You have a lyophilized peptide vial in one hand and two types of water on the counter: bacteriostatic water and sterile water. They look identical. Both are clear, colorless, and injectable. The label is the only thing that separates them, and that label determines whether your reconstituted peptide stays safe for weeks or becomes a contamination risk within hours.
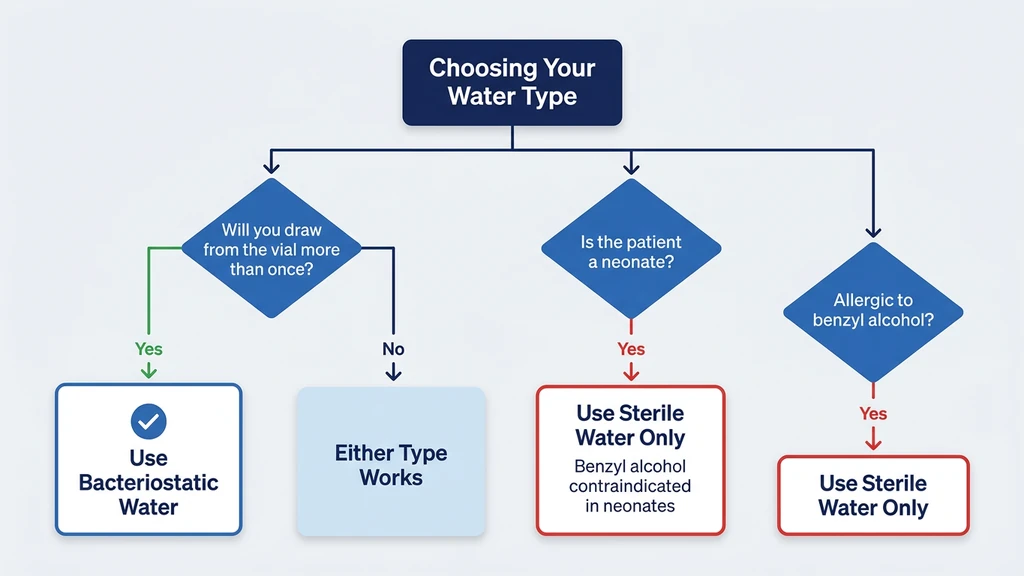
Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which inhibits bacterial growth and allows multiple draws from the same vial over 28 days. Sterile water contains no preservative. Once you puncture the vial, you must use the entire contents in a single draw and discard the remainder.

| Feature | Bacteriostatic Water | Sterile Water |
|---|---|---|
| Preservative | 0.9% benzyl alcohol | None |
| Multi-use | Yes, up to 28 days | No, single-use only |
| Storage after opening | Refrigerate at 2-8°C | Discard after single draw |
| Shelf life (sealed) | 1-3 years at room temp | 1-3 years at room temp |
| Shelf life (opened) | 28 days refrigerated | Use immediately |
| Peptide reconstitution | Preferred for multi-dose vials | Only for single-dose use |
| Neonatal safe | No (benzyl alcohol toxicity) | Yes |
| Cost per vial (30 mL) | $3-8 | $1-4 |
| USP classification | Multi-dose container | Single-dose container |
For most peptide users reconstituting semaglutide, tirzepatide, BPC-157, or any other research peptide intended for multiple doses, bacteriostatic water is the correct choice. Sterile water has specific, narrower use cases. The reconstitution calculator will help you determine exactly how much of either water type to add to your peptide vial.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Chemical Composition: What Makes Them Different
Both bacteriostatic water and sterile water start as the same product: purified water that has been sterilized to remove all microorganisms. The divergence happens at the last step of manufacturing. Bacteriostatic water receives a measured addition of benzyl alcohol. Sterile water receives nothing.
Bacteriostatic Water Composition
Bacteriostatic water for injection (BWFI) is USP-grade water containing 0.9% (9 mg/mL) benzyl alcohol. The benzyl alcohol serves one purpose: preventing bacterial replication inside the vial after the stopper is punctured.
Benzyl alcohol is a naturally occurring aromatic alcohol found in essential oils, fruits, and teas. At 0.9% concentration, it disrupts bacterial cell membranes by inserting into the lipid bilayer, increasing membrane fluidity until the cell can no longer maintain structural integrity (Nair, Int J Pharm, 2001). This mechanism is bacteriostatic, meaning it inhibits growth rather than killing bacteria outright. The distinction matters. If a large bacterial load enters the vial through a contaminated needle, benzyl alcohol may not eliminate it. It simply prevents a small inoculum from multiplying to dangerous levels.
