
You increased your tirzepatide dose to 10 mg last Friday. By Tuesday, you are lying awake at 2 a.m., stomach churning, mind racing through a mental list of everything you ate that day. Tirzepatide does not directly cause insomnia. No clinical trial in the SURMOUNT or SURPASS programs identified insomnia as a statistically significant adverse event compared to placebo. However, five indirect pathways explain why some users report sleep disruption, and all five are fixable.
| Quick Reference | Details |
|---|---|
| Clinical evidence of direct insomnia | None (not statistically significant vs. placebo in Phase 3 trials) |
| Indirect sleep disruption prevalence | Estimated 8-15% of users, based on GLP-1 RA class data |
| Primary indirect causes | GI side effects, blood sugar shifts, caloric deficit, rapid weight loss, adjustment anxiety |
| Onset | Typically within 1-2 weeks of starting or dose escalation |
| Peak | Weeks 2-6 at each new dose level |
| Resolution | 4-8 weeks at a stable dose for most users |
| Key fixes | Meal timing (3+ hrs before bed), sleep hygiene, electrolyte repletion |
| Evidence level | Phase 3 trials (SURMOUNT-1 through SURMOUNT-4, SURMOUNT-OSA, SURPASS-1 through SURPASS-5) |
Use our tirzepatide dosage calculator to plan a titration schedule that minimizes side effects. For a companion guide on daytime energy, see does tirzepatide make you tired. For broader safety context, our peptide safety guide covers the fundamentals.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What the Clinical Trials Actually Show
The most important fact first: across more than 20,000 participants studied in the combined SURMOUNT and SURPASS clinical programs, insomnia was not reported as a common adverse event at rates statistically different from placebo.
SURMOUNT-1: Obesity Without Diabetes
SURMOUNT-1 enrolled 2,539 adults with obesity (BMI 30+) or overweight (BMI 27+) with at least one weight-related comorbidity. The most frequently reported adverse events were gastrointestinal: nausea (24.6% at 5 mg, up to 33.3% at 10 mg), diarrhea, constipation, and vomiting (Jastreboff et al., NEJM 2022). Insomnia did not appear among the top adverse events at any dose level.
SURPASS-1 Through SURPASS-5: Type 2 Diabetes
The SURPASS trials evaluated tirzepatide in over 7,000 adults with type 2 diabetes. Adverse event profiles were dominated by GI complaints. Fatigue appeared in 5-8% of participants across dose levels, but insomnia was not broken out as a distinct endpoint (Rosenstock et al., Lancet 2021).
SURMOUNT-OSA: Sleep Apnea Population
The SURMOUNT-OSA trials specifically studied sleep outcomes. Rather than worsening sleep, tirzepatide-treated participants showed significantly improved Patient-Reported Outcomes Measurement Information System (PROMIS) Sleep Disturbance scores and PROMIS Sleep-related Impairment scores at Week 52 compared to placebo. The apnea-hypopnea index dropped by up to two-thirds (Malhotra et al., NEJM 2024). Tirzepatide improved sleep quality in people whose sleep was previously disrupted by obstructive sleep apnea.
| Trial | Population | Sleep-Related Findings |
|---|---|---|
| SURMOUNT-1 | Obesity (n=2,539) | Insomnia not among top adverse events |
| SURMOUNT-OSA | OSA + Obesity (n=469) | Significant improvement in sleep disturbance scores |
| SURPASS-1 to -5 | T2D (n=7,000+) | Fatigue 5-8%; insomnia not separately reported |
The clinical data tells a clear story: tirzepatide does not cause insomnia through a direct pharmacological mechanism. But clinical trials capture averages, not outliers. The question for the person staring at their ceiling at 2 a.m. is not whether the average participant slept well. It is why they, specifically, cannot sleep.
