdown 2026.
If you choose to go this route, understand the quantified risks. A 2024 FDA investigation of seized GLP-1 peptide samples found that 15% contained bacterial endotoxins exceeding safety limits. Another 8% had purity below 90%, meaning up to 10% of the vial contained unknown impurities. Some samples contained heavy metals including lead and cadmium at levels exceeding USP limits (FDA Safety Communication, 2025). For general safety guidance, see our peptide safety guide.
What Will Retatrutide Cost When It Becomes Available?
Nobody knows the exact price yet, but Eli Lilly's pricing history with tirzepatide provides a reasonable estimate.
Expected Retail Pricing
Zepbound (tirzepatide for weight loss) launched at $1,059.87 per month. Mounjaro (tirzepatide for diabetes) was priced at $1,023.04 per month. Retatrutide will almost certainly fall in a similar range, possibly slightly higher given the additional receptor target and superior efficacy data.
For context, Wegovy (semaglutide for weight loss) costs $1,349.02 per month. Ozempic (semaglutide for diabetes) costs $935.77 per month. Weight loss drugs from the same manufacturer tend to be priced at a premium over diabetes versions of similar drugs.
| Drug | Monthly List Price | Manufacturer |
|---|---|---|
| Wegovy (semaglutide) | $1,349 | Novo Nordisk |
| Ozempic (semaglutide) | $936 | Novo Nordisk |
| Zepbound (tirzepatide) | $1,060 | Eli Lilly |
| Mounjaro (tirzepatide) | $1,023 | Eli Lilly |
| Retatrutide (est.) | $1,000 to $1,200 | Eli Lilly |
Use the peptide cost calculator to compare costs across available options.
Insurance Coverage Expectations
Insurance coverage for weight loss drugs remains inconsistent. As of early 2026, most commercial insurance plans cover Mounjaro (diabetes indication) but fewer cover Zepbound (weight loss indication). Medicare does not cover any weight loss drugs.
Legislation is moving in the right direction. The Treat and Reduce Obesity Act has been reintroduced in Congress multiple times, and some state Medicaid programs have begun covering GLP-1 agonists for obesity. By the time retatrutide reaches market in late 2027 or 2028, coverage may have expanded significantly.
Eli Lilly will almost certainly offer a manufacturer savings card for retatrutide, similar to the Zepbound savings card that reduces the monthly cost to $25 for commercially insured patients. These programs typically exclude government insurance (Medicare, Medicaid, Tricare). For a breakdown of current insurance dynamics, see tirzepatide cost with insurance.
Will Compounding Pharmacies Make Generic Retatrutide?
Not legally. US law only permits compounding of drugs that are already FDA-approved and either on the drug shortage list or not commercially available in a needed form. Retatrutide has never been approved, so there is no legal basis for compounding it.
Even after approval, compounding would only be permitted if retatrutide entered the FDA's drug shortage list, which happened with both semaglutide and tirzepatide. Lilly's manufacturing investments suggest the company is trying to avoid shortage scenarios. For background on the compounding landscape, see is compound tirzepatide safe and compound semaglutide with B12.
True generic competition is even further away. Eli Lilly's patents on retatrutide extend into the 2040s. A generic version would require those patents to expire or be successfully challenged, plus a separate FDA approval process for the generic. You should not expect a generic retatrutide for at least 15 years after initial approval.
What Should You Do While Waiting for Retatrutide?
The 18 to 24 months between now and retatrutide availability is not wasted time. You have options that work today.
Consider Currently Available Medications
Two effective weight loss drugs are available by prescription right now. Tirzepatide (sold as Zepbound for weight loss) targets two of the three receptors that retatrutide hits and produces about 22.5% body weight loss. Semaglutide (sold as Wegovy for weight loss) targets one receptor and produces about 15% loss.
Starting one of these medications today does not prevent you from switching to retatrutide later. Both tirzepatide and retatrutide are made by Eli Lilly, and the transition would involve a standard dose titration protocol. See our retatrutide dosage guide for how titration works. For guidance on transitioning between GLP-1 drugs, see semaglutide to tirzepatide switching.
Your doctor can help determine which option fits your medical history, insurance coverage, and goals. If cost is a barrier, manufacturer savings cards can reduce Zepbound to $25 per month for eligible patients. For injection technique, see how to inject tirzepatide.
