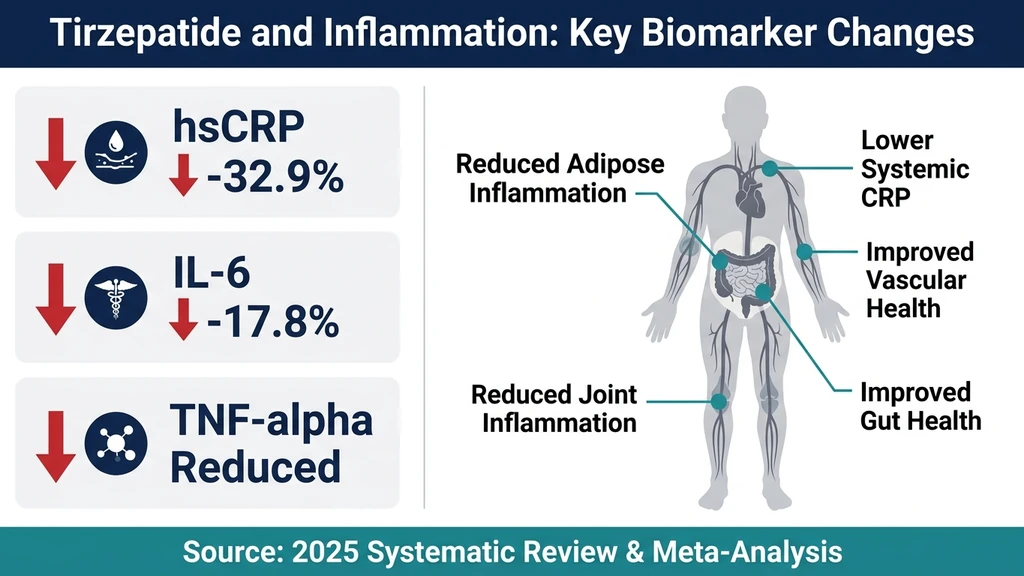
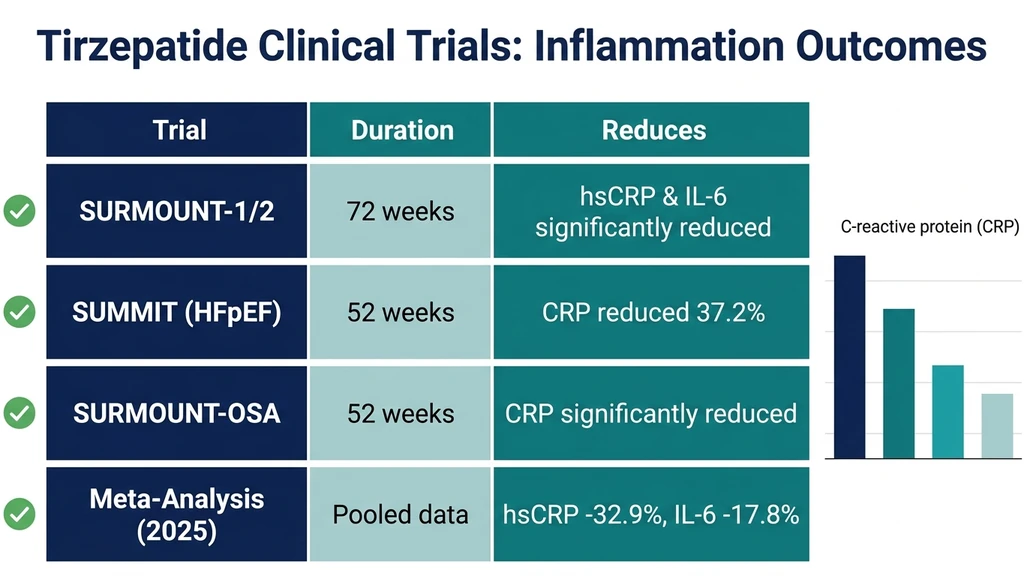
Your joints ache, your bloodwork shows elevated CRP, and your doctor mentioned chronic low-grade inflammation as a risk factor for heart disease. You are already considering tirzepatide for weight management. Yes, tirzepatide helps with inflammation. A 2025 systematic review and meta-analysis of multiple clinical trials found that tirzepatide reduced high-sensitivity C-reactive protein (hsCRP) by a mean of 32.9% and interleukin-6 (IL-6) by 17.8% compared to placebo (Abdelghany et al., Reviews in Endocrine and Metabolic Disorders, 2025). These reductions occurred across populations with obesity, type 2 diabetes, and heart failure.
