You found a vial of tirzepatide in the back of your refrigerator. The label says it was compounded four months ago, but you never opened it. Is it still safe to use? Yes, tirzepatide expires, and the timeline depends on its form. Lyophilized (powder) tirzepatide remains stable for 24 to 36 months when stored at -20°C to 2-8°C. Reconstituted tirzepatide lasts approximately 28 days refrigerated at 2-8°C. Pre-filled pens like Mounjaro and Zepbound carry a manufacturer-printed expiration date, typically 18 to 24 months from production.
Using tirzepatide past its expiration is unlikely to cause harm, but potency drops measurably. A vial that has lost 15-20% of its active peptide delivers a lower effective dose, which can stall weight loss or glucose control without an obvious explanation.
| Tirzepatide Form | Storage Temperature | Shelf Life | Key Condition |
|---|---|---|---|
| Lyophilized powder | -20°C (freezer) | 24-36 months | Sealed, dry, away from light |
| Lyophilized powder | 2-8°C (fridge) | 18-24 months | Sealed, dry, away from light |
| Reconstituted (bac water) | 2-8°C (fridge) | 28 days | Bacteriostatic water, sterile technique |
| Reconstituted (sterile water) | 2-8°C (fridge) | 24-48 hours | No preservative, use immediately |
| Mounjaro/Zepbound pen | 2-8°C (fridge) | Until printed date (18-24 months) | Unopened, in original carton |
| Mounjaro/Zepbound pen | Room temp (up to 30°C) | 21 days | After first use or removal from fridge |
For proper reconstitution technique, use the peptide reconstitution calculator. For dosing protocols, see the tirzepatide dosage chart in units.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Understanding Tirzepatide Expiration by Form
Tirzepatide reaches patients in three distinct forms. Each has its own stability profile because the chemical environment surrounding the peptide differs in each case. The active molecule is the same 39-amino-acid dual agonist peptide, but packaging, solvent, and preservatives change how fast degradation occurs.
Expiration dates are not arbitrary. Pharmaceutical manufacturers conduct accelerated stability testing under controlled heat and humidity, then project long-term stability from those results. The International Council for Harmonisation (ICH) Q1A guidelines require stability data at 25°C/60% relative humidity for 12 months and at 40°C/75% relative humidity for 6 months before assigning a shelf life (ICH Q1A(R2), 2003).
Lyophilized (Powder) Tirzepatide
Lyophilized tirzepatide is freeze-dried into a white or off-white cake or powder. Removing water halts nearly all chemical degradation pathways. In this form, the peptide is remarkably stable.
At -20°C (standard freezer), lyophilized tirzepatide retains greater than 95% potency for 24 to 36 months. At 2-8°C (refrigerator), expect 18 to 24 months. At room temperature (20-25°C), stability drops to 6 to 12 months depending on humidity and light exposure.
The reason powder lasts so long is straightforward: degradation reactions need water. Deamidation, the conversion of asparagine residues to aspartate, requires aqueous conditions. Oxidation of methionine residues accelerates in moisture. Remove the water, and both reactions slow to a crawl. A study on peptide pharmaceutical stability demonstrated that lyophilized formulations retain potency 5 to 10 times longer than their liquid counterparts under identical temperature conditions (Manning et al., Pharm Res, 2010).
Store lyophilized vials in their original packaging or wrapped in foil to block UV light. Do not store them in the refrigerator door where temperature fluctuates with each opening. A dedicated drawer or shelf in the back of the fridge or freezer is ideal. For comprehensive storage instructions, see our peptide storage guide.
Reconstituted Tirzepatide

Once you add bacteriostatic water to lyophilized tirzepatide, the clock starts. The peptide is now dissolved in aqueous solution, and every degradation pathway becomes active.
With bacteriostatic water (containing 0.9% benzyl alcohol as a preservative), reconstituted tirzepatide lasts approximately 28 days at 2-8°C — for a detailed breakdown, see how long does tirzepatide last in the fridge. The benzyl alcohol inhibits bacterial growth, which is why multi-use vials require it. Without that preservative, using plain sterile water, the solution is safe for only 24 to 48 hours before microbial contamination risk becomes unacceptable.
