You started tirzepatide two months ago, and your period arrived a week early. Or it vanished entirely. Or the flow changed from predictable to unpredictable. Yes, tirzepatide can affect your period, but not because it targets reproductive hormones. The changes stem from rapid fat loss, shifting estrogen levels, and improved insulin sensitivity. A Natural Cycles survey found 27% of GLP-1 users noticed menstrual changes after starting treatment, with 45% of those reporting more predictable cycles and 21% reporting more frequent periods.
| Quick Reference | Details |
|---|---|
| Direct hormonal effect | None confirmed |
| Indirect mechanism | Fat loss reduces estrogen production via aromatase |
| Users reporting changes | 27% in observational surveys |
| Most common change | More predictable cycles (45% of those affected) |
| PCOS effect | Cycle normalization as insulin sensitivity improves |
| Birth control warning | Oral contraceptive absorption reduced up to 66% |
| FDA recommendation | Use non-oral contraception or add barrier method for 4 weeks after each dose change |
| Timeline | Changes typically stabilize within 3-6 months |
For dosing guidance, use our tirzepatide dosage calculator. For a full overview of side effects, see our tirzepatide long-term side effects guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Why Tirzepatide Changes Your Period
Tirzepatide does not bind to estrogen receptors. It does not interact with the hypothalamic-pituitary-ovarian (HPO) axis directly. It does not suppress luteinizing hormone or follicle-stimulating hormone. The drug targets GIP and GLP-1 receptors to reduce appetite, slow gastric emptying, and improve insulin sensitivity. Period changes are a downstream consequence of what happens when the body loses fat rapidly and recalibrates metabolically.
Three mechanisms drive the menstrual disruption. Each one operates independently, but they compound when they overlap.
Mechanism 1: Estrogen Drops When Fat Drops
Adipose tissue is not inert storage. It functions as an endocrine organ. Fat cells contain aromatase, an enzyme that converts androgens (testosterone, androstenedione) into estrogen (estradiol). In women with higher body fat, adipose tissue can contribute 50-100% of circulating estrogen after menopause and a meaningful percentage during reproductive years.
When tirzepatide drives 15-22% body weight loss over 72 weeks (Jastreboff et al., NEJM 2022), the body's aromatase factory shrinks rapidly. Less fat means less aromatase. Less aromatase means less estrogen conversion. The drop is not gradual. It tracks with the speed of weight loss, and tirzepatide produces the fastest weight loss of any approved GLP-1 drug.
Estrogen orchestrates the menstrual cycle at every phase. It thickens the uterine lining during the follicular phase, triggers the LH surge that causes ovulation, and supports the corpus luteum during the luteal phase. A sudden estrogen dip can delay ovulation, shorten the luteal phase, thin the endometrial lining (lighter periods), or cause breakthrough bleeding. These changes are the same ones seen after bariatric surgery, crash diets, and extreme athletic training. The trigger is fat loss speed, not tirzepatide pharmacology.
For women already experiencing tirzepatide side effects, menstrual changes add another variable to track.
Mechanism 2: Insulin Sensitivity Recalibrates the HPO Axis
Insulin resistance and reproductive hormones are deeply entangled. Excess insulin stimulates ovarian androgen production, which disrupts the follicular development cycle and is the primary driver of anovulation in PCOS. Tirzepatide reduces fasting insulin by 30-50% in clinical trials, and that improvement starts within weeks of the first injection.
When insulin drops, the ovaries receive less androgen-stimulating signal. For women with PCOS, this can restore ovulation that was previously suppressed for months or years. For women without PCOS, the recalibration can still shift cycle timing temporarily while the HPO axis adjusts to a new metabolic baseline.
The adjustment period is typically 2-4 cycles. The body is not malfunctioning. It is finding a new equilibrium. Women who lose weight through diet and exercise alone experience the same transitional irregularity.
