You rolled up your sleeve, cleaned the skin with alcohol, and injected your weekly tirzepatide dose into your abdomen. Two hours later, a red welt the size of a quarter appeared at the injection site. It itched. By the next morning, the redness had spread to the diameter of a tennis ball. This is a classic injection site reaction, and it happens to 3-7% of tirzepatide users. The good news: most reactions are mild, resolve within 3-7 days, and rarely require treatment discontinuation.
The SURMOUNT-1 trial reported injection site reactions in 3.2% of participants on 5 mg, 4.5% on 10 mg, and 5.6% on 15 mg, compared to 1.4% on placebo (Jastreboff et al., NEJM 2022). In the SURPASS program for type 2 diabetes, rates ranged from 2.6% to 7.0% across doses, with a clear dose-response relationship (Rosenstock et al., 2021). Fewer than 0.3% of trial participants stopped treatment because of injection site reactions.
| Quick Reference | Details |
|---|---|
| Prevalence | 3.2% (5 mg), 4.5% (10 mg), 5.6% (15 mg) in SURMOUNT-1 |
| Reaction types | Redness, swelling, itching, nodules, bruising |
| Onset | 30 minutes to 24 hours after injection |
| Duration | 2-7 days for most reactions |
| Peak occurrence | First 8-12 weeks; decreases with continued use |
| Key prevention | Rotate sites, warm medication, proper technique |
| Evidence level | Phase 3 clinical trials (SURMOUNT-1 through SURMOUNT-4, SURPASS-1 through SURPASS-5) |
For dosing protocols, see our tirzepatide dosage chart. For safe injection practices, see our peptide safety guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Causes Injection Site Reactions to Tirzepatide?
Think of your skin as a gatekeeper. Every time you push a needle through the epidermis and deposit a foreign substance in the subcutaneous tissue, the immune system evaluates the intruder. Tirzepatide is a 39-amino-acid peptide with a C20 fatty acid side chain attached to a lysine residue. That fatty acid moiety, which extends the drug's half-life to approximately 5 days, also increases local tissue irritation compared to shorter-acting peptides.
Three distinct mechanisms drive injection site reactions to tirzepatide. Understanding them helps distinguish normal immune responses from reactions that warrant clinical attention.
1. Local immune activation and mast cell degranulation
When tirzepatide is deposited subcutaneously, local immune cells recognize the peptide as foreign. Mast cells in the dermis and subcutaneous tissue degranulate, releasing histamine, prostaglandins, and cytokines. This cascade produces the hallmark triad of redness, swelling, and itching within 30 minutes to 6 hours of injection. A study examining local immune responses to subcutaneous peptide injections found that mast cell density in the injection area correlates with reaction severity (Laurent et al., 2017). Histamine release explains why antihistamines like cetirizine effectively reduce symptoms in most cases.
2. Tissue trauma from needle insertion and fluid volume
The injection itself damages a small column of tissue. At higher tirzepatide doses, the injected fluid volume increases, creating more local tissue displacement and pressure. A 15 mg dose in a prefilled pen delivers a larger bolus than 2.5 mg, and the physical distension contributes to pain, tenderness, and bruising at the site. Needle gauge, insertion depth, and injection speed all modify the degree of tissue trauma (Frid et al., 2016).
3. Excipient and formulation factors
Tirzepatide's formulation contains sodium phosphate buffer, sodium chloride, and other excipients that maintain drug stability. The pH of the solution (approximately 4.5) is mildly acidic compared to the body's physiological pH of 7.4. This pH difference triggers a transient burning sensation during injection and contributes to local inflammation in the minutes following administration. Compounded tirzepatide formulations may use different buffers or concentrations, potentially altering the local tissue response. We cover compounded versus brand differences in detail below.
Types of Injection Site Reactions
Not every red mark at an injection site is the same. Identifying the specific type helps determine whether the reaction is expected, manageable at home, or worth discussing with a prescriber.
Redness (Erythema)
The most common injection site reaction. A flat or slightly raised red patch appears 1-6 hours after injection, typically 1-3 centimeters in diameter. The redness is warm to the touch but not hot. It blanches when pressed, meaning the red color temporarily disappears under finger pressure and returns when released. Blanching redness indicates dilated capillaries rather than deeper tissue damage.
In SURMOUNT-1, erythema accounted for approximately 60% of all reported injection site reactions (Jastreboff et al., 2022). Most erythematous reactions resolved within 48-72 hours without treatment. Applying a cool compress for 10-15 minutes after injection reduces the severity by constricting local blood vessels and limiting histamine-driven capillary dilation.
If the redness exceeds 10 centimeters in diameter, feels hot rather than warm, or spreads progressively over 24-48 hours, this is not a typical injection site reaction. Expanding redness with warmth and tenderness suggests cellulitis, a bacterial skin infection that requires antibiotic treatment.
Swelling and Induration
Swelling at the injection site ranges from a barely perceptible puffiness to a firm, raised nodule you can feel under the skin. Induration, a hardened area beneath the surface, occurs when the injected fluid creates a pocket that the body walls off with inflammatory tissue.
