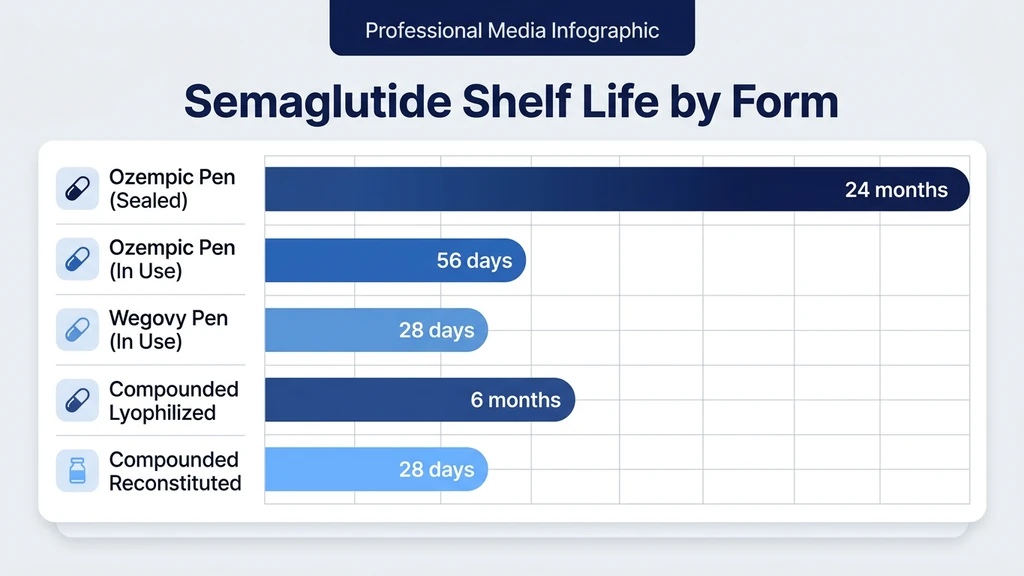
You picked up your semaglutide from the pharmacy, or a compounded vial arrived in an insulated mailer, and you need to know where it goes. Yes, semaglutide must be refrigerated at 2-8°C (36-46°F) before first use. After first use, the rules depend on the form: a brand-name Ozempic pen can stay at room temperature (up to 30°C / 86°F) for 56 days; a Wegovy pen gets 28 days at room temp; a compounded semaglutide vial lasts 28 days refrigerated at 2-8°C once reconstituted.
Semaglutide is a 31-amino-acid GLP-1 receptor agonist with a fatty acid side chain that extends its half-life to roughly 7 days. That side chain makes the molecule more stable than most peptides, but it does not make it immune to heat, light, or freezing. Proper storage protects the protein's tertiary structure, which is the folded shape that lets it bind its receptor and produce the appetite-suppressing, glucose-lowering effects you are paying for.

| Semaglutide Form | Before First Use | After First Use | Max Duration After Opening |
|---|---|---|---|
| Ozempic pen (1.34 mg/mL or 2.68 mg/mL) | Refrigerate 2-8°C | Room temp (up to 30°C) or fridge | 56 days |
| Wegovy pen (all strengths) | Refrigerate 2-8°C | Room temp (up to 30°C) or fridge | 28 days |
| Compounded lyophilized vial (pre-reconstitution) | Refrigerate 2-8°C or freezer | N/A | Until manufacturer expiration |
| Compounded vial (reconstituted) | N/A | Refrigerate 2-8°C | 28 days |
| Compounded pre-mixed liquid vial | Refrigerate 2-8°C | Refrigerate 2-8°C | 28 days after first draw |
If you need help calculating your dose in units or milliliters, use the semaglutide dosage calculator. For mixing compounded semaglutide with bacteriostatic water, see how much bacteriostatic water for semaglutide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Why Semaglutide Requires Refrigeration
Semaglutide is a protein. Proteins hold their function through a specific three-dimensional fold. Heat disrupts hydrogen bonds and hydrophobic interactions that maintain that fold. When the fold breaks, the molecule denatures, and a denatured semaglutide molecule cannot bind the GLP-1 receptor on pancreatic beta cells or hypothalamic neurons.
The fatty acid chain (a C-18 octadecandioic acid linker) that Novo Nordisk attached to the peptide backbone gives semaglutide an unusually long half-life by allowing it to bind albumin in the bloodstream. That same chain also provides modest thermal stability compared to shorter peptides like BPC-157 or GHK-Cu. But "more stable than most peptides" still means "fragile compared to a tablet of ibuprofen."
What Happens at the Molecular Level When Storage Fails
Three degradation pathways threaten semaglutide outside the recommended temperature range.
Deamidation. The asparagine residues in the peptide chain react with water at elevated temperatures, converting to aspartate. This changes the molecule's charge profile and reduces receptor binding affinity. At 37°C, deamidation rates increase roughly 3-fold compared to 5°C (Wakankar & Borchardt, J Pharm Sci, 2006).
