You just received a 30 mL bottle of bacteriostatic water and you are not sure whether it belongs in the fridge or the medicine cabinet. Unopened bacteriostatic water is stable at controlled room temperature (20-25°C / 68-77°F). Once you puncture the rubber stopper with a needle, refrigerate the vial at 2-8°C (36-46°F) and use the contents within 28 days.
That 28-day rule is not arbitrary. It comes from USP Chapter 797, the United States Pharmacopeia standard governing sterile compounding. After 28 days, the preservative (0.9% benzyl alcohol) can no longer guarantee sterility in a multi-puncture vial, even under refrigeration.
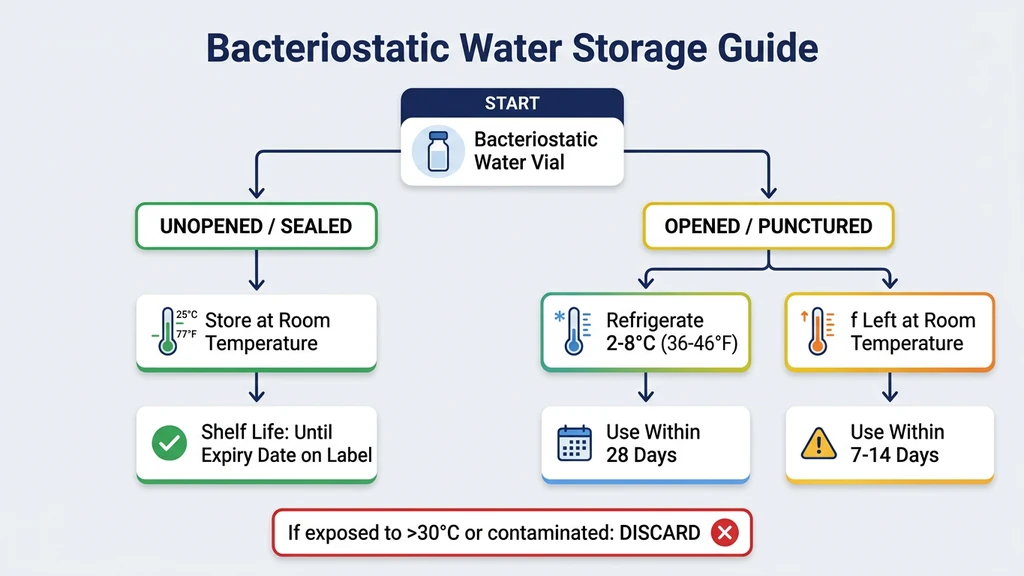
| Vial State | Storage Temperature | Shelf Life | Key Condition |
|---|---|---|---|
| Sealed, unopened | 20-25°C (room temp) | Until manufacturer expiration (1-3 years) | Away from light and heat |
| Sealed, unopened | 2-8°C (fridge) | Until manufacturer expiration | Acceptable but not required |
| Opened / punctured | 2-8°C (fridge) | 28 days from first puncture | Mark the date on the vial |
| Opened / punctured | 20-25°C (room temp) | 24-48 hours max | Discard if left out longer |
If you need help calculating how much bacteriostatic water to add to your peptide vial, use the reconstitution calculator. For complete peptide storage rules, see the peptide storage guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Why Bacteriostatic Water Needs Refrigeration After Opening
A sealed vial of bacteriostatic water is a closed system. No air enters. No bacteria enter. The 0.9% benzyl alcohol preservative has nothing to fight, and the water remains sterile indefinitely until the manufacturer expiration date.
The moment a needle punctures the stopper, that closed system breaks. Each puncture pushes a microscopic core of rubber aside and creates a channel, however brief, between the outside air and the sterile interior. Skin flora, airborne microbes, and particles on the needle tip all have a path in.
How Benzyl Alcohol Preserves Sterility
Benzyl alcohol disrupts bacterial cell membranes. At 0.9% concentration, it prevents the growth of most common contaminants: Staphylococcus epidermidis (skin flora), Escherichia coli, and Pseudomonas aeruginosa. It does not kill all organisms on contact. It inhibits replication, keeping bacterial counts below a dangerous threshold.