Mild swelling (less than 2 centimeters) is a normal inflammatory response and resolves within 2-5 days. Induration that persists beyond 7-10 days may indicate that the injection was deposited too superficially (intradermally rather than subcutaneously) or that the injection speed was too fast, causing tissue tearing. Users who reconstitute tirzepatide for research purposes should ensure proper needle length (typically 27-31 gauge, 0.5 inch for subcutaneous injection) and inject slowly over 5-10 seconds to minimize tissue disruption.
A warm compress applied 10 minutes before injection softens the subcutaneous tissue and improves fluid absorption, reducing post-injection swelling.
Itching (Pruritus)
Itching at the injection site reflects histamine release from mast cells in the dermis. It typically begins 1-4 hours after injection and persists for 12-48 hours. The itch can range from mild (noticeable but ignorable) to intense (disrupting sleep).
About 30% of patients who report injection site reactions specifically mention pruritus as the most bothersome symptom (Frias et al., 2021). The urge to scratch is strong, but scratching worsens the reaction by triggering additional mast cell degranulation in the surrounding skin, a process called dermatographism.
A single dose of oral cetirizine (10 mg) or loratadine (10 mg) taken 30-60 minutes before injection significantly reduces itch severity. Topical hydrocortisone 1% cream applied after injection provides localized anti-inflammatory relief without systemic effects. Do not apply hydrocortisone before injection, as it should not be pushed into the subcutaneous tissue.
Nodules and Lumps
Small, firm lumps under the skin at injection sites develop in approximately 1-2% of tirzepatide users. These nodules form when the injected fluid does not disperse evenly through the subcutaneous tissue. The body encapsulates the concentrated deposit with a thin layer of inflammatory tissue, creating a palpable lump 0.5-2 centimeters in diameter.
Nodules are more common when the same injection site is used repeatedly (a practice called "site abuse"), when the injection is given too quickly, or when cold medication is injected. Cold tirzepatide is more viscous, disperses poorly, and pools in a concentrated area. Allowing the medication to reach room temperature before injection (20-30 minutes outside the refrigerator) reduces nodule formation significantly.
Most nodules resolve within 1-3 weeks without intervention. Gentle massage of the area after injection helps distribute the medication through the subcutaneous tissue. If a nodule persists beyond 4 weeks, becomes painful, or grows in size, report it to your prescriber. For tirzepatide storage guidance, proper temperature management helps prevent this issue.
Bruising (Ecchymosis)
Bruising occurs when the needle nicks a small blood vessel during insertion. A blue, purple, or greenish discoloration appears at or near the injection site within hours and fades over 5-14 days as the body reabsorbs the extravasated blood.
Bruising is a mechanical event rather than an immunological reaction. It correlates with needle gauge (larger needles cause more vessel damage), insertion angle (inserting at less than 45 degrees increases the path through vascularized dermis), and individual factors like anticoagulant use, aspirin therapy, or genetic coagulation variants.
To minimize bruising: use the smallest effective needle gauge (29-31 gauge for subcutaneous injection), insert at a 90-degree angle into a pinched skin fold, and apply gentle pressure with a clean gauze for 30-60 seconds after needle withdrawal. Do not rub the site after injection, as rubbing increases tissue trauma and disperses blood from damaged capillaries into a larger bruise. Users who also take blood thinners or daily aspirin should inform their prescriber, as these medications increase bruising frequency and duration.
SURMOUNT and SURPASS Trial Data: How Common Are Injection Site Reactions?

Injection site reactions are among the most consistently reported adverse events across the tirzepatide clinical program. While less common than gastrointestinal effects like nausea or diarrhea, they affect a meaningful minority of users.
SURMOUNT-1: Obesity Without Diabetes
SURMOUNT-1 enrolled 2,539 adults with obesity or overweight and at least one weight-related condition. Participants received tirzepatide 5 mg, 10 mg, or 15 mg versus placebo for 72 weeks (Jastreboff et al., 2022).
| SURMOUNT-1 | 5 mg (n=630) | 10 mg (n=636) | 15 mg (n=630) | Placebo (n=643) |
|---|---|---|---|---|
| Injection site reaction | 3.2% | 4.5% | 5.6% | 1.4% |
| Nausea | 24.6% | 33.3% | 31.0% | 9.5% |
| Constipation | 6.2% | 9.2% | 11.7% | 3.9% |
| Headache | 8.7% | 10.2% | 13.8% | 6.2% |
| Discontinuation for ISR | <0.3% | <0.3% | <0.3% | 0% |
The dose-response pattern matches expectations: higher doses deliver a larger volume of peptide solution, increasing both immune stimulation and tissue displacement. The 15 mg group experienced injection site reactions at four times the placebo rate, yet discontinuation for this reason was extremely rare.
SURPASS Trials: Type 2 Diabetes
The SURPASS program evaluated tirzepatide across multiple diabetes populations and comparators.