Explore Clinical Trial Enrollment
Two TRIUMPH trials are still recruiting as of March 2026. The maintenance trial (NCT06859268) tests whether patients can reduce their retatrutide dose after achieving significant weight loss. The new enrollment trial (NCT07232719) has 250 spots for adults with obesity or overweight.
Search for open trials at ClinicalTrials.gov or trials.lilly.com. Be prepared to provide your BMI, medical history, and current medications during the screening process. If your nearest trial site is far away, some trials cover travel expenses. Ask the study coordinator when you call.
Build the Foundation That Amplifies Results
Weight loss drugs produce better outcomes when combined with behavioral changes. Clinical trials require participants to follow a reduced-calorie diet and increase physical activity, and the weight loss numbers reflect that combination. The drug alone produces less.
Start tracking measurable health markers now: weight, waist circumference, blood pressure, fasting blood sugar, and cholesterol. This creates a baseline so you can accurately measure the impact of any treatment you eventually start. It also documents your weight management efforts, which insurance companies and clinical trials look for.
For broader guidance on beginning a peptide-based protocol, see getting started with peptides. For preparation and storage fundamentals, see how to store peptides and how to reconstitute retatrutide.
What Could Delay or Accelerate the Timeline?
The mid-2027 approval estimate is based on current trajectory. Several factors could shift it in either direction.
Factors That Could Speed Things Up
Breakthrough Therapy Designation. If the FDA grants this designation (which it has not yet), Lilly would receive more intensive FDA guidance and a potentially faster review. The 28.7% weight loss and 82% liver fat reduction could justify this designation, particularly for MASLD, which currently has limited treatment options.
Positive TRIUMPH-1 and TRIUMPH-2 results. If both pivotal trials deliver results as strong as or stronger than TRIUMPH-4, Lilly could submit the NDA as early as Q3 2026 rather than Q4, shaving 1 to 3 months off the timeline.
Rolling submission. The FDA sometimes allows companies to submit sections of the NDA as they become ready, rather than waiting for the complete application. This can save 2 to 4 months. Lilly used this approach with tirzepatide.
Factors That Could Slow Things Down
Safety signals in larger trials. If TRIUMPH-1 or TRIUMPH-2 reveal a new safety concern not seen in smaller trials, the FDA could require additional studies. The dysesthesia signal from TRIUMPH-4 is already on the agency's radar. If the rate or severity increases in larger populations, it could prompt a complete response letter.
Manufacturing delays. Peptide manufacturing at pharmaceutical scale is technically demanding. If Lilly encounters production problems, it could delay the launch even after approval. The company's $4 billion manufacturing investment is designed to prevent this, but technical issues can still arise.
Regulatory environment changes. The FDA's approach to obesity drugs has been evolving. A change in FDA leadership, new safety requirements, or shifts in regulatory philosophy could add review time. The current FDA commissioner has been supportive of obesity drug development, but political changes are unpredictable.
Complete response letter. If the FDA issues a complete response letter (asking for more data rather than approving), the delay is typically 6 to 12 months. This happened to lorcaserin (Belviq) and naltrexone/bupropion (Contrave) before their eventual approvals.
International Availability: When Will Other Countries Approve Retatrutide?
FDA approval does not automatically make retatrutide available worldwide. Each country or region has its own regulatory process.
European Medicines Agency (EMA)
Eli Lilly typically files for European approval (through the EMA's centralized procedure) around the same time as the FDA submission, sometimes within a few months. The EMA review process takes approximately 12 to 15 months, longer than the FDA. If Lilly files with both agencies in late 2026, European approval could come in early to mid-2028.
However, individual European countries then negotiate pricing and reimbursement, which adds another 6 to 18 months before the drug is actually available at pharmacies in each country. UK approval through the MHRA may come separately and could be faster since Brexit allowed the UK to conduct independent reviews.
Other Markets
Japan, Australia, Canada, and several other countries have their own approval pathways. Japan's PMDA often requires Japan-specific clinical data, which could delay approval there. Australia's TGA and Canada's Health Canada sometimes rely on FDA or EMA reviews to expedite their own processes.
The bottom line: if you are outside the United States, expect retatrutide availability 6 to 18 months after FDA approval, depending on your country's regulatory timeline and Lilly's submission strategy.