Oxidation. Methionine residues in the semaglutide sequence are vulnerable to oxidation, particularly under light exposure or elevated temperature. Oxidized semaglutide shows reduced potency in preclinical binding assays (Li et al., Pharm Res, 1995).
Aggregation. Denatured semaglutide molecules clump together, forming visible or sub-visible particles. Aggregated protein cannot bind its receptor. Worse, injecting aggregated protein increases the risk of injection site reactions and, in rare cases, immunogenic responses where the body produces antibodies against the peptide (Rosenberg, AAPS J, 2006).
Think of it like an egg. A raw egg is functional: it can be fertilized, it has structure, it serves its biological purpose. Heat it to 70°C and the proteins denature irreversibly. You cannot "un-cook" an egg, and you cannot "un-denature" semaglutide. The damage is permanent. Once the molecule unfolds, refrigerating it again does not restore its shape. The literal fact: protein denaturation caused by sustained heat exposure is irreversible for injectable peptide formulations.
Storage Rules by Semaglutide Form
The correct storage protocol depends on whether you have a brand-name pen, a compounded lyophilized vial, or a compounded pre-mixed liquid. Each form has a different formulation, different excipients, and different stability data.
Ozempic Pens (Novo Nordisk)
Before first use: Refrigerate at 2-8°C. The pen ships cold and should go directly into the refrigerator. Ozempic pens carry a manufacturer expiration date, typically 24 months from production when stored continuously at 2-8°C.
After first use: You may keep the pen at room temperature (not exceeding 30°C / 86°F) or return it to the fridge. Each pen contains multiple doses, and Novo Nordisk designed the formulation with preservatives (phenol and polysorbate 80) that maintain sterility and stability for 56 days after first injection.
The 56-day window starts when you first attach a needle and inject. Mark the date on the pen. After 56 days, discard the pen even if solution remains. This timeline comes from Novo Nordisk's stability data submitted to the FDA as part of the Ozempic NDA (Novo Nordisk, Ozempic Prescribing Information, 2017).
Cap the pen after every use. The cap protects the solution from light. Polysorbate 80 in the formulation reduces aggregation, but prolonged UV exposure still degrades the peptide.
Wegovy Pens (Novo Nordisk)
Before first use: Refrigerate at 2-8°C. Same as Ozempic.
After first use: Room temperature (up to 30°C) or refrigerator, but only for 28 days. Wegovy pens are single-dose prefilled injectors at higher concentrations (0.25 mg through 2.4 mg), and the shorter stability window reflects the different formulation and the single-use design. Most patients use the entire Wegovy pen in one injection, so the 28-day window primarily matters if you remove the pen from the fridge and do not inject immediately.
Practical note: If you take your Wegovy pen out of the fridge in the morning and inject that evening, you are well within the safety margin. The 28-day limit covers situations where the pen sits in a travel bag or medicine cabinet for extended periods before use. For side effect management during Wegovy titration, see how long do semaglutide side effects last.
Compounded Lyophilized (Freeze-Dried) Semaglutide
Compounded semaglutide from 503B pharmacies often arrives as a white lyophilized powder in a glass vial. This is the most temperature-sensitive form at the point of reconstitution, but the most stable form for long-term storage.
Before reconstitution: Refrigerate at 2-8°C. Some pharmacies ship lyophilized semaglutide with instructions to store at -20°C (freezer) for extended shelf life. Lyophilized peptides tolerate freezing because the water has already been removed. No ice crystals form, and the protein structure is locked in a glassy matrix of stabilizing excipients (typically mannitol or trehalose). Check the pharmacy's label for the specific expiration date, which can range from 6 months to 2 years from compounding.
After reconstitution: Refrigerate at 2-8°C. Use within 28 days. This follows the same USP 797 guidelines that govern bacteriostatic water storage after opening. Once you add bacteriostatic water to the powder, you create a liquid solution with a preservative (0.9% benzyl alcohol from the bacteriostatic water). That preservative holds sterility for 28 days at refrigerator temperature.
For exact mixing volumes, use the reconstitution calculator or follow the semaglutide mixing guide.
Compounded Pre-Mixed Liquid Semaglutide
Some compounding pharmacies ship semaglutide already dissolved in a buffered solution, typically at concentrations of 2.5 mg/mL or 5 mg/mL. These vials arrive cold and must stay cold.
Before first draw: Refrigerate at 2-8°C continuously. The pharmacy's beyond-use date (BUD) on the label applies. Common BUDs range from 30 to 90 days from compounding, depending on the pharmacy's stability testing.
After first needle puncture: Continue refrigerating. The vial is now a multi-dose container subject to the 28-day rule from first puncture, identical to the rule for bacteriostatic water. If the pharmacy's BUD falls before 28 days from your first puncture, follow the shorter of the two dates.
Never leave compounded liquid semaglutide at room temperature. Unlike Ozempic pens, which contain preservatives and excipients optimized for room-temperature stability, compounded liquid formulations may lack polysorbate 80 or other aggregation inhibitors. Room-temperature storage of compounded vials has not been validated by the stability testing that Novo Nordisk performed for Ozempic.