SURPASS-1 (monotherapy) reported injection site reactions in 2.6% (5 mg), 4.8% (10 mg), and 7.0% (15 mg) versus 0.5% on placebo (Rosenstock et al., 2021). The higher rates compared to SURMOUNT-1 may reflect differences in adipose tissue distribution, injection technique education, or reporting standards across trial sites.
| SURPASS-1 (Diabetes) | 5 mg | 10 mg | 15 mg | Placebo |
|---|---|---|---|---|
| Injection site reaction | 2.6% | 4.8% | 7.0% | 0.5% |
| HbA1c reduction | -1.87% | -1.89% | -2.07% | +0.04% |
SURPASS-2, which compared tirzepatide head-to-head against semaglutide, reported injection site reactions in 3.4-5.1% of tirzepatide groups versus 0.4% with semaglutide 1 mg (Frias et al., 2021). This is one of the few adverse event categories where tirzepatide rates clearly exceeded semaglutide rates, likely due to differences in formulation pH, excipient profile, and the C20 fatty acid moiety unique to tirzepatide.
SURMOUNT-2, studying tirzepatide for weight management in adults with type 2 diabetes and obesity, confirmed the injection site reaction signal at 4.7% (10 mg) and 5.9% (15 mg) versus 1.7% on placebo over 72 weeks (Garvey et al., 2023). The consistency across four major trials strengthens confidence that the 3-7% range is a reliable estimate of injection site reaction prevalence.
Key takeaway: Expect injection site reactions in roughly 1 out of 15 to 1 out of 20 users. The reactions are predominantly mild, self-limiting, and rarely lead to treatment discontinuation. Higher doses carry slightly higher risk due to increased injection volume and peptide concentration at the depot site.
How Long Do Injection Site Reactions Last?
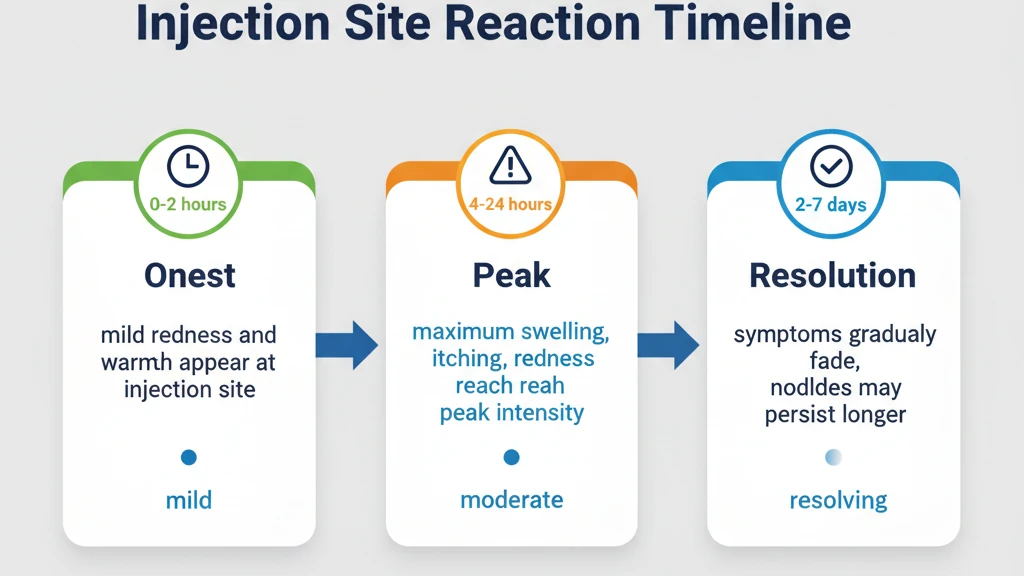
Injection site reactions to tirzepatide follow a predictable timeline. Knowing what to expect at each phase helps distinguish a normal reaction from one that needs medical evaluation.
Minutes 0-30: Immediate Response
A mild stinging or burning sensation during and immediately after injection is caused by the slightly acidic pH of the tirzepatide solution contacting subcutaneous tissue. This sensation fades within 5-15 minutes in most people. A small wheal (raised white area) may appear at the needle entry point. This is a normal tissue response to fluid injection and disappears within 30 minutes.
If itching, hives, or swelling begins spreading beyond the injection site within the first 30 minutes, this is not a local reaction. Rapid-onset widespread symptoms may indicate a systemic allergic response. We cover this distinction in the allergy section below.
Hours 1-24: Peak Reaction
The local inflammatory response peaks between 2 and 12 hours after injection. Redness, swelling, and itching reach maximum intensity during this window. The affected area may feel warm and tender. The reaction typically covers a 2-5 centimeter diameter, though larger reactions up to 10 centimeters are still within the expected range.
Users who experience their worst symptoms in this phase benefit from pretreatment with an oral antihistamine 30-60 minutes before injection. Applying ice wrapped in a cloth for 10-15 minutes after injection constricts blood vessels and limits the inflammatory cascade. Avoid applying heat during the first 24 hours, as warmth dilates blood vessels and amplifies redness and swelling.