Five Indirect Pathways from Tirzepatide to Lost Sleep
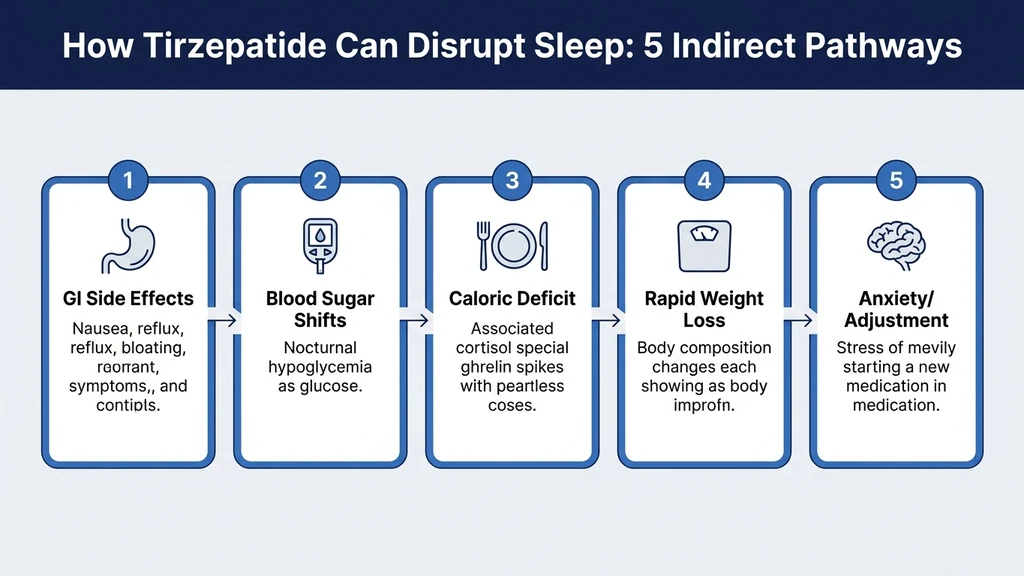
Think of sleep as a house built on five pillars: comfortable digestion, stable blood sugar, adequate caloric intake, hormonal equilibrium, and psychological calm. Tirzepatide can shake each of these pillars without ever touching the sleep centers of the brain directly. Most users who report insomnia on tirzepatide are experiencing disruption in two or three of these pathways simultaneously.
Pathway 1: Gastrointestinal Disruption at Night
Tirzepatide slows gastric emptying through GLP-1 and GIP receptor activation. Food that would normally leave your stomach in 2-3 hours may linger for 4-6 hours (Samms et al., Diabetes 2023). When you eat dinner at 7 p.m. and lie down at 10 p.m., that meal is still sitting in your stomach. The result: acid reflux, bloating, nausea, and abdominal discomfort that make falling asleep difficult and staying asleep harder.
Nausea alone affects 24-33% of tirzepatide users in clinical trials. Reflux may not be captured under that label but follows the same mechanism. A meta-analysis of GLP-1 receptor agonist trials found GI-related sleep disturbances in 8-15% of active treatment groups versus 3-5% on placebo (Nauck et al., Lancet Diabetes Endocrinol 2021).
The fix is mechanical, not pharmacological. Stop eating 3-4 hours before bed. Keep evening meals small and low in fat. Elevate the head of your bed by 6 inches if reflux is the primary issue. For comprehensive nausea strategies, see how to relieve nausea from semaglutide, which applies equally to tirzepatide's GI effects.
Pathway 2: Blood Sugar Fluctuations and Nocturnal Hypoglycemia
Tirzepatide lowers fasting glucose by 35-60 mg/dL in people with type 2 diabetes (Rosenstock et al., 2021). Even in non-diabetic users, the combination of reduced food intake and enhanced insulin sensitivity can push blood sugar into the low-normal range (65-80 mg/dL) during overnight fasting.
When blood glucose drops below 70 mg/dL, the body mounts a counterregulatory response: adrenaline and cortisol surge to mobilize stored glucose. This response triggers nighttime awakenings, sweating, heart pounding, and anxiety. You may wake at 3 a.m. feeling wired and unable to return to sleep. Mild nocturnal hypoglycemia can fragment sleep without producing obvious symptoms, reducing sleep quality without full awakening (Pillar et al., Diabetes Care 2003).
The risk is highest when tirzepatide is combined with insulin or sulfonylureas. The tirzepatide drug interactions guide covers these combinations in detail. Non-diabetic users can mitigate this pathway by including a small protein-containing snack (10-15 g of protein, such as Greek yogurt or cottage cheese) 1-2 hours before bed to provide slow-release glucose overnight.
Pathway 3: Caloric Deficit and Hormonal Disruption
Tirzepatide reduces daily caloric intake by 500-1,000 calories in most users. SURMOUNT-1 participants on 15 mg lost 22.5% of body weight over 72 weeks, requiring a sustained daily deficit of 750-1,000 calories (Jastreboff et al., 2022). That deficit triggers a cascade of hormonal changes that directly affect sleep.