The 0.9% concentration was chosen because it sits below the threshold for significant tissue irritation at subcutaneous injection volumes (typically 0.5-2 mL) while remaining effective against common environmental bacteria including Staphylococcus epidermidis, Escherichia coli, and Pseudomonas aeruginosa (Drapeau et al., J Pharm Sci, 2005).
Sterile Water Composition
Sterile water for injection (SWFI) is purified water and nothing else. No preservatives, no buffers, no antimicrobial agents. It meets USP standards for sterility at the time of manufacture, and that sterility is guaranteed only as long as the vial remains sealed.
The absence of a preservative creates a binary situation. Sealed, sterile water is as safe as bacteriostatic water. Punctured, it becomes a growth medium for any bacteria introduced through the needle. Water at room temperature with no antimicrobial agent is a near-ideal environment for bacterial replication. A single colony-forming unit introduced during a draw can multiply to millions within 24 hours at 25°C (Cundell et al., PDA J Pharm Sci Technol, 2002).
This is why USP Chapter 797 classifies sterile water vials as single-dose containers. One puncture, one draw, then discard the vial regardless of remaining volume.
How Benzyl Alcohol Preserves Sterility
Think of benzyl alcohol like chlorine in a swimming pool. A pool with chlorine can tolerate swimmers introducing bacteria from their skin, sweat, and hair. The chlorine holds contamination in check between cleanings. A pool with no chlorine becomes a bacterial breeding ground after the first swimmer enters.
The literal fact: benzyl alcohol at 0.9% concentration maintains bacterial counts below infectious thresholds across 28 days and approximately 15 needle punctures, provided the vial is stored at 2-8°C. At room temperature (20-25°C), this timeline shortens because bacteria replicate faster in warmer environments, narrowing the preservative's margin of control (Akers et al., J Pharm Sci, 1984).
The preservative is not indestructible. It slowly degrades through oxidation and evaporation over weeks. After 28 days, the USP standard no longer guarantees sufficient preservative concentration to maintain the bacteriostatic effect. This is why the 28-day rule exists for opened vials.
When to Use Bacteriostatic Water
Bacteriostatic water is the standard diluent for any injectable product that will be drawn from more than once. In the peptide community, this covers nearly every use case. A reconstituted peptide vial is typically used over days or weeks, with each dose requiring a separate needle puncture.
Peptide Reconstitution (Multi-Dose Vials)
Most peptides ship as lyophilized (freeze-dried) powder in 3 mL or 5 mL vials. You add bacteriostatic water to dissolve the powder, then draw individual doses from the reconstituted vial over the following days or weeks.
Common peptides that require bacteriostatic water for reconstitution:
- BPC-157: 5 mg vials, typically 20-40 daily doses per vial
- Semaglutide: 3-5 mg vials, weekly dosing for 4-12 weeks per vial. See how much bacteriostatic water for semaglutide for exact volumes.
- Tirzepatide: 5-15 mg vials, weekly dosing for multiple weeks. See the tirzepatide reconstitution guide.
- TB-500: 5 mg vials, 2-3 draws per week during loading phase
- GHK-Cu: 50-200 mg vials, daily dosing for 4-12 weeks. See how to reconstitute GHK-Cu.
- Retatrutide: weekly dosing from compounded vials. See the retatrutide reconstitution guide.
In every case above, the reconstituted vial will be punctured multiple times. Without the 0.9% benzyl alcohol preservative, each draw introduces a contamination risk that accumulates with no antimicrobial defense. Bacteriostatic water is not optional for multi-dose use. It is the only safe choice.
HCG Reconstitution
Human chorionic gonadotropin (HCG) is reconstituted with bacteriostatic water for the same reason as peptides: the vial is used over multiple doses. A typical HCG protocol involves drawing 250-500 IU every 2-3 days from a 5,000 or 10,000 IU vial. That translates to 10-20 punctures per vial over 20-40 days.