Think of it like salt in a jar of pickles. The salt does not destroy every microbe instantly. It creates an environment where bacteria cannot multiply fast enough to spoil the food. Benzyl alcohol does the same in water: it holds contamination in check rather than sterilizing on demand. The literal fact: benzyl alcohol is bacteriostatic (growth-inhibiting), not bactericidal (bacteria-killing). This distinction matters because it means the preservative can be overwhelmed.
At room temperature (20-25°C), bacteria that survive the preservative can still multiply slowly. At refrigerator temperature (2-8°C), that replication rate drops further, giving the benzyl alcohol a wider margin. Refrigeration and the preservative work together. Neither is sufficient alone in a punctured vial over multiple weeks (Akers et al., J Pharm Sci, 1984).
The USP 797 28-Day Rule
USP Chapter 797 sets standards for compounding sterile preparations in the United States. It classifies bacteriostatic water as a multi-dose container once the vial is punctured. The beyond-use date (BUD) for a multi-dose container stored at 2-8°C is 28 days after first entry.
This 28-day limit accounts for the cumulative microbial risk from repeated punctures, the slow degradation of benzyl alcohol over time, and the possibility that a single contamination event introduces organisms that grow gradually. After 28 days, the USP standard considers the risk-to-benefit ratio unacceptable for injectable use.
Compounding pharmacies and research facilities follow this rule strictly. For individual users reconstituting peptides at home, whether preparing semaglutide, tirzepatide, or BPC-157, the same principle applies. Write the date of first puncture on the vial with a marker. After 28 days, discard the remaining water and open a new vial. The cost of a fresh 30 mL vial (typically $3-8) does not justify the infection risk of using expired stock.
Opened vs. Unopened Storage: What Changes
The storage rules for bacteriostatic water split cleanly at one event: the first needle puncture. Everything before that puncture is simple. Everything after requires more discipline.
Unopened Vials: Room Temperature Is Fine
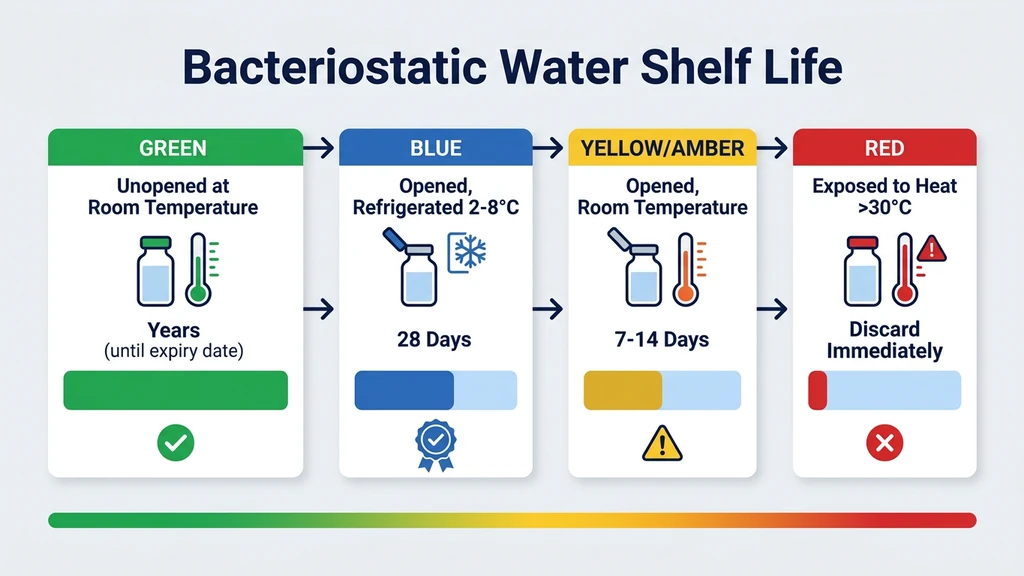
Sealed bacteriostatic water vials are stable at controlled room temperature (20-25°C). The USP defines this as the acceptable range, though brief excursions up to 40°C during shipping do not compromise the product. Most manufacturers assign a 2-to-3-year expiration date from the production date under these conditions.
You can also store unopened vials in the refrigerator if you prefer. Cold storage will not harm the product. Some users keep their entire supply in the fridge for convenience so that opened and unopened vials sit in the same location. This is a reasonable approach but not a requirement.