Days 2-5: Resolution
Most injection site reactions begin fading on day 2 and resolve completely by day 5. The redness transitions from bright red to pink to the surrounding skin tone. Itching subsides before redness in most cases. Bruising, if present, may persist longer (7-14 days) but is a cosmetic rather than symptomatic issue.
A firm, non-tender nodule may remain at the injection site for 1-3 weeks after the visible redness resolves. This represents a small depot of tirzepatide being slowly absorbed from the subcutaneous tissue. The depot effect is actually desirable from a pharmacokinetic standpoint. Tirzepatide's long half-life of approximately 5 days depends partly on this slow release from the injection site depot, which is why it only needs to be administered once weekly.
Beyond Day 7: When to Seek Evaluation
A reaction that worsens after day 3, persists beyond day 7, or is accompanied by fever, pus, or expanding redness with a sharp border is not a typical injection site reaction. These features suggest secondary infection (cellulitis) at the injection site, which requires prompt antibiotic treatment.
Granulomas, firm nodules that persist beyond 4 weeks, may develop in rare cases (fewer than 0.1% of users). These represent a foreign body reaction where the immune system walls off the injected material with a dense inflammatory capsule. Granulomas are not dangerous but may require corticosteroid injection or surgical excision if they cause discomfort or cosmetic concern.
Injection Site Reaction vs Allergic Reaction: How to Tell the Difference
This distinction is clinically critical. A local injection site reaction is a nuisance. A systemic allergic reaction is a medical emergency. Knowing the difference could save your life.
Local injection site reaction (common, not dangerous)
A localized area of redness, swelling, and itching confined to within 10 centimeters of the injection point. The reaction develops gradually over 1-12 hours. There are no systemic symptoms. Vital signs are normal. The reaction improves with time and does not spread beyond the local area.
Systemic allergic reaction (rare, potentially dangerous)
Any of the following symptoms occurring within minutes to hours of injection indicate a potential allergic response: hives or raised itchy welts appearing on skin distant from the injection site, swelling of the lips, tongue, or throat, difficulty breathing or wheezing, rapid heartbeat, dizziness or lightheadedness, nausea and vomiting unrelated to the typical GI effects of tirzepatide.
The SURMOUNT and SURPASS trials reported serious hypersensitivity events in fewer than 0.1% of participants. Anaphylaxis was exceedingly rare, with isolated case reports documented in post-marketing surveillance (Heise et al., 2023). The FDA prescribing information for Mounjaro and Zepbound lists anaphylaxis and angioedema as potential risks, consistent with any injectable biologic.
| Feature | Local Reaction | Allergic Reaction |
|---|---|---|
| Location | At injection site only | Spreads beyond injection site |
| Onset | 1-12 hours | Minutes to 1 hour |
| Hives/welts elsewhere | No | Yes |
| Breathing difficulty | No | Possible |
| Throat swelling | No | Possible |
| Duration | 2-7 days | Escalates without treatment |
| Action | Home management | Call 911 immediately |
What to do if you suspect an allergic reaction:
Stop all tirzepatide injections. If symptoms include throat swelling, difficulty breathing, or dizziness, call 911 immediately. Use an epinephrine auto-injector (EpiPen) if available. Even if symptoms seem mild, seek same-day medical evaluation because allergic reactions can progress rapidly from mild to severe. Do not re-administer tirzepatide without allergist evaluation and clearance.
Users who experience a confirmed allergic reaction to tirzepatide should discuss alternative GLP-1 receptor agonists with their prescriber. Cross-reactivity between tirzepatide and semaglutide is uncommon because the peptide sequences differ significantly. A referral to an allergist for skin testing can determine whether the reaction was to the active peptide or to an excipient in the formulation.
Compounded vs Brand Tirzepatide: Injection Site Reaction Differences
Compounded tirzepatide has grown in popularity as an alternative to branded Mounjaro and Zepbound, particularly during supply shortages. However, compounded formulations differ from FDA-approved products in ways that directly affect injection site reaction risk.
pH and buffer differences
FDA-approved tirzepatide (Mounjaro/Zepbound) uses a carefully optimized phosphate buffer system. Compounded versions may use different buffer systems depending on the compounding pharmacy. Sodium acetate buffers at lower pH values (3.5-4.5) produce more injection site stinging and erythema than phosphate-buffered formulations closer to physiological pH. Users who switch from branded to compounded tirzepatide and notice increased injection site reactions should ask their compounding pharmacy about the formulation's pH.
Concentration and injection volume
Compounded tirzepatide arrives as a lyophilized powder or concentrated solution that requires reconstitution. The final concentration depends on the reconstitution volume. A higher concentration means a smaller injection volume but a more concentrated depot at the injection site, which can increase local irritation. Conversely, a lower concentration means a larger injection volume that causes more tissue displacement. Finding the balance matters. A concentration of 5-10 mg/mL, injecting 0.25-1.0 mL per dose, typically minimizes both concentration-dependent irritation and volume-dependent tissue trauma.