Mechanism 3: Energy Deficit Signals the Brain to Pause Reproduction
The hypothalamus monitors energy availability. When caloric intake drops below what the body perceives as safe for reproduction, it can suppress gonadotropin-releasing hormone (GnRH) pulsatility. Fewer GnRH pulses means less FSH and LH. Less FSH and LH means delayed or absent ovulation.
Tirzepatide suppresses appetite aggressively. Some users report eating 800-1,200 calories daily without feeling hungry. At that intake, a 180-pound woman is in a 40-50% caloric deficit. The body interprets this as a survival scenario and deprioritizes reproduction. Missing a period under these conditions is a protective mechanism, not a pathology.
This is the same phenomenon seen in female athletes with relative energy deficiency in sport (RED-S). The solution is the same: increase caloric intake, particularly protein. Aim for at least 1,200 calories daily with a minimum of 0.8g protein per pound of body weight. For nutrition strategies, see our guide on what to eat on tirzepatide.
What Changes to Expect by Cycle Phase
Not all menstrual changes are equal. Understanding which phase is disrupted helps predict what you will experience and when to be concerned.
| Cycle Phase | Normal Duration | How Tirzepatide Affects It | What You Notice |
|---|---|---|---|
| Follicular (Days 1-13) | 10-16 days | Lower estrogen slows follicle maturation | Period starts later than expected |
| Ovulation (Day 14) | 24-36 hours | Delayed or absent LH surge | Missed ovulation, no mid-cycle pain |
| Luteal (Days 15-28) | 12-14 days | Shortened progesterone phase | Spotting before expected period |
| Menstruation (Days 1-5) | 3-7 days | Thinner endometrial lining | Lighter flow, shorter duration |
Lighter Periods
The most frequently reported change. When estrogen drops, the endometrial lining grows thinner during the follicular phase. A thinner lining produces less flow during menstruation. Women who previously had heavy periods may find this a welcome change. It is not dangerous. It reflects reduced estrogen stimulation of the endometrium.
The SURMOUNT post hoc analysis on reproductive-age women confirmed that premenopausal participants on tirzepatide experienced metabolic improvements without evidence of reproductive harm (PMC, 2025). Lighter periods in this context are a metabolic consequence, not a toxicity signal.
Irregular Timing
Cycles may shorten to 21-24 days or lengthen to 35-45 days during the first 3-4 months of treatment. The irregularity tracks with weight loss velocity. The fastest weight loss occurs during the dose escalation phase (weeks 4-20), which is also when menstrual irregularity peaks. Once you reach a stable dose and weight loss decelerates, cycles typically regularize.
If your cycle becomes unpredictable, tracking basal body temperature or using ovulation predictor kits can help identify whether you are still ovulating. Anovulatory cycles are common during rapid weight loss and are usually temporary.
Missed Periods
A missed period during tirzepatide treatment has three possible explanations: pregnancy (tirzepatide can improve fertility, see below), severe caloric deficit suppressing ovulation, or the normal variation that occurs during metabolic recalibration. Always rule out pregnancy first. Use a home test if your period is more than 7 days late, especially if you rely on oral contraceptives (whose effectiveness may be reduced, see the contraception section below).
If you miss three consecutive periods and pregnancy is excluded, consult your prescriber. Prolonged amenorrhea from energy deficit can affect bone density over time.
Heavier or More Painful Periods
Less common but reported. In some women, the hormonal recalibration temporarily increases prostaglandin production, intensifying cramps. Others experience a "rebound" heavier period after a lighter cycle, as the endometrium overcompensates. These changes are transient and should stabilize within 2-3 cycles. If heavy bleeding soaks through a pad or tampon hourly for more than 2 hours, seek medical evaluation.
