
You are 8 weeks into tirzepatide and the nausea is fading, but you want to know what lies ahead at month 6, month 12, and beyond. Most tirzepatide side effects peak during weeks 1 to 8 and resolve by week 12. In the 72-week SURMOUNT-1 trial of 2,539 participants, GI side effects (nausea, diarrhea, vomiting) occurred in 39 to 49% of participants depending on dose. Serious events remained rare: gallbladder disease 1 to 3%, pancreatitis below 0.2%, thyroid C-cell tumors observed only in rodents (Jastreboff et al., NEJM, 2022). The FAERS database (65,974 real-world reports from 2022 to 2025) confirms the clinical trial safety profile (Healthcare, 2025).
| Side Effect | Incidence (72 wk) | Peak Timing | Resolution | Severity |
|---|---|---|---|---|
| Nausea | 24 to 31% | Weeks 1 to 8 | By week 12 | Mild to moderate |
| Diarrhea | 17 to 23% | Weeks 1 to 8 | By week 12 | Mild to moderate |
| Constipation | 6 to 17% | Weeks 4 to 12 | Variable | Mild |
| Vomiting | 5 to 12% | Weeks 1 to 8 | By week 12 | Mild to moderate |
| Gallbladder disease | 1 to 3% | Months 3 to 12+ | Requires treatment | Serious |
| Pancreatitis | <0.2% | Unpredictable | Requires treatment | Serious |
| GI-related discontinuation | 2.7% (vs 5.6% semaglutide) | Weeks 1 to 12 | N/A | N/A |
For dosing strategy during titration, use our tirzepatide dosage calculator. For medication interactions, see our tirzepatide drug interactions guide.
Not FDA-approved for use outside labeled indications. Consult a healthcare provider before starting tirzepatide or adjusting your dose.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
GI Side Effects: Timeline and Resolution
Gastrointestinal side effects dominate the tirzepatide safety profile. They appear early, peak during dose escalation, and resolve for the vast majority of users who continue treatment. The pooled analysis of SURMOUNT-1 through SURMOUNT-4 confirms this pattern across more than 5,000 participants (DOM, 2025).
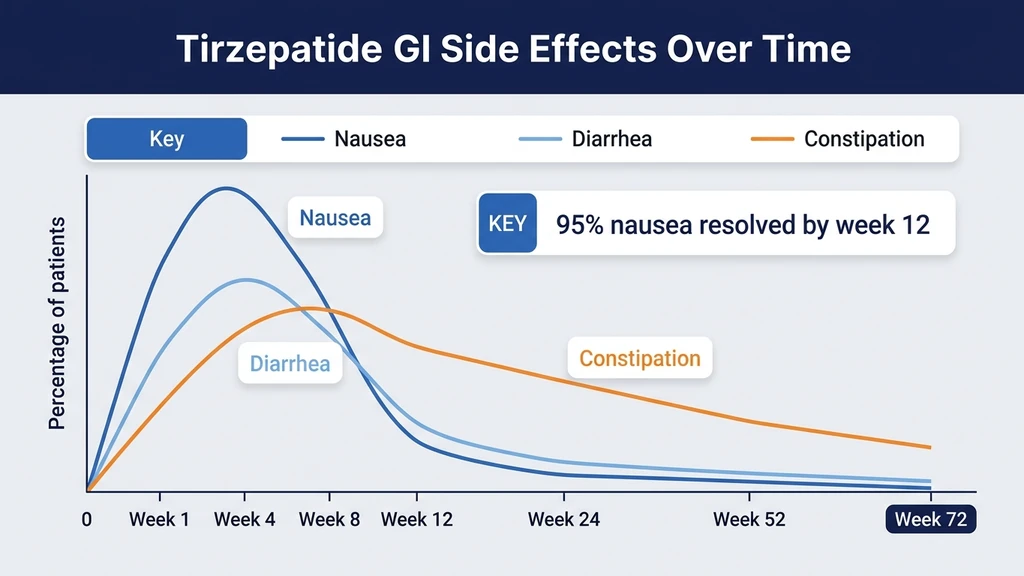
Nausea (24 to 31%)
Nausea is the most common side effect across all doses. In SURMOUNT-1, incidence was dose-dependent: 24.6% at 5 mg, 33.3% at 10 mg, and 31.0% at 15 mg (Jastreboff et al., NEJM, 2022). The slight drop at 15 mg compared to 10 mg reflects the slow titration schedule: patients reaching 15 mg have already adapted through months at 5 mg and 10 mg.