Each needle puncture through the rubber stopper introduces a potential contamination pathway. After 15 to 20 punctures, the stopper may no longer seal properly, allowing air and bacteria to enter even with bacteriostatic water. Keep puncture counts low by drawing accurate doses. Use our tirzepatide dosage calculator to determine exact volumes before drawing.
For a detailed breakdown of reconstituted peptide timelines, see how long do reconstituted peptides last.
Pre-Filled Pens: Mounjaro and Zepbound
Manufacturer pre-filled pens (Mounjaro for type 2 diabetes, Zepbound for weight management) contain tirzepatide in a liquid formulation engineered for stability. Eli Lilly's proprietary buffer system and excipients extend shelf life beyond what a compounding pharmacy typically achieves.
Unopened pens stored at 2-8°C remain potent until the printed expiration date, usually 18 to 24 months from manufacture. Once removed from refrigeration or after first use, pens are stable for 21 days at room temperature (up to 30°C / 86°F). After 21 days at room temperature, discard the pen regardless of remaining medication.
Never freeze a pre-filled pen. The freeze-thaw cycle can damage the protein structure and alter the delivery mechanism. If a pen has been frozen accidentally, do not use it.
The 21-day room temperature window is clinically validated through stability testing. Eli Lilly's prescribing information specifies that pens should not be stored above 30°C and should be protected from direct sunlight.
How Tirzepatide Degrades Over Time
Peptide degradation is not an on-off switch. Tirzepatide does not go from fully potent to worthless on its expiration date. Potency declines gradually through specific chemical pathways, and the rate depends on temperature, light, moisture, and pH.
Understanding these pathways helps you recognize when a vial is still usable and when it has crossed the line.
Deamidation
Asparagine residues in the tirzepatide peptide chain convert to aspartate through a process called deamidation. This is the primary degradation pathway for most peptides in aqueous solution.
The reaction rate doubles for every 10°C increase in temperature. At 2-8°C, deamidation proceeds slowly enough that the peptide retains greater than 90% potency for 28 days. At 25°C, the same loss occurs in roughly 7 to 10 days. At 37°C (body temperature), degradation accelerates to the point where 10-15% potency loss can occur within 3 to 5 days.
A comprehensive review of peptide pharmaceutical degradation documented deamidation as the most common chemical instability in therapeutic peptides, affecting virtually every peptide containing asparagine-glycine sequences (Manning et al., Pharm Res, 2010).
Oxidation
Methionine and tryptophan residues in tirzepatide are vulnerable to oxidation. Exposure to dissolved oxygen, light (especially UV), and trace metals catalyzes the reaction.
Oxidized tirzepatide binds less effectively to GLP-1 and GIP receptors. A 2019 study on GLP-1 receptor agonist stability found that oxidation of a single methionine residue reduced receptor binding affinity by 40-60%, translating directly to reduced efficacy (Houen & Jakobsen, J Pharm Sci, 2019).
Protecting vials from light is one of the simplest ways to slow oxidation. Store in the original carton or wrapped in aluminum foil. Amber vials provide some UV protection, but they are not a substitute for dark storage.
Aggregation
When peptide molecules unfold or partially degrade, they can clump together into aggregates. These aggregates appear as cloudiness, visible particles, or a gel-like substance in the solution.
Aggregated tirzepatide is not only less potent but potentially immunogenic. The immune system can recognize peptide aggregates as foreign and mount an antibody response. This is one reason manufacturers set conservative expiration dates: to keep aggregate levels below clinically significant thresholds.
Temperature cycling (repeated warming and cooling) accelerates aggregation. Every time you take a vial out of the fridge, warm it in your hands, draw a dose, and return it, you create a micro-cycle. Minimize handling time. Draw your dose, return the vial, and close the refrigerator door within 60 seconds.