Sterility and endotoxin considerations
503B outsourcing facilities operate under FDA oversight and must meet cGMP standards, including endotoxin testing. However, quality varies between compounding pharmacies. Endotoxins, bacterial cell wall fragments that can contaminate pharmaceutical products, trigger intense local inflammatory reactions even at levels below the detection threshold of standard sterility testing (Venter et al., 2021). If injection site reactions are consistently more severe with compounded tirzepatide than with branded product, endotoxin contamination is one possible explanation.
Reconstitution solvent
Bacteriostatic water (containing 0.9% benzyl alcohol) is the standard reconstitution solvent for compounded peptides. Benzyl alcohol is a mild tissue irritant and can contribute to injection site pain and redness, particularly in users sensitive to preservatives. Sterile water for injection (without benzyl alcohol) causes less local irritation but does not inhibit microbial growth in multi-use vials. For users experiencing significant injection site reactions with bacteriostatic water, single-use reconstitution with sterile water may reduce local symptoms.
Practical recommendation: If you experience more injection site reactions with compounded tirzepatide than you did with branded product (or vice versa), the formulation difference is the most likely explanation. Discuss alternative reconstitution protocols or pharmacy sources with your prescriber rather than assuming the reactions are an inherent property of tirzepatide itself.
9 Strategies to Prevent and Minimize Injection Site Reactions
These nine strategies address the three root causes: immune activation, tissue trauma, and formulation factors. Most users eliminate or significantly reduce reactions by implementing the first four.
1. Rotate Injection Sites Systematically
Site rotation is the single most important prevention measure. Using the same location repeatedly causes cumulative tissue damage, increases local immune sensitization, and promotes nodule formation. A study on subcutaneous injection best practices found that systematic rotation reduced injection site reactions by over 40% compared to habitual single-site use (Frid et al., 2016).
Use a four-quadrant rotation system: divide the abdomen into four zones (upper left, upper right, lower left, lower right) and rotate clockwise each week. Each injection should be at least 2 inches (5 cm) from the previous week's site and at least 2 inches from the navel. Alternative sites include the front of the thighs and the back of the upper arms. The abdomen is generally preferred because subcutaneous tissue is thickest and most consistent in this area.
Keep a simple log: write the date and location (e.g., "3/15, lower right abdomen") on your calendar or phone. This prevents accidental site reuse and helps identify if certain locations produce more reactions than others.
2. Warm the Medication Before Injection
Cold tirzepatide straight from the refrigerator (36-46 degrees Fahrenheit) is more viscous, disperses poorly in tissue, and causes more local pain and reactions than room-temperature medication. Warming the syringe or pen to room temperature (68-77 degrees Fahrenheit) before injection reduces viscosity, improves tissue distribution, and decreases reaction severity.
Remove the medication from the refrigerator 20-30 minutes before your planned injection time. Do not microwave, boil, or place the medication in hot water. Gentle warming to room temperature is sufficient. Roll the vial or pen between your palms for 30-60 seconds to promote even warming. Refer to our tirzepatide storage guide for safe temperature handling. Note that tirzepatide stability is not compromised by brief room-temperature exposure.
3. Use Proper Injection Technique
Technique errors account for a substantial proportion of avoidable injection site reactions. The following checklist covers optimal subcutaneous injection technique.
Clean the injection site with an alcohol swab and allow it to dry completely (15-30 seconds). Injecting through wet alcohol pushes the alcohol into the subcutaneous tissue, causing a burning sensation and increased local irritation. Pinch a 1-2 inch fold of skin between your thumb and forefinger. Insert the needle at a 90-degree angle into the skin fold in a single, swift motion. Slow or hesitant insertion increases tissue trauma.
Inject the medication slowly and steadily over 5-10 seconds. Rapid injection (under 2 seconds) creates a high-pressure fluid jet that tears tissue planes and increases pain, bruising, and nodule formation. After the full dose is delivered, wait 5-10 seconds with the needle still in place before withdrawing. This allows the tissue to begin absorbing the medication and prevents leakback through the needle track.
Remove the needle at the same angle it was inserted. Apply gentle pressure with a clean gauze pad for 10-30 seconds. Do not rub or massage the site immediately. Our guide on how to inject BPC-157 covers subcutaneous injection technique in detail, and the same principles apply to tirzepatide.
4. Apply Ice After Injection
A cold compress applied to the injection site for 10-15 minutes immediately after injection constricts local blood vessels, reduces histamine release, and limits the inflammatory cascade. Wrap ice or a gel pack in a thin cloth to prevent cold injury to the skin. Direct ice contact can cause frostbite, especially in the small area of tissue already stressed by injection.
Do not apply ice before injection, as vasoconstriction makes the subcutaneous tissue firmer and more resistant to needle insertion, increasing tissue trauma. Warmth before, cold after is the optimal sequence: warm compress 5-10 minutes pre-injection to soften tissue, then ice for 10-15 minutes post-injection to limit inflammation.