But the picture is more nuanced than a single statistic. The anti-inflammatory effect operates through at least two distinct pathways, and the clinical relevance depends on your starting point, your dose, and how long you stay on treatment. This article breaks down every piece of published evidence so you can make an informed decision with your prescriber.
| Inflammatory Marker | Reduction vs. Placebo | Trial Source | Timeline |
|---|---|---|---|
| hsCRP | -32.9% (mean) | Pooled meta-analysis | 24-72 weeks |
| IL-6 | -17.8% (mean) | Pooled meta-analysis | 24-72 weeks |
| CRP (HFpEF) | -37.2% | SUMMIT trial | 52 weeks |
| CRP (OSA) | Significant reduction | SURMOUNT-OSA | 52 weeks |
Use our tirzepatide dosage calculator to determine your appropriate starting dose before discussing anti-inflammatory goals with your prescriber.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Inflammation Markers Does Tirzepatide Lower?
Tirzepatide's anti-inflammatory profile has been measured across several biomarkers in randomized controlled trials. The two most studied are hsCRP and IL-6, both of which are established predictors of cardiovascular disease and metabolic dysfunction.
High-sensitivity C-reactive protein (hsCRP) is produced by the liver in response to IL-6 signaling. Elevated hsCRP above 3.0 mg/L indicates high cardiovascular risk according to American Heart Association guidelines. Tirzepatide reduced hsCRP significantly at all three approved doses (5 mg, 10 mg, and 15 mg), with greater reductions at higher doses (Abdelghany et al., 2025).
Interleukin-6 (IL-6) is a pro-inflammatory cytokine secreted by adipose tissue, macrophages, and endothelial cells. It drives the acute-phase response and contributes to insulin resistance. The same meta-analysis found tirzepatide reduced IL-6 by a pooled mean of 17.8% versus placebo. IL-6 reduction correlated with but was not fully explained by weight loss, suggesting a direct pharmacological effect.
Beyond hsCRP and IL-6, emerging data suggest tirzepatide may reduce tumor necrosis factor-alpha (TNF-alpha) and monocyte chemoattractant protein-1 (MCP-1), though these markers have been studied less rigorously in human trials. Animal models show tirzepatide suppresses TNF-alpha induction through pathways that do not rely exclusively on neuronal GLP-1 receptors (Drucker et al., JCI, 2025). For a broader overview of peptides studied for their anti-inflammatory properties, see our guide on peptides for inflammation.
The SURMOUNT Trials: Inflammatory Biomarker Data
The SURMOUNT clinical trial program provides the most detailed evidence on tirzepatide's effects on inflammation in people with obesity. A post-hoc analysis of SURMOUNT-1 (obesity without diabetes) and SURMOUNT-2 (obesity with type 2 diabetes) examined changes in IL-6 and hsCRP at 24 and 72 weeks (Jastreboff et al., Circulation, 2024).
Early vs. Late Inflammatory Changes
At 24 weeks, tirzepatide-treated participants showed hsCRP reductions that were not fully associated with weight loss. This is a critical finding. It suggests tirzepatide exerts direct anti-inflammatory effects before the full magnitude of weight reduction occurs.
By 72 weeks, the relationship shifted. Changes in both IL-6 and hsCRP were predominantly weight-reduction-associated. In SURMOUNT-2 specifically, 78% of IL-6 changes and 57% of hsCRP changes at 72 weeks correlated with body weight reduction.
This two-phase pattern matters for clinical decision-making. Early inflammation reduction may reflect direct receptor-mediated effects of GLP-1 and GIP agonism. Late inflammation reduction is largely a consequence of losing 15-22% of body weight and the resulting decrease in visceral adipose tissue. Both contribute, but through different timelines.
