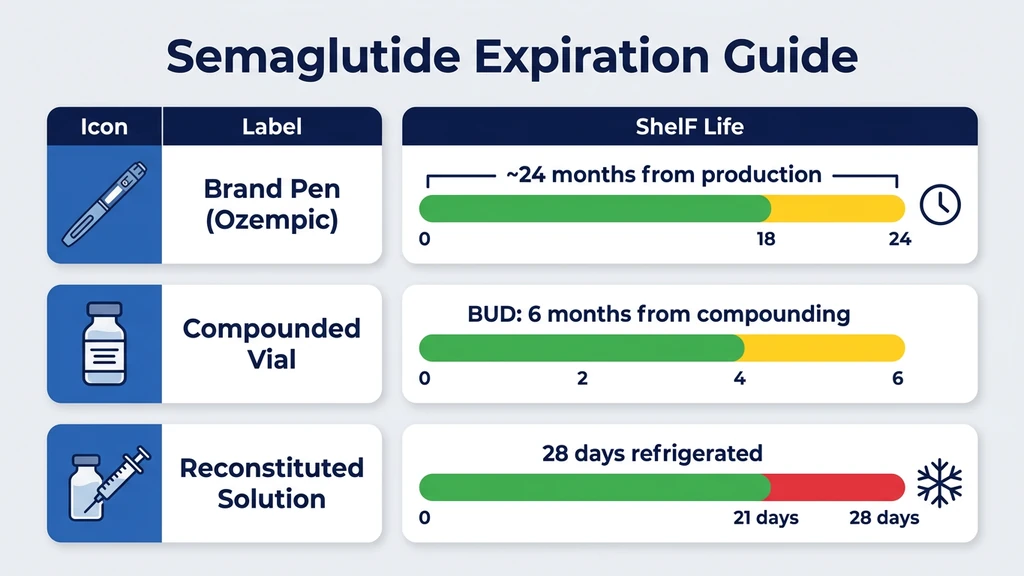
You pulled an Ozempic pen from the back of your refrigerator. The box has been sitting there for months, and now you need to check whether it is still good. Yes, semaglutide expires. Unopened Ozempic and Wegovy pens are potent until the label date, typically two years from manufacture. Compounded semaglutide vials carry a beyond-use date of six months from compounding. Reconstituted lyophilized semaglutide lasts 28 days refrigerated at 2-8°C when mixed with bacteriostatic water.
The expiration date is not decoration. Semaglutide is a 31-amino-acid peptide, and every form degrades through predictable chemical pathways once the clock starts. Using a pen or vial past its window means injecting a lower dose than intended, which can stall weight loss or destabilize blood glucose without a clear explanation.
| Semaglutide Form | Storage Temperature | Shelf Life | Key Condition |
|---|---|---|---|
| Ozempic/Wegovy pen (unopened) | 2-8°C (fridge) | Until printed date (typically 2 years) | Original carton, away from light |
| Ozempic/Wegovy pen (in use) | Room temp (up to 30°C) | 56 days | Cap on, away from direct heat/light |
| Compounded vial (lyophilized) | 2-8°C (fridge) | 6 months (BUD) | Sealed, protected from light |
| Compounded vial (liquid) | 2-8°C (fridge) | 28-45 days (BUD) | Per pharmacy label |
| Reconstituted (bacteriostatic water) | 2-8°C (fridge) | 28 days | Sterile technique, minimize punctures |
| Reconstituted (sterile water) | 2-8°C (fridge) | 24-48 hours | No preservative, use immediately |
| Lyophilized powder | -20°C (freezer) | 24-36 months | Sealed, dry, away from light |
For precise mixing volumes, use the peptide reconstitution calculator. For dosing protocols across all concentrations, see the semaglutide dosage chart in mL.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
How Long Does Semaglutide Last by Product Type
Every semaglutide product reaches patients in a different physical form. The active molecule is identical across all of them: the same GLP-1 receptor agonist with a C-18 fatty acid side chain that extends its half-life to approximately seven days in the body. But the packaging, solvent, and preservatives surrounding that molecule determine how fast it breaks down on the shelf.
Novo Nordisk, the manufacturer of Ozempic and Wegovy, conducts stability testing under International Council for Harmonisation (ICH) Q1A guidelines. These protocols expose the product to elevated temperature and humidity, then project long-term stability from the degradation rate (ICH Q1A(R2), 2003). Compounding pharmacies follow USP <797> and USP <795> standards, which are less product-specific but still conservative.