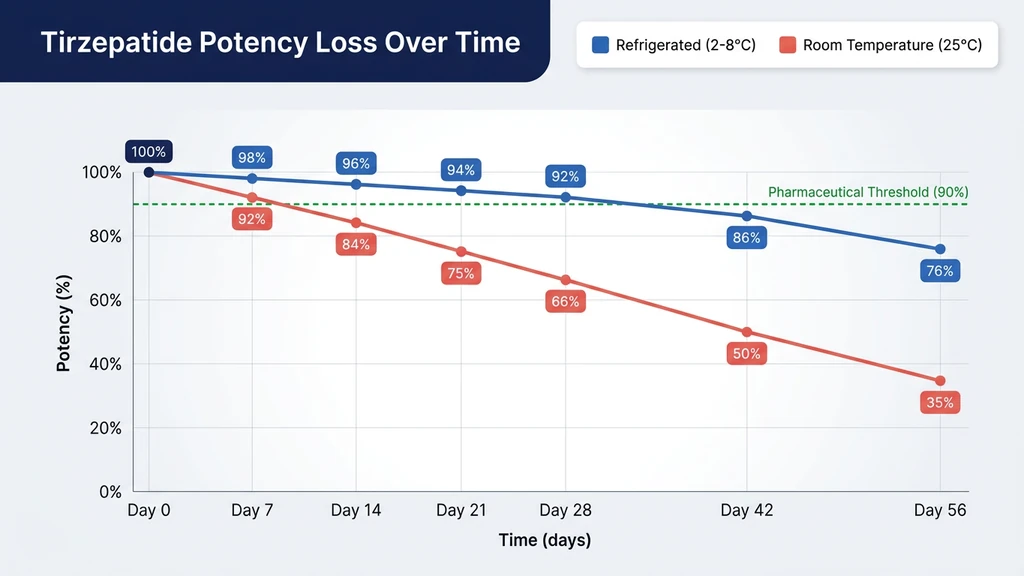
The Degradation Curve
Tirzepatide does not degrade linearly. Potency loss follows an exponential decay curve, with the rate accelerating as degradation products themselves catalyze further breakdown.
| Time After Reconstitution | Estimated Potency at 2-8°C | Estimated Potency at 25°C |
|---|---|---|
| Day 0 | 100% | 100% |
| Day 7 | 97-99% | 90-95% |
| Day 14 | 95-98% | 80-88% |
| Day 21 | 92-96% | 70-80% |
| Day 28 | 90-95% | 60-72% |
| Day 42 | 82-90% | 45-55% |
| Day 56 | 70-82% | 30-40% |
At refrigerator temperature, tirzepatide remains within acceptable pharmaceutical limits (greater than 90% potency) for approximately 28 days. At room temperature, it drops below that threshold by day 14. This is why reconstituted vials carry a 28-day refrigerated shelf life and why pre-filled pens are limited to 21 days outside the fridge.
For context on how other peptides compare, see the shelf life data in how long do reconstituted peptides last.
How to Tell If Tirzepatide Has Gone Bad
You cannot measure potency at home with laboratory precision. But several visible and physical signs indicate that a vial has degraded beyond acceptable use.
Visual Inspection
Cloudiness or turbidity. Fresh tirzepatide solution is clear and colorless. Any haziness, cloudiness, or opalescence indicates protein aggregation. Do not use a cloudy solution.
Visible particles. Hold the vial up to a light source and gently swirl. Floating particles, flakes, or fibers indicate contamination or aggregation. A single small fiber may be a stopper fragment and is less concerning, but multiple particles mean the vial should be discarded.
Color change. Tirzepatide solution should be colorless to very slightly yellow. A shift toward amber, brown, or any other color indicates oxidation or chemical breakdown. Discard the vial.
Gel formation. In advanced degradation, peptide aggregates can form a gel-like substance on the vial walls or at the bottom. This represents extensive structural damage. The peptide is no longer viable.