Cortisol elevation. Caloric restriction increases evening cortisol levels by 10-20% (Tomiyama et al., Psychosom Med 2010). Cortisol is the body's primary wakefulness hormone. Elevated cortisol at bedtime delays sleep onset, reduces slow-wave (deep) sleep, and increases early-morning awakening. The body interprets caloric deficit as a mild stressor, and stress hormones do not respect your bedtime.
Ghrelin dysregulation. The hunger hormone ghrelin rises during caloric restriction, particularly at night. A study of moderate caloric restriction combined with sleep restriction found that 24-hour ghrelin concentrations increased significantly (Spiegel et al., Ann Intern Med 2004). Tirzepatide suppresses appetite through central mechanisms, but ghrelin's peripheral effects on arousal may persist. The result: you do not feel hungry, but you feel restless.
Leptin decline. As body fat decreases, leptin production drops. Low leptin signals the hypothalamus that energy reserves are depleted, triggering a survival response that includes lighter, more fragmented sleep (Rosenbaum & Leibel, Int J Obes 2010). This explains why sleep disruption often intensifies during the phase of most rapid weight loss (months 2-4).
For strategies on maintaining energy during weight loss, see does tirzepatide make you tired.
Pathway 4: Rapid Body Composition Changes
Losing 15-25% of body weight in under a year changes more than the number on the scale. Rapid weight loss alters thermoregulation, respiratory mechanics, and body positioning during sleep.
Temperature regulation. Adipose tissue acts as insulation. As fat mass decreases, many users report feeling cold at night, leading to difficulty falling asleep or frequent awakenings. Metabolic adaptation reduces resting energy expenditure by 10-15% beyond what weight loss alone predicts (Rosenbaum & Leibel, 2010), and a lower metabolic rate generates less body heat.
Sleep position discomfort. People who have carried significant weight for years develop sleeping positions adapted to that weight. Losing 40-60 pounds changes pressure points. Hip bones and shoulders press into the mattress differently. Side sleepers may need a different pillow height as neck and shoulder geometry changes. These physical discomforts are real sleep disruptors that have nothing to do with tirzepatide pharmacology.
Sleep apnea improvement paradox. Counterintuitively, as obstructive sleep apnea improves with weight loss, some users initially feel like their sleep is worse. They were accustomed to the heavy, sedated sleep that characterizes untreated OSA. Normal-architecture sleep feels lighter and less "deep" by comparison, even though it is healthier. The SURMOUNT-OSA data confirms that objective sleep metrics improve with tirzepatide, even if subjective perception lags behind (Malhotra et al., 2024).
Track your progress with tirzepatide before and after to contextualize body composition changes against the weight loss timeline.
Pathway 5: Psychological Adjustment and Anxiety
Starting a new injectable medication triggers hypervigilance in many people. You monitor every sensation. You check forums at 11 p.m. to see if your symptoms are normal. You lie awake wondering whether the injection site lump is a problem. This anxiety-driven insomnia is independent of tirzepatide's pharmacology but inseparable from the treatment experience.
A subset of tirzepatide users report increased anxiety during the first weeks of treatment. Whether this is a pharmacological effect of GLP-1/GIP receptor activation in the amygdala and prefrontal cortex, or a psychological response to starting an injectable, remains debated. A 2023 analysis of FDA Adverse Event Reporting System data found anxiety reports associated with GLP-1 receptor agonists, though the signal was weak and confounded by baseline anxiety rates in the obesity population (McIntyre et al., Mol Psychiatry 2024).
For users experiencing anxiety alongside insomnia, our guide on can tirzepatide cause anxiety covers the pharmacological evidence and management strategies.
Sleep Architecture: What Actually Happens When You Sleep on Tirzepatide
No study has placed tirzepatide users in a sleep lab to measure polysomnographic changes. The data we have comes from the SURMOUNT-OSA trials, which used patient-reported sleep questionnaires, and from the broader GLP-1 receptor agonist literature.