Ozempic and Wegovy Pre-Filled Pens
Ozempic (for type 2 diabetes) and Wegovy (for weight management) are liquid formulations engineered for maximum shelf stability. Novo Nordisk's proprietary buffer system, precise pH control, and pharmaceutical-grade excipients protect the peptide from degradation far longer than a compounding pharmacy can achieve.
Unopened pens stored at 2-8°C remain potent until the printed expiration date. That date is typically 24 months from manufacture, though it can vary by batch. After the first injection or after removing the pen from refrigeration, it remains stable for 56 days at room temperature (up to 30°C / 86°F). This 56-day window is longer than tirzepatide pens, which allow only 21 days at room temperature.
Never freeze an Ozempic or Wegovy pen. The freeze-thaw cycle disrupts the protein structure and can alter the injection mechanism. If a pen has been frozen, even briefly, discard it. Novo Nordisk's prescribing information is explicit on this point: frozen pens should not be used, even if thawed.
The 56-day room-temperature stability is clinically validated. Novo Nordisk submitted accelerated stability data to the FDA demonstrating that semaglutide in the pen formulation retains greater than 95% potency for 8 weeks at 30°C (Novo Nordisk Ozempic Prescribing Information, 2024).
Compounded Semaglutide Vials
Compounded semaglutide comes in two forms: lyophilized powder requiring reconstitution, and pre-mixed liquid solutions. Each carries a beyond-use date (BUD) assigned by the compounding pharmacy.
Lyophilized compounded semaglutide typically receives a 6-month BUD when stored at 2-8°C. Pre-mixed liquid formulations get 28 to 45 days depending on the pharmacy's stability data. These dates are often based on USP general chapter standards rather than product-specific accelerated stability testing, so they tend to be conservative.
The practical gap between manufacturer and compounded products matters. An Ozempic pen can sit in your refrigerator for two years and remain fully potent. A compounded vial has a fraction of that window. If you are switching from brand-name to compounded semaglutide, plan your purchasing around how fast you will use each vial. A cheaper vial that expires before you finish it saves nothing.
For guidance on mixing compounded semaglutide, see how much bacteriostatic water for semaglutide.
Reconstituted Lyophilized Semaglutide
Adding bacteriostatic water to lyophilized semaglutide powder starts the degradation clock. The peptide is now in aqueous solution, and every chemical breakdown pathway activates simultaneously.
With bacteriostatic water (containing 0.9% benzyl alcohol), reconstituted semaglutide lasts approximately 28 days at 2-8°C. The benzyl alcohol suppresses bacterial growth, allowing multiple draws from the same vial. Without that preservative, using plain sterile water for injection, the solution is safe for only 24 to 48 hours.
Each needle puncture through the rubber stopper introduces potential contamination. After 15 to 20 punctures, the stopper may no longer seal properly. Use our semaglutide dosage calculator to calculate exact draw volumes and minimize unnecessary vial entries.
For a broader look at reconstituted peptide timelines across multiple compounds, see how long do reconstituted peptides last.
How Semaglutide Degrades Over Time
Semaglutide does not flip from potent to useless on its expiration date. Degradation is a continuous chemical process. Potency drops gradually, accelerating as breakdown products catalyze further reactions. Understanding the pathways tells you what to watch for and why storage conditions matter so much.
The C-18 fatty acid chain that gives semaglutide its long in-vivo half-life also provides some protection against degradation. This acylation shields vulnerable amino acid residues from solvent exposure. But the protection is partial, not absolute.
Deamidation
Asparagine residues in the semaglutide peptide chain convert to aspartate through deamidation. This is the dominant degradation pathway for GLP-1 receptor agonists in aqueous solution.