Many HCG kits ship with a separate ampule of sodium chloride diluent or sterile water. If the included diluent contains no preservative, transfer the reconstituted HCG into a sterile vial and add bacteriostatic water instead, or use bacteriostatic water from the start. The kit diluent is adequate only if you plan to use the entire vial in a single session.
Once reconstituted with bacteriostatic water, HCG is stable for 28 days at 2-8°C, the same rule that applies to all multi-dose bacteriostatic water preparations. For proper storage guidelines, see how to store peptides.
HGH Reconstitution
Human growth hormone (HGH) vials contain 4-12 IU per vial and are drawn from daily. A 10 IU vial at a dose of 2 IU per day lasts 5 days, requiring 5 separate draws. Bacteriostatic water is the appropriate diluent.
Some pharmaceutical HGH products (Genotropin, Norditropin) come with proprietary diluent cartridges that already contain a preservative (often metacresol rather than benzyl alcohol). These should be used as supplied. Compounded or generic HGH without a proprietary diluent system should always be reconstituted with bacteriostatic water.
Reconstituted HGH with bacteriostatic water maintains potency for approximately 21-28 days at 2-8°C. At room temperature, HGH degrades faster than the water's preservative expires. Refrigeration is essential. For shelf life data across all peptides, see the peptide dosage chart.
When to Use Sterile Water
Sterile water has a smaller but critical set of use cases. In each, the absence of benzyl alcohol is a feature, not a limitation.
Single-Dose Preparations
If you are reconstituting a peptide vial that you will use entirely in one session, sterile water is acceptable. There is no need for a preservative in a solution that will be drawn and injected within minutes.
Practical examples: reconstituting a 2 mg vial of a peptide for a single intramuscular injection, dissolving a lyophilized medication in a hospital setting for immediate IV administration, or preparing a single-dose subcutaneous injection.
Sterile water costs less than bacteriostatic water ($1-4 vs. $3-8 per 30 mL vial), so it saves money when multi-use capability is unnecessary. However, most peptide users work with multi-dose vials. If you are uncertain, bacteriostatic water is always the safer default.
Neonatal and Pediatric Applications
Benzyl alcohol is toxic to neonates (newborns under 28 days old). In 1982, the FDA documented a series of neonatal deaths linked to intravenous flush solutions containing benzyl alcohol. The preservative accumulates in neonatal tissue because newborns lack the enzyme alcohol dehydrogenase needed to metabolize it efficiently. The resulting "gasping syndrome" includes metabolic acidosis, respiratory depression, seizures, and cardiovascular collapse (Gershanik et al., N Engl J Med, 1982).
The FDA subsequently required that all injectable products containing benzyl alcohol carry a warning against neonatal use. This is the primary clinical reason sterile water exists as a separate product category. In neonatal intensive care units and pediatric settings, sterile water is used exclusively for reconstitution and dilution.
For adult peptide users, this restriction is not relevant. A 70 kg adult metabolizes benzyl alcohol from a 1-2 mL subcutaneous injection without measurable accumulation (Nair, Int J Pharm, 2001). The daily benzyl alcohol exposure from a typical peptide injection (0.9-1.8 mg) is far below the 5 mg/kg/day threshold associated with adverse effects in adults.
Certain Vaccine Formulations
Some vaccines are reconstituted with sterile water rather than bacteriostatic water because the vaccine is administered as a single dose from a single vial. The MMR vaccine (measles, mumps, rubella) and varicella vaccine are common examples.
Additionally, certain live attenuated vaccines may be incompatible with benzyl alcohol, which could reduce viral viability. Vaccine manufacturers specify the exact diluent for each product, and substituting bacteriostatic water for sterile water (or vice versa) without manufacturer guidance is not recommended.
This is a specialized use case. For peptide users, vaccine reconstitution protocols are managed by healthcare providers and do not affect personal peptide preparation decisions.
Benzyl Alcohol Sensitivity
A small percentage of adults are sensitive or allergic to benzyl alcohol. Symptoms include localized redness, swelling, and itching at the injection site that exceeds normal injection-site irritation. True anaphylactic reactions to benzyl alcohol are extremely rare but have been documented (Wilson et al., Ann Allergy Asthma Immunol, 2005).
If you suspect benzyl alcohol sensitivity, test with a small subcutaneous injection of bacteriostatic water (0.1 mL) without any peptide. If the site develops significant redness or a raised welt within 30 minutes, you may need to reconstitute with sterile water and use the entire vial in a single session. Alternatively, discuss sodium chloride 0.9% (normal saline) with a healthcare provider as a preservative-free diluent option.