Differences Between Diabetic and Non-Diabetic Populations
The relative contribution of weight loss to inflammatory marker reduction was more prominent in participants without type 2 diabetes. In people with diabetes, tirzepatide may have additional anti-inflammatory mechanisms related to improved glycemic control and reduced glucotoxicity.
This distinction is relevant if you are considering tirzepatide primarily for inflammation rather than weight loss. People with type 2 diabetes may derive a larger proportion of their anti-inflammatory benefit from the drug's direct metabolic effects rather than from weight reduction alone. For those switching from another GLP-1 medication, our semaglutide to tirzepatide switching guide covers the transition protocol.
The SUMMIT Trial: Inflammation in Heart Failure
The SUMMIT trial tested tirzepatide in patients with heart failure with preserved ejection fraction (HFpEF) and obesity. At 52 weeks, tirzepatide reduced C-reactive protein by an estimated 37.2% compared to placebo (Kosiborod et al., The Lancet, 2024).
HFpEF is increasingly understood as a disease driven in part by systemic inflammation and microvascular dysfunction. Excess adipose tissue secretes pro-inflammatory cytokines that impair cardiac function, increase vascular stiffness, and drive fluid retention. The 37.2% CRP reduction in SUMMIT suggests tirzepatide addresses one of the root mechanisms of obesity-related heart failure.
This finding has implications beyond cardiac patients. If tirzepatide reduces inflammation enough to improve heart failure outcomes, the same anti-inflammatory effect likely benefits other inflammatory conditions driven by obesity: metabolic syndrome, non-alcoholic fatty liver disease (NAFLD), and chronic joint pain. For those dealing with joint inflammation specifically, peptides for joint pain covers additional research-backed options.
How Tirzepatide Reduces Inflammation: Two Pathways

Tirzepatide's anti-inflammatory effects operate through two distinct but overlapping pathways. Understanding both helps explain why only 20-60% of the measured CRP reduction can be attributed to weight loss alone.
Direct Anti-Inflammatory Pathway (Receptor-Mediated)
Tirzepatide is a dual GLP-1/GIP receptor agonist. Both receptor types are expressed on immune cells, including macrophages, monocytes, and T cells. When tirzepatide activates these receptors, several downstream effects occur:
AMPK pathway activation. AMP-activated protein kinase (AMPK) is a master regulator of cellular energy sensing and inflammation. GLP-1 receptor agonists activate AMPK, which in turn suppresses the NF-kB signaling pathway. NF-kB is the primary transcription factor driving production of pro-inflammatory cytokines including TNF-alpha, IL-6, and IL-1beta (Drucker et al., JCI, 2025).
Macrophage polarization. Tirzepatide promotes a shift from M1 (pro-inflammatory) macrophages to M2 (anti-inflammatory) macrophages. M1 macrophages produce TNF-alpha and IL-6 and drive tissue damage. M2 macrophages produce IL-10 and TGF-beta and promote tissue repair. A 2024 study found tirzepatide inhibited M1-type macrophage infiltration in visceral adipose tissue and promoted M1 macrophage apoptosis through the ERK signaling pathway (Luo et al., International Immunopharmacology, 2024).
GIP-specific effects. Unlike pure GLP-1 agonists such as semaglutide, tirzepatide also activates GIP receptors. GIP receptor agonism contributes to adipose tissue remodeling and may enhance mitochondrial oxidative capacity in fat cells, reducing the inflammatory profile of adipose tissue independently of total fat mass reduction.
Indirect Anti-Inflammatory Pathway (Weight-Mediated)
Visceral adipose tissue is not inert storage. It functions as an endocrine organ, secreting pro-inflammatory adipokines including TNF-alpha, IL-6, MCP-1, and resistin. Every kilogram of visceral fat lost reduces the circulating pool of these inflammatory mediators.