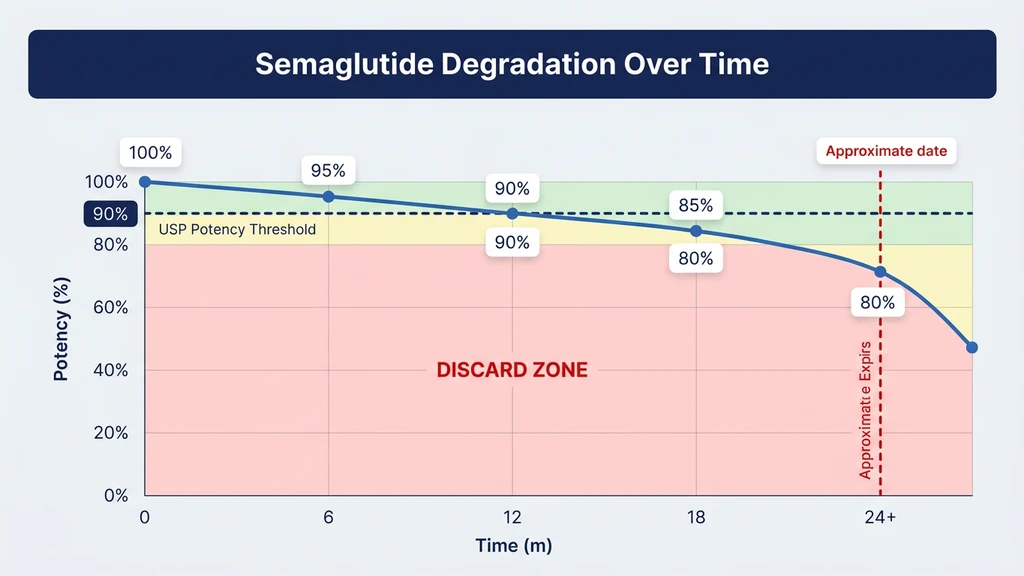
The reaction rate roughly doubles with every 10°C temperature increase. At 2-8°C, deamidation is slow enough that the peptide retains greater than 90% potency for 28 days in solution. At 25°C, that same level of degradation occurs within 7 to 10 days. At 37°C, significant potency loss happens in 3 to 5 days.
A comprehensive review of peptide pharmaceutical stability identified deamidation as the most common chemical instability, affecting virtually every peptide containing asparagine-glycine sequences (Manning et al., Pharm Res, 2010). Semaglutide's sequence includes multiple asparagine residues, making it susceptible.
Oxidation
Methionine and tryptophan residues in semaglutide are vulnerable to oxidation. Dissolved oxygen, UV light, and trace metals all catalyze the reaction.
Oxidized semaglutide binds less effectively to the GLP-1 receptor. Research on GLP-1 receptor agonist stability found that oxidation of a single methionine residue reduced receptor binding affinity by 40-60% (Houen & Jakobsen, J Pharm Sci, 2019). That reduction translates directly to a weaker dose.
A vial sitting on a countertop in direct sunlight for 8 hours can lose 5-8% potency from photo-oxidation alone. Combine that with heat exposure if the room is warm, and you compound the damage. Store vials in the original carton, in amber glass, or wrapped in aluminum foil. Never leave semaglutide on a windowsill, bathroom counter, or anywhere exposed to natural light.
Aggregation
When semaglutide molecules unfold or partially degrade, they can clump into aggregates. These appear as cloudiness, visible particles, or a gel-like film in the solution.
Aggregated semaglutide is not just less potent. It is potentially immunogenic. The immune system can recognize peptide aggregates as foreign and mount an antibody response. Anti-drug antibodies against semaglutide could reduce the efficacy of future doses or, in rare cases, trigger allergic reactions. In the STEP trials, anti-semaglutide antibody formation occurred in approximately 1-2% of subjects at therapeutic doses (Wilding et al., NEJM, 2021). Degraded formulations with higher aggregate content could push that number higher.
Temperature cycling accelerates aggregation. Every time you take a vial out of the fridge, warm it in your hands, draw a dose, and return it, you create a micro-cycle that stresses the peptide. Minimize handling time. Draw your dose and return the vial within 60 seconds.
The Degradation Curve
Potency loss follows an exponential decay pattern. Early degradation is slow; later degradation accelerates as breakdown products catalyze further reactions.
| Time After Reconstitution | Estimated Potency at 2-8°C | Estimated Potency at 25°C |
|---|---|---|
| Day 0 | 100% | 100% |
| Day 7 | 97-99% | 90-95% |
| Day 14 | 95-98% | 82-90% |
| Day 21 | 93-97% | 72-82% |
| Day 28 | 90-95% | 62-74% |
| Day 42 | 83-91% | 48-58% |
| Day 56 | 72-84% | 32-42% |
At refrigerator temperature, reconstituted semaglutide stays within the pharmaceutical threshold (greater than 90% potency) for approximately 28 days. At room temperature, it drops below that threshold by day 14 to 18. Pre-filled pens perform better because Novo Nordisk's formulation includes stabilizing excipients that slow all three degradation pathways simultaneously.
For context on how tirzepatide compares, its reconstituted shelf life is also 28 days refrigerated, but pen stability at room temperature is only 21 days versus semaglutide's 56.
How to Read Expiration Dates on Semaglutide Products
Different semaglutide products display their expiration information differently. Misreading the date is a common and avoidable mistake.
