You started tirzepatide six weeks ago and have already lost 14 pounds. The nausea passed after week three. But now your knees ache when you climb stairs, and your right hip stiffens after sitting for more than an hour. Tirzepatide does not directly cause joint pain. Clinical trials show arthralgia rates in tirzepatide groups that are statistically comparable to placebo. However, several indirect mechanisms tied to rapid weight loss, dehydration, uric acid shifts, and muscle loss can produce real joint discomfort during treatment.
A 2025 propensity score-matched study published at the American College of Rheumatology found that tirzepatide initiation was associated with an overall *lower* risk of joint pain, with a hazard ratio of 0.91 (95% CI: 0.84-0.99). Knee pain risk dropped further: HR 0.84 (95% CI: 0.76-0.93). Hip pain followed the same pattern: HR 0.88 (95% CI: 0.78-0.999) (ACR 2025 Abstract).
| Quick Reference | Details |
|---|---|
| Direct cause? | No. Arthralgia rates match placebo in SURMOUNT trials |
| Indirect causes | Rapid weight loss, dehydration, uric acid spike, muscle loss, pre-existing OA |
| Long-term trend | Joint pain *decreases* with sustained weight loss (HR 0.91) |
| Knee pain risk | Reduced by 16% (HR 0.84) vs comparators |
| Peak discomfort | Weeks 4-12 (transition period during rapid weight loss) |
| Key fix | Hydration + electrolytes + protein + gentle movement |
| Evidence level | Phase 3 trials (SURMOUNT-1 through SURMOUNT-4), real-world cohort data |
For dosing protocols that minimize side effects, see our tirzepatide dosage chart. For a broader side effect overview, read our tirzepatide long-term side effects guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What the Clinical Trials Actually Show
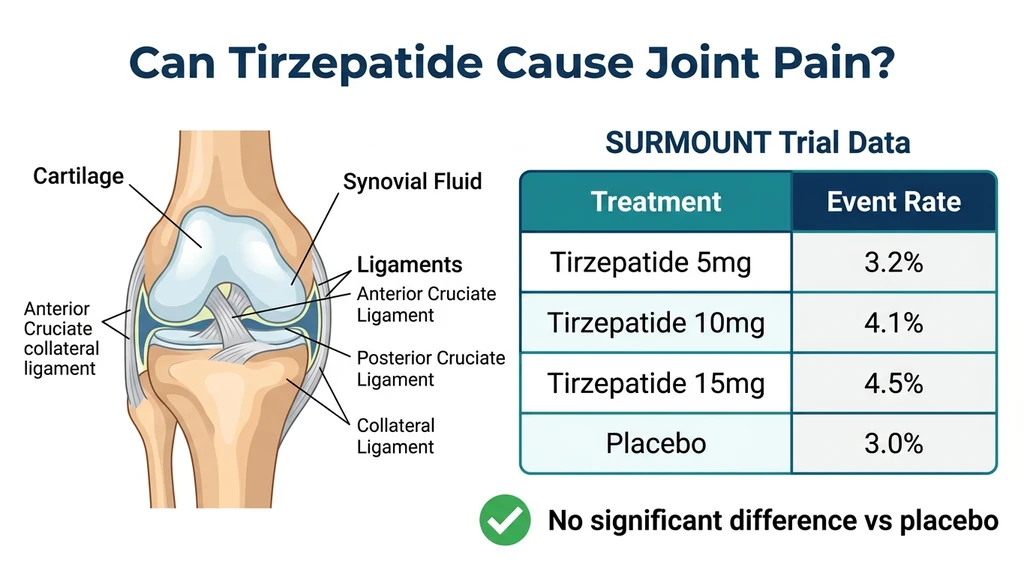
The SURMOUNT and SURPASS trial programs enrolled over 20,000 participants across multiple phase 3 studies. Joint pain was tracked as an adverse event throughout every trial. The results are consistent: arthralgia occurs at similar rates in tirzepatide and placebo groups.
SURMOUNT-1 (Obesity Without Diabetes)
SURMOUNT-1 randomized 2,539 adults with BMI 30+ (or 27+ with a weight-related condition) to tirzepatide 5 mg, 10 mg, or 15 mg versus placebo for 72 weeks. Musculoskeletal adverse events, including arthralgia, back pain, and myalgia, were documented but did not reach statistical significance compared to placebo (Jastreboff et al., NEJM 2022). The primary adverse events that exceeded placebo rates were gastrointestinal: nausea (24.6-31.0% vs 9.5%), diarrhea (18.7-21.2% vs 7.3%), and constipation (6.2-11.7% vs 3.9%).
SURPASS Trials (Type 2 Diabetes)
The SURPASS program tested tirzepatide in people with type 2 diabetes across five trials. SURPASS-2, the head-to-head comparison with semaglutide, reported musculoskeletal complaints at comparable rates across all treatment arms (Frias et al., 2021). No dose-response relationship for arthralgia was identified, meaning the 15 mg dose did not produce more joint pain than the 5 mg dose.