For users with confirmed benzyl alcohol sensitivity, switching to sterile water means you will need multiple smaller vials (3 mL or 5 mL) rather than a single 30 mL vial. Each vial is a single-use event. This increases cost and waste but eliminates the preservative exposure.
Shelf Life and Storage: Side-by-Side Comparison
The storage rules for both water types follow the same dividing line as bacteriostatic water alone: sealed vs. punctured. The difference is what happens after the first needle puncture.
Sealed Vial Storage
Both bacteriostatic water and sterile water, when sealed, are stable at controlled room temperature (20-25°C) until the manufacturer's expiration date. This is typically 1-3 years from the date of manufacture.
| Condition | Bacteriostatic Water | Sterile Water |
|---|---|---|
| Room temperature (20-25°C) | Stable until expiration | Stable until expiration |
| Refrigerated (2-8°C) | Acceptable, not required | Acceptable, not required |
| Avoid | Temps above 40°C, direct sunlight | Temps above 40°C, direct sunlight |
| Freezing | Not recommended (uneven benzyl alcohol distribution) | Not recommended (vial cracking risk) |
Store sealed vials of either type in a cool, dark cabinet. Both tolerate shipping temperatures and brief heat exposure without degradation. Prolonged storage above 40°C can compromise rubber stopper integrity over months.
Opened Vial Storage
This is where the two products diverge sharply.
Bacteriostatic water (opened): Refrigerate at 2-8°C. Use within 28 days of the first puncture. Label the vial with the date of first use. Discard after 28 days or 15 punctures, whichever comes first. For full storage guidance, see does bacteriostatic water need to be refrigerated.
Sterile water (opened): Use immediately. Discard the remaining contents. There is no "storage after opening" protocol because storage after opening is not safe. Any remaining sterile water in a punctured vial should be discarded within 1 hour.
The cost difference makes this clear. A 30 mL vial of bacteriostatic water at $5 provides enough diluent for 10-30 peptide reconstitutions over 28 days. Achieving the same with sterile water requires 10-30 individual single-use vials, costing $10-120 depending on vial size.
For guidance on how long your reconstituted peptides stay potent, see how long do reconstituted peptides last.
Contamination Risks: What Happens When You Use the Wrong Water
Using sterile water in place of bacteriostatic water for a multi-dose peptide vial is one of the most common preparation mistakes in the peptide community. The consequences are predictable and preventable.
Scenario 1: Sterile Water in a Multi-Dose Peptide Vial
You reconstitute a 5 mg vial of BPC-157 with 2 mL of sterile water instead of bacteriostatic water. The BPC-157 dissolves normally. The solution looks clear. You draw your first 250 mcg dose (0.1 mL) and refrigerate the vial.
Day 1-3: The solution appears fine. No visible changes. You inject 250 mcg daily.
Day 5-7: Bacteria introduced during the first draw have been replicating without any preservative to slow them down. At 2-8°C, bacterial doubling time is slower than at room temperature (12-24 hours instead of 20-30 minutes), but replication still occurs. After 5 days, bacterial counts in the vial may exceed 10,000 CFU/mL, the threshold at which contamination becomes clinically relevant for injectable preparations (USP Chapter 71).
Day 10-14: You notice slight cloudiness in the solution. This indicates bacterial growth has reached visible levels (approximately 1,000,000 CFU/mL). By this point, every injection from the vial has delivered bacteria directly into your subcutaneous tissue. The likely outcome: injection site infection requiring antibiotics. The wasted BPC-157 ($30-50 per vial) is the lesser cost.
The fix: always use bacteriostatic water for any vial you plan to draw from more than once. If you already reconstituted with sterile water by mistake, draw the entire volume into pre-loaded syringes and store them capped and refrigerated for no more than 48 hours.
Scenario 2: Bacteriostatic Water for a Neonatal Preparation
A pharmacy technician reconstitutes an IV antibiotic with bacteriostatic water instead of sterile water for a 3 kg premature infant. The 0.9% benzyl alcohol enters the bloodstream at a dose that an adult would clear in hours.