Nausea peaks during weeks 1 to 8 of each new dose level. It occurs most often within 48 hours of injection and fades over the following days. By week 12 at a stable dose, fewer than 5% of participants still report nausea. The mechanism: GLP-1 receptor activation slows gastric emptying, creating a fullness signal the brain interprets as nausea. For detailed management strategies, see our guide on does tirzepatide cause nausea.
Diarrhea (17 to 23%)
Diarrhea follows the same early-peak, late-resolution pattern as nausea. SURMOUNT-1 rates: 17.4% at 5 mg, 21.2% at 10 mg, and 23.0% at 15 mg. Episodes are typically mild (1 to 3 loose stools per day) and self-limiting within the first 8 weeks.
Hydration is critical. Tirzepatide-induced diarrhea combined with reduced food intake can cause dehydration, the primary pathway to the rare but serious side effect of acute kidney injury. Drink at least 64 ounces of water daily. For a focused discussion, see our article on tirzepatide and diarrhea.
Constipation (6 to 17%)
Constipation appears later than nausea and diarrhea, typically emerging at weeks 4 to 12. It occurs because slowed gastric emptying extends transit time through the entire GI tract. Rates in SURMOUNT-1 ranged from 6.0% at 5 mg to 17.1% at 15 mg.
Fiber supplementation (psyllium husk, methylcellulose), adequate water intake, and regular physical activity help manage constipation effectively. For details, see our guide on tirzepatide constipation.
Vomiting (5 to 12%)
Vomiting is less common than nausea but follows the same timeline. SURMOUNT-1 rates: 5.1% at 5 mg, 9.0% at 10 mg, 12.2% at 15 mg. Most vomiting episodes occur during weeks 1 to 4 of a new dose and resolve without intervention. Persistent vomiting beyond 48 hours after injection warrants medical contact to assess hydration status.
Sulfur Burps and GERD
Sulfur burps (eructation with a hydrogen sulfide odor) are not extensively captured in clinical trial data but appear prominently in the FAERS database and patient communities. The mechanism: slowed gastric emptying allows food to ferment longer in the stomach, producing hydrogen sulfide gas.
Management strategies include eating smaller portions, avoiding sulfur-rich foods (eggs, cruciferous vegetables, dairy), and simethicone for gas relief. Proton pump inhibitors may help if GERD symptoms (heartburn, acid reflux) accompany the eructation.
Serious Long-Term Side Effects
Rare but clinically significant side effects require awareness and monitoring. The FAERS database (65,974 reports from 2022 to 2025) provides real-world context beyond the controlled trial environment (Healthcare, 2025).
Gallbladder Disease (1 to 3%)
A meta-analysis of 9 trials and 9,871 participants found composite gallbladder and biliary disease significantly associated with tirzepatide compared to placebo. The 10 mg dose showed the highest risk for the composite gallbladder endpoint. Cholelithiasis (gallstones) alone did not reach statistical significance (Frontiers, 2023).
Rapid weight loss is the primary driver. When the body mobilizes stored fat quickly, the liver excretes more cholesterol into bile, increasing gallstone formation. This risk applies to any intervention producing rapid weight loss, not just tirzepatide.
Symptoms: right upper abdominal pain radiating to the shoulder, nausea and vomiting after fatty meals, and fever with jaundice. These warrant urgent medical evaluation.
Pancreatitis (Below 0.2%)
The same meta-analysis found no significant increase in pancreatitis with tirzepatide versus all control groups. Incidence remained below 0.2% across the entire SURMOUNT and SURPASS trial programs (Frontiers, 2023).