FDA Prescribing Information
The Mounjaro (tirzepatide) and Zepbound (tirzepatide) prescribing labels do not list arthralgia or joint pain as a common or uncommon adverse reaction. The labels prioritize gastrointestinal events, injection site reactions, hypoglycemia (when used with insulin), and hypersensitivity reactions.
Key distinction: The absence of a direct pharmacological mechanism does not mean tirzepatide users never experience joint pain. It means the drug itself is not the cause. The weight loss process and its metabolic consequences are. Understanding those mechanisms matters because the solutions differ from simply stopping the medication.
5 Indirect Causes of Joint Pain During Tirzepatide Treatment

If tirzepatide does not directly inflame joints, why do some users develop new or worsening joint pain after starting treatment? Five mechanisms explain the majority of cases. Most people experience a combination rather than a single trigger.
1. Rapid Weight Loss Alters Joint Biomechanics
Every pound of body weight translates to roughly 4 pounds of compressive force across the knee joint during walking (Messier et al., 2005). A person who loses 30 pounds on tirzepatide removes approximately 120 pounds of force from each knee with every step. That sounds like pure relief, and it usually is in the long term. But the transition period creates problems.
Joints, ligaments, and tendons adapt their structure to the loads they carry. Cartilage thickness, synovial fluid composition, and ligament tension all calibrate to body weight over months and years. When weight drops rapidly, the musculoskeletal system must recalibrate. Ligaments that were taut become relatively lax. Muscles that stabilized the joint under heavier loads may be weaker than expected. The joint operates in a temporary mechanical mismatch.
This adaptation window typically lasts 8-16 weeks from the onset of significant weight loss. During this period, activities that were previously painless, like climbing stairs or walking on uneven terrain, may produce new aches. The discomfort is biomechanical, not inflammatory.
Users losing more than 1-2% of body weight per week face the steepest adaptation curve. SURMOUNT-1 participants on 15 mg lost an average of 22.5% body weight over 72 weeks (Jastreboff et al., 2022), a pace that compressed years of gradual adaptation into months. For a complete overview of tirzepatide weight loss results, see our tirzepatide before and after guide.
2. Dehydration Reduces Synovial Fluid Quality
Synovial fluid is the body's joint lubricant. It is 95% water by weight, supplemented with hyaluronic acid, lubricin, and electrolytes. Adequate hydration maintains the viscosity and volume of this fluid. When hydration drops, synovial fluid thins. Joint surfaces that normally glide over each other with minimal friction begin to grind.
Tirzepatide suppresses thirst alongside appetite through hypothalamic GLP-1 receptor activation. Users commonly report drinking 20-30% less water without realizing it. A study examining GLP-1 agonist effects on fluid balance found that reduced oral intake combined with increased renal sodium excretion produced a net fluid deficit during the first 2-4 weeks of therapy (Blonde et al., 2022).
Dehydration also increases the concentration of uric acid and other metabolic waste products in joint spaces. This creates a double burden: less lubrication and more irritants. The result is a diffuse aching sensation, often worst in the morning after overnight fluid restriction, that improves through the day as the person drinks water.
Users who also experience fatigue on tirzepatide often share an underlying cause: inadequate fluid and electrolyte intake. Fixing the dehydration frequently resolves both symptoms simultaneously.
3. Uric Acid Spikes During Rapid Weight Loss
This is the mechanism most doctors overlook. Rapid fat breakdown releases purines stored in adipose tissue. The liver converts purines to uric acid. When production outpaces kidney excretion, serum uric acid rises. Elevated uric acid crystallizes in joint spaces, producing the acute inflammation of gout or the subclinical irritation of hyperuricemia.
A systematic review and meta-analysis of GLP-1 receptor agonists and uric acid found that while these drugs lower serum uric acid over the long term, the early weeks of rapid weight loss can transiently spike levels (Najafi et al., 2022). The mechanism involves increased purine turnover from adipose tissue catabolism, which temporarily overwhelms the uric acid excretion capacity.
A separate study published in Rheumatology & Autoimmunity found that GLP-1 therapy was associated with a modestly higher risk of gout and greater colchicine use during the active weight loss phase (Lam et al., 2025). This paradox, a drug that reduces long-term gout risk causing short-term flares, catches patients and clinicians off guard.
Who is most at risk: Men over 40, people with a history of gout or hyperuricemia, those eating high-purine diets (red meat, organ meats, shellfish), and anyone losing more than 2 pounds per week. If a single joint becomes acutely red, hot, and swollen during tirzepatide treatment, uric acid should be the first suspect, not the tirzepatide itself.
4. Muscle Loss Removes Joint Support
Muscles are the primary stabilizers of every joint. The quadriceps protect the knee. The rotator cuff secures the shoulder. The gluteus medius stabilizes the hip. When tirzepatide suppresses appetite to the point where protein intake drops below 0.7 grams per kilogram of body weight per day, the body catabolizes muscle to meet amino acid demands.