Odor and Physical Clues
Fresh bacteriostatic water has a faint, slightly medicinal smell from the benzyl alcohol. A foul, sour, or unusual odor when the stopper is pierced indicates bacterial contamination. This is more common in vials stored beyond 28 days or punctured many times.
If you notice that your injection stings or burns more than usual at the injection site, the solution pH may have shifted due to degradation. Degradation products can lower the pH of the solution, increasing tissue irritation. While not definitive proof of expiration, increased injection site discomfort in a previously tolerated preparation warrants caution.
Functional Signs
The most telling sign of degraded tirzepatide is reduced efficacy. If you have been on a stable dose producing consistent appetite suppression, weight loss, or glucose control, and those effects suddenly diminish without any change in your routine, the vial may have lost significant potency.
A 15-20% potency loss means your 10 mg dose is effectively delivering 8 to 8.5 mg. That difference is meaningful. In the SURMOUNT trials, each dose tier (5 mg, 10 mg, 15 mg) produced measurably different weight loss outcomes. Dropping from one effective dose to a lower one due to degradation will show in results.
For dosing details and how units translate to milligrams, see how many units is 2.5 mg of tirzepatide.
What Happens If You Use Expired Tirzepatide
This is the question that matters most. Pharmaceutical expiration dates carry a margin of safety, and the answer depends on how far past the date you are and how the product was stored.
Reduced Efficacy Is the Primary Risk
Expired tirzepatide is not typically dangerous. The peptide breaks down into smaller, inactive fragments and altered amino acids. These degradation products are generally recognized and cleared by the body without adverse effects.
The real consequence is reduced efficacy. If you are using tirzepatide for weight management and your vial has lost 20% potency, your actual dose is 20% lower than intended. That gap can mean the difference between losing 1.5 pounds per week and losing nothing at all.
A study on the stability of GLP-1 receptor agonists found that degraded formulations showed reduced receptor binding but no increase in adverse events compared to fresh formulations (Kapitza et al., 2015). The risk is therapeutic failure, not toxicity.
When Expired Tirzepatide Becomes Genuinely Dangerous
Three scenarios cross the line from "less effective" to "potentially harmful."
Bacterial contamination. A reconstituted vial stored beyond 28 days, especially one mixed with sterile water rather than bacteriostatic water, can harbor bacteria. Injecting contaminated solution risks abscess formation, cellulitis, or systemic infection. In a worst case, injection of heavily contaminated solution can cause sepsis. Bacteriostatic water's benzyl alcohol preservative reduces this risk but does not eliminate it after extended storage.
Aggregate-driven immune response. Peptide aggregates formed during prolonged storage can trigger antibody formation. Anti-drug antibodies against tirzepatide could reduce the efficacy of future doses or, in rare cases, cause allergic reactions ranging from injection site reactions to anaphylaxis. Clinical trials report anti-tirzepatide antibody formation in approximately 2.1% of treated subjects at standard doses. Degraded formulations with higher aggregate content could increase that percentage.
Unpredictable dosing. If a vial has degraded unevenly (more potent at the bottom where sediment settles, less potent at the top), individual doses become unpredictable. One injection might deliver 70% of the intended dose while the next delivers 110%. This inconsistency can cause unexpected blood sugar swings in diabetic patients, which is clinically dangerous.
Storage Best Practices to Maximize Shelf Life
Proper storage is the single most important factor in preserving tirzepatide potency. The difference between careful storage and careless handling can mean weeks of additional shelf life.
Temperature Control
Lyophilized powder: Store at -20°C for maximum longevity. A standard kitchen freezer is adequate. If freezer space is limited, the refrigerator at 2-8°C is acceptable for up to 24 months.
Reconstituted solution: Refrigerate at 2-8°C immediately after mixing. Place the vial in the main body of the refrigerator, never in the door. Door shelves experience temperature swings of 5-8°C with each opening.
Pre-filled pens: Refrigerate until first use. After the first injection, the pen can stay at room temperature (up to 30°C) for 21 days. Do not return a room-temperature pen to the fridge and restart the clock. The 21-day countdown begins the moment the pen leaves refrigeration.