The neonate's immature liver cannot metabolize benzyl alcohol efficiently. The preservative accumulates, causing metabolic acidosis, respiratory depression, and central nervous system toxicity. This scenario has caused documented deaths (Gershanik et al., N Engl J Med, 1982), which led to the FDA's 1982 warning against benzyl alcohol in neonatal products.
This scenario is specific to neonatal medicine. For adult peptide users, the benzyl alcohol exposure from subcutaneous injections of 0.5-2 mL of bacteriostatic water is metabolized without complication. The concern applies only to newborns and, in some guidelines, children under 2 years old weighing less than 10 kg.
Scenario 3: Expired Bacteriostatic Water Used for Reconstitution
You find a vial of bacteriostatic water that was opened 6 weeks ago. It has been refrigerated. The solution looks clear. You decide to use it because "it still looks fine."
The benzyl alcohol concentration has declined below the 0.9% threshold through oxidation and repeated exposure to air during draws. Bacterial counts in the water itself may be subclinical (invisible to the naked eye) but present. You reconstitute tirzepatide with this water. The bacteria transfer into the peptide vial. Over the next 7 days, bacterial growth in the tirzepatide solution accelerates because the already-weakened preservative is now diluted further.
By day 5 of injections, you develop redness, warmth, and tenderness at your injection site. A course of oral antibiotics costs $30-50 and interrupts your dosing schedule. The expired bacteriostatic water cost $0 (it was already in the fridge). A new vial would have cost $5.
The rule: discard opened bacteriostatic water after 28 days. Always. For sourcing fresh vials, see where to buy bacteriostatic water for injection.
Which Peptides Require Which Water Type
The choice between bacteriostatic water and sterile water depends on how the reconstituted peptide will be used, not on the peptide itself. Any peptide drawn from more than once requires bacteriostatic water.
Peptides That Always Need Bacteriostatic Water
Any peptide administered in daily or multiple-weekly doses from a single reconstituted vial requires bacteriostatic water. This includes the majority of peptides used for research purposes:
| Peptide | Typical Dosing | Draws Per Vial | Water Type |
|---|---|---|---|
| BPC-157 | 250-500 mcg daily | 10-40 | Bacteriostatic |
| TB-500 | 2.5-5 mg, 2-3x/week | 4-12 | Bacteriostatic |
| Semaglutide | 0.25-2.4 mg weekly | 4-20 | Bacteriostatic |
| Tirzepatide | 2.5-15 mg weekly | 4-12 | Bacteriostatic |
| GHK-Cu | 1-4 mg daily | 12-50 | Bacteriostatic |
| Ipamorelin | 200-300 mcg, 2-3x daily | 20-60 | Bacteriostatic |
| CJC-1295 | 100-300 mcg, 2-3x/week | 8-24 | Bacteriostatic |
| Retatrutide | 1-12 mg weekly | 4-12 | Bacteriostatic |
| Selank | 250-500 mcg daily | 10-40 | Bacteriostatic |
| Semax | 200-600 mcg daily | 10-30 | Bacteriostatic |
The pattern is simple: if the vial holds more than one dose, use bacteriostatic water. For specific reconstitution volumes, use the reconstitution calculator.
When Sterile Water Is Acceptable for Peptides
Sterile water is acceptable only when the entire reconstituted volume will be used in a single session. Practical scenarios:
- 1.Single-use research preparations. You reconstitute a 2 mg peptide vial, draw the full volume, and inject it in one dose. No remaining solution to store.
- 1.Pre-loaded syringe protocols. You reconstitute with sterile water, immediately draw the entire volume into individual syringes, cap and label each syringe, and discard the empty vial. The pre-loaded syringes should be used within 24-48 hours.
- 1.Hospital or clinical settings. IV medications reconstituted for immediate infusion. The vial is punctured once, the full volume is drawn, and the vial is discarded per standard protocol.
For home peptide reconstitution, scenario 2 is the only practical use of sterile water. Most users find it more convenient and safer to use bacteriostatic water and draw from the vial as needed over 28 days. For beginners learning the reconstitution process, see getting started with peptides.
Cost Comparison and Practical Sourcing
The price difference between bacteriostatic water and sterile water is small per vial but compounds significantly when sterile water is used incorrectly (one vial per draw instead of one vial per month).