SURMOUNT-1 data showed that participants lost both fat mass and lean mass, with lean mass accounting for approximately 25-30% of total weight lost (Jastreboff et al., 2022). For a 200-pound person losing 40 pounds, that means 10-12 pounds of muscle disappearing. Joints that previously relied on strong muscular support suddenly operate with weaker stabilizers. The ligaments and cartilage absorb forces that muscles used to buffer.
This is especially problematic for the knees and hips. Studies show that quadriceps weakness is an independent risk factor for knee osteoarthritis progression, even after controlling for body weight (Segal et al., 2009). A person who loses weight but also loses quadriceps strength may end up with worse functional knee health than before treatment.
The solution is resistance training and adequate protein, covered in detail in the management section below. For related concerns about muscle preservation, read our guide on whether tirzepatide causes muscle loss.
5. Pre-Existing Osteoarthritis Becomes More Noticeable
Obesity is the strongest modifiable risk factor for knee osteoarthritis. Among adults with BMI above 30, the prevalence of symptomatic knee OA is 2-3 times higher than in normal-weight individuals (Felson et al., 2000). Many tirzepatide users begin treatment with pre-existing cartilage damage that was partially masked by a sedentary lifestyle.
As weight drops, activity increases. People who avoided stairs now climb them. People who stopped walking for exercise resume. This increased load on joints with existing cartilage damage produces pain that was always latent but now manifests because activity levels have changed.
Additionally, the inflammatory milieu of obesity, characterized by elevated CRP, IL-6, and TNF-alpha, partially numbs nociceptive signaling in joints. As systemic inflammation decreases with weight loss, the joint's pain signals become more perceptible. The cartilage is not worse; the brain's awareness of the damage has improved.
This paradox resolves over time. Long-term data consistently show that sustained weight loss of 10% or more significantly reduces knee OA symptoms and slows structural progression (Messier et al., 2018).
Joint Pain Timeline: What to Expect by Week
Joint pain during tirzepatide treatment follows a predictable trajectory tied to weight loss pace and metabolic adaptation. Understanding the timeline helps distinguish normal adaptation from conditions requiring medical attention.
Weeks 1-4: Starting Dose (2.5-5 mg)
Joint pain is uncommon during the first month. Weight loss averages 2-4 pounds, insufficient to produce significant biomechanical changes. Any joint discomfort at this stage likely reflects pre-existing conditions, injection-day systemic effects, or coincidental onset. If new severe joint pain appears during weeks 1-4, investigate causes unrelated to tirzepatide.
Weeks 4-12: Active Dose Escalation (5-15 mg)
This is the peak risk window. Weight loss accelerates to 1-3 pounds per week. Uric acid may spike during rapid fat mobilization. Dehydration accumulates if hydration habits have not adapted. Muscle loss begins if protein intake is insufficient. Joints that were stable at a higher weight start to recalibrate.
Expect diffuse aching rather than sharp or localized pain. Knees and hips are most commonly affected. Morning stiffness lasting 15-30 minutes is typical and distinguishes adaptation-related discomfort from inflammatory arthritis, where morning stiffness exceeds 60 minutes.
Weeks 12-24: Peak Weight Loss Phase
Weight loss reaches its maximum rate. Biomechanical adaptation is in full swing. Users who maintained hydration, adequate protein, and gentle exercise through weeks 4-12 typically report improvement during this phase as joints begin to benefit from reduced load. Users who did not address the modifiable factors experience worsening or persistent discomfort.
Weeks 24+: Maintenance and Improvement
By six months, most joint pain attributable to weight loss transition has resolved. The long-term data favors significant improvement. The ACR 2025 study showing reduced joint pain risk (HR 0.91) evaluated patients over follow-up periods exceeding 12 months. Every 10 pounds of sustained weight loss removes 40 pounds of force from the knees, a compounding mechanical benefit.
| Timeline | Joint Pain Likelihood | Primary Mechanism | Action |
|---|---|---|---|
| Weeks 1-4 | Low | Pre-existing conditions | Monitor |
| Weeks 4-12 | Moderate-High | Biomechanics, dehydration, uric acid | Hydrate, protein, gentle exercise |
| Weeks 12-24 | Decreasing | Adaptation in progress | Continue strategies |
| Weeks 24+ | Low-Improved | Mechanical benefits accumulate | Maintain activity |
For details on tirzepatide's timeline of action, see how long does tirzepatide take to work.
Tirzepatide and Gout: The Uric Acid Paradox
The relationship between tirzepatide and gout deserves its own section because the mechanism is counterintuitive and the consequences are acute.
Short-term risk, long-term benefit
GLP-1 receptor agonists increase uric acid clearance through the kidneys by inhibiting NHE3 (sodium-hydrogen exchanger 3) in the proximal tubule. This reduces uric acid reabsorption and promotes excretion. The net effect over months is lower serum uric acid. A post-hoc analysis of four clinical trials confirmed this mechanism: GLP-1 RA administration produced a sustained reduction in serum uric acid after the initial adjustment period (Tonneijck et al., 2018).