Tirzepatide produces weight loss of 15-22% in clinical trials, significantly more than diet and exercise alone (typically 3-5%). This magnitude of fat loss translates directly to measurable reductions in inflammatory biomarkers. The SURMOUNT-2 data showing 78% of IL-6 changes at 72 weeks being weight-associated confirms this pathway is the dominant long-term mechanism.
The indirect pathway also involves improved insulin sensitivity. Insulin resistance itself promotes inflammation through elevated free fatty acids, oxidative stress, and endoplasmic reticulum stress in adipose tissue. By resolving insulin resistance, tirzepatide breaks a self-reinforcing cycle between metabolic dysfunction and chronic inflammation. If you want to understand how tirzepatide promotes fat loss specifically, see does tirzepatide burn fat.
Tirzepatide vs. Semaglutide: Anti-Inflammatory Comparison
Both tirzepatide and semaglutide reduce inflammatory markers, but head-to-head anti-inflammatory comparisons remain limited. The available data suggest differences in mechanism and potentially in magnitude.
Semaglutide is a pure GLP-1 receptor agonist. Its anti-inflammatory effects are well-documented in the SELECT trial, which showed reduced cardiovascular events in people with obesity. Semaglutide reduces hsCRP and other inflammatory markers primarily through GLP-1 receptor activation and secondary weight loss effects.
Tirzepatide adds GIP receptor agonism to the equation. In mice lacking neuronal GLP-1 receptors, semaglutide lost its acute anti-inflammatory effects while tirzepatide still prevented TNF-alpha induction (Drucker et al., JCI, 2025). This suggests tirzepatide has anti-inflammatory pathways that are independent of neuronal GLP-1 signaling.
| Feature | Tirzepatide | Semaglutide |
|---|---|---|
| Receptor targets | GLP-1 + GIP | GLP-1 only |
| Weight loss (mean) | 15-22% | 12-17% |
| hsCRP reduction | -32.9% (meta) | Significant (SELECT) |
| TNF-alpha suppression | GLP-1R independent | GLP-1R dependent |
| GIP-mediated adipose remodeling | Yes | No |
| Cardiovascular outcomes trial | Pending | SELECT (positive) |
The greater weight loss with tirzepatide may translate to larger indirect anti-inflammatory effects. The additional GIP receptor pathway may contribute direct anti-inflammatory effects not available through semaglutide alone. For a detailed comparison of these two drugs for weight loss specifically, see retatrutide vs. tirzepatide, which also covers the emerging triple-agonist class.
Adipose Tissue Inflammation: The Central Mechanism
Chronic low-grade inflammation in obesity originates primarily from visceral adipose tissue. Understanding this process clarifies why tirzepatide's effects on inflammation are so closely tied to its effects on body composition.
In lean individuals, adipose tissue macrophages represent about 10% of the cell population in fat deposits and are predominantly the anti-inflammatory M2 phenotype. In people with obesity, macrophages can comprise 40-50% of adipose tissue cells, and the majority shift to the pro-inflammatory M1 phenotype. These M1 macrophages produce TNF-alpha, IL-6, IL-1beta, and MCP-1 in quantities sufficient to raise systemic inflammatory markers.
How Tirzepatide Reverses Adipose Inflammation
Tirzepatide addresses adipose tissue inflammation through multiple mechanisms operating simultaneously:
1. Visceral fat reduction. By reducing total adipose tissue mass, tirzepatide decreases the absolute number of inflammatory macrophages. Fewer macrophages means less cytokine production.
2. Macrophage phenotype shift. Beyond reducing total macrophage numbers, tirzepatide promotes the conversion of remaining macrophages from M1 to M2 phenotype. The ERK signaling pathway modulation documented in animal studies drives M1 apoptosis while sparing M2 cells (Luo et al., 2024).
3. Adipocyte health improvement. Tirzepatide improves mitochondrial function in adipocytes, reducing the oxidative stress that triggers inflammatory signaling. Healthier adipocytes produce less MCP-1, which means fewer monocytes are recruited from the bloodstream to infiltrate fat tissue.
4. Crown-like structure reduction. In advanced obesity, dead adipocytes become surrounded by macrophages in structures called crown-like structures (CLS). These are hotspots of inflammatory cytokine production. Weight loss with tirzepatide reduces both adipocyte death and CLS density, directly lowering local and systemic inflammation.