A separate analysis of pancreatic safety showed tirzepatide actually preserved islet cell function in type 2 diabetes patients, with no evidence of pancreatic damage (PMC, 2024). Report severe, persistent abdominal pain radiating to the back to your prescriber immediately.
Thyroid C-Cell Tumors (Rodent Data Only)
Tirzepatide carries a black box warning for thyroid C-cell tumors based on rodent studies. In rats, GLP-1 receptor agonists caused dose-dependent, duration-dependent C-cell hyperplasia and medullary thyroid carcinoma (MTC). Human relevance is unknown: human thyroid C-cells express far fewer GLP-1 receptors than rodent C-cells.
Tirzepatide is contraindicated in patients with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). No case of MTC has been attributed to tirzepatide in human clinical trials or post-marketing surveillance (Jastreboff et al., NEJM, 2022).
Kidney Concerns
Acute kidney injury (AKI) signals appear in the FAERS database, primarily linked to dehydration from severe GI symptoms (vomiting, diarrhea). The FAERS analysis found renal signals consistent with volume depletion rather than direct nephrotoxicity (Healthcare, 2025).
Prevention: maintain hydration (64+ ounces of water daily), report persistent vomiting or diarrhea to your prescriber, and monitor kidney function if you have pre-existing renal disease. Tirzepatide does not appear to cause direct kidney damage.
Body Composition Changes at 72 Weeks
Weight loss is the intended effect. The composition of that weight loss matters for long-term health.
Muscle Loss and Sarcopenia Risk
Approximately 30 to 35% of total weight lost on tirzepatide comes from lean mass (muscle, bone, water) rather than fat. This ratio is consistent with other caloric-deficit interventions, including diet alone and bariatric surgery.
The concern: at the 15 mg dose producing 22.5% body weight loss, roughly 7 to 8% is lean tissue. For a 100 kg individual losing 22.5 kg, approximately 7 to 8 kg is lean mass. In older adults or those with low baseline muscle mass, this can accelerate sarcopenia and increase fall risk.
Resistance training is the primary mitigation. Studies consistently show that structured resistance exercise 2 to 3 times per week preserves 50 to 80% of lean mass during caloric restriction. Protein intake of 1.2 to 1.6 g per kg body weight per day provides the amino acid substrate for muscle maintenance. For the full discussion, see our article on tirzepatide muscle loss.
Bone Density Considerations
Modest reductions in bone mineral density (BMD) at the hip and lumbar spine have been observed with significant weight loss from GLP-1 receptor agonists. The mechanical unloading hypothesis: less body weight means less gravitational stress on bones, signaling the body to reduce bone density.
This concern is most relevant for postmenopausal women with pre-existing osteopenia. Baseline DEXA scanning before starting tirzepatide and follow-up at 12 months is a reasonable monitoring strategy for at-risk individuals. Weight-bearing exercise and adequate calcium and vitamin D intake support bone health during rapid weight loss.
Cardiovascular Safety: Reassuring Data
Cardiovascular safety is the best-documented long-term outcome for tirzepatide. The data is reassuring across multiple analyses.