5. Take an Antihistamine Before Injection
For users who consistently develop itching or redness at injection sites, taking a second-generation antihistamine 30-60 minutes before injection is effective and safe. Cetirizine (Zyrtec, 10 mg) or loratadine (Claritin, 10 mg) blocks H1 histamine receptors on mast cells, reducing the itch-redness-swelling cascade.
Second-generation antihistamines are preferred over first-generation options (like diphenhydramine/Benadryl) because they cause less drowsiness and last 24 hours. A single pre-injection dose of cetirizine is sufficient for most users. If reactions persist despite pre-treatment, adding famotidine (Pepcid, 20 mg), an H2 histamine blocker, provides additional coverage through a complementary receptor pathway.
This approach is supported by clinical experience with other injectable biologics, where antihistamine premedication is standard practice for injection site reactions (Rosen et al., 2019).
6. Avoid Injecting Into Scars, Stretch Marks, or Tattoos
Scarred tissue, stretch marks, and tattooed skin have altered vascularity, nerve distribution, and immune cell density compared to normal skin. Injecting into these areas increases pain, slows drug absorption, and may produce more pronounced reactions. Tattoo pigments in the dermis can trigger foreign body reactions when disturbed by needle insertion.
Choose injection sites with smooth, intact skin and a minimum of 1 inch of pinchable subcutaneous tissue. Avoid areas with visible veins, moles, skin lesions, or recent sunburn. The lower abdomen (below the navel, above the pubic bone, lateral to the midline) typically offers the largest area of suitable injection territory.
7. Use the Correct Needle Gauge and Length
For subcutaneous injection, a 27-31 gauge needle with a length of 0.5 inches (12.7 mm) is standard. Finer gauges (30-31) create a smaller puncture, reduce tissue trauma, and decrease bruising frequency. Shorter needles (0.5 inch) are sufficient for subcutaneous delivery in most body compositions, as the subcutaneous layer in the abdomen is typically 1-3 centimeters thick.
Users with very lean body composition (BMI under 20) should consider 0.5-inch needles and shallower angles (45 degrees rather than 90 degrees) to avoid intramuscular injection, which increases pain and alters drug absorption kinetics. Users with higher body mass can use 0.5-inch needles at 90 degrees with confidence. The tirzepatide dosage calculator helps determine appropriate dosing, and matching the injection technique to the dose volume optimizes local tolerability.
8. Keep a Reaction Log and Identify Patterns
Record each injection: date, time, dose, site location, needle gauge, whether the medication was warmed, and any reaction (type, size, duration, severity on a 1-10 scale). After 4-6 weeks, patterns emerge. You may discover that abdominal injections produce fewer reactions than thigh injections, or that reactions are worse during your dose escalation weeks.
This log is invaluable for prescriber discussions. Objective data about reaction frequency, severity, and relationship to specific techniques or sites helps guide decisions about premedication, dose adjustment, or formulation changes. Users who find that reactions occur exclusively at higher doses may benefit from splitting the dose into two smaller injections at different sites, a strategy sometimes used for high-volume subcutaneous injections.
9. Consider Injection Site Numbing for Severe Reactions
For the small percentage of users whose injection site reactions significantly impact quality of life, topical anesthetic application before injection can reduce both the acute pain and the subsequent inflammatory reaction. Apply a lidocaine-prilocaine cream (EMLA cream, available over the counter in many formulations) to the planned injection site under an occlusive bandage 30-60 minutes before injection. The cream numbs the skin and superficial subcutaneous tissue, reducing pain perception and the neurogenic inflammatory response.
This is not necessary for most users and should be reserved for cases where standard prevention strategies (rotation, warming, antihistamine, ice) have failed to provide adequate relief. Discuss topical anesthetic use with your prescriber before starting.
Injection Site Reactions During Dose Escalation
Injection site reactions are not constant throughout treatment. They follow a pattern tied to the tirzepatide titration schedule and the body's developing tolerance to the injected peptide.
Starting dose (2.5 mg, weeks 1-4)
Injection site reactions at the 2.5 mg starting dose are uncommon (fewer than 2% of users). The low dose means a small injection volume and a modest immune stimulus. Users who do experience reactions at 2.5 mg are more likely to develop them at higher doses and should implement preventive strategies early. For context on how long tirzepatide takes to work, effects begin building during this initiation phase.
First escalation (5 mg, weeks 5-8)
The doubling from 2.5 to 5 mg is the point where many users first notice injection site reactions. The increased peptide concentration and volume triggers a stronger local immune response. This is also when most users begin noticing other side effects like fatigue and headaches.
Mid-range doses (7.5-10 mg, weeks 9-16)
Injection site reaction rates plateau in the mid-range doses. The body develops partial local immune tolerance through repeated exposure, a process called desensitization. Mast cells exposed to the same antigen repeatedly release progressively less histamine with each encounter. Users who had reactions at 5 mg often report that reactions decrease in severity at 7.5 mg and 10 mg despite the higher doses.