Ozempic and Wegovy Pens
The expiration date is printed on the pen label and on the outer carton. It appears as "EXP" followed by a month and year (e.g., EXP 03/2028). This means the pen is potent through the last day of that month when stored at 2-8°C.
Two separate timelines apply. The printed expiration date governs the unopened pen in the refrigerator. The 56-day clock starts the moment you remove the pen from the fridge or give your first injection, whichever comes first. Both timelines matter. If your pen's printed date is next month but you took it out of the fridge 50 days ago, you have 6 days left regardless of the printed date.
Write the "first use" date on the pen with a permanent marker. Without it, you are guessing. A study on medication adherence found that patients who labeled their injectable medications with start dates were 34% more likely to discard expired product on time (Patel et al., Patient Prefer Adherence, 2013).
Compounded Semaglutide Vials
Compounding pharmacies print a beyond-use date (BUD) on the vial label. This is not the same as a manufacturer expiration date. The BUD is typically more conservative because it is based on general USP standards rather than product-specific stability testing.
A compounded vial might read "BUD: 09/2026" for lyophilized powder or "BUD: 04/14/2026" for pre-mixed liquid. The liquid BUD counts from the compounding date, not from when you received it. Check how many days remain when your order arrives. If a pre-mixed liquid vial arrives with only 15 of its 28-day window remaining, you need to use it faster or request fresher batches.
For lyophilized vials, the BUD applies to the unopened powder. Once you reconstitute, a new 28-day window begins (with bacteriostatic water). Write the reconstitution date on the vial. This date overrides the printed BUD if it comes first.
Batch Numbers and Lot Codes
Every semaglutide product carries a lot number linking it to a specific manufacturing batch. If Novo Nordisk or a compounding pharmacy issues a recall, the lot number identifies affected products.
Record your lot number when you start a new pen or vial. Keep a simple note on your phone: product name, lot number, start date. In the event of a recall, this information lets you confirm immediately whether your product is affected. The FDA's MedWatch system publishes recall notices searchable by lot number.
Signs That Your Semaglutide Has Expired or Degraded
You cannot measure potency at home with laboratory instruments. But several visible and functional signs indicate a vial or pen has degraded past acceptable use.
Visual Warning Signs
Cloudiness or turbidity. Fresh semaglutide solution is clear and colorless. Any haziness or opalescence indicates protein aggregation. Do not inject a cloudy solution.
Visible particles. Hold the vial or pen up to a light source and gently swirl. Floating specks, flakes, or fibers signal contamination or aggregation. A single tiny fiber could be a stopper fragment, but multiple particles mean the product should be discarded.
Color change. Semaglutide solution is colorless to very slightly yellow. Any shift toward amber, brown, or pink indicates oxidation or chemical breakdown. Discard immediately.
Gel formation. In advanced degradation, peptide aggregates can form a gel-like layer on vial walls or at the bottom. This represents severe structural damage. The peptide is not salvageable.
Functional Warning Signs
The most telling sign of degraded semaglutide is reduced clinical effect. If you have been on a stable dose producing consistent appetite suppression and weight loss, and those effects suddenly weaken without any change in your habits, the product may have lost significant potency.
Consider the dose-response data. In the STEP 1 trial, 2.4 mg weekly semaglutide produced 14.9% mean body weight reduction versus 2.4% with placebo (Wilding et al., NEJM, 2021). A 15-20% potency loss in your vial effectively drops your 2.4 mg dose to 1.9-2.0 mg. That gap could reduce your weekly weight loss from 0.5 kg to 0.2 kg or less.
If you are using semaglutide for type 2 diabetes and notice blood glucose readings creeping upward on a previously stable dose, degraded medication is one possible explanation. Check the vial visually, confirm the reconstitution date, and contact your prescriber if suspicion persists. For other reasons weight loss may stall, see why am I not losing weight on semaglutide.
Increased Injection Site Reactions
Degradation products can lower the pH of semaglutide solution, increasing tissue irritation at the injection site. If your injections begin stinging or burning more than usual with a previously tolerated preparation, the solution chemistry may have shifted.
Aggregated peptide fragments can also trigger localized immune responses, producing more redness, swelling, or induration than you experienced with fresh product. This is not definitive proof of expiration, but it warrants caution. Switch to a new vial or pen and see if the reaction resolves.
Is Expired Semaglutide Dangerous or Just Less Effective
This is the question that drives most searches. The answer depends on how far past the date you are, how the product was stored, and which form you are using.
Reduced Efficacy: The Common Outcome
Expired semaglutide is not typically toxic. The peptide breaks down into smaller inactive fragments and altered amino acids. The body recognizes and clears these degradation products without adverse effects in most cases.