Price Per Vial
A 30 mL vial of bacteriostatic water costs $3-8 from reputable suppliers. This single vial provides enough diluent for 10-30 peptide reconstitutions, depending on how much water each peptide requires. A typical user reconstituting one peptide per week (2 mL per reconstitution) uses 8 mL per month from a single 30 mL vial. At $5 per vial, the cost per reconstitution is $0.17.
Sterile water costs $1-4 per single-use vial (typically 5-10 mL). If you reconstitute one peptide per week and discard the sterile water vial after each use, the monthly cost is $4-16 for 4 vials. At the higher end, this is 3x the cost of a single bacteriostatic water vial that would last the entire month.
For a comprehensive guide to sourcing quality bacteriostatic water, see where to buy bacteriostatic water for injection.
Total Cost Over a Typical Peptide Protocol
Consider a 12-week semaglutide protocol with weekly reconstitution from a multi-dose vial:
With bacteriostatic water: - 1-2 vials of bacteriostatic water (30 mL each): $5-10 total - Each vial covers 4+ weeks of weekly draws
With sterile water (incorrect multi-dose use): - Contamination risk starts after the first draw - Potential infection requiring antibiotics: $30-50 for the prescription, plus lost peptide ($50-200 per vial) - A single contamination event costs more than a year's supply of bacteriostatic water
With sterile water (correct single-dose use): - 12 individual sterile water vials: $12-48 - Additional syringes for pre-loading: $5-10 - Total: $17-58 vs. $5-10 for bacteriostatic water
The math is unambiguous. Bacteriostatic water costs less and provides better contamination protection. The only reason to choose sterile water is a medical contraindication (benzyl alcohol sensitivity or neonatal use).
For help converting units and calculating doses accurately, use the peptide unit converter.
Safety Considerations and Special Populations
Both water types are pharmaceutical-grade products with strong safety profiles when used correctly. The safety concerns center on misuse: wrong water type for the application, expired products, or contaminated preparation technique.
Benzyl Alcohol Daily Exposure Limits
The FDA sets a maximum recommended daily intake of benzyl alcohol at 5 mg/kg of body weight for adults. For a 70 kg person, this is 350 mg per day.
A typical peptide injection uses 0.5-2 mL of bacteriostatic water. At 0.9% (9 mg/mL), this delivers 4.5-18 mg of benzyl alcohol per injection. Even at 3 injections per day (an aggressive peptide stacking protocol), total daily benzyl alcohol exposure is 13.5-54 mg, well below the 350 mg limit for a 70 kg adult.
For context: a single glass of wine contains approximately 10-20 mg of benzyl alcohol as a natural component of the fermentation process. The amount delivered by peptide injections is comparable to dietary exposure.
Users running multiple peptides simultaneously should still calculate their total daily benzyl alcohol intake. If you are stacking 3 or more peptides with separate reconstituted vials, see the peptide stacking guide for protocol design that minimizes total injection volume. The peptide interaction checker can help identify any combination concerns.
Populations That Should Avoid Benzyl Alcohol
Neonates (birth to 28 days): Absolute contraindication. Use only sterile water or preservative-free sodium chloride for any injectable preparation.
Premature infants: Higher risk than full-term neonates due to even more immature hepatic enzyme systems. No benzyl alcohol exposure in any form.
Children under 2 years / under 10 kg: Some guidelines extend the benzyl alcohol restriction to this age group, though the evidence for toxicity in older infants is less clear than for neonates. Consult a pediatrician.
Pregnant women (high-dose IV exposure): Standard subcutaneous peptide injection volumes of bacteriostatic water do not deliver benzyl alcohol at doses associated with fetal toxicity. However, many peptides themselves are contraindicated during pregnancy. If you are pregnant, peptide use should be discussed with your healthcare provider.
Adults with documented benzyl alcohol allergy: Rare but real. Switch to sterile water with single-use protocols or preservative-free normal saline.
For a broader safety overview covering all peptide use, see the peptide safety guide.
Injection Site Reactions: Bacteriostatic vs. Sterile Water
Benzyl alcohol causes a mild stinging sensation at the injection site in some users. This is not an allergic reaction. The alcohol irritates nerve endings briefly upon injection. The sensation fades within 30-60 seconds.
Sterile water injections sting more, not less. Water without any solute is hypotonic relative to body fluids, meaning it creates an osmotic gradient that causes cells at the injection site to swell. This swelling triggers a sharper, more prolonged sting than the brief benzyl alcohol burn (Arendt-Nielsen et al., Br J Anaesth, 1994).