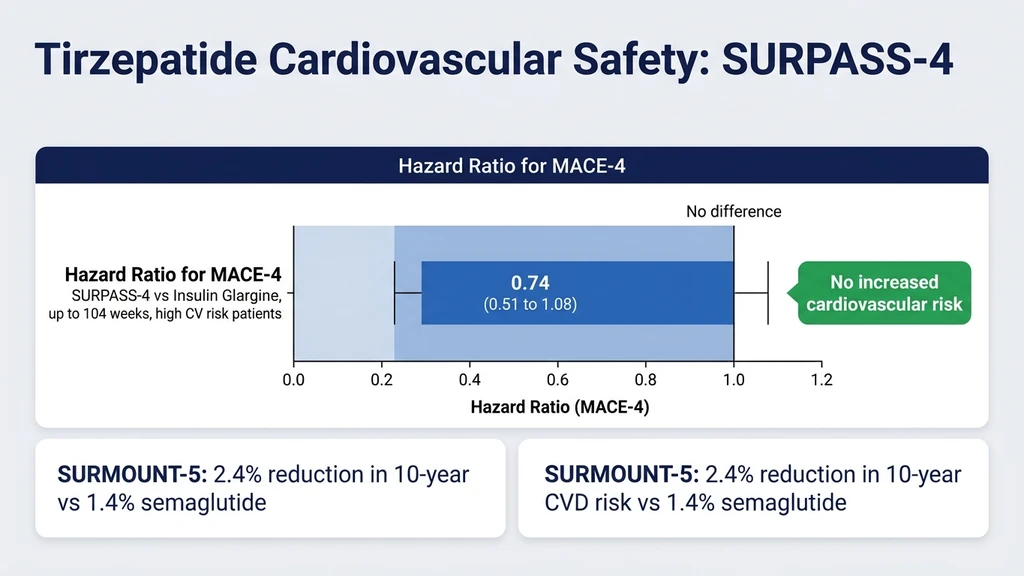
The SURPASS-4 trial followed high-cardiovascular-risk type 2 diabetes patients for up to 104 weeks. The hazard ratio for MACE-4 (cardiovascular death, myocardial infarction, stroke, unstable angina) was 0.74 (95% CI 0.51 to 1.08) versus insulin glargine (Del Prato et al., Lancet, 2021). The confidence interval crossed 1.0, meaning the reduction was not statistically significant, but the point estimate suggests a cardiovascular benefit trend.
A pre-specified cardiovascular meta-analysis across the SURPASS program confirmed no increased cardiovascular risk (Nature Medicine, 2022). The SURMOUNT-5 head-to-head trial showed a 2.4% reduction in 10-year cardiovascular disease risk with tirzepatide versus 1.4% with semaglutide (NEJM, 2025). FAERS cardiovascular analysis found no disproportionate signals for heart failure, atrial fibrillation, or ischemic events (Frontiers, 2026).
Psychiatric and Neurological Effects
Fatigue is common during the first 4 to 8 weeks. It correlates with reduced caloric intake rather than a direct drug effect. As the body adapts to lower energy intake, fatigue typically resolves. Ensuring adequate protein and micronutrient intake accelerates recovery. For detail, see our guide on tirzepatide fatigue.
Mood changes have not been identified as a significant signal in any randomized controlled trial. Individual reports of irritability or mood swings during rapid weight loss are consistent with caloric restriction effects.
Suicide risk: A real-world cohort study specifically evaluated this concern and found no increased risk with tirzepatide compared to non-GLP-1 controls (PMC, 2025). The FDA continues to monitor this signal for the entire GLP-1 receptor agonist class but has not issued warnings for tirzepatide.
Hair loss (telogen effluvium) occurs in some users experiencing rapid weight loss. This is a stress response of the hair follicle cycle, not a direct drug effect. Hair loss typically begins 2 to 4 months after significant weight loss onset and resolves within 6 to 12 months. For more on tirzepatide hair loss, see our focused guide. For anxiety-related concerns, see tirzepatide and anxiety.
Real-World Safety Data (FAERS 2022 to 2025)
The FDA Adverse Event Reporting System collected 65,974 tirzepatide-related reports from 2022 to 2025. This real-world data extends beyond the controlled clinical trial population (Healthcare, 2025).
| FAERS Finding | Detail |
|---|---|
| Total reports analyzed | 65,974 |
| Demographics | 96% US, 67% female, primarily age 40 to 59 |
| Top adverse event | Incorrect dose administration (8x increase 2022 to 2024) |
| GI events | Confirm clinical trial profile (nausea, diarrhea, vomiting) |
| Cardiovascular signals | No disproportionate signals for HF, AF, or ischemic events |
| Renal signals | Dehydration-related AKI, not direct nephrotoxicity |
| Psychiatric signals | No suicide or self-harm disproportionality |
The most striking FAERS finding: incorrect dose administration was the number one reported adverse event, with an 8-fold increase from 2022 to 2024. This reflects the complexity of auto-injector pen use and the surge in new users unfamiliar with the device. Many "side effects" in real-world data are preventable dosing errors.