High doses (12.5-15 mg, weeks 17-24+)
At the highest doses, the increased injection volume partially offsets the desensitization effect. SURMOUNT-1 data shows that the highest injection site reaction rate (5.6%) occurred at 15 mg (Jastreboff et al., 2022). Users who reach 15 mg and experience new or worsening reactions should reassess injection technique, ensure adequate site rotation, and consider premedication with antihistamines.
Maintenance phase
After reaching the target dose and entering maintenance, injection site reactions typically stabilize and often improve further. The immune system has been exposed to weekly tirzepatide for months, and local tolerance is well established. Users who are not losing weight on tirzepatide at their current dose should discuss the situation with their prescriber before assuming that injection site reactions warrant a dose change.
Impact of Lifestyle Factors on Injection Site Reactions
Several modifiable factors outside of injection technique influence injection site reaction severity. Addressing these can meaningfully reduce reactions even when technique is already optimized.
Alcohol consumption
Alcohol consumption around injection time affects injection site reactions through two mechanisms. First, alcohol is a vasodilator. Drinking within 2-4 hours of injection increases blood flow to the skin and subcutaneous tissue, amplifying redness and swelling. Second, alcohol impairs immune regulation, potentially prolonging the duration of the local inflammatory response. Avoid alcohol for 4-6 hours before and after tirzepatide injection.
Hydration status
Dehydration concentrates circulating inflammatory mediators and reduces tissue turgor, making the subcutaneous tissue less compliant and more prone to mechanical trauma during injection. Well-hydrated tissue absorbs injected fluid more efficiently, reducing the depot concentration and the resulting local reaction. Ensure adequate hydration on injection day, aiming for at least 64-80 ounces of water. Tirzepatide stays in your system for weeks, but injection day hydration specifically affects the local tissue response.
Exercise timing
Vigorous exercise within 2 hours of injection increases blood flow to skeletal muscle and skin, which can amplify injection site redness and speed drug absorption (altering pharmacokinetics). Light activity is fine, but avoid intense cardio or resistance training targeting the injection area (e.g., abdominal exercises after an abdominal injection) for at least 2 hours post-injection.
Skin temperature
Injecting into cold skin (e.g., after coming inside from winter temperatures) reduces subcutaneous blood flow and slows drug dispersion, increasing local concentration and reaction severity. Warm the injection site with your hand or a warm cloth for 1-2 minutes before cleaning with alcohol if your skin is cold.
Fat-burning considerations
Users interested in how tirzepatide burns fat should understand that as subcutaneous fat decreases with weight loss, the injection site tissue composition changes. Areas that previously had ample subcutaneous fat may become thinner, requiring adjusted needle depth or new site selection to maintain subcutaneous delivery and avoid intramuscular injection.
Important Warnings
Any injection site reaction accompanied by fever above 100.4 degrees Fahrenheit, pus or purulent drainage, rapidly expanding redness with sharp borders, or red streaking extending away from the injection site requires same-day medical evaluation for cellulitis (bacterial skin infection). Do not treat suspected cellulitis at home.
Widespread hives, throat or tongue swelling, difficulty breathing, or lightheadedness occurring within minutes to hours of tirzepatide injection is a medical emergency suggesting anaphylaxis. Call 911 immediately. Do not drive yourself to the hospital. Do not administer another tirzepatide injection until cleared by an allergist.
Users who take anticoagulants (warfarin, apixaban, rivarelbaan) or daily aspirin should discuss injection technique modifications with their prescriber. These medications increase bruising risk and may require longer post-injection pressure application.
Do not reuse needles. Single-use needles become dulled after one insertion, and reuse increases tissue trauma, infection risk, and injection site reaction severity. Dispose of used needles in an FDA-cleared sharps container.
Compounded tirzepatide users should source from 503B outsourcing facilities that follow cGMP standards. Request certificates of analysis showing sterility testing, endotoxin levels, and potency verification. Contaminated products cause severe injection site reactions that may mimic cellulitis or allergic reactions.
For users switching from semaglutide to tirzepatide or vice versa, review our semaglutide to tirzepatide switching guide for information on how injection site reaction profiles differ between the two medications.
Frequently Asked Questions
How long do tirzepatide injection site reactions last?
Most injection site reactions resolve within 2-7 days. Redness and itching typically peak 2-12 hours after injection and fade over 48-72 hours. Bruising may persist 7-14 days. Nodules can last 1-3 weeks. If any reaction worsens after day 3 or persists beyond 10 days, contact your prescriber to rule out infection.
Are injection site reactions more common with compounded tirzepatide?
Compounded tirzepatide may cause more injection site reactions than branded Mounjaro or Zepbound due to differences in pH, buffer systems, reconstitution solvents, and quality control standards. Benzyl alcohol in bacteriostatic water is a known tissue irritant. If reactions increase after switching to a compounded product, discuss alternative pharmacies or reconstitution protocols with your prescriber.
Can I take Benadryl before my tirzepatide injection?
Diphenhydramine (Benadryl) works but causes drowsiness. Second-generation antihistamines like cetirizine (Zyrtec, 10 mg) or loratadine (Claritin, 10 mg) are preferred because they provide 24-hour histamine blockade without sedation. Take one dose 30-60 minutes before injection. Add topical hydrocortisone 1% cream after injection if itching persists.