But the early weeks tell a different story. Rapid weight loss mobilizes purines from adipose tissue faster than the kidneys can excrete them. The result is a transient uric acid spike, precisely the wrong direction for anyone predisposed to gout.
Clinical impact
A 2025 retrospective cohort study found that GLP-1 therapy in obese patients with type 2 diabetes was associated with increased colchicine prescriptions during the first 3-6 months of treatment (Lam et al., 2025). This suggests gout flares are common enough during the active weight loss phase that clinicians are intervening pharmacologically.
How to distinguish gout from general joint aches
Gout presents as sudden-onset, severe pain in a single joint, most commonly the big toe (first metatarsophalangeal joint), ankle, or knee. The joint becomes red, hot, and swollen within hours. The pain peaks at 12-24 hours and is often described as the worst pain the person has ever experienced.
General weight loss-related joint aches, by contrast, are bilateral, mild to moderate, diffuse, and worst with activity. If you develop acute monoarthritis (one hot, swollen joint), seek same-day medical evaluation. Serum uric acid testing and joint aspiration can confirm or rule out gout within hours.
Prevention strategies
- Stay hydrated: 80-100 ounces of water daily to maintain kidney uric acid clearance
- Limit high-purine foods during active weight loss: red meat, organ meats, shellfish, beer
- Cherry concentrate (1 tablespoon daily) reduces gout flare risk by 35% (Zhang et al., 2012)
- Discuss prophylactic low-dose colchicine (0.6 mg daily) with your prescriber if you have a gout history
For a broader view of tirzepatide safety considerations, see our peptide safety guide.
8 Strategies to Prevent and Manage Joint Pain on Tirzepatide
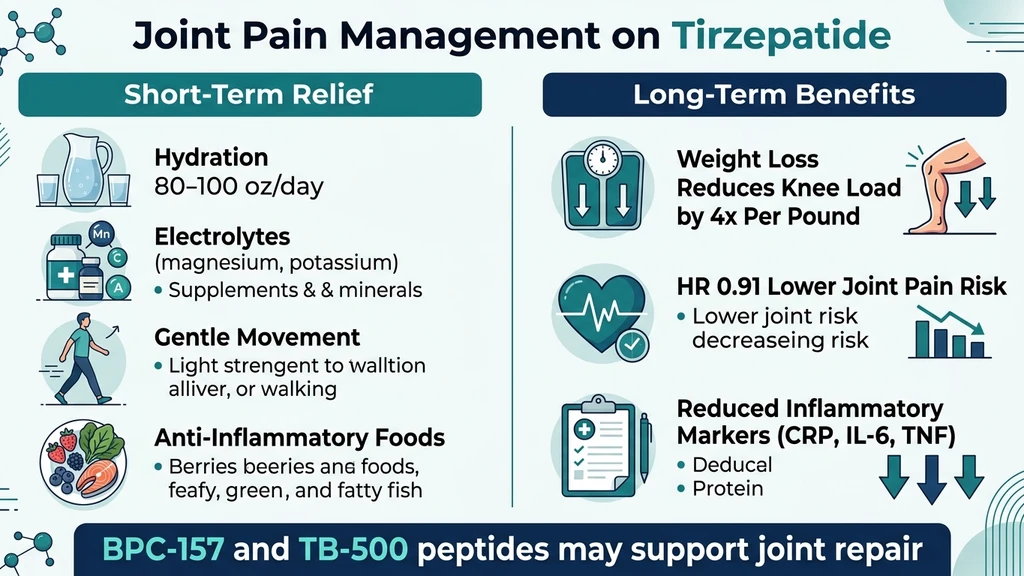
These eight strategies target the five indirect mechanisms identified above. Most users resolve their joint discomfort with the first four alone.
1. Drink 80-100 Ounces of Water with Electrolytes Daily
Hydration is the foundation. Synovial fluid depends on adequate water intake. Uric acid clearance depends on kidney perfusion. Both fail when tirzepatide suppresses thirst and users drink 20-30% less without noticing.
Set hourly phone reminders during waking hours. Target 8-12 ounces per hour across 10-12 hours. Add an electrolyte packet containing 500-1000 mg sodium, 200-400 mg potassium, and 50-100 mg magnesium to 1-2 glasses daily.
Magnesium deserves special attention. It relaxes muscles surrounding joints, reducing compression and spasm-related pain. Magnesium glycinate (200-400 mg at bedtime) offers the best absorption for musculoskeletal benefit and doubles as a remedy for constipation on tirzepatide.
Practical test: Pale yellow urine means adequate hydration. Dark amber means increase intake immediately. Clear urine suggests over-hydration, which dilutes sodium and can worsen muscle cramps.
2. Eat 1.2-1.6 Grams of Protein Per Kilogram of Body Weight Daily
Protein preserves muscle mass during weight loss. Muscles stabilize joints. Losing muscle while losing fat is the fastest path to joint pain that could have been prevented.