Normal sleep cycles through four stages roughly every 90 minutes:
| Sleep Stage | Duration (per cycle) | Function | Potential Tirzepatide Impact |
|---|---|---|---|
| N1 (Light sleep) | 5-10 min | Transition from wakefulness | GI discomfort may prolong this stage |
| N2 (True sleep) | 20-30 min | Memory consolidation begins | Blood sugar drops may fragment N2 |
| N3 (Deep/slow-wave) | 20-40 min | Physical repair, growth hormone release | Cortisol elevation may reduce N3 duration |
| REM | 10-20 min | Emotional processing, dreaming | Some users report vivid dreams on GLP-1 RAs |
A 2026 review in the International Journal of Molecular Sciences positioned GLP-1 receptor agonists as "chronometabolic modulators," noting that GLP-1 signaling is under circadian regulation and may influence central and peripheral clock systems. Tirzepatide's half-life exceeds 5 days, providing near-continuous receptor engagement that could affect circadian-aligned eating patterns and, by extension, sleep timing (Timmermans et al., Int J Mol Sci 2026).
The practical implication: tirzepatide likely does not degrade sleep architecture directly. But when GI distress wakes you at 1 a.m., a cortisol spike wakes you at 3 a.m., and an alarm goes off at 6 a.m., you get fragments of every sleep stage without completing enough full cycles. The result is morning fatigue that feels like insomnia even if total time in bed was adequate.
For peptides that actively promote sleep, see our guide on peptides for sleep, covering DSIP, epitalon, and other sleep-specific compounds.
Who Is Most at Risk for Sleep Disruption?
Not everyone on tirzepatide loses sleep. Based on clinical trial subgroup analyses and the mechanistic pathways described above, certain profiles carry higher risk.
Higher-Risk Profiles
Aggressive dose escalators. Users who increase from 2.5 mg to 7.5 mg or higher within 6-8 weeks experience more severe GI side effects, which peak during the first 2 weeks at each new dose level. Slower titration (staying at each dose for 4-6 weeks instead of the minimum 4 weeks) reduces GI disruption and its downstream sleep effects.
Evening eaters. People whose largest meal is dinner face the worst gastric emptying delays. Tirzepatide slows stomach emptying by 40-60% compared to baseline. A large 8 p.m. dinner on a 10 mg dose may not clear the stomach until 2-3 a.m.
Concurrent insulin or sulfonylurea users. The risk of nocturnal hypoglycemia increases substantially when tirzepatide is combined with insulin secretagogues. The tirzepatide drug interactions guide covers dose adjustment protocols for these combinations.
Pre-existing anxiety or insomnia. Roughly 30% of adults have baseline insomnia (Roth, J Clin Sleep Med 2007). Starting a new medication provides a convenient attribution target for sleep problems that predate tirzepatide.
Rapid weight losers. Those losing more than 1.5% of body weight per week experience more pronounced cortisol and leptin shifts. The SURMOUNT-1 top responders (losing 25%+ of body weight) likely experienced more sleep disruption than average responders, though this was not separately analyzed.
Lower-Risk Profiles
Slow titrators with good GI tolerance. Users who experience minimal nausea and follow the standard 4-week escalation typically report no sleep changes.
Morning or midday largest-meal eaters. When the main caloric load is consumed by 1-2 p.m., gastric emptying delays resolve well before bedtime.
Users with pre-existing OSA. As demonstrated in SURMOUNT-OSA, tirzepatide actually improves sleep quality in this population by reducing apnea events. Weight loss of 10-15% can reduce the AHI by 50% or more.
Those with established sleep hygiene. Users who already maintain consistent sleep-wake times, cool bedroom temperatures, and screen-free wind-down routines experience less disruption from the indirect pathways.
How to Fix Sleep Problems on Tirzepatide

The approach follows a tiered strategy: start with behavioral fixes (free, effective, no side effects), move to nutritional support, and escalate to medical consultation only if insomnia persists beyond 4-6 weeks.
Tier 1: Behavioral and Timing Fixes
Move your largest meal to lunch. This single change addresses the GI pathway directly. When your biggest meal clears the stomach by 6-7 p.m. instead of 1-2 a.m., nighttime reflux, bloating, and nausea drop dramatically. Many tirzepatide users find that appetite is strongest in the first half of the day anyway, making this shift natural.
Stop eating 3-4 hours before bed. If dinner must remain your larger meal, finish eating by 6-7 p.m. for a 10-11 p.m. bedtime. On tirzepatide, the old rule of "no food 2 hours before bed" needs extending because gastric emptying is slower.