For those interested in other peptides studied for adipose tissue effects, BPC-157 has shown anti-inflammatory properties in animal models, though through entirely different mechanisms. Our best peptides for weight loss guide covers the full landscape.
Clinical Implications: Who Benefits Most?
Not everyone with inflammation will benefit equally from tirzepatide's anti-inflammatory effects. The evidence points to specific populations where the benefit is most pronounced.
Obesity With Elevated CRP
People with BMI above 30 and hsCRP above 3.0 mg/L represent the strongest evidence base. The SURMOUNT trials enrolled this exact population and demonstrated consistent CRP reductions across all dose levels. If your inflammation is driven primarily by excess adipose tissue, tirzepatide addresses the root cause rather than masking the symptom.
The practical implication: request hsCRP testing before starting tirzepatide and at 6-month intervals during treatment. A declining CRP confirms the anti-inflammatory benefit and can motivate continued adherence during the dose titration phase, when tirzepatide side effects are most noticeable.
Type 2 Diabetes With Inflammatory Comorbidities
Type 2 diabetes creates a pro-inflammatory environment through glucotoxicity, lipotoxicity, and insulin resistance. Tirzepatide addresses all three metabolic drivers while simultaneously reducing inflammatory markers. The SURMOUNT-2 data showed that anti-inflammatory effects in diabetic participants had a larger weight-independent component than in non-diabetic participants.
If you have both type 2 diabetes and elevated inflammatory markers, tirzepatide offers dual benefit. The anti-inflammatory effects are additive to glycemic control. For guidance on maintaining your dose once target weight is reached, see our guide on tirzepatide maintenance dose after weight loss.
Heart Failure With Preserved Ejection Fraction
The SUMMIT trial established tirzepatide as the first incretin therapy to show benefit in HFpEF. The 37.2% CRP reduction is a key part of this benefit, alongside improvements in cardiac filling pressures and exercise capacity. If you have HFpEF and obesity, the anti-inflammatory effect of tirzepatide is not a side benefit. It is central to the therapeutic mechanism.
Who May Not Benefit
Inflammation driven by autoimmune conditions (rheumatoid arthritis, lupus, inflammatory bowel disease) involves different pathways than obesity-driven inflammation. While some preliminary interest exists in GLP-1 agonists for autoimmune conditions, no clinical trial has demonstrated tirzepatide reduces disease activity in these populations. Do not replace disease-modifying antirheumatic drugs (DMARDs) or biologics with tirzepatide for autoimmune inflammation.
Similarly, acute infection-driven inflammation (elevated CRP from pneumonia, COVID-19, surgical recovery) will not respond to tirzepatide. The drug targets chronic, low-grade, metabolically-driven inflammation specifically.
Dose-Response Relationship: Does More Tirzepatide Mean Less Inflammation?
The meta-analysis found dose-dependent anti-inflammatory effects. Higher tirzepatide doses produced greater reductions in hsCRP and IL-6 (Abdelghany et al., 2025).
5 mg dose: Significant hsCRP reduction versus placebo. This is the standard starting maintenance dose after titration from 2.5 mg.
10 mg dose: Greater hsCRP reduction than 5 mg. Corresponds to approximately 15-18% body weight loss in clinical trials.
15 mg dose: Greatest hsCRP reduction. Corresponds to 20-22% body weight loss. The maximum approved dose.
The dose-response relationship is partially but not fully explained by greater weight loss at higher doses. At each dose level, the anti-inflammatory effect exceeds what weight loss alone would predict, consistent with direct receptor-mediated anti-inflammatory action.
For practical dose selection, the decision should not be based on anti-inflammatory goals alone. Titrate based on tolerability and weight management targets as your prescriber directs. The anti-inflammatory benefit follows. Use our tirzepatide dosage calculator to understand your current dose in the correct units.
Timeline: When Do Anti-Inflammatory Effects Appear?