Avoid storing unopened vials in direct sunlight, on a windowsill, in a car, or near a heat source. While benzyl alcohol is chemically stable across a wide temperature range, sustained heat above 40°C accelerates any trace degradation and can compromise the stopper seal over months. A cool, dark cabinet is the ideal location for your unopened supply.
Opened Vials: Refrigerate Immediately
Once you draw from the vial for the first time, move it to the refrigerator. The back of the middle shelf provides the most consistent temperature (2-8°C). Avoid the fridge door, where temperature swings 5-10°C every time you open it.
Label the vial with the date of first puncture. Many users write directly on the vial with a fine-tip permanent marker. Others place a small adhesive label. The method does not matter. What matters is that 28 days from that date, you discard the vial.
If you leave a punctured vial at room temperature overnight (8-12 hours), the contents are likely still safe to use. Benzyl alcohol provides a buffer. But every hour at room temperature narrows that buffer. A punctured vial left on a countertop for 48 hours or more should be discarded. The $5 cost of replacement is trivial compared to the risk of injecting contaminated water into a peptide vial that you will then inject subcutaneously.
For guidance on handling your reconstituted peptide vials after mixing, see how long do reconstituted peptides last.
Temperature Sensitivity and What Kills Bacteriostatic Water
Bacteriostatic water is more resilient than the peptides it dissolves. Where a reconstituted peptide may lose 15% potency after 12 hours at room temperature, bacteriostatic water itself tolerates broader conditions. The risks are different: you are not worried about the water losing potency. You are worried about contamination and preservative failure.
Heat Exposure
Benzyl alcohol is stable up to approximately 60°C. Normal room temperature, shipping conditions, and brief exposure to warm environments pose no threat to the preservative.
Sustained storage above 40°C (104°F) creates two problems. First, bacterial growth accelerates. Organisms that benzyl alcohol suppresses at 25°C may overcome the preservative at 40°C because their replication rate increases faster than the preservative's inhibitory effect. Second, prolonged heat can degrade the rubber stopper, causing it to lose elasticity and seal integrity, especially after multiple punctures.
Practical scenarios: A vial left in a hot car for several hours during summer (interior temperatures reaching 50-70°C) should be discarded. A vial shipped in a package that sat on a porch on a warm day is fine, as internal package temperatures rarely exceed 40°C for the duration needed to cause harm.
Freezing: Avoid It
Do not freeze bacteriostatic water. Water expands approximately 9% when it freezes. In a glass vial, this expansion can crack the glass. In a plastic vial, it may deform the container and compromise the seal.
Freezing also drives benzyl alcohol out of uniform distribution. As ice crystals form, the benzyl alcohol concentrates in the remaining liquid fraction. When thawed, the distribution may not fully re-homogenize, leaving some portions under-preserved and vulnerable to contamination.
If a vial froze accidentally, inspect it after thawing. If the glass is intact and the stopper is undamaged, the water may still be usable. Swirl gently and allow it to reach refrigerator temperature before drawing. If you see cracks, leaks, or cloudiness, discard it.
Light Exposure
Benzyl alcohol is not significantly light-sensitive. Unlike certain peptide amino acids (tryptophan, tyrosine) that degrade under UV radiation, benzyl alcohol maintains its preservative function under normal indoor lighting and brief sun exposure.
Storing vials in a dark location is still best practice. UV light can degrade the rubber stopper over years and may promote trace chemical reactions in the water itself. A fridge, cabinet, or drawer provides sufficient light protection. There is no need to wrap bacteriostatic water vials in foil the way you would for light-sensitive peptides like MOTS-c or Selank.
Multi-Use Vial Protocol: Getting 28 Clean Days
A 30 mL vial of bacteriostatic water provides enough solvent for multiple peptide reconstitutions. Most users draw 1-3 mL per peptide vial, meaning a single bottle of bacteriostatic water can last through 10 to 30 reconstitutions. The challenge: keeping it sterile across all those draws.
Sterile Drawing Technique
1. Swab the stopper. Before every draw, wipe the rubber stopper with a fresh alcohol swab for 10 seconds. Allow it to air-dry for 5 seconds. Residual alcohol evaporates quickly and does not contaminate the water.