Tirzepatide, PCOS, and Fertility
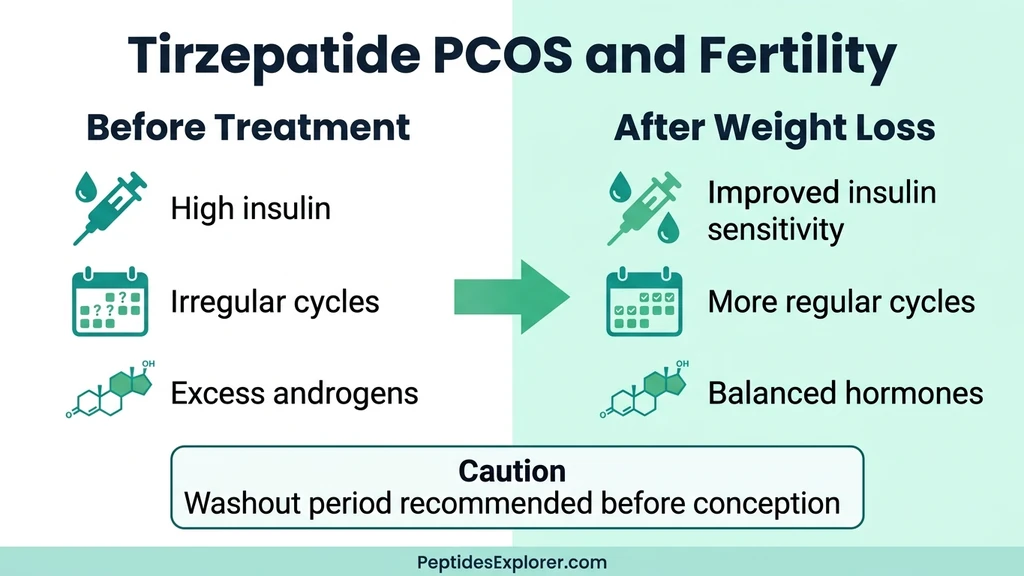
Polycystic ovary syndrome affects 8-13% of reproductive-age women. The hallmark triad: insulin resistance, excess androgens, and irregular or absent ovulation. Tirzepatide addresses the root metabolic dysfunction more effectively than any previous GLP-1 drug, and the fertility implications are significant.
How Tirzepatide Improves PCOS Symptoms
A 2023 review in the Journal of Clinical Medicine evaluated the potential utility of tirzepatide for PCOS management (Jensterle et al., JCM 2023). The theoretical framework is compelling:
Insulin reduction: Tirzepatide reduces hyperinsulinemia, which directly lowers ovarian androgen production. Lower androgens mean less acne, less hirsutism, and restored follicular development.
Weight loss: Every 5% body weight reduction improves menstrual regularity in PCOS. Tirzepatide produces 15-22% weight loss. Women with PCOS on GLP-1 drugs report cycle normalization rates of up to 43% in observational data.
GIP receptor activation: Unlike pure GLP-1 agonists like semaglutide, tirzepatide also activates GIP receptors. GIP signaling in adipose tissue may enhance fat mobilization and improve the hormonal milieu beyond what GLP-1 alone achieves.
Inflammation reduction: Chronic low-grade inflammation drives PCOS progression. Weight loss from tirzepatide reduces inflammatory markers (CRP, IL-6), which may further restore ovarian function.
For women exploring peptide options specifically designed for their needs, see our guide on peptides for women over 40.
The Surprise Pregnancy Risk
Here is the paradox that catches women off guard: tirzepatide can restore fertility in women who believed they could not conceive. A woman with PCOS who has not ovulated in 18 months starts tirzepatide. Within 3-4 months, insulin sensitivity improves, androgens drop, and ovulation resumes silently. If she is relying on her history of anovulation as de facto contraception, she may become pregnant without realizing her fertility window reopened.
This is not theoretical. Clinicians have reported unplanned pregnancies in women with long-standing PCOS anovulation after starting GLP-1 drugs. The FDA label for tirzepatide (Mounjaro/Zepbound) recommends discontinuing the drug at least 2 months before a planned pregnancy due to insufficient safety data in human pregnancy.