Based on the SURMOUNT post-hoc analysis and broader clinical trial data, the anti-inflammatory timeline follows a predictable pattern:
Weeks 0-12 (Titration Phase): During dose escalation from 2.5 mg to your target dose, inflammatory markers may begin to decline. The early hsCRP changes at 24 weeks that were not weight-associated suggest direct receptor-mediated effects begin within the first months. However, this is also the period when gastrointestinal side effects are most common. If you are considering tirzepatide and already track what you eat, our guide on what to eat on tirzepatide can help during this transition.
Weeks 12-24 (Early Treatment): Weight loss accelerates, and the indirect anti-inflammatory pathway begins contributing meaningfully. You may see hsCRP changes on bloodwork even before reaching your maximum dose.
Weeks 24-72 (Sustained Treatment): Both direct and indirect pathways are fully active. The largest measured CRP reductions occur during this window. The SUMMIT trial's 37.2% CRP reduction was measured at 52 weeks. The SURMOUNT post-hoc analysis showed the most significant IL-6 and hsCRP reductions at 72 weeks.
Beyond 72 weeks: Limited data exist beyond 72 weeks. Given that weight loss plateaus around 60-72 weeks for most patients, the anti-inflammatory benefit likely stabilizes in this timeframe as well. Maintaining the dose is critical. Stopping tirzepatide leads to weight regain, which would predictably reverse the indirect anti-inflammatory effects.
Practical Considerations for Inflammation Monitoring
If reducing inflammation is one of your treatment goals, structured monitoring helps you and your prescriber assess the response objectively.
Testing Protocol
Baseline (before starting tirzepatide): - hsCRP (not standard CRP; the high-sensitivity assay detects low-grade inflammation) - Fasting IL-6 (if available; not all labs offer this routinely) - Fasting lipid panel (inflammation markers correlate with lipid abnormalities) - HbA1c (if diabetic or prediabetic) - Body weight and waist circumference
At 6 months (24 weeks): - Repeat hsCRP and IL-6 - Compare against baseline - Document weight loss percentage - Assess whether CRP reduction exceeds what weight loss alone predicts
At 12 months (52 weeks): - Full repeat panel - Most patients will show maximum anti-inflammatory benefit by this point - Discuss long-term maintenance plan with prescriber
For understanding how tirzepatide before and after results look in practice, including inflammatory marker improvements reported by users, see our documentation of real-world treatment outcomes.
What Counts as a Clinically Meaningful CRP Reduction?
The American Heart Association classifies cardiovascular risk by hsCRP level: - Low risk: below 1.0 mg/L - Average risk: 1.0-3.0 mg/L - High risk: above 3.0 mg/L
Moving from above 3.0 mg/L to below 3.0 mg/L represents a meaningful risk category change. Based on the 32.9% mean CRP reduction seen with tirzepatide, a person starting at 5.0 mg/L could expect to reach approximately 3.4 mg/L, approaching the risk threshold. A person starting at 8.0 mg/L might reach 5.4 mg/L, still above threshold but substantially improved.
The higher your baseline CRP, the more absolute reduction you can expect. This is consistent with the general principle that people with more adipose-driven inflammation have more to gain from tirzepatide's dual mechanism.
Tirzepatide and Specific Inflammatory Conditions
While the primary evidence base involves obesity and diabetes, researchers are investigating tirzepatide's anti-inflammatory effects in several specific conditions.
Non-Alcoholic Fatty Liver Disease (NAFLD/MASH)
NAFLD is driven by hepatic inflammation and fat accumulation. Elevated liver enzymes (ALT, AST) and hepatic steatosis are hallmarks. Tirzepatide's combination of weight loss, insulin sensitization, and direct anti-inflammatory effects positions it as a promising therapy. The SURPASS trials showed significant improvements in liver enzyme levels and hepatic fat fraction. Dedicated MASH trials are ongoing.
Obstructive Sleep Apnea
The SURMOUNT-OSA trial demonstrated that tirzepatide significantly reduced CRP in people with obesity and moderate-to-severe obstructive sleep apnea, alongside improvements in the apnea-hypopnea index (Malhotra et al., NEJM, 2024). Sleep apnea itself promotes systemic inflammation through intermittent hypoxia and sympathetic activation. By reducing both body weight and inflammation, tirzepatide may break the cycle between sleep apnea, inflammation, and cardiometabolic risk.