Some users switch from bacteriostatic water to sterile water thinking they will reduce injection site discomfort. The opposite is usually true. If injection site irritation is a concern, the better solution is proper injection technique (slower injection speed, pinching the skin, rotating sites) rather than changing diluent type. For injection technique guidance, see the peptide injections guide.
Common Mistakes When Choosing Between the Two
These errors account for the most frequent bacteriostatic water vs. sterile water problems reported in peptide communities.
Using Sterile Water for Multi-Dose Peptide Vials
The most dangerous mistake. A user reconstitutes tirzepatide with sterile water and plans to draw weekly doses for 4-8 weeks. By the second or third draw, bacteria from the environment or needle surface are replicating in a preservative-free solution. The user sees no cloudiness because bacterial counts below 1,000,000 CFU/mL are invisible to the naked eye. Each subsequent injection delivers an increasing bacterial load.
The fix: always use bacteriostatic water for any vial you will draw from more than once. Write "BAC" on the bacteriostatic water vial with a marker if you keep both types in the same storage area. For proper reconstitution technique, see how to reconstitute tirzepatide.
Confusing Bacteriostatic Water with Normal Saline
Bacteriostatic water (0.9% benzyl alcohol, no sodium chloride) and bacteriostatic normal saline (0.9% sodium chloride plus 0.9% benzyl alcohol) are different products. Plain bacteriostatic water is hypotonic. Bacteriostatic normal saline is isotonic.
For subcutaneous peptide injections, bacteriostatic water is the standard diluent. Bacteriostatic normal saline is used primarily for IV flush lines and medication dilution in clinical settings. Using bacteriostatic normal saline for peptide reconstitution is not harmful, but the added sodium chloride may affect the solubility or stability of certain peptides. Stick with plain bacteriostatic water unless a peptide manufacturer specifically recommends saline.
Reusing Sterile Water Vials
Some users puncture a sterile water vial, draw what they need, and save the rest for later. This is the worst-case contamination scenario. Sterile water has no defense against bacteria introduced through the needle. The remaining water in that vial is no longer sterile.
If you use sterile water, treat each vial as a single event. Puncture, draw the full contents, discard the vial. Any remaining volume is waste. This is the intended use outlined in USP Chapter 797. If wasting water bothers you, buy smaller vials (3-5 mL) that match your reconstitution volume, or switch to bacteriostatic water and avoid the problem entirely.
Storing Both Types Together Without Labels
Bacteriostatic water and sterile water vials look nearly identical. Both contain clear, colorless liquid. Both have similar vial sizes and rubber stoppers. The label is the only differentiator.
Keep the two types in separate locations. Store bacteriostatic water in the refrigerator (where it belongs after opening). Store unopened sterile water vials in a cabinet. If you must keep both in the same area, mark bacteriostatic water vials with colored tape or a marker. Grabbing the wrong vial for a multi-dose peptide reconstitution has real consequences.
Bacteriostatic Water vs Sterile Water FAQ
Frequently Asked Questions
Can I use sterile water instead of bacteriostatic water for peptides?
Only if you use the entire reconstituted vial in a single draw. Sterile water has no preservative, so bacteria introduced through the needle will replicate freely. For multi-dose peptide vials (BPC-157, semaglutide, tirzepatide), always use bacteriostatic water. A 30 mL vial costs $3-8 and lasts 28 days.
What is the difference between bacteriostatic water and sterile water?
Bacteriostatic water contains 0.9% benzyl alcohol, a preservative that inhibits bacterial growth for 28 days after opening. Sterile water contains no preservative and must be discarded after a single use. Both are pharmaceutical-grade purified water. The benzyl alcohol is the only chemical difference.
Is bacteriostatic water safe to inject?
Yes. Bacteriostatic water is USP-grade and approved for subcutaneous, intramuscular, and intravenous injection in adults. The 0.9% benzyl alcohol preservative is metabolized by the liver. A typical 1 mL injection delivers 9 mg of benzyl alcohol, far below the FDA's 350 mg/day limit for a 70 kg adult. The one exception: never use it for neonates.
Why is benzyl alcohol bad for babies?