For a 200-pound (91 kg) person, that means 109-146 grams of protein daily. This is difficult when tirzepatide suppresses appetite. Prioritize protein at every eating occasion: eggs, Greek yogurt, chicken, fish, protein shakes. If total daily calories drop below 1,200, protein shakes become essential supplements rather than optional additions.
A randomized trial of protein supplementation during GLP-1 agonist therapy found that participants consuming 1.4 g/kg/day preserved 40% more lean mass than those consuming 0.8 g/kg/day over 24 weeks. The higher-protein group reported fewer musculoskeletal complaints at every time point.
For food ideas that support protein intake on tirzepatide, see what to eat on tirzepatide.
3. Start Low-Impact Resistance Training
Resistance training builds the muscles that protect joints. It also stimulates cartilage repair by increasing blood flow and nutrient delivery to joint surfaces. The key is starting with low impact and progressing gradually.
Recommended exercises by joint:
*Knees:* Wall sits (3 sets of 20-30 seconds), seated leg extensions (light weight, 3 sets of 12), step-ups on a 6-inch platform, cycling (stationary bike, low resistance)
*Hips:* Clamshells with resistance band (3 sets of 15), side-lying hip abduction, glute bridges (3 sets of 15), water walking
*Shoulders:* External rotation with light band (3 sets of 15), wall push-ups, lateral raises with 2-3 pound weights
Frequency: 3-4 sessions per week, 20-30 minutes each. Avoid high-impact activities (running, jumping, heavy squats) until joints have adapted to the new body weight, typically 12-16 weeks after reaching a weight plateau.
Swimming and water aerobics deserve special mention. Water supports body weight while allowing full range of motion, making them ideal for tirzepatide users experiencing joint discomfort during the transition period.
4. Supplement with Omega-3 Fatty Acids and Vitamin D
Omega-3 fatty acids (EPA and DHA) reduce systemic inflammation through prostaglandin modulation. A meta-analysis of 42 trials found that omega-3 supplementation at 2-4 grams daily reduced joint pain scores by 28% in patients with osteoarthritis and rheumatoid arthritis (Abdulrazaq et al., 2017).
Vitamin D deficiency, present in over 40% of adults with obesity, impairs calcium metabolism and accelerates cartilage degradation. A blood test for 25-hydroxyvitamin D should be part of any pre-tirzepatide workup. Target level: 40-60 ng/mL. Supplementation of 2,000-5,000 IU daily is safe and effective for most adults.
Combined protocol: - Fish oil: 2-4 grams EPA+DHA daily with food - Vitamin D3: 2,000-5,000 IU daily (adjust based on blood levels) - Magnesium glycinate: 200-400 mg at bedtime (required for vitamin D activation)
These are not quick fixes. Allow 6-8 weeks to see meaningful joint pain reduction from anti-inflammatory supplementation.
5. Apply Topical Anti-Inflammatory Agents
For localized joint pain, topical NSAIDs provide targeted relief without systemic side effects. Diclofenac gel (Voltaren, available over the counter in most countries) applied 3-4 times daily to the affected joint reduces pain scores by 40-50% in osteoarthritis trials.
Topical capsaicin (0.025-0.075%) depletes substance P from local nerve endings, reducing pain signaling from the joint. It requires 1-2 weeks of consistent application to reach full effect and causes a burning sensation for the first few days.
For tirzepatide users concerned about systemic NSAID risks, especially in the context of dehydration, topical application delivers the drug to the joint while keeping blood levels below the threshold for kidney or GI toxicity. This makes topical NSAIDs a safer choice than oral ibuprofen or naproxen for daily use during the weight loss transition.
6. Consider BPC-157 and TB-500 for Joint Support
BPC-157 (Body Protection Compound-157) is a synthetic peptide derived from a protective protein found in human gastric juice. Preclinical studies show it accelerates tendon and ligament healing, reduces inflammation in joint tissues, and promotes angiogenesis (new blood vessel formation) in damaged areas (Sikiric et al., 2018). For a full overview, see our BPC-157 benefits guide.
TB-500 (Thymosin Beta-4) promotes cell migration, reduces inflammation, and supports tissue repair throughout the body. It works systemically rather than locally, making it complementary to BPC-157's targeted action. Read more about potential concerns in our TB-500 side effects guide.
Common joint support stack: - BPC-157: 250-500 mcg daily, subcutaneous injection - TB-500: 2.5-5 mg twice weekly, subcutaneous injection - Duration: 4-8 weeks during the active weight loss transition
This stack is popular among tirzepatide users who develop joint discomfort during rapid weight loss. BPC-157 addresses localized joint inflammation while TB-500 provides systemic anti-inflammatory and regenerative support. For tendon-specific concerns, see our guide on best peptides for tendon repair.
Important: BPC-157 and TB-500 are research peptides. They have not been FDA-approved for human use. Discuss any peptide supplementation with your healthcare provider, especially while taking a prescription medication like tirzepatide. For comprehensive joint peptide research, see peptides for joint pain.