How Retatrutide Fits Into the Weight Loss Drug Pipeline
Retatrutide is not the only next-generation weight loss drug in development. Several competitors are pursuing similar or alternative approaches, and the competitive landscape could influence pricing, availability, and insurance coverage.
Orforglipron (Eli Lilly): The Oral Option
Eli Lilly is also developing orforglipron, an oral (pill) GLP-1 agonist. In Phase 3 trials, orforglipron produced approximately 12 to 14% weight loss. That is less than injectable retatrutide but comes in a pill format that many patients prefer over weekly injections. Orforglipron is on a similar timeline to retatrutide, with potential approval in 2027 or 2028.
Lilly may position retatrutide as the maximum-efficacy injectable option and orforglipron as the more convenient oral option, giving doctors and patients a choice within Lilly's own portfolio.
Amycretin (Novo Nordisk): The Dual Approach
Novo Nordisk, the maker of Ozempic and Wegovy, is developing amycretin, a dual GLP-1 and amylin agonist. Early data showed approximately 22% weight loss in 36 weeks, which extrapolates to potentially 25% or more at 68 weeks. This positions amycretin as a direct competitor to retatrutide, likely reaching market around the same time.
The existence of multiple competitors is actually good for patients. Competition typically drives prices down, expands insurance coverage, and gives doctors more options to match individual patient needs. For a perspective on existing drug comparisons, see retatrutide vs tirzepatide.
Survodutide (Boehringer Ingelheim): Another Glucagon Approach
Survodutide is a dual GLP-1 and glucagon agonist (it skips GIP). Phase 2 results showed approximately 19% weight loss at 46 weeks. This drug targets the same glucagon receptor as retatrutide but without the GIP component. If survodutide reaches market before retatrutide, it could provide early real-world evidence on glucagon receptor safety, which would benefit the retatrutide review process.
Key Dates to Watch in 2026 and 2027
If you are tracking retatrutide's progress toward availability, these are the dates and events that matter most.
| When | What to Watch For |
|---|---|
| Q2 2026 | TRIUMPH-1 and TRIUMPH-2 top-line results |
| Q3 2026 | Lilly earnings call commentary on NDA timeline |
| Q4 2026 | Potential NDA submission to FDA |
| December 2026 | Head-to-head vs tirzepatide results |
| Q1 2027 | FDA acceptance of NDA, PDUFA date announced |
| Q2-Q3 2027 | Potential advisory committee meeting |
| Mid to Late 2027 | FDA approval decision |
| Late 2027 to Q1 2028 | Commercial availability at pharmacies |
Set up free alerts on ClinicalTrials.gov for "retatrutide" to be notified of trial updates. Follow Eli Lilly's investor relations page for press releases. We will update this article as each milestone is reached.
For the current regulatory landscape affecting all peptides, see FDA peptide crackdown 2026. For dosing reference across available peptides, see the peptide dosage chart. For reconstitution guidance, see the peptide reconstitution calculator.
Frequently Asked Questions
When will retatrutide be available at pharmacies?
The most realistic estimate is late 2027 to early 2028. Eli Lilly needs to finish Phase 3 trials (mid-2026), submit a New Drug Application (late 2026), and wait for the FDA to review it (6 to 10 months). Best case with Priority Review is Q3 2027. Standard Review pushes availability to Q4 2027 or Q1 2028.
Is retatrutide FDA-approved?
No. As of March 2026, retatrutide has not been approved by the FDA or any other regulatory agency worldwide. It is in Phase 3 clinical trials under Eli Lilly's TRIUMPH program. No doctor can prescribe it and no pharmacy can legally fill it. The only legal access is through clinical trials or, in extreme cases, expanded access.
How much weight can you lose on retatrutide?
In the Phase 3 TRIUMPH-4 trial, participants on the highest dose (12 mg) lost an average of 28.7% of their body weight over 68 weeks. That translates to roughly 71 pounds from a 249-pound starting weight. Participants were still losing weight when the trial ended, suggesting the final number could be higher with longer use.
Can I buy retatrutide online right now?
Some vendors sell retatrutide labeled 'for research use only.' These products are not manufactured to pharmaceutical standards and are not regulated by the FDA. Testing of seized samples has found bacterial endotoxins, heavy metals, and purity levels below 90%. The FDA has issued dozens of warning letters to peptide vendors since 2024.