If you have PCOS and are not planning pregnancy, use reliable contraception. If you are planning pregnancy, work with your prescriber to time discontinuation. The recommendation is a washout period of at least 2 months (5 half-lives) before attempting conception.
Tirzepatide as a Fertility Preparation Strategy
Some reproductive endocrinologists are prescribing tirzepatide as a pre-conception weight loss strategy for women with obesity-related infertility. The logic: lose 15-20% body weight, normalize insulin and androgens, restore ovulation, then discontinue tirzepatide and attempt conception with a healthier metabolic profile.
Early observational data supports this approach. Women who achieved 10-15% weight loss before IVF had higher clinical pregnancy rates and lower miscarriage rates in retrospective analyses. Tirzepatide accelerates this weight loss timeline from 12-18 months (diet and exercise) to 4-6 months.
Discuss this strategy with a reproductive endocrinologist. Tirzepatide is not FDA-approved for fertility treatment, and its use in this context is off-label. For a broader look at weight loss outcomes, see tirzepatide before and after results.
The Critical Birth Control Warning

This section demands attention. Tirzepatide reduces the absorption of oral hormonal contraceptives. The FDA prescribing information includes a specific drug interaction warning. Ignoring it risks unplanned pregnancy.
What the Data Shows
A pharmacokinetic study measured the impact of a single 5 mg tirzepatide dose on oral contraceptive absorption (Sahbaz et al., JAPhA 2023). The results:
| Contraceptive Component | Peak Concentration Reduction | Total Exposure Reduction |
|---|---|---|
| Ethinyl estradiol | 59% | 20% |
| Norgestimate | 66% | 21% |
| Norelgestromin | 55% | 23% |
A 66% reduction in peak concentration of norgestimate is clinically meaningful. Peak levels drive the contraceptive effect. If the pill's active component never reaches therapeutic concentration, ovulation suppression fails.
The mechanism is delayed gastric emptying. Tirzepatide slows stomach emptying by 20-30%, which alters when and how much of the oral contraceptive is absorbed in the small intestine. This effect is most pronounced after the first dose and after each dose escalation, then diminishes as the body adapts.
What Makes Tirzepatide Different From Semaglutide
This interaction is unique to tirzepatide among GLP-1 drugs. Studies on semaglutide, liraglutide, and dulaglutide showed no clinically significant reduction in oral contraceptive bioavailability. The likely explanation: tirzepatide's dual GIP/GLP-1 mechanism produces more pronounced gastric emptying delay than pure GLP-1 agonists.
This distinction matters. If you previously used oral birth control while on semaglutide without issues, do not assume the same applies to tirzepatide. The drugs are pharmacologically different. For a broader comparison, see our semaglutide vs tirzepatide conversion guide.
FDA Recommendations
The Mounjaro and Zepbound prescribing labels state:
- 1.Patients using oral hormonal contraceptives should switch to a non-oral method (IUD, implant, injection, patch, ring) OR
- 2.Add a barrier method (condoms) for 4 weeks after tirzepatide initiation AND for 4 weeks after each dose escalation
The 4-week window accounts for the adjustment period during which gastric emptying delay is most pronounced. After 4 weeks at a stable dose, the body partially adapts and absorption normalizes, though individual variation exists.
Non-oral contraceptives are unaffected because they bypass the GI tract entirely. An IUD, hormonal implant (Nexplanon), injectable (Depo-Provera), patch, or vaginal ring delivers hormones directly into the bloodstream or locally, avoiding the gastric emptying interaction.
Check our tirzepatide drug interactions guide for other medications affected by delayed gastric emptying.
Cycle Changes by Treatment Phase
Menstrual changes follow a predictable pattern that mirrors the tirzepatide dose escalation schedule.