Time your injection for morning. While tirzepatide is a once-weekly injection with a 5-day half-life (making injection timing less critical than with shorter-acting peptides), some users report that injecting in the morning rather than evening reduces the intensity of GI side effects during the first 48 hours post-injection. For optimal timing strategies, see best time to take tirzepatide.
Lock in sleep hygiene basics. Keep the bedroom at 65-68F (18-20C). Maintain consistent sleep-wake times (even on weekends). Stop screens 30-60 minutes before bed. These rules matter more on tirzepatide because the indirect pathways erode your sleep margin. Good hygiene provides the buffer.
Block light and noise. Weight loss changes how your body regulates temperature and arousal. Blackout curtains and white noise machines compensate for the lighter sleep that accompanies reduced body fat and lower metabolic rate.
Tier 2: Nutritional Support
Magnesium glycinate (200-400 mg before bed). Magnesium supports GABAergic neurotransmission, the same calming pathway targeted by prescription sleep aids. Caloric restriction often depletes magnesium stores. Glycinate is the preferred form because it crosses the blood-brain barrier more effectively than citrate or oxide and causes less GI distress, an important consideration when tirzepatide is already taxing the digestive system (Abbasi et al., J Res Med Sci 2012).
Protein-containing evening snack. A small snack with 10-15 g of protein (Greek yogurt, cottage cheese, a handful of almonds) provides slow-release amino acids that stabilize overnight blood glucose. This addresses the nocturnal hypoglycemia pathway without adding significant calories. Pair with what to eat on tirzepatide for a comprehensive nutrition plan.
Electrolyte repletion. Sodium, potassium, and magnesium depletion from reduced food intake contributes to muscle cramps, restless legs, and general restlessness at night. An electrolyte supplement or homemade solution (1/4 tsp salt, 1/4 tsp potassium chloride, squeeze of lemon in water) taken in the afternoon can help.
Adequate total protein (1.0-1.2 g/kg/day). Protein is not just about muscle preservation during weight loss. Tryptophan, an essential amino acid found in protein-rich foods, is the precursor to serotonin and melatonin. Insufficient protein intake on tirzepatide may reduce melatonin production, further disrupting the sleep-wake cycle. For information on preserving muscle during weight loss, see does tirzepatide cause muscle loss.
Tier 3: Medical Consultation
Consult your prescribing physician if:
Insomnia persists beyond 4-6 weeks at a stable dose. Indirect pathway disruption should resolve as your body adapts to the new caloric intake and GI tolerance improves. Persistent insomnia suggests either a pre-existing sleep disorder (now unmasked by medication-related hypervigilance) or a separate medical issue.
You suspect nocturnal hypoglycemia. Waking with sweating, palpitations, tremor, or confusion between 2-4 a.m. warrants blood glucose monitoring. Your physician may adjust the tirzepatide dose, modify concurrent diabetes medications, or recommend a continuous glucose monitor for a 2-week diagnostic period.
Sleep disruption is severe (fewer than 4 hours per night). Severe insomnia impairs driving, cognitive function, and immune response. It may warrant temporary pharmacological intervention while the underlying causes are addressed.
New symptoms accompany the insomnia. Racing thoughts, persistent anxiety, mood changes, or manic symptoms alongside insomnia require urgent evaluation. GLP-1 receptor agonists interact with central nervous system pathways, and while serious psychiatric effects are rare, they demand prompt attention.
For a comprehensive overview of long-term considerations, see tirzepatide long-term side effects.
Tirzepatide vs. Semaglutide: Sleep Effects Compared
Both tirzepatide and semaglutide are GLP-1 receptor agonists. Both slow gastric emptying, reduce caloric intake, and lower blood sugar. The question is whether tirzepatide's additional GIP receptor agonism changes the sleep equation.
| Factor | Tirzepatide | Semaglutide |
|---|---|---|
| GLP-1 receptor activation | Yes | Yes |
| GIP receptor activation | Yes | No |
| Gastric emptying delay | More pronounced | Moderate |
| Nausea incidence | 24-33% | 20-44% (dose-dependent) |
| Weight loss magnitude | 20-25% (SURMOUNT-1) | 15-17% (STEP-1) |
| Half-life | ~5 days | ~7 days |
| OSA improvement data | SURMOUNT-OSA (positive) | STEP trials (limited) |
| Fatigue incidence | 12-20% | 8-15% |
The GIP receptor component is interesting. GIP receptors are present in the hypothalamus, a brain region that regulates both appetite and circadian rhythms. Whether dual agonism produces different sleep effects than GLP-1 agonism alone has not been studied head-to-head. The available data suggests comparable indirect sleep disruption profiles, with tirzepatide potentially producing more GI-related sleep disturbance due to stronger gastric emptying suppression.