Invest in a simple refrigerator thermometer. Consumer refrigerators frequently run warmer than their dial suggests. A reading above 8°C means your peptides are degrading faster than expected.
For reconstitution technique, see how to reconstitute GHK-Cu (the same principles apply to tirzepatide). Use our peptide reconstitution calculator for precise volumes.
Light Protection
UV and visible light catalyze oxidation of tryptophan and methionine residues. Always store tirzepatide in its original carton, in an amber vial, or wrapped in aluminum foil.
A vial left on a countertop in direct sunlight for 8 hours can lose 5-8% potency from photo-oxidation alone. That loss compounds with heat exposure if the room is warm. Never store peptides on a windowsill, bathroom counter, or anywhere exposed to natural light.
Contamination Prevention
Sterile technique during reconstitution and dose drawing determines whether microbial contamination shortens your vial's life.
Swab the vial stopper with a fresh alcohol pad before every needle insertion. Use a new needle for each draw. Do not touch the needle to any surface before piercing the stopper. Wash hands thoroughly before handling vials.
If you reconstitute with sterile water (no preservative), draw all planned doses immediately and store individual doses in separate sterile syringes in the refrigerator. Do not re-enter a sterile-water vial after 24 hours. Bacteriostatic water is strongly preferred for multi-dose vials. Learn more about mixing ratios in how much bacteriostatic water for semaglutide (the principles apply across GLP-1 agonists).
Travel Considerations
Traveling with tirzepatide requires maintaining the cold chain. Pre-filled pens tolerate 21 days at room temperature, making them travel-friendly for trips under three weeks. Reconstituted vials need insulated bags with cold packs.
Use a medical-grade cooler bag with gel packs pre-chilled to 2-8°C. Avoid direct contact between the ice pack and the vial, which can freeze the solution. A washcloth or small towel between the pack and the vial provides adequate insulation.
For air travel, carry peptides in your cabin bag with a letter from your prescriber. Checked luggage undergoes temperature extremes in the cargo hold. TSA allows injectable medications with proper documentation. The peptide safety guide covers travel protocols in greater detail.
Peptide Shelf Life Comparison Table
Tirzepatide's shelf life is comparable to other engineered GLP-1 agonists and longer than many research peptides. This comparison helps you plan storage and purchasing timelines if you use multiple compounds.
| Peptide | Lyophilized (Freezer) | Lyophilized (Fridge) | Reconstituted (Fridge) | Room Temp Tolerance |
|---|---|---|---|---|
| Tirzepatide | 24-36 months | 18-24 months | 28 days | 21 days (pen) |
| Semaglutide | 24-36 months | 18-24 months | 28-56 days | 56 days (pen) |
| BPC-157 | 36-48 months | 24-36 months | 28-30 days | 4-6 hours |
| GHK-Cu | 24-36 months | 12-18 months | 21-28 days | 2-4 hours |
| TB-500 | 36-48 months | 24-36 months | 28-30 days | 4-6 hours |
| Ipamorelin | 24-36 months | 18-24 months | 28-30 days | 4-6 hours |
| Retatrutide | 24-36 months | 18-24 months | 28-30 days | 4-6 hours |
| CJC-1295 | 24-36 months | 18-24 months | 21-28 days | 4-6 hours |
| IGF-1 LR3 | 18-24 months | 12-18 months | 14-21 days | 2-3 hours |
| NAD+ | 12-18 months | 6-12 months | 7-14 days | 1-2 hours |
Several patterns emerge from this table. Engineered pharmaceutical peptides like tirzepatide and semaglutide are designed for stability, incorporating modifications like fatty acid side chains that protect against degradation. Research peptides without these modifications have shorter shelf lives. Peptides with metal coordination (GHK-Cu) or extreme pH sensitivity (IGF-1 LR3) degrade fastest.
For daily management of multiple peptides, our peptide half-life tracker helps you schedule doses around each compound's pharmacokinetics. Convert between units with the peptide unit converter.