2. Use a new needle. Never reinsert a used needle into a bacteriostatic water vial. Each used needle carries bacteria from the previous injection site, your skin, or the peptide vial stopper. A fresh, sealed needle costs pennies and eliminates the primary contamination vector.
3. Draw only what you need. Pull the exact volume of water required for your reconstitution. Excess handling increases contamination risk. If you are reconstituting semaglutide, use the semaglutide mixing guide to determine your volume before opening anything. For tirzepatide, see the tirzepatide reconstitution guide.
4. Minimize stopper punctures. Each puncture creates a new micro-channel. After 15 to 20 punctures, the stopper may not reseal completely. Some users transfer bacteriostatic water into smaller sterile vials to limit punctures on the original container.
5. Return to the fridge immediately. Do not leave the vial on the counter while you complete your reconstitution. Draw the water, cap the vial, refrigerate it, then proceed to reconstitute your peptide.
Tracking Puncture Count and Expiration
A simple tracking system prevents ambiguity. Write two things on the vial the first time you puncture it:
- 1.The date (e.g., "Opened 03/14/26")
- 2.A tally of punctures (add a mark each time you draw)
When either condition is met, 28 days have passed or the puncture count exceeds 15, open a new vial. This dual-tracking approach is used in clinical settings and translates directly to home use.
Some users pre-draw their bacteriostatic water into individual syringes at the start. For example, if you know you need 2 mL for each of four peptide vials over the next month, you could draw four 2 mL syringes on day one, cap them, and store them in the fridge. This minimizes stopper punctures to a single session. The pre-drawn syringes remain sterile for the same 28-day window, provided they stay capped and refrigerated.
How Storage Affects Peptide Reconstitution Quality
Bacteriostatic water is a vehicle. Its job is to dissolve your peptide powder while keeping the solution sterile. When the water is compromised, the peptide solution inherits every problem.
Contaminated water introduces bacteria directly into your peptide vial. You will not see it. You will not smell it. Bacteria at low colony counts are invisible in a clear solution. But once injected subcutaneously, they encounter warm tissue with no benzyl alcohol barrier, and they multiply.
What Happens When You Use Compromised Water
Scenario 1: Water left at room temperature for 3 days after opening. You reconstitute BPC-157 with this water. The BPC-157 dissolves normally. The solution looks clear. But bacteria that multiplied during those 3 days are now in your peptide vial. Over the next week of daily injections, bacterial counts rise further. By day 7, you notice redness and warmth at your injection site. By day 10, you have a localized skin infection requiring antibiotics.
Scenario 2: Water used 45 days after opening, kept refrigerated. The benzyl alcohol has partially degraded. Bacterial inhibition is weaker. You use it to reconstitute GHK-Cu. The copper peptide solution develops slight cloudiness by day 5, earlier than its usual 21-28 day window. The cloudiness indicates bacterial growth or peptide aggregation triggered by contaminants. The vial is unusable.
Both scenarios cost more in wasted peptide, medical treatment, or lost research time than a $5 replacement vial. For detailed signs of peptide degradation, see how to store peptides.
Reconstitution Best Practices with Fresh Water
Start every reconstitution with water you trust. Check the date on the vial. Inspect the solution: bacteriostatic water should be perfectly clear and colorless. Any cloudiness, particulates, or color suggests contamination. Discard and open a new vial.
Once your water passes inspection, follow the standard reconstitution procedure. Aim the stream of water down the inside wall of the peptide vial, not directly onto the powder cake. Let gravity and gentle swirling dissolve the peptide. Never shake.
For peptide-specific reconstitution instructions: - How to reconstitute GHK-Cu - How to reconstitute tirzepatide - How much bacteriostatic water for semaglutide - How to take BPC-157
Use the reconstitution calculator to determine exact water volumes for your vial size and desired concentration.
Signs Your Bacteriostatic Water Is Contaminated
Contamination can be visible or invisible. Visible contamination is straightforward: you see something wrong and you discard the vial. Invisible contamination is the real danger, because you use the water assuming it is safe. Your best defense is strict adherence to the storage and handling rules above. Your second defense is knowing what to look for.
Visual Warning Signs
Cloudiness or haziness. Fresh bacteriostatic water is as clear as tap water. Any turbidity indicates microbial growth or particulate contamination. Hold the vial against a white background under bright light. Even faint haziness is grounds for disposal.