Weeks 1-4 (Starting Dose: 2.5 mg)
Weight loss is minimal at the starting dose. Most women notice no menstrual changes during this period. Gastric emptying delay begins, which may cause nausea or bloating but rarely affects the cycle. If you are on oral contraceptives, this is when to implement your backup method.
Weeks 5-16 (Dose Escalation: 5-10 mg)
This is the window of maximum disruption. Weight loss accelerates. The body sheds fat rapidly. Estrogen levels shift. Insulin sensitivity improves. The HPO axis recalibrates. Expect the highest likelihood of cycle irregularity during this phase. Periods may arrive early, late, lighter, or be skipped entirely.
Women with PCOS may notice their first regular period in months during this phase. Women with previously regular cycles may experience their first irregular one. Both are the same phenomenon viewed from opposite metabolic starting points.
Weeks 17-36 (Maintenance: 10-15 mg)
Weight loss velocity decreases as you approach your plateau. Estrogen levels stabilize at their new lower baseline. The HPO axis adapts. Most women report cycle normalization during this phase. Periods become predictable again, though they may be lighter than pre-treatment baseline due to lower body fat and estrogen.
If cycles remain irregular after 6 months at a stable dose, request hormonal testing (FSH, LH, estradiol, progesterone, thyroid panel). Persistent irregularity may indicate an underlying condition unmasked by weight loss rather than a treatment side effect. For long-term management strategies, see tirzepatide maintenance dose after weight loss.
Beyond Week 36 (Long-Term Treatment)
SURMOUNT-4 (88 weeks) showed that side effects, including those affecting reproductive-age women, stabilized after the first year (Wadden et al., 2023). Women who remain on tirzepatide long-term typically settle into a new menstrual pattern that reflects their lower body weight and improved metabolic status.
The new pattern is not abnormal. It is the pattern appropriate for your current physiology. A woman who weighed 220 pounds and now weighs 175 pounds has a different hormonal profile. Her cycles reflect that change.
How to Manage Menstrual Changes on Tirzepatide
Five strategies reduce disruption and help you distinguish normal adjustment from concerning symptoms.
Track Your Cycle Meticulously
Use an app (Clue, Flo, Natural Cycles) to log cycle start dates, flow intensity, symptoms, and basal body temperature. This data helps your prescriber distinguish tirzepatide-related changes from other causes. Track your tirzepatide dose changes alongside cycle data. Correlations will emerge quickly.
Maintain Adequate Caloric Intake
The hypothalamus suppresses ovulation when energy availability drops too low. Eating at least 1,200-1,400 calories daily with adequate protein (0.8-1.0g per pound body weight) preserves reproductive function while still allowing weight loss. Extreme caloric restriction (below 1,000 calories) invites amenorrhea.
Protein is especially critical. It provides the amino acids needed for hormone synthesis and helps preserve lean mass during tirzepatide weight loss. See our nutrition guide for tirzepatide for meal planning strategies.
Address Micronutrient Deficiencies
Rapid weight loss on tirzepatide can deplete nutrients essential for menstrual health:
Iron: Heavy periods deplete iron. Tirzepatide-related appetite suppression reduces dietary iron intake. Request a ferritin test. Supplement if below 30 ng/mL.
Vitamin D: Levels below 30 ng/mL correlate with menstrual irregularity and worse PCOS symptoms. Supplement 2,000-4,000 IU daily if deficient.
Zinc: Essential for progesterone production and luteal phase support. 15-30 mg daily if dietary intake is low.
Magnesium: Reduces menstrual cramps and supports progesterone. 200-400 mg daily (glycinate form for best absorption).
Consult your prescriber before starting supplements. For broader safety guidance, see our peptide safety guide.
Time Your Dose Strategically
Some women report that injecting tirzepatide on the same day each week, ideally just after their period ends, minimizes mid-cycle disruption. The pharmacokinetic peak occurs 24-72 hours post-injection, and timing it outside the ovulatory window may reduce interference. This is anecdotal, not evidence-based, but logistically simple to implement.