Tirzepatide vs Semaglutide Side Effects: SURMOUNT-5 Head-to-Head
The 72-week SURMOUNT-5 trial directly compared tirzepatide 15 mg to semaglutide 2.4 mg in 751 adults with obesity. This is the only randomized head-to-head tolerability comparison available (NEJM, 2025).

| Parameter | Tirzepatide 15 mg | Semaglutide 2.4 mg |
|---|---|---|
| GI-related discontinuation | 2.7% | 5.6% |
| Nausea | Similar rates | Similar rates |
| Diarrhea | Similar rates | Similar rates |
| Vomiting | Similar rates | Similar rates |
| Weight loss achieved | 20.2% | 13.7% |
| 10-year CVD risk reduction | 2.4% | 1.4% |
Tirzepatide showed better GI tolerability despite producing 47% more weight loss than semaglutide. The GI-related discontinuation rate of 2.7% versus 5.6% is clinically meaningful: fewer patients abandoned tirzepatide because of GI symptoms.
The dual GIP/GLP-1 mechanism may explain this tolerability advantage. GIP receptor activation may modulate the nausea pathway differently than GLP-1 alone, potentially buffering the intensity of GI symptoms while enhancing weight loss efficacy.
How to Minimize Long-Term Side Effects
Four strategies reduce side effect burden and improve long-term adherence.
Slow Dose Titration
Follow the standard escalation: 2.5 mg for 4 weeks, then 5 mg for 4 weeks, increasing by 2.5 mg every 4 weeks as tolerated. Some prescribers extend to 6 or 8 weeks at each level for patients with severe GI symptoms. Rushing titration is the single most common cause of avoidable side effects in the real world. Use our tirzepatide dosage calculator to plan your schedule. See the full dosage chart for the complete escalation timeline.
Dietary Adjustments
Eat smaller, more frequent meals (4 to 6 per day instead of 2 to 3). Avoid high-fat, greasy, and fried foods during dose escalation. These sit in the already-slow stomach longest and worsen nausea. Prioritize lean protein, complex carbohydrates, and vegetables. The BRAT diet (bananas, rice, applesauce, toast) provides relief during acute nausea episodes.
Hydration
Aim for 64 ounces of water minimum per day. Increase to 80 or more ounces if you experience diarrhea or vomiting. Dehydration is the gateway to the serious side effects (AKI, electrolyte imbalance). Use a marked water bottle to track intake. Electrolyte supplements help when fluid losses are significant.
Resistance Training for Muscle Preservation
Start or maintain a resistance training program before or concurrent with tirzepatide. Two to three sessions per week targeting major muscle groups preserves lean mass during rapid weight loss. Combine with 1.2 to 1.6 g protein per kg body weight daily. This dual approach reduces the 30 to 35% lean mass loss ratio toward 15 to 20%.
For maintenance dosing after reaching your target weight, the same exercise and protein principles prevent weight regain. For injection technique to minimize site reactions, see our best injection sites guide.
Frequently Asked Questions
What are the long-term side effects of tirzepatide after 1 year?
After 72 weeks, the most common persisting effects are constipation (6 to 17%), reduced appetite, and body composition changes (30 to 35% of weight loss from lean mass). GI side effects like nausea and diarrhea resolve by week 12 in over 95% of users. Gallbladder disease (1 to 3%) and telogen effluvium hair loss may emerge after 3 to 6 months of significant weight loss. For related side effects, see our tirzepatide headaches guide.
Does tirzepatide cause cancer?
No human cancer cases have been attributed to tirzepatide. Rodent studies showed thyroid C-cell tumors at high doses, prompting a black box warning. Human thyroid C-cells express far fewer GLP-1 receptors than rodent cells. A meta-analysis of 9 trials (9,871 participants) found no increased pancreatitis or cancer signal (Frontiers, 2023). Tirzepatide is contraindicated in patients with personal or family history of medullary thyroid carcinoma or MEN 2.
Is tirzepatide safe for long-term use?