Neonates lack sufficient alcohol dehydrogenase enzyme activity to metabolize benzyl alcohol. The preservative accumulates in their tissues, causing gasping syndrome: metabolic acidosis, respiratory depression, seizures, and potentially death. This toxicity was documented in a 1982 FDA investigation (Gershanik et al., N Engl J Med, 1982). Always use preservative-free sterile water for neonatal injections.
How long does bacteriostatic water last after opening?
28 days when refrigerated at 2-8°C, per USP Chapter 797 guidelines. After 28 days, the benzyl alcohol preservative may have degraded below effective concentration. Discard the vial regardless of remaining volume. A new 30 mL vial costs $3-8. For full storage rules, see the bacteriostatic water refrigeration guide.
Can I use bacteriostatic water for HCG?
Yes. Bacteriostatic water is the preferred diluent for HCG when the vial will be used for multiple doses. A 5,000 IU vial drawn at 250 IU every 2-3 days requires 10-20 punctures over 20-40 days. Without the benzyl alcohol preservative, contamination risk increases with each draw. Reconstituted HCG stays potent for 28 days at 2-8°C.
Does sterile water expire?
Sealed sterile water expires 1-3 years from manufacture, similar to bacteriostatic water. Once opened (punctured), sterile water should be used immediately and the vial discarded. There is no post-opening shelf life because no preservative exists to prevent bacterial contamination. Never store a punctured sterile water vial for later use.
Can I mix bacteriostatic water and sterile water together?
Technically yes, but doing so dilutes the benzyl alcohol concentration below 0.9%, reducing its preservative effectiveness. If you mix 1 mL of bacteriostatic water with 1 mL of sterile water, the resulting solution has only 0.45% benzyl alcohol, which may not inhibit bacterial growth reliably over 28 days. Use one type or the other, not both.
The Bottom Line
Bacteriostatic water and sterile water differ by one ingredient: 0.9% benzyl alcohol. That single ingredient determines whether your reconstituted peptide stays safe for 28 days of multi-dose use or becomes a contamination risk after a single draw.
For peptide reconstitution, the rule is straightforward. If the vial will be punctured more than once, use bacteriostatic water. If the entire volume is drawn in a single session, sterile water is acceptable. For nearly all home peptide protocols involving BPC-157, semaglutide, tirzepatide, or any other multi-dose peptide, bacteriostatic water is the correct choice.
Sterile water has its place: neonatal medicine, single-dose preparations, and users with documented benzyl alcohol sensitivity. Outside those categories, bacteriostatic water provides better safety at lower total cost. A $5 vial of bacteriostatic water protects every reconstituted peptide you prepare for 28 days. Use the reconstitution calculator to determine exact water volumes for your vial size and desired concentration. For sourcing quality bacteriostatic water, see where to buy bacteriostatic water for injection. For proper storage after opening, follow the bacteriostatic water storage guide.
Related articles: - How Long Does Bacteriostatic Water Last? - complete shelf life guide with the 28-day USP 797 rule - Is Reconstitution Solution the Same as Bacteriostatic Water? - comparing all diluent categories including normal saline and manufacturer diluents - How to Store Peptides - complete storage protocols for lyophilized and reconstituted peptides - How Long Do Reconstituted Peptides Last? - shelf life data for all major peptides after mixing - How to Inject BPC-157 - injection technique guide covering reconstitution and sterile handling - Does Semaglutide Need to Be Refrigerated? - storage rules for Ozempic, Wegovy, and compounded semaglutide
Related Articles
Bacteriostatic Water Refrigeration
Unopened bacteriostatic water stores at room temperature. Once opened or punctured, refrigerate at 2-8°C and use within 28 days.
Reconstitution Solution vs Bac Water
Reconstitution solution is a broad category. Bacteriostatic water is one type. Compare bac water, sterile water, and saline for peptide mixing.
How Long Does Bac Water Last?
Unopened bacteriostatic water lasts until manufacturer expiry (2-3 years). After puncture, it lasts 28 days refrigerated at 2-8°C per USP 797.
CJC-1295 vs Sermorelin: Half-Life, Dosing, and GH Output Compared
CJC-1295 vs sermorelin: half-life (6-8 days vs 10-20 min), GH output (2-10x vs physiologic pulse), cost, FDA status, and who should choose which. PubMed clinical data with decision framework.