How does retatrutide compare to tirzepatide and semaglutide?
Retatrutide targets three receptors (GLP-1, GIP, glucagon) and produced 28.7% weight loss. Tirzepatide targets two receptors (GLP-1, GIP) and produced 22.5% weight loss. Semaglutide targets one receptor (GLP-1) and produced 15% weight loss. More receptors means more weight loss but also more side effects. Tirzepatide and semaglutide are available now. Retatrutide is not.
Will insurance cover retatrutide?
Unknown, but expectations are mixed. Coverage for weight loss drugs remains inconsistent across insurers. Based on tirzepatide precedent, diabetes-related indications will likely receive better coverage than obesity indications. Eli Lilly will almost certainly offer a manufacturer savings card to reduce out-of-pocket costs, similar to the $25/month Zepbound card.
What are the main side effects of retatrutide?
Side effect rates vary significantly by trial population. In TRIUMPH-4 (obesity + knee OA): nausea 43%, diarrhea 33%, dysesthesia 20.9%. In TRANSCEND-T2D-1 (type 2 diabetes): nausea 26.5%, diarrhea 22.8%, dysesthesia 4.4%. Heart rate increased by 4 to 7 beats per minute. Most side effects were mild to moderate and improved over time. See our retatrutide side effects guide for the full breakdown.
Can a compounding pharmacy make retatrutide?
No. US law only allows compounding of FDA-approved drugs. Since retatrutide has never been approved, compounding it is illegal. Even after eventual approval, compounding would only be permitted during a drug shortage. Eli Lilly is investing billions in manufacturing to prevent shortages like those that affected semaglutide and tirzepatide.
The Bottom Line
Retatrutide is the most powerful weight loss drug in development. The 28.7% body weight loss recorded in Phase 3 exceeds every other drug on the market or in testing. But power without availability is just a number on a research poster.
The realistic timeline: pivotal trial results by mid-2026, NDA submission by late 2026, FDA decision by mid to late 2027, and pharmacy availability by late 2027 or early 2028. That is 18 to 24 months from now.
Your options today are clear. If you qualify, enroll in a clinical trial for free pharmaceutical-grade access. If you need weight management now, tirzepatide (Zepbound) and semaglutide (Wegovy) are FDA-approved, available by prescription, and produce meaningful results (22.5% and 15% weight loss, respectively). Starting one now does not prevent switching to retatrutide later. For a side-by-side, see retatrutide vs tirzepatide.
Avoid unregulated research peptides unless you fully understand the risks: no pharmaceutical-quality standards, no FDA oversight, and documented contamination in seized samples. For the regulatory picture, see FDA peptide crackdown 2026.
We will update this article as each milestone is reached. Key dates: TRIUMPH-1 and TRIUMPH-2 results (mid-2026), head-to-head vs tirzepatide (December 2026), and any NDA filing announcement from Eli Lilly.
Related Articles: - Retatrutide: Complete Guide - Retatrutide Dosage Guide - Retatrutide Side Effects - How Does Retatrutide Work - Retatrutide: How to Get It - Retatrutide vs Tirzepatide - FDA Peptide Crackdown 2026 - Where to Buy Peptides 2026 - Getting Started with Peptides - Peptide Safety Guide
Helpful Tools
Related Articles
Retatrutide: Mechanism & Trial Results
Retatrutide (LY3437943) is a triple agonist weight loss drug by Eli Lilly. 28.7% weight loss in trials, 82% liver fat reduction. Not yet FDA-approved.
How to Get Retatrutide in 2026
How to access retatrutide in 2026: clinical trial enrollment, compounding pharmacy status, FDA approval timeline (est. 2027). Updated March 2026.
How Does Retatrutide Work?
Retatrutide activates 3 receptors (GLP-1, GIP, glucagon) to reduce appetite, improve metabolism, and burn fat. 28.7% weight loss in trials.
Retatrutide Side Effects: What 2 Phase 3 Trials Show (2026)
Retatrutide side effects from 2 Phase 3 trials (1,200+ patients): nausea 26-43%, dysesthesia 4-21%, heart rate +6.7 bpm. Dose-by-dose rates, comparison vs tirzepatide, and management strategies.