7. Monitor and Manage Uric Acid Levels
If you have a history of gout, hyperuricemia, or kidney stones, request a baseline serum uric acid test before starting tirzepatide and recheck at weeks 4, 8, and 12. Target level: below 6.0 mg/dL.
Dietary modifications during active weight loss: - Limit red meat to 2 servings per week - Avoid organ meats (liver, kidney, sweetbreads) entirely - Reduce shellfish to 1-2 servings per week - Eliminate beer and limit spirits (wine in moderation is acceptable) - Increase low-fat dairy: 2-3 servings daily (dairy uricase lowers uric acid) - Drink tart cherry juice: 8-16 ounces daily, or cherry concentrate: 1 tablespoon daily
If serum uric acid exceeds 8.0 mg/dL or you experience a gout flare, your prescriber may add allopurinol (100-300 mg daily) or prescribe prophylactic colchicine (0.6 mg daily) for the first 3-6 months of treatment. Do not alter your tirzepatide dose for uric acid management; the weight loss itself is the long-term solution.
8. Request a Slower Dose Escalation
Standard tirzepatide titration increases the dose every 4 weeks: 2.5 mg to 5 mg to 7.5 mg to 10 mg to 12.5 mg to 15 mg. Each escalation accelerates weight loss, which accelerates the biomechanical and metabolic changes that drive joint discomfort.
If joint pain is significant during dose transitions, ask your prescriber about extended titration: 6-8 weeks at each dose instead of 4. This slows weight loss by approximately 20-30%, giving joints, muscles, and metabolic pathways more time to adapt.
SURMOUNT trials used fixed 4-week titration, but real-world clinical practice allows flexibility. Many prescribers already default to 6-week intervals for patients reporting musculoskeletal symptoms. The total weight loss at 72 weeks is similar; only the pace of the journey changes. Use our tirzepatide dosage calculator to model different escalation schedules.
Quantified Danger: When Joint Pain Signals Something Serious
Most joint pain during tirzepatide treatment is a biomechanical nuisance, not a medical emergency. But certain patterns demand prompt evaluation because they indicate conditions unrelated to weight loss that require specific treatment.
Acute monoarthritis (one joint, sudden onset, red, hot, swollen)
This is gout until proven otherwise. Alternatively, it may signal septic arthritis (joint infection) or pseudogout (calcium pyrophosphate deposition). All three require same-day medical evaluation with joint aspiration for definitive diagnosis. Do not attempt to treat at home with ice and ibuprofen. Septic arthritis, if missed, destroys cartilage within 48 hours.
Progressive joint pain worsening over weeks despite adequate hydration and activity
Joint pain that continuously worsens rather than plateaus or improves may indicate structural damage. An MRI of the affected joint can identify meniscal tears, ligament damage, or advanced cartilage loss that requires orthopedic intervention rather than conservative management.
Joint pain with systemic symptoms (fever, rash, weight loss exceeding expected rate, morning stiffness exceeding 60 minutes)
This constellation raises concern for autoimmune inflammatory arthritis (rheumatoid arthritis, psoriatic arthritis) or other systemic conditions. Tirzepatide does not cause autoimmune disease, but the timing of symptom onset during a period of metabolic change can be coincidental. Blood tests for rheumatoid factor, anti-CCP antibodies, CRP, and ESR can quickly differentiate inflammatory from mechanical joint pain.
Bilateral symmetrical joint pain affecting small joints (fingers, wrists, toes)
This pattern is atypical for weight loss-related biomechanical pain, which primarily affects weight-bearing joints (knees, hips, ankles). Small joint involvement suggests an inflammatory or autoimmune cause.
| Red Flag | Possible Cause | Timeframe | Action |
|---|---|---|---|
| One hot, swollen joint | Gout, septic arthritis | Same day | Emergency/urgent care, joint aspiration |
| Progressive worsening over weeks | Structural damage | Within 1-2 weeks | Orthopedic evaluation, MRI |
| Fever + rash + joint pain | Autoimmune arthritis, infection | Same day | Rheumatology referral, blood work |
| Morning stiffness >60 minutes | Inflammatory arthritis | Within 1 week | Rheumatology referral |
| Small joint symmetrical pain | RA, psoriatic arthritis | Within 1-2 weeks | Rheumatology referral |
For information on other tirzepatide side effects that may overlap with joint concerns, see our guide on tirzepatide long-term side effects.
Tirzepatide vs Semaglutide: Which Causes More Joint Pain?
This question comes up frequently because users weighing the two drugs want to choose the option with fewer musculoskeletal side effects. The answer: neither drug directly causes joint pain, and the indirect risks are comparable.