For injection timing and technique, see our guide on how to inject tirzepatide. For the best injection locations, see best injection sites for tirzepatide.
When to Contact Your Doctor
Normal adjustment: lighter periods, cycle shifting by 3-7 days, one missed period during dose escalation.
Contact your prescriber if you experience: - Three or more consecutive missed periods (after ruling out pregnancy) - Heavy bleeding that soaks a pad hourly for 2+ hours - Severe pelvic pain not associated with your typical menstrual pattern - New onset of mid-cycle bleeding lasting more than 3 days - Signs of pregnancy (especially if using oral contraceptives)
These symptoms may indicate conditions unmasked by weight loss (fibroids, endometriosis, thyroid dysfunction) rather than tirzepatide effects.
What the Research Says
SURMOUNT-1 (Jastreboff et al., 2022) 2,539 participants. Primary endpoints were weight loss and glycemic control. Menstrual changes were not a pre-specified endpoint. Adverse event reporting captured general categories but did not systematically track cycle irregularity. The 22.5% weight loss at 15 mg is the most aggressive of any approved GLP-1 drug (PubMed).
SURMOUNT Post Hoc Analysis (2025) Retrospective analysis of reproductive-age women in the SURMOUNT program found weight loss was consistent across premenopausal, perimenopausal, and postmenopausal women. Premenopausal women showed metabolic improvements without evidence of reproductive harm (PMC).
Tirzepatide and PCOS Review (Jensterle et al., JCM 2023) Evaluated the theoretical framework for tirzepatide in PCOS management. Concluded that the dual GIP/GLP-1 mechanism may offer superior insulin sensitization compared to pure GLP-1 agonists, with potential benefits for ovulatory function (PubMed).
Oral Contraceptive Interaction Study (Sahbaz et al., 2023) Demonstrated that tirzepatide reduced peak concentrations of ethinyl estradiol by 59% and norgestimate by 66%. This effect was specific to tirzepatide and not observed with semaglutide, liraglutide, or dulaglutide (PubMed).
Tirzepatide and Hormone Therapy in Menopause (Lancet, 2025) Postmenopausal women on concurrent hormone therapy achieved greater weight loss with tirzepatide than those without hormone therapy, suggesting estrogen and GLP-1 signaling interact synergistically (Lancet Obstetrics00145-1/abstract)).
The gap in the literature is clear: no prospective, randomized trial has specifically examined tirzepatide's effects on menstrual cycle parameters as a primary endpoint. The SURMOUNT and SURPASS programs were designed for weight loss and glycemic outcomes. Menstrual data comes from adverse event reports, post hoc analyses, and observational surveys.
Tirzepatide vs. Semaglutide: Period Effects Compared
Both drugs cause menstrual changes through the same weight-loss-driven mechanisms. The key differences:
| Factor | Tirzepatide | Semaglutide |
|---|---|---|
| Weight loss magnitude | 15-22.5% | 10-14.9% |
| Speed of weight loss | Faster | Moderate |
| Estrogen disruption potential | Higher (more fat lost faster) | Lower |
| Birth control interaction | Yes (FDA warning) | No significant interaction |
| Insulin sensitivity improvement | Greater (dual GIP/GLP-1) | Moderate (GLP-1 only) |
| PCOS cycle normalization | Potentially stronger | Documented benefit |
| Gastric emptying delay | More pronounced | Less pronounced |
For women where menstrual stability is a priority, semaglutide may cause fewer cycle disruptions simply because it produces slower, less dramatic weight loss. For women with PCOS seeking cycle normalization, tirzepatide's stronger insulin sensitization may be advantageous. Neither drug is inherently "better" for menstrual health. The choice depends on your clinical goals.
For a complete drug comparison, see best peptides for weight loss.
Important Safety Warnings
Pregnancy: Tirzepatide is not approved for use during pregnancy. Animal studies showed adverse fetal effects. Discontinue at least 2 months before planned conception. If you discover you are pregnant while on tirzepatide, stop immediately and contact your prescriber.