Compounded vs. Manufacturer Tirzepatide: Expiration Differences
The expiration date on a compounded tirzepatide vial is not the same as the date on a Mounjaro pen, and the gap matters.
Eli Lilly conducts extensive stability testing under ICH guidelines and assigns expiration dates backed by months of accelerated degradation data. Mounjaro pens benefit from proprietary buffers, precise pH control, and controlled manufacturing environments. Their 18 to 24 month shelf life reflects rigorous validation.
Compounding pharmacies operate under different constraints. Many assign a beyond-use date (BUD) of 6 months for lyophilized preparations and 28 to 45 days for reconstituted solutions. These dates are often based on USP <797> and USP <795> standards rather than product-specific stability testing. Some compounding pharmacies conduct their own stability studies; many do not.
The practical implication: a compounded tirzepatide vial labeled with a 6-month BUD may actually retain potency for longer, but there is no data to confirm it. Use it within the labeled date. If you are comparing costs between compounded and brand-name tirzepatide, factor in how quickly you will use each vial. A cheaper vial that expires before you finish it is not a bargain.
For guidance on navigating the current peptide landscape, see getting started with peptides.
The Science of Peptide Expiration Dating
Pharmaceutical expiration dates are built on stability testing protocols defined by the ICH and enforced by the FDA. Understanding how these dates are derived helps you interpret them correctly.
Accelerated Stability Testing
Manufacturers store samples at elevated temperatures (40°C / 75% relative humidity) for 6 months and use the degradation rate to project shelf life at recommended storage conditions (2-8°C or 25°C). The Arrhenius equation models the relationship between temperature and reaction rate, allowing extrapolation from short-term high-temperature data to long-term cold-storage predictions.
This method is conservative by design. The projected shelf life represents the point at which the peptide is expected to retain at least 90% of its labeled potency with 95% statistical confidence. Most products remain above 90% potency for weeks or months beyond the printed date. However, "most" is not "all," and the margin of safety exists because individual vials within a batch can degrade at different rates.
Why the 90% Threshold Matters
The pharmaceutical industry uses 90% of labeled potency as the lower acceptable limit. Below 90%, dose-response relationships become unpredictable. For a drug like tirzepatide, where specific doses (5 mg, 10 mg, 15 mg) are titrated against clinical response, a 10%+ potency loss effectively drops you into a sub-therapeutic dose range without your knowledge.
The SURMOUNT-1 trial documented that 15 mg tirzepatide produced 22.5% mean body weight reduction compared to 15.0% at 5 mg. That dose-response curve is steep enough that a 15-20% potency loss would materially affect outcomes (Jastreboff et al., NEJM 2022).
The semaglutide dosage chart in mL illustrates how precise dosing affects clinical outcomes across another GLP-1 agonist.
Common Mistakes That Shorten Tirzepatide Shelf Life
Most premature tirzepatide degradation comes from avoidable handling errors. Correcting these habits can add days or weeks to a vial's useful life.
Leaving the vial out during dose preparation. Reconstituted tirzepatide left at room temperature for 30 minutes during a dose draw experiences more degradation in that half hour than in a full day in the refrigerator. Get the vial out, draw your dose, and return it within 60 seconds.
Storing in the bathroom. Bathrooms are the worst location for any medication. Showers create humidity spikes that penetrate even sealed containers. Temperature fluctuations from hot showers compound the problem. Store peptides in a bedroom or kitchen refrigerator.
Shaking the vial. Agitation creates air-liquid interfaces that accelerate protein aggregation. Swirl gently. Never shake.
Using the wrong water. Reconstituting with tap water or non-pharmaceutical-grade water introduces contaminants that catalyze degradation and microbial growth. Use only bacteriostatic water or sterile water for injection, and make sure you know whether bacteriostatic water needs refrigeration after opening. For mixing details, see how much bacteriostatic water for semaglutide.