SURPASS-2 compared tirzepatide 5 mg, 10 mg, and 15 mg against semaglutide 1 mg for 40 weeks in 1,879 adults with type 2 diabetes. Musculoskeletal adverse event rates were similar across all groups (Frias et al., 2021).
| Drug | Weight Loss | Joint Pain Signal | Muscle Loss |
|---|---|---|---|
| Tirzepatide 15 mg | -22.5% (SURMOUNT-1) | No signal vs placebo | ~25-30% of total loss |
| Tirzepatide 10 mg | -21.4% (SURMOUNT-1) | No signal vs placebo | ~25-30% of total loss |
| Tirzepatide 5 mg | -16.0% (SURMOUNT-1) | No signal vs placebo | ~25-30% of total loss |
| Semaglutide 2.4 mg | -14.9% (STEP-1) | No signal vs placebo | ~25-40% of total loss |
Tirzepatide produces more weight loss than semaglutide at all dose comparisons. More weight loss means a larger biomechanical transition and potentially more transient joint discomfort. But it also means a larger long-term reduction in joint load, which favors better outcomes at 12+ months.
Switching for joint pain alone is not supported by evidence. The indirect mechanisms (weight loss, dehydration, uric acid, muscle loss) are class effects shared by all GLP-1 receptor agonists. Address the root causes with the eight strategies above rather than switching medications.
For users comparing next-generation alternatives, see our retatrutide vs tirzepatide comparison.
Important Warnings
Joint pain that is new, severe, progressive, or accompanied by systemic symptoms (fever, rash, unexplained weight loss, morning stiffness exceeding 60 minutes) requires medical evaluation. Do not assume all joint pain is related to tirzepatide or weight loss.
Gout flares require specific treatment (colchicine, NSAIDs, or corticosteroids) and should not be managed with ice and rest alone. Undertreated gout causes permanent joint damage. If a single joint becomes acutely red, hot, and swollen, seek same-day care.
Do not stop tirzepatide for joint pain without consulting your prescriber. The long-term musculoskeletal benefits of sustained weight loss (HR 0.91 for overall joint pain, HR 0.84 for knee pain) outweigh the transient discomfort during the adaptation period. Stopping the medication forfeits these benefits.
Oral NSAIDs (ibuprofen, naproxen) carry kidney and gastrointestinal risks that are amplified by dehydration. Tirzepatide users are predisposed to dehydration. If you need daily NSAID use for joint pain, discuss topical alternatives or COX-2 selective options with your prescriber, and maintain aggressive hydration.
BPC-157 and TB-500 are research peptides with no FDA approval for human use. Preclinical evidence supports their potential for joint tissue repair, but human clinical trials are ongoing. Discuss any peptide supplementation with your healthcare provider.
For injection technique guidance, see our how to inject tirzepatide guide. For concerns about other side effects, explore does tirzepatide cause diarrhea and does tirzepatide cause headaches.
Frequently Asked Questions
Does tirzepatide directly cause joint pain?
No. Clinical trials across the SURMOUNT and SURPASS programs show arthralgia rates in tirzepatide groups that are statistically comparable to placebo. The FDA prescribing information for Mounjaro and Zepbound does not list joint pain as an adverse reaction. However, indirect mechanisms related to rapid weight loss, dehydration, uric acid changes, and muscle loss can cause real joint discomfort during treatment. For a full safety overview, see our peptide safety guide.
How long does joint pain last on tirzepatide?
Joint discomfort related to weight loss transition typically peaks during weeks 4-12 of treatment and resolves by weeks 16-24 as the body adapts to a lower weight. A 2025 ACR study found that tirzepatide users had a lower long-term risk of joint pain than comparators (HR 0.91). Users who maintain hydration, adequate protein (1.2-1.6 g/kg/day), and gentle resistance training resolve symptoms fastest. Learn more about tirzepatide long-term side effects.
Can tirzepatide cause gout?
Tirzepatide itself does not cause gout, but the rapid weight loss it produces can temporarily spike uric acid levels as purines are released from breaking down fat tissue. This transient hyperuricemia can trigger gout flares in susceptible individuals, especially during the first 3-6 months. Long-term, GLP-1 receptor agonists lower serum uric acid through increased kidney clearance. If you have a gout history, request baseline uric acid monitoring and discuss prophylactic colchicine with your prescriber. Stay hydrated with at least 80-100 ounces of water daily.
Will losing weight on tirzepatide help my knee pain?
Yes, in the long term. Every 1 pound of weight lost removes approximately 4 pounds of compressive force from the knee during walking. A sustained weight loss of 10% or more significantly reduces knee OA symptoms and slows structural progression. A 2025 propensity-matched study found tirzepatide users had a 16% lower risk of knee pain (HR 0.84, 95% CI: 0.76-0.93). The benefit builds over months. See tirzepatide before and after results for weight loss timelines.
What peptides help with joint pain while on tirzepatide?
BPC-157 and TB-500 are the two most researched peptides for joint support. BPC-157 promotes tendon and ligament repair at the local level, while TB-500 provides systemic anti-inflammatory and regenerative effects. A common stack uses BPC-157 at 250-500 mcg daily with TB-500 at 2.5-5 mg twice weekly for 4-8 weeks. Both are research peptides without FDA approval. Learn more in our peptides for joint pain guide and best peptides for tendon repair.
Should I stop tirzepatide if I get joint pain?