Floating particles. Fibers, specks, or sediment visible to the naked eye mean the seal has been compromised or a contaminated needle was used. These particles may be rubber fragments from a degraded stopper, fibers from a cotton swab, or microbial colonies large enough to see.
Color change. Bacteriostatic water should be colorless. A yellow, brown, or green tint indicates chemical degradation or biological contamination. Green is particularly concerning, as it suggests Pseudomonas aeruginosa, a common environmental bacterium.
Damaged stopper. Look at the rubber stopper under light. If you see visible tears, excessive puncture marks, or pieces of rubber floating inside, the seal is no longer reliable. Discard the vial.
When to Discard Even If It Looks Fine
Discard the vial under any of these conditions, regardless of appearance:
- More than 28 days since first puncture
- More than 15 punctures through the stopper
- Vial was left at room temperature for more than 48 hours after opening
- Vial was exposed to temperatures above 40°C after opening
- Vial was frozen and thawed
- You are unsure when it was first opened
Bacteriostatic water is inexpensive. A 30 mL vial costs less than a single dose of most research peptides. Replacing it is not wasteful. Using questionable water to reconstitute a $50-100 peptide vial is wasteful, because you risk destroying the peptide and your health in the process. For expiration timelines on specific peptides, see does tirzepatide expire and the peptide dosage chart for shelf life data across all 29 peptides.
Common Bacteriostatic Water Storage Mistakes
These errors account for the majority of contamination events in home peptide preparation. Each is preventable.
Reusing Needles Between Vials
A needle used to draw from a bacteriostatic water vial and then insert into a peptide vial is a one-way trip. That same needle should never return to the bacteriostatic water bottle. The peptide vial stopper, your fingers near the needle hub, and brief air exposure all deposit bacteria on the needle surface.
The fix: use a fresh needle for every draw from the bacteriostatic water vial. For a typical reconstitution, you need two needles: one to draw water, one to inject it into the peptide vial (or one needle used for both in sequence, then discarded). The cost is under $0.10 per needle. For BPC-157 injection guidance, this needle discipline is outlined step by step.
Skipping the Alcohol Swab
The rubber stopper on a bacteriostatic water vial sits exposed to ambient air. Dust, skin cells, and airborne bacteria settle on the surface between uses. Inserting a needle through an unswabbed stopper pushes surface contaminants directly into the sterile water.
One alcohol swab, 10 seconds of wiping, 5 seconds of air drying. This step alone prevents the most common contamination pathway in home reconstitution. Keep a box of individually wrapped alcohol prep pads next to your peptide storage area. If you are new to peptide preparation, the getting started with peptides guide covers all the supplies you need.
Ignoring the 28-Day Window
Some users keep a single vial of bacteriostatic water for months, drawing from it sporadically. "It still looks clear" is their justification. Clarity is not proof of sterility. Bacterial counts below 10,000 colony-forming units per milliliter are invisible to the naked eye but still represent a contamination risk for subcutaneous injection.
The USP 28-day limit exists precisely because visual inspection is unreliable. Clinical pharmacies perform microbial testing with culture plates. Home users do not have that option. The 28-day cutoff replaces laboratory testing with a time-based safety margin. Respect it.
Storing Near Heat Sources
Placing the vial on top of the refrigerator (warm), next to a stove, near a window, or in a bathroom cabinet where shower steam raises temperatures repeatedly is a slow-motion contamination setup. These locations expose the vial to temperature cycling between 25-40°C multiple times per day.
Stable, cold, dark. The back of the middle refrigerator shelf. That is the correct location for any opened vial of bacteriostatic water, just as it is the correct location for reconstituted peptides.
Bacteriostatic Water Storage FAQ
Frequently Asked Questions
Does unopened bacteriostatic water need to be refrigerated?
No. Unopened bacteriostatic water is stable at controlled room temperature (20-25°C) until the manufacturer's expiration date, typically 1-3 years. Refrigeration is acceptable but not required. Store in a cool, dark location away from direct sunlight and heat sources above 40°C.
How long does bacteriostatic water last after opening?
Bacteriostatic water lasts 28 days after the first needle puncture when stored at 2-8°C (refrigerated). This limit comes from USP Chapter 797 guidelines for multi-dose sterile containers. After 28 days, discard the vial regardless of remaining volume. At room temperature, a punctured vial should be discarded after 48 hours.