Ignoring the first-use date. Write the reconstitution date on the vial with a permanent marker. Without a date, you are guessing at the 28-day window. Guessing leads to using degraded peptide.
Reusing needles. Each needle puncture widens the stopper hole slightly. After multiple uses of the same needle, the stopper may leak, admitting air and bacteria. Use a fresh needle every time.
Extending Shelf Life: What Works and What Does Not
Researchers and clinicians have investigated several approaches to extending peptide shelf life. Some are effective; others are myths.
What Works
Freezing lyophilized powder. Moving from 2-8°C to -20°C genuinely extends shelf life from 18-24 months to 24-36 months or beyond. This is the single most effective intervention for unopened vials.
Adding stabilizers during compounding. Professional compounding pharmacies may add trehalose, mannitol, or other lyoprotectants that shield the peptide during freeze-drying and storage. These additives measurably improve stability. You cannot add them at home.
Minimizing headspace. Replacing air in the vial with nitrogen gas after each draw reduces oxidation. Some compounding pharmacies ship vials nitrogen-purged. At home, you can partially replicate this by storing vials inverted (solution against the stopper, air pocket at the bottom), though this is not standard practice.
What Does Not Work
Freezing reconstituted peptide. Freezing a solution creates ice crystals that physically shear peptide bonds and cause irreversible aggregation. The freeze-thaw cycle destroys the very structure you are trying to preserve. Never freeze reconstituted tirzepatide.
Adding extra bacteriostatic water. Diluting the solution does not extend its life. It lowers concentration, making dosing less accurate, while the same degradation pathways continue at the same rate.
Wrapping in plastic wrap. Sometimes suggested as a moisture barrier for lyophilized vials, plastic wrap does not seal tightly enough to matter. The original rubber stopper and crimp seal are designed for this purpose. Plastic wrap adds nothing.
Important Safety Warnings
Never use tirzepatide that shows visible cloudiness, particles, or color change. These signs indicate structural degradation and potential immunogenicity risk.
Do not use reconstituted tirzepatide beyond 28 days regardless of appearance. Microbial contamination is not always visible. A clear solution can still harbor dangerous levels of bacteria.
If you are using tirzepatide for type 2 diabetes and suspect your medication has lost potency, monitor blood glucose closely and contact your prescriber. A sudden loss of glycemic control from degraded medication can cause hyperglycemia requiring medical intervention.
Do not share vials between individuals. Multi-dose vials from compounding pharmacies are intended for single-patient use. Sharing increases contamination risk and is medically and legally inappropriate.
For a comprehensive overview of safety considerations across all peptides, see the peptide safety guide. For information on how tirzepatide compares to similar compounds, see our semaglutide profile.
Frequently Asked Questions
How long does tirzepatide last in the fridge after reconstitution?
Reconstituted tirzepatide lasts 28 days in the refrigerator at 2-8°C when mixed with bacteriostatic water. If mixed with sterile water (no preservative), use within 24-48 hours. After 28 days, potency drops below the 90% threshold considered pharmaceutically acceptable. Write the mixing date on the vial to track this window.
Can you use Mounjaro after the expiration date?
Eli Lilly does not recommend using Mounjaro past its printed expiration date. Most pens retain greater than 90% potency for 2-4 weeks beyond the date if stored at 2-8°C, but this is not guaranteed. Potency testing requires laboratory equipment. If the pen has been at room temperature for more than 21 days, discard it regardless of the printed date.
Does tirzepatide go bad at room temperature?
Pre-filled pens tolerate room temperature (up to 30°C) for 21 days. Reconstituted tirzepatide degrades rapidly at room temperature, losing 10-15% potency within 7 days and 28-40% within 14 days. Lyophilized powder is more stable, lasting 6-12 months at room temperature. Always refrigerate reconstituted solutions immediately after each use.
How can you tell if tirzepatide has expired?
Check for cloudiness, visible particles, color change (should be clear to slightly yellow), or unusual odor. Functionally, decreased appetite suppression or worsening blood glucose on a previously effective dose suggests potency loss. Degradation beyond 15-20% produces measurable reductions in clinical effect. When in doubt, replace the vial rather than risk subtherapeutic dosing.