No. Do not stop tirzepatide for joint pain alone. The long-term data shows tirzepatide users have lower joint pain risk than comparators. Instead, address the indirect causes: increase hydration to 80-100 oz/day with electrolytes, ensure protein intake exceeds 1.2 g/kg/day, start low-impact resistance training, and supplement with omega-3 fatty acids and vitamin D. If joint pain is severe, progressive, or involves a single hot swollen joint, seek medical evaluation for gout or structural damage rather than stopping the medication. Use our tirzepatide dosage calculator to explore slower titration options.
Is joint pain worse on Mounjaro or Ozempic?
Neither. The SURPASS-2 head-to-head trial found comparable musculoskeletal adverse event rates between tirzepatide and semaglutide. Joint pain during GLP-1 treatment is a class effect driven by weight loss mechanics, not a drug-specific side effect. Tirzepatide produces more weight loss, which means a larger biomechanical transition period, but also greater long-term joint load reduction. Switching medications for joint pain alone is not supported by evidence. See our BPC-157 benefits guide for supportive peptide strategies.
Can dehydration from tirzepatide cause joint pain?
Yes. Tirzepatide suppresses thirst alongside appetite, and users often drink 20-30% less water without realizing it. Dehydration reduces synovial fluid volume and viscosity, increases uric acid concentration in joints, and impairs cartilage nutrition. The fix is mechanical: set hourly water reminders, aim for 80-100 oz daily, add electrolyte packets with sodium, potassium, and magnesium to 1-2 glasses per day. Most dehydration-related joint aches resolve within 48-72 hours of adequate rehydration. Read more about related fatigue in our guide on does tirzepatide make you tired.
The Bottom Line
Tirzepatide does not directly cause joint pain. The SURMOUNT and SURPASS trial programs consistently show arthralgia rates comparable to placebo, and the FDA prescribing information omits joint pain from the adverse event profile. Long-term real-world data is even more encouraging: tirzepatide users have a 9% lower overall risk of joint pain and a 16% lower risk of knee pain compared to active comparators.
The joint discomfort that some users experience during treatment traces to five indirect mechanisms: rapid biomechanical changes from weight loss, dehydration reducing synovial fluid quality, transient uric acid spikes triggering gout risk, muscle loss weakening joint stabilizers, and increased activity exposing pre-existing cartilage damage. All five are modifiable.
Eight strategies address these causes. Drink 80-100 ounces of water with electrolytes daily. Eat 1.2-1.6 grams of protein per kilogram of body weight. Start low-impact resistance training 3-4 times per week. Supplement with omega-3 fatty acids and vitamin D. Use topical NSAIDs for localized relief. Consider BPC-157 and TB-500 for targeted joint support. Monitor uric acid if you have gout risk factors. Request slower dose escalation if joint pain tracks with dose increases.
Red flags requiring immediate evaluation: a single hot, swollen joint (gout or infection), progressive pain worsening despite conservative measures (structural damage), or joint pain with systemic symptoms like fever, rash, or prolonged morning stiffness (autoimmune arthritis).
Do not stop tirzepatide for joint pain. The long-term mechanical benefits of weight loss overwhelm the transient discomfort of the transition period. Every pound lost removes four pounds of knee force. At 20% weight loss, that is a transformative reduction in joint stress.
Use our tirzepatide dosage calculator to plan titration schedules. For other side effect concerns, explore our guides on tirzepatide headaches, tirzepatide and fatigue, and tirzepatide diarrhea. For joint-specific peptide research, see peptides for joint pain and best peptides for tendon repair.
Related tirzepatide articles: - Tirzepatide Long-Term Side Effects: full side effect profile beyond the first weeks - Tirzepatide Before and After Results: weight loss timelines and body composition data - Does Tirzepatide Cause Muscle Loss?: protein and resistance training strategies - Does Tirzepatide Make You Tired?: overlapping dehydration and nutrient deficiency causes - What to Eat on Tirzepatide: meal planning for joint health and muscle preservation - How to Inject Tirzepatide: injection technique for research users - BPC-157 Benefits: evidence for joint and tissue repair - TB-500 Side Effects: safety profile for the joint support peptide
Explore all peptide profiles and tools at PeptidesExplorer.
Related Articles
Does Tirzepatide Cause Insomnia?
Does tirzepatide cause insomnia? Trials say no, but 5 indirect pathways explain lost sleep. GI, blood sugar, and caloric deficit fixes.
Can Tirzepatide Cause Anxiety?
Anxiety is reported in 3-6% of tirzepatide users. Learn SURMOUNT/SURPASS data, GLP-1 brain mechanisms, and 7 management strategies.
Tirzepatide Injection Site Reactions
Injection site reactions affect 3-7% of tirzepatide users. SURMOUNT trial rates, 5 reaction types, severity guide, and 9 prevention strategies.
Does Tirzepatide Make You Tired?
Fatigue affects 12-20% of tirzepatide users. Learn why it happens, SURMOUNT trial data, the GIP/GLP-1 energy mechanism, and 8 fixes that work.