Tirzepatide users who want to compare their experience may find our semaglutide vs tirzepatide conversion guide useful for understanding equivalent dose levels.
Can Melatonin Be Taken with Tirzepatide?
Yes, but with caveats. No direct drug interaction between melatonin and tirzepatide has been documented. Melatonin does not affect GLP-1 or GIP receptor signaling, and tirzepatide does not alter melatonin metabolism through the CYP1A2 enzyme pathway.
However, melatonin addresses only one of the five indirect pathways (circadian timing). If your insomnia stems from GI discomfort, blood sugar drops, or cortisol elevation, melatonin will provide minimal benefit. Think of melatonin as a timing signal, not a sedative. It tells your body when to sleep but does not force sleep in the presence of nausea or hypoglycemia.
Practical guidance:
- Dose: 0.5-3 mg, 30-60 minutes before target bedtime. Start with 0.5 mg. Higher is not better for melatonin.
- Form: Extended-release if you have trouble staying asleep; immediate-release if your issue is falling asleep.
- Timing: Consistent nightly use at the same time reinforces circadian rhythm.
- Caveat: Melatonin can slow gastric motility in some individuals, potentially worsening the GI pathway. If nausea increases after adding melatonin, discontinue it.
For other potential interactions, consult our tirzepatide drug interactions guide.
When Tirzepatide Actually Improves Sleep
The discussion so far has focused on sleep disruption, but the clinical evidence points in the opposite direction for many users. Tirzepatide improves sleep quality in several populations.
Obstructive sleep apnea. The SURMOUNT-OSA trials demonstrated a 55-63% reduction in the apnea-hypopnea index with tirzepatide versus placebo. Participants reported significantly improved sleep disturbance scores, sleep-related impairment scores, and overall quality of life (Malhotra et al., 2024). For the estimated 30 million Americans with OSA (many of whom are undiagnosed), tirzepatide-driven weight loss may resolve or substantially improve their sleep disorder.
Chronic pain reduction. Weight loss of 10-20% reduces mechanical load on joints, spine, and soft tissues. Users with chronic back pain, knee osteoarthritis, or plantar fasciitis often report improved sleep as pain diminishes. This mirrors the paradox seen with BPC-157 and insomnia, where healing a pain condition can improve sleep even when the peptide has theoretical stimulatory properties.
Reduced nocturnal reflux. Obesity is the strongest risk factor for gastroesophageal reflux disease (GERD). As body weight decreases, intra-abdominal pressure drops, and the lower esophageal sphincter functions more effectively. Users who had been waking with heartburn for years may find that tirzepatide's long-term weight loss eliminates the problem, even though short-term gastric emptying delays initially make it worse.
Improved metabolic health. Stable blood sugar, reduced systemic inflammation (CRP drops by 30-40% in clinical trials), and improved cardiovascular function all contribute to better sleep architecture. These benefits accumulate over months and often outweigh the acute disruptions experienced during the first 6-8 weeks.
A Timeline of Sleep Changes on Tirzepatide
Understanding the typical trajectory helps you distinguish expected adjustment from a problem that needs intervention.
Weeks 1-2 (2.5 mg starting dose): Most users notice no sleep changes at this dose. Appetite suppression is mild. GI side effects are usually minimal. If insomnia appears at this stage, it is likely psychological (adjustment anxiety) rather than physiological.
Weeks 3-6 (5 mg dose): Appetite suppression intensifies. GI side effects peak. This is when most sleep disruption reports emerge. Nausea, bloating, and reflux are strongest during the first 2 weeks at 5 mg. Users eating late dinners are most affected.
Weeks 7-12 (7.5-10 mg dose): The caloric deficit deepens. Weight loss accelerates. Cortisol and ghrelin shifts become more pronounced. Users losing more than 1.5% of body weight per week may notice lighter sleep, more early-morning awakening, and reduced deep sleep. GI tolerance usually improves, but the metabolic pathways now drive disruption.