The real consequence is reduced efficacy. A pen or vial that has lost 20% potency delivers 20% less active drug per injection. For someone on a weight management protocol, that shortfall can stall progress without an obvious cause. You eat the same, exercise the same, inject on schedule, yet the scale stops moving.
A study on the stability of GLP-1 receptor agonists found that degraded formulations showed reduced receptor binding but no increase in adverse events compared to fresh formulations (Kapitza et al., 2015). The risk is therapeutic failure, not poisoning.
When Expired Semaglutide Becomes Genuinely Risky
Three scenarios cross the line from "weaker dose" to "potential harm."
Bacterial contamination. A reconstituted vial stored beyond 28 days, especially one mixed with sterile water instead of bacteriostatic water, can harbor bacteria. Injecting contaminated solution risks abscess formation, cellulitis, or systemic infection. In severe cases, heavily contaminated injections can cause sepsis. Bacteriostatic water's benzyl alcohol preservative reduces but does not eliminate this risk after extended storage.
Aggregate-driven immune response. Peptide aggregates formed during prolonged storage can trigger antibody formation. Anti-drug antibodies against semaglutide could reduce the effectiveness of future doses or cause allergic reactions ranging from injection site swelling to, in rare cases, anaphylaxis. The SUSTAIN and STEP trials reported anti-semaglutide antibody formation in approximately 1-2% of treated subjects at standard doses. Degraded formulations with higher aggregate content could increase that percentage.
Unpredictable dosing in multi-dose vials. If a vial has degraded unevenly, with more concentrated solution near the bottom and dilute solution near the top, individual doses become inconsistent. One draw might deliver 70% of the intended dose. The next might deliver 110%. For diabetic patients relying on semaglutide for glycemic control, these swings can cause dangerous blood sugar fluctuations.
Storage Best Practices to Maximize Semaglutide Shelf Life
Proper storage is the single most important factor in preserving semaglutide potency. The difference between careful handling and casual storage can add weeks to a product's useful life.
Temperature Control
Ozempic/Wegovy pens: Refrigerate at 2-8°C until first use. After the first injection, the pen can remain at room temperature (up to 30°C) for 56 days. Do not return a room-temperature pen to the fridge and assume the clock resets. The 56-day countdown begins when the pen first leaves refrigeration.
Lyophilized powder: Store at -20°C for maximum longevity (24-36 months). A standard kitchen freezer works. If freezer space is unavailable, 2-8°C is acceptable for the duration of the printed BUD. Never store vials in the refrigerator door, where temperature fluctuates 5-8°C with each opening.
Reconstituted solution: Refrigerate at 2-8°C immediately after mixing. Place the vial in the main body of the refrigerator on a dedicated shelf. A simple refrigerator thermometer costs a few dollars and confirms your fridge is actually at 2-8°C. Consumer refrigerators frequently run warmer than the dial indicates.
For step-by-step reconstitution technique, see how much bacteriostatic water for semaglutide. For broader storage principles, consult the peptide storage guide.
Light Protection
UV and visible light catalyze oxidation of tryptophan and methionine residues. Always store semaglutide in its original carton, in an amber vial, or wrapped in aluminum foil.
Novo Nordisk packages Ozempic and Wegovy pens in opaque cartons for this reason. Removing the pen from its carton and leaving it on a kitchen counter exposes it to ambient light throughout the day. Even indoor fluorescent lighting contributes to photo-oxidation over weeks. Keep the pen in its box between injections.
For compounded vials, request amber glass from your pharmacy. Clear glass vials transmit the UV wavelengths (290-320 nm) most responsible for peptide oxidation. If your vial is clear glass, wrap it in foil and store in a closed container inside the fridge.
Sterile Technique for Multi-Dose Vials
Every needle entry into a vial is a potential contamination event. Sterile technique determines whether your vial lasts the full 28 days or becomes a bacterial culture.
Swab the rubber stopper with a fresh alcohol pad before every needle insertion. Wait 10 seconds for the alcohol to dry. Use a new needle for each draw. Do not touch the needle to any surface before piercing the stopper. Wash hands thoroughly before handling vials and syringes.
If you reconstitute with sterile water (no preservative), draw all planned doses immediately into separate sterile syringes and store them individually in the refrigerator. Do not re-enter a sterile-water vial after 24 hours. Bacteriostatic water is strongly preferred for any multi-dose vial. For details on bacteriostatic water storage, see does bacteriostatic water need to be refrigerated.