Breastfeeding: Insufficient human data on tirzepatide excretion in breast milk. The prescribing label advises weighing risks versus benefits. Most prescribers recommend discontinuation during breastfeeding.
Oral contraceptives: The absorption reduction is not theoretical. It is measured, dose-dependent, and clinically significant. Switch to a non-oral method or add barrier contraception. This applies to combined oral contraceptives and progestin-only pills.
Thyroid: Tirzepatide carries a boxed warning for thyroid C-cell tumors (observed in rodents). Rapid weight loss can also unmask subclinical thyroid dysfunction. If menstrual irregularity is accompanied by fatigue, cold intolerance, or hair loss, request thyroid function tests.
Bone density: Prolonged amenorrhea (6+ months) from any cause, including severe caloric restriction on GLP-1 drugs, can reduce bone mineral density. If you miss multiple cycles, discuss bone health screening with your prescriber.
This information does not replace medical advice. Discuss any menstrual changes with your healthcare provider, especially if you have pre-existing gynecological conditions.
Related Side Effects
Does Tirzepatide Cause Hair Loss? Hair thinning (2.8-5.7%) and menstrual changes share the same root cause: rapid fat loss and nutritional shifts. Management strategies overlap. Adequate protein and micronutrients protect both hair follicles and reproductive function.
Does Tirzepatide Cause Constipation? Constipation (6-12%) reduces nutrient absorption, which can compound the deficiencies driving menstrual irregularity. Adequate fiber, hydration, and magnesium address both.
Does Tirzepatide Make You Tired? Fatigue alongside menstrual changes may signal thyroid dysfunction unmasked by weight loss. If both symptoms appear together, request a thyroid panel.
Tirzepatide Drug Interactions The oral contraceptive interaction is one of several drug absorption issues caused by delayed gastric emptying. Thyroid medications and antibiotics may also be affected.
Frequently Asked Questions
Does Mounjaro affect your period?
Yes, Mounjaro (tirzepatide) can cause menstrual changes including lighter periods, irregular timing, and missed cycles. These changes result from rapid fat loss reducing estrogen production, improved insulin sensitivity recalibrating hormones, and caloric deficit signaling the brain to deprioritize reproduction. About 27% of GLP-1 users report cycle changes. Most normalize within 3-6 months. See our tirzepatide long-term side effects guide for the full side effect profile.
Can tirzepatide cause missed periods?
Yes. Missed periods on tirzepatide most commonly result from severe caloric deficit suppressing ovulation, rapid hormonal recalibration during dose escalation, or (paradoxically) restored fertility in women with PCOS. Always rule out pregnancy first, especially if you use oral contraceptives, whose absorption tirzepatide reduces by up to 66%. Use our tirzepatide dosage calculator to ensure your dose escalation follows the recommended schedule.
Does tirzepatide affect birth control pills?
Yes. Tirzepatide reduces peak absorption of oral contraceptive components by 55-66% due to delayed gastric emptying. The FDA recommends switching to a non-oral method (IUD, implant, patch, ring) or adding a barrier method for 4 weeks after starting tirzepatide and after each dose increase. This interaction is unique to tirzepatide and does not occur with semaglutide or other GLP-1 drugs.
Can you get pregnant on tirzepatide?
Yes, and more easily than expected. Tirzepatide improves insulin sensitivity and reduces androgens, which can restore ovulation in women with PCOS who previously did not ovulate regularly. If you are not planning pregnancy, use reliable contraception (preferably non-oral). If you are planning pregnancy, discontinue tirzepatide at least 2 months before attempting conception. See peptides for women over 40 for additional reproductive health considerations.
Does tirzepatide lower estrogen levels?