Weeks 13-24 (10-15 mg dose): Body composition changes become significant (20-40 lbs lost). Temperature regulation shifts. Sleep positions may need adjustment. Paradoxically, OSA-related sleep improvements begin offsetting the negative pathways for users with pre-existing sleep apnea.
Months 6-12+ (maintenance phase): GI tolerance is established. The body adapts to the lower caloric intake. Hormonal markers stabilize at new setpoints. Most users report that sleep quality returns to baseline or improves compared to pre-treatment, particularly if they have lost enough weight to resolve OSA, chronic pain, or GERD.
Track your weight loss trajectory and compare with tirzepatide before and after results to understand where you fall on the response curve.
Important Safety Warnings
Tirzepatide (Mounjaro, Zepbound) is an FDA-approved prescription medication. All dosing decisions should be made in consultation with your prescribing physician.
Do not take prescription sleep medications without discussing tirzepatide. Benzodiazepines (diazepam, lorazepam), Z-drugs (zolpidem, eszopiclone), and antihistamine sleep aids (diphenhydramine, doxylamine) can all slow gastric motility. Combined with tirzepatide's gastric emptying delay, this increases the risk of gastroparesis-like symptoms, severe constipation, and potentially intestinal obstruction in rare cases.
Monitor blood glucose if you take insulin or sulfonylureas. The combination of these medications with tirzepatide increases nocturnal hypoglycemia risk. Your physician should reduce the dose of insulin or sulfonylurea when starting tirzepatide, per the prescribing information.
Report psychiatric symptoms promptly. While causation has not been established, post-marketing surveillance has identified anxiety, depression, and (rarely) suicidal ideation reports among GLP-1 receptor agonist users. Insomnia accompanied by racing thoughts, agitation, or mood changes warrants same-week evaluation by your prescriber.
Do not use compound tirzepatide from unregulated sources. The FDA has issued warnings about compounded tirzepatide products that may contain incorrect doses or impurities. Sleep disruption from an impure product may have nothing to do with tirzepatide itself. Use only FDA-approved brand-name products or compounds from FDA-registered 503B outsourcing facilities. For more on compound safety, see is compound tirzepatide safe.
Frequently Asked Questions
Does tirzepatide cause insomnia?
Tirzepatide does not directly cause insomnia. No clinical trial in the SURMOUNT or SURPASS programs reported insomnia as a statistically significant adverse event versus placebo. However, five indirect pathways (GI disruption, blood sugar shifts, caloric deficit, rapid weight loss, and adjustment anxiety) can disrupt sleep in 8-15% of users. All five pathways are manageable with behavioral and nutritional strategies. For dosing that minimizes side effects, use our tirzepatide dosage calculator.
Why can't I sleep after taking tirzepatide?
The most common cause is gastrointestinal discomfort from delayed gastric emptying. Tirzepatide slows stomach emptying by 40-60%, so food eaten at dinner may still be in your stomach at bedtime, causing nausea, reflux, and bloating. The fix: stop eating 3-4 hours before bed and shift your largest meal to lunch. Blood sugar fluctuations and caloric-deficit-driven cortisol elevation are the second and third most common causes. See best time to take tirzepatide for timing strategies.
Will tirzepatide insomnia go away?
For most users, yes. GI-related sleep disruption typically resolves within 4-6 weeks at a stable dose as the body adapts to delayed gastric emptying. Metabolic sleep disruption from caloric deficit and weight loss may take longer (2-4 months) as hormonal markers stabilize at new setpoints. The SURMOUNT-OSA trials showed that tirzepatide users reported significantly improved sleep disturbance scores at Week 52. If insomnia persists beyond 6 weeks at a stable dose, consult your physician. For long-term expectations, see tirzepatide long-term side effects.
Can I take melatonin with tirzepatide?
Yes. No direct drug interaction between melatonin and tirzepatide has been documented. Start with 0.5 mg of melatonin 30-60 minutes before bedtime and increase only if needed. However, melatonin addresses circadian timing, not GI discomfort or blood sugar fluctuations. If nausea is the primary cause of your insomnia, melatonin will not help. Note that melatonin can slow gastric motility in some people, potentially worsening GI symptoms. For other interactions, see tirzepatide drug interactions.
Does tirzepatide improve sleep for people with sleep apnea?