Travel with Semaglutide
Semaglutide pens have a generous travel window. The 56-day room temperature stability means you can carry an Ozempic or Wegovy pen in your bag for nearly two months without refrigeration, as long as the temperature stays below 30°C.
For reconstituted vials, use a medical-grade insulated bag with gel packs pre-chilled to 2-8°C. Place a small towel between the gel pack and the vial to prevent accidental freezing. Direct contact with an ice pack can freeze the solution, destroying the peptide.
For air travel, carry semaglutide in your cabin bag with a prescription label or letter from your prescriber. Checked luggage is exposed to extreme temperature swings in the cargo hold. TSA permits injectable medications with documentation. The peptide safety guide covers travel protocols in greater detail.
FDA vs. Compounding Pharmacy Expiration Standards
The expiration date on an Ozempic pen and the beyond-use date on a compounded semaglutide vial are generated through fundamentally different processes. Understanding the gap helps you interpret both dates correctly.
Novo Nordisk submits Ozempic and Wegovy to ICH stability testing: 6 months at 40°C/75% relative humidity, plus 12-24 months of real-time data at recommended storage conditions. The Arrhenius equation models the relationship between temperature and degradation rate, allowing extrapolation from accelerated data to cold-storage predictions. The resulting expiration date represents the point at which at least 90% of labeled potency is retained with 95% statistical confidence. Most individual pens exceed this threshold by weeks or months.
Compounding pharmacies operate under different constraints. USP <797> (sterile preparations) and USP <795> (non-sterile preparations) provide default BUD frameworks. Many pharmacies assign BUDs based on these general standards rather than conducting product-specific stability studies. A 503B outsourcing facility may perform its own accelerated testing and assign longer BUDs; a traditional 503A pharmacy typically does not.
The practical implication: a compounded vial labeled "BUD: 6 months" may actually retain potency longer, but no validated data confirms it. Respect the printed date. If cost-effectiveness matters, calculate whether you will finish the vial before its BUD. If not, request a smaller vial size or adjust your purchasing frequency.
For guidance on navigating the peptide supply landscape, see getting started with peptides. For information on sourcing, see where to buy peptides in 2026.
Common Mistakes That Shorten Semaglutide Shelf Life
Most premature semaglutide degradation stems from avoidable handling errors. Correcting these habits protects both your investment and your results.
Leaving the pen or vial out during dose preparation. Reconstituted semaglutide at room temperature for 30 minutes experiences more degradation in that half hour than in a full day in the refrigerator. Retrieve the product, draw your dose, and return it within 60 seconds. Think of it like leaving milk on the counter. Brief exposure is fine. Forgetting it for an hour is not.
Storing in the bathroom. Bathrooms generate humidity spikes from showers that penetrate even sealed containers. Temperature swings from hot water compound the problem. Store semaglutide in a bedroom or kitchen refrigerator, never a bathroom cabinet.
Shaking the vial. Agitation creates air-liquid interfaces that accelerate protein aggregation. Swirl gently to mix. Never shake. This distinction matters more for peptides than for small-molecule drugs because the protein structure is physically disrupted by vigorous mixing.
Using the wrong reconstitution water. Tap water, distilled water from the grocery store, and non-pharmaceutical-grade water all introduce contaminants that catalyze degradation and microbial growth. Use only bacteriostatic water or sterile water for injection. For sourcing, see where to buy bacteriostatic water.
Forgetting to label the first-use date. Write the date you opened the pen or reconstituted the vial with a permanent marker on the product itself. Without a date, you are estimating the 28-day or 56-day window. Estimates drift. Labels do not.
Reusing needles. Each puncture through the stopper widens the entry point slightly. After multiple uses of the same needle, the stopper can leak, admitting air and bacteria. Use a fresh needle every time. The cost of a new needle is negligible compared to discarding a contaminated vial.
Semaglutide Shelf Life Compared to Other Peptides
Semaglutide's engineered stability places it among the longest-lasting GLP-1 agonists. The C-18 fatty acid acylation that extends its pharmacological half-life also provides some structural protection during storage. This comparison helps you plan storage and purchasing if you use multiple compounds.