The SURMOUNT-1 trial (72 weeks, 2,539 participants) and SURPASS-4 (up to 104 weeks) showed an acceptable safety profile. Treatment discontinuation for side effects ranged from 4.3% to 7.1% in SURMOUNT-1, and only 2.7% for GI reasons in the head-to-head SURMOUNT-5 trial. The FAERS database (65,974 reports) confirms the trial findings. Ongoing cardiovascular outcomes trials will provide additional long-term data.
Does tirzepatide cause muscle loss?
Approximately 30 to 35% of weight lost on tirzepatide is lean mass, consistent with any caloric deficit intervention. For a 22.5 kg weight loss, roughly 7 to 8 kg is lean tissue. Resistance training (2 to 3 sessions per week) and protein intake of 1.2 to 1.6 g per kg per day preserve 50 to 80% of lean mass. Start resistance training before or simultaneously with tirzepatide for maximum benefit. See our dedicated tirzepatide muscle loss article.
What is the dropout rate due to tirzepatide side effects?
In SURMOUNT-1, treatment discontinuation due to adverse events ranged from 4.3% (5 mg) to 7.1% (15 mg). In the head-to-head SURMOUNT-5 trial, only 2.7% of tirzepatide users discontinued for GI reasons, compared to 5.6% on semaglutide. The FAERS database identifies incorrect dose administration as the most common adverse event, suggesting many real-world issues are preventable dosing errors.
Is tirzepatide safer than semaglutide?
The SURMOUNT-5 head-to-head trial found tirzepatide had a lower GI-related discontinuation rate (2.7% vs 5.6%) despite producing greater weight loss (20.2% vs 13.7%) (NEJM, 2025). Both drugs share similar GI side effect types. Tirzepatide's dual GIP/GLP-1 mechanism may buffer GI intensity. Long-term serious adverse event profiles are comparable between both drugs.
Does tirzepatide affect the heart?
Cardiovascular data is reassuring. SURPASS-4 showed a hazard ratio of 0.74 for MACE-4 (95% CI 0.51 to 1.08), trending toward cardiovascular benefit (Lancet, 2021). FAERS found no disproportionate signals for heart failure, atrial fibrillation, or ischemic events. SURMOUNT-5 showed a 2.4% reduction in 10-year CVD risk with tirzepatide. The definitive SURPASS-CVOT trial is ongoing.
The Bottom Line
Tirzepatide's 72-week safety profile is well-characterized: GI side effects peak early and resolve by week 12, serious events (gallbladder disease 1 to 3%, pancreatitis below 0.2%) are rare, and cardiovascular data trends positive. The FAERS database of 65,974 real-world reports confirms rather than contradicts the clinical trial findings.
Slow titration, adequate hydration, dietary adjustments, and resistance training address the major manageable concerns. Lean mass loss (30 to 35% of total weight lost) deserves attention through structured exercise and protein intake. The SURMOUNT-5 head-to-head showed tirzepatide achieves more weight loss with better GI tolerability than semaglutide.
Use our tirzepatide dosage calculator to plan your titration schedule. For medication safety during treatment, see our tirzepatide drug interactions guide. For injection technique that reduces site reactions, review our injection sites guide. For tirzepatide maintenance dosing after reaching target weight, see our dedicated guide.
Related Articles
Does Tirzepatide Cause Insomnia?
Does tirzepatide cause insomnia? Trials say no, but 5 indirect pathways explain lost sleep. GI, blood sugar, and caloric deficit fixes.
Does Tirzepatide Affect Your Period?
Does tirzepatide affect your period? Yes, via rapid fat loss and estrogen shifts. Cycle changes, PCOS fertility, and birth control warning.
Can Tirzepatide Cause Joint Pain? Clinical Data
Can tirzepatide cause joint pain? SURMOUNT arthralgia rates match placebo. Review 5 indirect causes, uric acid risks, and 8 relief tactics.
Does Tirzepatide Cause Muscle Loss?
SURMOUNT-1 DEXA data: 25% of tirzepatide weight loss is lean mass (5.6 kg at 72 weeks). Protein targets, resistance training, and supplements.