Should I stop tirzepatide if I get injection site reactions?
No. Injection site reactions alone are not a reason to discontinue tirzepatide. Fewer than 0.3% of clinical trial participants stopped treatment because of injection site reactions. Implement rotation, warming, proper technique, and antihistamine pretreatment. Only stop if you experience signs of systemic allergy: widespread hives, throat swelling, or breathing difficulty.
Is a lump at the tirzepatide injection site normal?
Small, painless lumps (1-2 centimeters) that form at the injection site are common and represent a normal depot of tirzepatide being slowly absorbed. They typically resolve within 1-3 weeks. Inject slowly, warm the medication, and rotate sites to minimize nodule formation. Report lumps that persist beyond 4 weeks, grow in size, or become painful.
Where is the best place to inject tirzepatide to avoid reactions?
The lower abdomen, at least 2 inches from the navel, offers the thickest subcutaneous tissue and the most consistent absorption. Rotate across four abdominal quadrants weekly. Front thighs and upper arms are alternatives. Avoid areas with scars, stretch marks, tattoos, or visible veins. Each injection should be at least 2 inches from the previous site.
Does the injection site reaction mean I am allergic to tirzepatide?
No. Local injection site reactions are immune responses confined to the injection area and occur in 3-7% of users. True allergy causes systemic symptoms: hives distant from the injection site, throat swelling, breathing difficulty, or dizziness. These occur in fewer than 0.1% of users. A local reaction that stays within 10 centimeters of the injection point is not an allergy.
Do injection site reactions get better over time on tirzepatide?
Yes, for most users. The immune system develops local tolerance through repeated exposure, a process called desensitization. Mast cells at regularly used injection sites release progressively less histamine over weeks to months. Many users who experienced reactions during the first 8-12 weeks report significant improvement or complete resolution by month 4-6 of treatment.
The Bottom Line
Injection site reactions affect 3-7% of tirzepatide users, making them one of the more common but least problematic adverse events in the clinical program. Three mechanisms drive these reactions: local immune activation with mast cell degranulation, tissue trauma from the injection itself, and formulation-specific factors including pH, excipients, and the drug's C20 fatty acid moiety.
Five distinct reaction types occur at varying frequencies: redness (most common), swelling and induration, itching, nodules, and bruising. Most reactions peak within 2-12 hours of injection and resolve within 2-7 days. The dose-response relationship is consistent across trials, with 15 mg doses producing approximately four times the placebo rate.
Nine prevention strategies target these mechanisms. Site rotation, medication warming, proper technique, and post-injection ice address the majority of reactions. Antihistamine premedication adds a pharmacological layer for persistent cases. Compounded tirzepatide may differ from branded product in local tolerability due to pH, buffer, and solvent differences.
The critical distinction is between local injection site reactions and systemic allergic reactions. Local reactions are confined to the injection area, self-limiting, and manageable at home. Systemic reactions, including widespread hives, throat swelling, and breathing difficulty, require immediate emergency care. Fewer than 0.1% of trial participants experienced systemic hypersensitivity.
Use our tirzepatide dosage calculator to plan your titration schedule. For dosing details, see our tirzepatide dosage chart in units. For reconstitution guidance, see how to reconstitute tirzepatide.
Related tirzepatide articles: - How Long Does Tirzepatide Take to Work? — week-by-week timeline of effects and side effects - Does Tirzepatide Cause Headaches? — another common side effect with overlapping prevention strategies - Does Tirzepatide Cause Hair Loss? — side effect management during rapid weight loss - Tirzepatide Maintenance Dose After Weight Loss — reducing side effects at maintenance doses - Semaglutide to Tirzepatide Switching Guide — comparing injection site profiles between medications - Can You Drink Alcohol on Tirzepatide? — alcohol's effect on injection site reactions and other side effects - How to Reconstitute Tirzepatide — proper preparation for research peptide users
Explore all peptide profiles and tools at PeptidesExplorer.
Related Articles
Does Tirzepatide Cause Insomnia?
Does tirzepatide cause insomnia? Clinical trials say no, but 5 indirect pathways explain why some users lose sleep. GI disruption, blood sugar, caloric deficit fixes inside.
Can Tirzepatide Cause Joint Pain? Clinical Data and Relief Strategies
Tirzepatide does not directly cause joint pain. SURMOUNT trial arthralgia rates match placebo. Learn 5 indirect causes, uric acid risks, and 8 relief strategies backed by clinical evidence.
Can Tirzepatide Cause Anxiety?
Anxiety is reported in 3-6% of tirzepatide users. Learn SURMOUNT/SURPASS data, GLP-1 brain mechanisms, and 7 management strategies.
Does Tirzepatide Make You Tired?
Fatigue affects 12-20% of tirzepatide users. Learn why it happens, SURMOUNT trial data, the GIP/GLP-1 energy mechanism, and 8 fixes that work.