| Peptide | Lyophilized (Freezer) | Lyophilized (Fridge) | Reconstituted (Fridge) | Room Temp Tolerance |
|---|---|---|---|---|
| Semaglutide | 24-36 months | 18-24 months | 28 days | 56 days (pen) |
| Tirzepatide | 24-36 months | 18-24 months | 28 days | 21 days (pen) |
| Retatrutide | 24-36 months | 18-24 months | 28-30 days | 4-6 hours |
| BPC-157 | 36-48 months | 24-36 months | 28-30 days | 4-6 hours |
| GHK-Cu | 24-36 months | 12-18 months | 21-28 days | 2-4 hours |
| TB-500 | 36-48 months | 24-36 months | 28-30 days | 4-6 hours |
| Ipamorelin | 24-36 months | 18-24 months | 28-30 days | 4-6 hours |
| CJC-1295 | 24-36 months | 18-24 months | 21-28 days | 4-6 hours |
| IGF-1 LR3 | 18-24 months | 12-18 months | 14-21 days | 2-3 hours |
| NAD+ | 12-18 months | 6-12 months | 7-14 days | 1-2 hours |
The 56-day room temperature tolerance of semaglutide pens stands out. That window exists because of Novo Nordisk's proprietary stabilizing excipients and the inherent protection from the fatty acid side chain. Research peptides without these modifications degrade far faster outside the fridge.
For daily management of multiple peptides, the peptide half-life tracker helps schedule doses around pharmacokinetic windows. Convert between measurement systems with the peptide unit converter. If you are considering switching between GLP-1 agonists, see the semaglutide to tirzepatide conversion guide.
Proper Disposal of Expired Semaglutide
Expired semaglutide should not be thrown in the household trash or flushed down the toilet. Both methods pose environmental and safety risks.
Pens. Place used Ozempic or Wegovy pens in an FDA-cleared sharps disposal container. If you do not have one, use a heavy-duty plastic container with a tight-fitting lid, such as a laundry detergent bottle. Label it "sharps" and bring it to a pharmacy or community sharps collection site. Many pharmacies accept used pens at no charge.
Vials and syringes. Place needles and syringes in the same sharps container. Empty vials can go in regular trash in most jurisdictions, but check local regulations. Some states require pharmaceutical waste to go through specific disposal channels.
Drug take-back programs. The DEA's National Prescription Drug Take Back Day, held twice yearly, accepts expired medications including injectables. Many pharmacies and hospitals also operate year-round take-back programs. The FDA maintains a searchable database of authorized collection sites.
Do not share expired semaglutide with others. Beyond the degradation and contamination risks, sharing prescription medications is illegal in most jurisdictions and medically inappropriate. Each patient requires individualized dosing, and a vial that has been in your refrigerator under your storage conditions cannot be verified as safe for someone else.
For broader safety considerations during treatment, see the peptide safety guide. If you experience side effects that persist after switching to a fresh product, consult how long do semaglutide side effects last.
What to Do If You Accidentally Used Expired Semaglutide
If you injected semaglutide that you later realized was past its expiration date, the appropriate response depends on how far past the date and the product's storage history.
Recently expired (1-4 weeks past date), properly stored. The most likely outcome is a slightly weaker dose. Most products retain greater than 85% potency for several weeks beyond their labeled date when stored correctly. No immediate medical action is needed, but switch to a fresh product for your next dose.
Significantly expired (2+ months past date) or improperly stored. Monitor for signs of infection at the injection site: increasing redness, warmth, swelling, or drainage over 24-72 hours. Mild soreness at the injection site is normal and not concerning. If you develop fever, spreading redness, or purulent discharge, seek medical attention. These signs could indicate bacterial contamination.
Visibly degraded (cloudy, discolored, particles). If you noticed the degradation only after injecting, monitor closely for allergic reactions over the next 4-6 hours: hives, facial swelling, difficulty breathing, or widespread rash. Aggregated peptide fragments carry the highest immunogenic risk. If you experience any of these symptoms, seek emergency care.
In all cases, discard the remaining product and open a new pen or vial. Do not continue drawing from a product you suspect is compromised. For ongoing dosing guidance, use the semaglutide dosage calculator. For information on how semaglutide clears your body, see how long does semaglutide stay in your system.
Frequently Asked Questions
How long does semaglutide last after reconstitution?
Reconstituted semaglutide lasts 28 days refrigerated at 2-8°C when mixed with bacteriostatic water. With sterile water (no preservative), use within 24-48 hours. After 28 days, potency drops below the 90% pharmaceutical threshold. Write the reconstitution date on the vial with a permanent marker to track the window accurately.
Can you use Ozempic after the expiration date?
Novo Nordisk does not recommend using Ozempic past its printed expiration date. Most pens retain greater than 90% potency for 2-4 weeks beyond the date if stored at 2-8°C, but this is not guaranteed. If the pen has been at room temperature longer than 56 days, discard it regardless of the printed date. Potency testing requires laboratory equipment unavailable to patients.
Does semaglutide go bad at room temperature?