Yes, significantly. The SURMOUNT-OSA trials demonstrated a 55-63% reduction in the apnea-hypopnea index with tirzepatide compared to placebo. Participants also reported improved sleep disturbance scores and better overall quality of life. The mechanism is primarily weight loss: reducing body mass by 15-20% decreases pharyngeal fat and upper airway collapsibility. For sleep-promoting peptides, see peptides for sleep.
What sleep supplements are safe with tirzepatide?
Magnesium glycinate (200-400 mg before bed) has the strongest evidence for sleep support during caloric restriction and is generally well-tolerated alongside tirzepatide. L-theanine (100-200 mg) promotes relaxation without sedation. Melatonin (0.5-3 mg) is safe but may worsen GI symptoms in some users. Avoid diphenhydramine (Benadryl, ZzzQuil) and doxylamine (Unisom), which slow gastric motility and compound tirzepatide's emptying delay. Review all supplements with your physician. For comprehensive safety information, see our peptide safety guide.
Is insomnia worse at higher doses of tirzepatide?
Indirect sleep disruption tends to worsen with dose escalation because the mechanisms that drive it intensify. GI side effects peak during the first 2 weeks at each new dose level. Caloric restriction deepens as appetite suppression increases. Weight loss accelerates, triggering greater cortisol and leptin shifts. SURMOUNT-1 showed fatigue rates climbing from 12% at 5 mg to 20% at 15 mg. Slow titration (staying at each dose for 4-6 weeks before increasing) and proactive meal timing reduce dose-dependent sleep disruption. Use our tirzepatide dosage calculator for a personalized schedule.
Should I take tirzepatide in the morning or at night to avoid sleep problems?
Morning injection is preferred by most users who experience sleep disruption, though the evidence is indirect. Tirzepatide's 5-day half-life means the drug remains active regardless of injection timing. However, GI side effects tend to peak in the 24-48 hours following injection. A Monday morning injection means the worst GI effects occur during the daytime Monday and Tuesday, leaving nights relatively undisturbed. See best time to take tirzepatide for a detailed timing analysis.
The Bottom Line
Tirzepatide does not cause insomnia through any direct pharmacological mechanism. The combined SURMOUNT and SURPASS clinical programs, enrolling over 20,000 participants, did not identify insomnia as a significant adverse event. The SURMOUNT-OSA trials showed tirzepatide actively improves sleep quality in people with obstructive sleep apnea.
The disconnect between clinical trial data and real-world sleep complaints comes down to five indirect pathways: gastrointestinal disruption at night, blood sugar fluctuations during overnight fasting, cortisol and ghrelin changes from caloric deficit, physical discomfort from rapid body composition changes, and psychological adjustment to a new medication. Each pathway has a specific, actionable fix.
Start with behavioral changes: shift your largest meal to midday, stop eating 3-4 hours before bed, and lock in sleep hygiene fundamentals. Add magnesium glycinate (200-400 mg) and a small protein snack before bed if sleep does not improve within 2 weeks. Consult your physician if insomnia persists beyond 6 weeks at a stable dose or is accompanied by signs of nocturnal hypoglycemia.
For most users, sleep disruption is a temporary side effect of the transition, not a permanent feature of tirzepatide treatment. The same mechanisms that cause short-term sleep disturbance (weight loss, improved metabolic health) produce long-term sleep improvement.
Use our tirzepatide dosage calculator to plan a titration schedule that minimizes side effects. For a broader understanding of tirzepatide, explore our tirzepatide profile page and tirzepatide long-term side effects guide.
Related Articles
Can Tirzepatide Cause Joint Pain? Clinical Data
Can tirzepatide cause joint pain? SURMOUNT arthralgia rates match placebo. Review 5 indirect causes, uric acid risks, and 8 relief tactics.
Can Tirzepatide Cause Anxiety?
Anxiety is reported in 3-6% of tirzepatide users. Learn SURMOUNT/SURPASS data, GLP-1 brain mechanisms, and 7 management strategies.
Tirzepatide Injection Site Reactions
Injection site reactions affect 3-7% of tirzepatide users. SURMOUNT trial rates, 5 reaction types, severity guide, and 9 prevention strategies.
Does Tirzepatide Make You Tired?
Fatigue affects 12-20% of tirzepatide users. Learn why it happens, SURMOUNT trial data, the GIP/GLP-1 energy mechanism, and 8 fixes that work.