Lipedema
A narrative review explored tirzepatide as a potential disease-modifying therapy in lipedema, a condition characterized by painful fat accumulation and chronic inflammation (Crosa et al., IJMS, 2025). The authors proposed that tirzepatide's ability to attenuate pro-inflammatory cytokine signaling, promote M2 macrophage polarization, enhance mitochondrial oxidative capacity, and partially reverse fibrosis could address multiple pathological mechanisms in lipedema simultaneously. Clinical trial data in this population are not yet available.
Limitations of the Current Evidence
The evidence for tirzepatide's anti-inflammatory effects is strong but has important gaps that honest reporting requires addressing.
No dedicated anti-inflammatory trial exists. All inflammation data come from post-hoc analyses or secondary endpoints of trials designed to study weight loss, diabetes, or heart failure. A trial specifically designed to test tirzepatide as an anti-inflammatory agent would provide more rigorous evidence.
Placebo-controlled, not active-comparator. The meta-analysis compared tirzepatide to placebo, not to other anti-inflammatory treatments. We cannot say whether tirzepatide reduces CRP as effectively as methotrexate, colchicine, or canakinumab in their respective target populations.
Weight loss confounding. Despite evidence of weight-independent anti-inflammatory effects, separating the direct from indirect pathways in humans is difficult. The SURMOUNT post-hoc analysis used statistical mediation models, not mechanistic experiments. The direct anti-inflammatory contribution may be smaller or larger than current estimates suggest.
Limited follow-up beyond 72 weeks. Long-term maintenance of anti-inflammatory effects during sustained tirzepatide use has not been studied. Given that weight regain is common after discontinuation, the durability of anti-inflammatory benefits likely depends on continued treatment.
No autoimmune disease data. Claims about tirzepatide's potential in autoimmune conditions are extrapolated from mechanistic reasoning, not clinical evidence. Exercise caution with such claims until trial data emerge.
Frequently Asked Questions
How much does tirzepatide reduce CRP levels?
A 2025 systematic review and meta-analysis found tirzepatide reduced hsCRP by a mean of 32.9% compared to placebo across clinical trials. The SUMMIT trial in heart failure patients showed a 37.2% CRP reduction at 52 weeks. Higher doses (10 mg and 15 mg) produced greater reductions than the 5 mg dose. You can determine your appropriate dose using the tirzepatide dosage calculator.
Is tirzepatide anti-inflammatory independent of weight loss?
Partially. At 24 weeks in the SURMOUNT trials, hsCRP reductions were not fully explained by weight loss, suggesting direct receptor-mediated anti-inflammatory effects. By 72 weeks, 57-78% of inflammatory marker changes were weight-associated. The remaining 22-43% represents weight-independent anti-inflammatory action through GLP-1/GIP receptor pathways, AMPK activation, and NF-kB suppression. For full details on tirzepatide's metabolic effects, see our guide on does tirzepatide burn fat.
How long does it take for tirzepatide to reduce inflammation?
Direct anti-inflammatory effects may begin within the first 12-24 weeks of treatment. The largest measured reductions in hsCRP and IL-6 occurred at 52-72 weeks in clinical trials. The timeline depends on your dose, starting weight, and baseline inflammation level. Monitoring hsCRP at baseline and 6-month intervals provides the clearest picture of your personal response. Learn more about dosing timelines in our tirzepatide before and after guide.
Does tirzepatide help with joint inflammation?
Tirzepatide reduces systemic inflammatory markers (CRP, IL-6) that contribute to joint inflammation in people with obesity. Weight loss of 15-22% also reduces mechanical stress on joints. However, tirzepatide has not been tested specifically for joint conditions like osteoarthritis or rheumatoid arthritis. For joint-specific peptide research, see peptides for joint pain and the BPC-157 profile.
Is tirzepatide better than semaglutide for inflammation?