Pre-filled pens tolerate room temperature (up to 30°C) for 56 days. Reconstituted semaglutide degrades rapidly at room temperature, losing 5-10% potency within 7 days and 26-38% within 14 days. Lyophilized powder lasts 6-12 months at room temperature. Always refrigerate reconstituted solutions and return them to the fridge within 60 seconds of drawing each dose.
How can you tell if semaglutide has gone bad?
Check for cloudiness, visible particles, color change (should be clear to slightly yellow), or unusual odor. Functionally, reduced appetite suppression or worsening blood glucose on a previously effective dose suggests potency loss. A 15-20% degradation produces measurable reductions in clinical effect. When in doubt, replace the product rather than risk subtherapeutic dosing.
Is expired semaglutide dangerous to inject?
Expired semaglutide from potency loss alone is not typically dangerous. The primary risk is microbial contamination in reconstituted vials stored beyond 28 days, which can cause injection site infections or sepsis. Aggregated peptide fragments may also trigger immune responses in 1-2% of users. Reduced efficacy, not toxicity, is the most common consequence of using expired product.
What is the difference between an expiration date and a beyond-use date for semaglutide?
Expiration dates on manufacturer products (Ozempic, Wegovy) are backed by 12-24 months of ICH-standard stability testing. Beyond-use dates on compounded semaglutide follow USP <797> standards and are often 6 months for lyophilized and 28-45 days for liquid formulations. The manufacturer date is more rigorously validated, but both should be respected as the outer limit for safe use.
How long can Ozempic stay out of the fridge?
An Ozempic pen can stay at room temperature (up to 30°C / 86°F) for 56 days after first use or first removal from refrigeration. After 56 days at room temperature, discard the pen even if medication remains. Do not expose the pen to temperatures above 30°C or direct sunlight. Never freeze the pen. The 56-day window does not reset if you return the pen to the fridge.
Should you freeze semaglutide to extend its shelf life?
Freeze lyophilized (powder) semaglutide at -20°C to extend shelf life from 18-24 months to 24-36 months. Never freeze liquid semaglutide, whether reconstituted or in a pre-filled pen. Ice crystals shear peptide bonds and cause irreversible aggregation, destroying 30-50% of active peptide in a single freeze-thaw cycle. This applies to Ozempic pens, Wegovy pens, and reconstituted vials alike.
The Bottom Line
Semaglutide expires across every form. Ozempic and Wegovy pens last until the printed date (typically two years) when refrigerated, and 56 days at room temperature after first use. Compounded vials carry a 6-month beyond-use date for lyophilized powder and 28-45 days for liquid formulations. Reconstituted semaglutide lasts 28 days refrigerated with bacteriostatic water.
Using expired semaglutide usually means reduced efficacy rather than danger. A vial or pen that has lost 15-20% potency delivers a lower effective dose, undermining weight loss or glycemic control. The exception is microbial contamination in reconstituted vials stored too long, which can cause infections requiring medical treatment.
Four habits protect your semaglutide: store cold, store dark, label dates, and use sterile technique. Use the semaglutide dosage calculator to plan precise doses and the peptide reconstitution calculator to mix accurately. For a broader overview of peptide storage principles, read the complete storage guide.
Related reading: - Does Semaglutide Need to Be Refrigerated?: complete storage rules by product form - How Long Does Bacteriostatic Water Last?: shelf life of your reconstitution solvent - How Long Does Semaglutide Take to Work?: week-by-week timeline to monitor during storage window - How Much Bacteriostatic Water for Semaglutide: mixing ratios and technique - Semaglutide Dosage Chart in mL: dosing across all concentrations - Does Tirzepatide Expire: comparison guide for the other major GLP-1 agonist - Compound Semaglutide With B12: shelf life and storage for B12-compounded formulations
Related Articles
Semaglutide Refrigeration Guide
Yes, semaglutide must be refrigerated at 2-8°C before first use. Ozempic pens last 56 days at room temp after opening. Compounded vials: 28 days.
What Happens If You Use Expired Semaglutide?
Expired semaglutide usually means a weaker dose, not a dangerous one. Real risks by timeframe: 1 week, 1 month, 3 months past expiry and signs.
Does Tirzepatide Expire? Shelf Life
Tirzepatide expires in 24-36 months as powder and 28 days once reconstituted. Learn storage temps, degradation signs, and potency loss timelines.
Does Semaglutide Cause Heartburn?
Semaglutide causes heartburn in 1.9-5% of users depending on dose. Learn the delayed gastric emptying mechanism, GERD vs heartburn differences, and 7 management strategies backed by clinical data.