Can you use bacteriostatic water after 28 days?
Using bacteriostatic water beyond 28 days post-opening is not recommended. The 0.9% benzyl alcohol preservative loses effectiveness over time, and cumulative contamination from repeated needle punctures may exceed the preservative's capacity to inhibit bacterial growth. A 30 mL replacement vial costs $3-8.
What happens if bacteriostatic water is not refrigerated after opening?
Bacteria multiply faster at room temperature (20-25°C) than at refrigerator temperature (2-8°C). A punctured vial left unrefrigerated for 24-48 hours carries increased contamination risk. Using this water to reconstitute a peptide transfers those bacteria into the peptide solution, potentially causing injection site infections.
Can you freeze bacteriostatic water?
Avoid freezing bacteriostatic water. Water expands approximately 9% when frozen, which can crack glass vials. Freezing also causes benzyl alcohol to distribute unevenly in the solution. If accidentally frozen and the vial is intact, thaw at room temperature, swirl gently, and inspect for cloudiness before use.
How should you store bacteriostatic water for peptide reconstitution?
Before opening, store at room temperature in a cool, dark location. After the first needle puncture, refrigerate at 2-8°C on the back of the middle shelf. Label the vial with the date of first use. Swab the stopper with alcohol before each draw. Use a fresh needle every time. Discard after 28 days or 15 punctures.
Does bacteriostatic water go bad?
Sealed bacteriostatic water expires 1-3 years from manufacture, depending on the brand. Opened vials expire 28 days from first puncture. Signs of compromised water include cloudiness, floating particles, color changes (yellow, green, or brown tint), and a damaged or excessively punctured stopper. Clear water past the 28-day mark is still considered expired.
Is bacteriostatic water the same as sterile water?
No. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which allows multi-dose use over 28 days. Sterile water has no preservative and must be used within 24 hours of opening (single use only). For reconstituting peptides intended for multiple doses, always use bacteriostatic water. For reconstitution basics, see the reconstitution calculator.
The Bottom Line
Bacteriostatic water storage follows one dividing line: the first needle puncture. Before that puncture, store at room temperature and forget about it until you need it. After that puncture, refrigerate at 2-8°C and discard within 28 days.
The 0.9% benzyl alcohol preservative keeps bacteria in check but does not sterilize on demand. It needs the support of cold temperature, clean needles, alcohol-swabbed stoppers, and a hard 28-day limit. Skip any one of these steps and you transfer risk into every peptide vial you reconstitute with that water.
Mark the date. Refrigerate. Discard at 28 days. The cost of a fresh vial is a fraction of the cost of a wasted peptide or a preventable infection. For exact reconstitution volumes, use the reconstitution calculator. For complete peptide storage protocols, see the peptide storage guide. For shelf life data on reconstituted peptides, see how long do reconstituted peptides last.
Related articles: - How Long Does Bacteriostatic Water Last? — complete shelf life guide with USP 797 rules - Bacteriostatic Water vs Sterile Water — detailed comparison of the two diluent types - Where to Buy Bacteriostatic Water for Injection — sourcing guide with price comparison and red flags - How to Reconstitute Retatrutide — step-by-step mixing guide using bacteriostatic water - How Long Does Tirzepatide Last in the Fridge? — refrigeration timelines for reconstituted and lyophilized tirzepatide - How Long Do Semaglutide Side Effects Last? — clinical timelines for GI side effects during titration
Related Articles
How Long Does Bac Water Last?
Unopened bacteriostatic water lasts until manufacturer expiry (2-3 years). After puncture, it lasts 28 days refrigerated at 2-8°C per USP 797.
Bacteriostatic vs Sterile Water
Bacteriostatic water contains 0.9% benzyl alcohol for 28-day multi-use. Sterile water has no preservative and is single-use only.
Semaglutide Refrigeration Guide
Yes, semaglutide must be refrigerated at 2-8°C before first use. Ozempic pens last 56 days at room temp after opening. Compounded vials: 28 days.
Tirzepatide Fridge Storage Guide
Tirzepatide lasts until its expiry date unreconstituted at 2-8°C and 28 days once reconstituted. Refrigeration guide for Mounjaro pens and compounded vials.