Is expired tirzepatide dangerous to inject?
Expired tirzepatide from potency loss alone is not typically dangerous. The greater risk is microbial contamination in reconstituted vials stored beyond 28 days, which can cause injection site infections or, in severe cases, sepsis. Aggregated peptide fragments may also trigger immune responses in approximately 2% of users. Reduced efficacy is the most common consequence.
How long does lyophilized tirzepatide last in the freezer?
Lyophilized tirzepatide stored at -20°C retains greater than 95% potency for 24-36 months. Some stability data suggests viability beyond 36 months under ideal conditions (sealed, protected from light, consistent temperature). At refrigerator temperature (2-8°C), expect 18-24 months. Always check for discoloration or unusual texture before reconstituting older vials.
Should you freeze tirzepatide to make it last longer?
Freeze lyophilized (powder) tirzepatide, yes. Storage at -20°C extends shelf life to 24-36 months versus 18-24 months refrigerated. Never freeze reconstituted (liquid) tirzepatide. Ice crystals physically shear peptide bonds and cause irreversible aggregation, destroying 30-50% of active peptide in a single freeze-thaw cycle. This distinction is critical.
What is the difference between expiration date and beyond-use date for tirzepatide?
Expiration dates on manufacturer products (Mounjaro, Zepbound) are backed by 12-24 months of stability testing under ICH guidelines. Beyond-use dates on compounded tirzepatide follow USP <797> standards and are often 6 months for lyophilized and 28-45 days for reconstituted. The manufacturer date is more rigorously validated, but both should be respected.
The Bottom Line
Tirzepatide expires across every form. Lyophilized powder lasts 24 to 36 months frozen, 18 to 24 months refrigerated. Reconstituted solution lasts 28 days at 2-8°C with bacteriostatic water. Pre-filled pens follow the printed date when refrigerated and 21 days at room temperature.
The consequences of using expired tirzepatide are usually limited to reduced efficacy rather than safety hazards. A vial that has lost 15-20% potency delivers a lower effective dose, undermining the weight loss or glucose control you are working toward. The exception is microbial contamination in reconstituted vials stored too long, which can cause serious infections.
Store cold, store dark, label dates, and use sterile technique. These four habits protect your investment and your health. Use the tirzepatide dosage calculator to plan exact doses, and the peptide reconstitution calculator to mix accurately. For a broader view of peptide storage principles, read our complete storage guide.
Related articles: - Does Semaglutide Expire? — parallel expiration guide for the other major GLP-1 agonist - Does Semaglutide Need to Be Refrigerated? — complete storage guide for Ozempic, Wegovy, and compounded forms - How to Reconstitute Tirzepatide — step-by-step guide that starts the 28-day clock - How to Take BPC-157 — storage principles apply to all reconstituted peptides - Does Tirzepatide Burn Fat? — what you gain from proper storage and consistent dosing - Can You Drink Alcohol on Tirzepatide? — safety considerations while on treatment - Why Am I Not Losing Weight on Semaglutide? — degraded peptide from poor storage can reduce efficacy
Related Articles
Does Semaglutide Expire? Storage
Semaglutide expires: Ozempic pens last until the label date (typically 2 years), compounded vials 6 months, reconstituted vials 28 days refrigerated.
Tirzepatide Fridge Storage Guide
Tirzepatide lasts until its expiry date unreconstituted at 2-8°C and 28 days once reconstituted. Refrigeration guide for Mounjaro pens and compounded vials.
What to Eat on Tirzepatide: Diet Guide
What to eat on tirzepatide to minimize nausea, prevent muscle loss, and maximize results. Protein targets, meal plans, foods to avoid, and hydration.
How to Reconstitute Tirzepatide
How to reconstitute tirzepatide step by step for 5mg, 10mg, 15mg, and 30mg vials. Concentration tables, BAC water volumes, syringe units, and storage guide.