No head-to-head anti-inflammatory comparison exists. Tirzepatide's dual GLP-1/GIP agonism provides an additional anti-inflammatory pathway. In animal models, tirzepatide maintained anti-inflammatory effects even when GLP-1 neuronal receptors were blocked, while semaglutide did not. Tirzepatide also produces greater weight loss (15-22% vs. 12-17%), which amplifies the indirect anti-inflammatory effect. See our semaglutide to tirzepatide switching guide for transition details.
Can tirzepatide replace anti-inflammatory medications?
No. Tirzepatide is not approved as an anti-inflammatory drug. It should not replace NSAIDs, corticosteroids, DMARDs, or biologic therapies prescribed for specific inflammatory conditions. The anti-inflammatory effect is a secondary benefit for people taking tirzepatide for obesity or diabetes. Discuss any medication changes with your prescriber. For a full safety overview, read about tirzepatide long-term side effects.
Does stopping tirzepatide reverse the anti-inflammatory benefits?
Likely yes, over time. Weight regain after stopping tirzepatide would restore adipose-driven inflammation. The direct receptor-mediated anti-inflammatory effects would cease when the drug clears from your system. This is why most prescribers recommend long-term treatment. For strategies to maintain results, see our guide on tirzepatide maintenance dose after weight loss.
What dose of tirzepatide is best for reducing inflammation?
Higher doses produce greater anti-inflammatory effects. The 15 mg dose showed the largest CRP reductions in clinical trials, followed by 10 mg and 5 mg. However, dose selection should be based on overall treatment goals and tolerability, not inflammation alone. Start at 2.5 mg and titrate as directed by your prescriber. The best peptides for weight loss guide compares dosing strategies across GLP-1 and dual agonist therapies.
The Bottom Line
Tirzepatide reduces chronic low-grade inflammation through two converging pathways: direct receptor-mediated suppression of NF-kB and inflammatory cytokines, and indirect reduction of adipose-driven inflammation through substantial weight loss. The clinical evidence is consistent across the SURMOUNT program, the SUMMIT heart failure trial, and a 2025 meta-analysis showing 32.9% hsCRP reduction and 17.8% IL-6 reduction versus placebo.
The anti-inflammatory effect is real, measurable, and clinically meaningful for people with obesity-driven inflammation. It is not a replacement for dedicated anti-inflammatory therapies in autoimmune disease, nor is it a reason to start tirzepatide solely for inflammation in the absence of obesity or metabolic dysfunction.
If you are already taking or considering tirzepatide for weight management or diabetes, the anti-inflammatory benefit is a substantial added value. Monitor hsCRP at baseline and every six months to track your personal response. Discuss inflammation goals with your prescriber alongside weight and metabolic targets.
Take the PeptidesExplorer quiz to find out which peptide therapy aligns with your specific health goals.
Related Articles: - Tirzepatide Before and After: Real Results - treatment outcomes and timeline expectations - Peptides for Inflammation: Research Guide - BPC-157, GHK-Cu, and other anti-inflammatory peptides - Tirzepatide Long-Term Side Effects - safety profile beyond 52 weeks - Does Tirzepatide Burn Fat? - adipose tissue reduction mechanisms - Retatrutide vs. Tirzepatide - dual vs. triple agonist comparison
Related Articles
Does Compounded Tirzepatide Work? Real Data
Does compounded tirzepatide work? Review SURMOUNT data, salt form differences (base, acetate, sodium), potency gaps, and FDA status in 2026.
Does Oral Tirzepatide Work? The Facts
Oral tirzepatide is not FDA-approved. Why peptide pills face absorption barriers, sublingual drops vs injections, and oral GLP-1 alternatives in 2026.
Does Tirzepatide Cause Insomnia?
Does tirzepatide cause insomnia? Trials say no, but 5 indirect pathways explain lost sleep. GI, blood sugar, and caloric deficit fixes.
Can Tirzepatide Cause Joint Pain? Clinical Data
Can tirzepatide cause joint pain? SURMOUNT arthralgia rates match placebo. Review 5 indirect causes, uric acid risks, and 8 relief tactics.