Indirectly, yes. Adipose tissue produces estrogen via aromatase. When tirzepatide causes rapid fat loss (15-22% body weight), the aromatase factory shrinks, reducing circulating estrogen. This is most pronounced during active weight loss and stabilizes once weight plateaus. The effect is identical to estrogen changes after bariatric surgery. See tirzepatide before and after for real-world weight loss outcomes and timelines.
How long do period changes last on tirzepatide?
Most menstrual changes resolve within 3-6 months of reaching a stable dose. The worst irregularity occurs during the dose escalation phase (weeks 5-16) when weight loss is fastest. By 6 months at maintenance dose, cycles have typically established a new pattern consistent with your lower body weight. If irregularity persists beyond 6 months, consult your prescriber for hormonal testing. Read our peptide safety guide for monitoring recommendations.
Is tirzepatide safe for women with PCOS?
Tirzepatide is not FDA-approved for PCOS treatment, but its metabolic effects align with PCOS management goals: reduced insulin resistance, lower androgens, weight loss, and potential ovulation restoration. A 2023 review in the Journal of Clinical Medicine highlighted its theoretical utility for PCOS. The key caution: restored fertility means you need contraception if pregnancy is not desired. Discuss with your endocrinologist. For dose guidance, use our tirzepatide dosage calculator.
Should I stop tirzepatide if my period changes?
No. Menstrual changes alone are not a reason to discontinue tirzepatide. They reflect metabolic improvement, not harm. Continue treatment and track your cycles. Contact your prescriber if you miss 3+ consecutive periods (after excluding pregnancy), experience heavy bleeding requiring hourly pad changes, or develop pelvic pain unrelated to your normal pattern. For managing other side effects, see our tirzepatide maintenance dose guide.
The Bottom Line
Tirzepatide does not directly disrupt reproductive hormones. The menstrual changes women experience are downstream consequences of rapid fat loss reducing estrogen, improved insulin sensitivity recalibrating the HPO axis, and caloric deficit signaling the hypothalamus to deprioritize reproduction. These are the same changes that follow bariatric surgery, crash diets, and intense athletic training. The difference is speed: tirzepatide produces the fastest weight loss of any approved GLP-1 drug, which compresses the adjustment period.
For most women, cycle changes are temporary and resolve within 3-6 months at a stable dose. For women with PCOS, the changes may actually be therapeutic, restoring ovulation that was suppressed for years. The birth control interaction is the one non-negotiable action item: switch to a non-oral method or add barrier contraception.
Track your cycles. Maintain adequate calories and protein. Address micronutrient deficiencies. Rule out pregnancy if your period is late. Report persistent changes to your prescriber.
Use our tirzepatide dosage calculator for titration planning. For broader context on side effects, read our tirzepatide long-term side effects guide.
Related Articles: - Tirzepatide Before and After - real-world weight loss timelines and outcomes - Does Tirzepatide Cause Hair Loss? - another estrogen-linked side effect - Peptides for Women Over 40 - comprehensive guide for women's peptide use - Best Peptides for Weight Loss - comparing all weight loss peptide options - Tirzepatide Maintenance Dose After Weight Loss - long-term management strategies
Related Articles
Does Tirzepatide Cause Insomnia?
Does tirzepatide cause insomnia? Trials say no, but 5 indirect pathways explain lost sleep. GI, blood sugar, and caloric deficit fixes.
Can Tirzepatide Cause Joint Pain? Clinical Data
Can tirzepatide cause joint pain? SURMOUNT arthralgia rates match placebo. Review 5 indirect causes, uric acid risks, and 8 relief tactics.
Does Tirzepatide Cause Muscle Loss?
SURMOUNT-1 DEXA data: 25% of tirzepatide weight loss is lean mass (5.6 kg at 72 weeks). Protein targets, resistance training, and supplements.
Can Tirzepatide Cause Anxiety?
Anxiety is reported in 3-6% of tirzepatide users. Learn SURMOUNT/SURPASS data, GLP-1 brain mechanisms, and 7 management strategies.