Before vs. After First Use: What Changes
The storage rules split at one event: first use. For pens, "first use" means the first injection. For vials, "first use" means the first needle puncture through the rubber stopper.
Why Unopened Semaglutide Is More Stable
A sealed pen or sealed vial is a closed sterile system. No oxygen enters. No bacteria enter. No additional light penetrates beyond what hits the outer packaging. The manufacturer's expiration date, which can be 18-24 months for Ozempic and Wegovy, reflects stability under these sealed conditions at 2-8°C.
Lyophilized powder in a sealed vial under vacuum or inert gas (nitrogen) is even more stable. Without water, deamidation cannot occur. Without oxygen, oxidation slows to near zero. A properly sealed lyophilized semaglutide vial stored at -20°C to 2-8°C can maintain potency for years, which is why compounding pharmacies use lyophilization as their primary stabilization strategy.
Why First Use Starts the Clock
Opening the system introduces three threats simultaneously.
Oxygen exposure. Every time you uncap a pen or puncture a vial stopper, ambient air (21% oxygen) contacts the solution. Dissolved oxygen accelerates methionine oxidation in the peptide.
Microbial exposure. Bacteria from the needle, your skin, and the air have a path into the solution. Preservatives (phenol in Ozempic, benzyl alcohol in bacteriostatic water) inhibit but do not eliminate microbial growth. The inhibition weakens over time, which is why every semaglutide product has a finite beyond-use window after first use.
Mechanical stress. Attaching and removing needles from a pen, or repeated punctures through a vial stopper, creates micro-channels and agitation. Agitation promotes protein aggregation, especially in formulations without polysorbate 80 as a surfactant.
These three factors compound over time. The 28-day and 56-day limits account for their cumulative effect. Extending beyond those limits introduces risk that no preservative system was designed to manage. For general peptide storage principles, see how to store peptides.
Temperature Excursions: What to Do When Storage Goes Wrong
Real life does not always cooperate with pharmaceutical storage requirements. Packages sit on porches. Refrigerators malfunction. Pens travel in bags during summer flights. Knowing which temperature excursions are survivable and which are not can save you from discarding good medication or injecting degraded product.
Semaglutide Left Out of the Fridge Overnight
A single overnight at room temperature (20-25°C) is unlikely to cause meaningful degradation for any form of semaglutide. Ozempic pens are explicitly rated for room temperature up to 30°C for 56 days. Compounded vials tolerate brief excursions less well, but 8-12 hours at 22°C falls within the transient excursion range permitted by USP 797.
Action: Return to the fridge. Continue using. No need to discard.
A compounded vial left out for 24-48 hours at room temperature is more concerning. If the ambient temperature stayed below 25°C, the solution is likely intact. Above 25°C, or if you are unsure of the duration, err on the side of replacement. Semaglutide is expensive, but injecting degraded peptide wastes the dose entirely. You lose both the medication cost and the week of treatment.
Semaglutide Exposed to Heat (Above 30°C)
Sustained exposure above 30°C accelerates all three degradation pathways. The higher the temperature and the longer the exposure, the greater the potency loss.
In a hot car (interior: 50-70°C during summer): Discard. Even 30 minutes at 50°C can cause irreversible aggregation. A pen left in a car's glove compartment on a July afternoon is no longer reliable.
On a porch in summer (30-40°C for several hours): Inspect the solution. If clear and colorless, it may still be usable, but potency loss of 5-15% is plausible depending on duration. If cloudy, discard immediately. For Ozempic pens, Novo Nordisk's stability data does not cover exposures above 30°C, so there is no manufacturer guarantee of potency after such events.
Shipped without cold packing: Reputable pharmacies ship semaglutide with ice packs or gel packs in insulated containers. If your package arrived warm with no cold packs, or the cold packs were fully thawed and warm to the touch, contact the pharmacy. Most will replace the shipment. Do not inject semaglutide that arrived at ambient temperature during summer months without cold chain protection.
Semaglutide That Froze
Freezing is more dangerous to liquid semaglutide than moderate heat. When water freezes, ice crystals form at the molecular interface between the protein and the solvent. These crystals mechanically shear the protein's tertiary structure, causing irreversible denaturation and aggregation (Bhatnagar et al., J Pharm Sci, 2007).
Ozempic or Wegovy pens that froze: Discard. Novo Nordisk states this explicitly in the prescribing information. A frozen pen may look normal after thawing, but the solution likely contains aggregated protein that will not bind the GLP-1 receptor effectively and may provoke injection site reactions.
Compounded liquid semaglutide that froze: Discard. Same rationale. Ice crystal damage to dissolved proteins is mechanical, not chemical, and is not reversible by thawing.
Lyophilized semaglutide that was stored in the freezer: This is fine. Lyophilized powder contains no water, so no ice crystals form. Freezer storage (-20°C) actually extends the shelf life of lyophilized peptides. Many compounding pharmacies recommend freezer storage for their lyophilized products. The critical distinction: freezing a dry powder is safe, freezing a liquid solution is destructive.
To prevent accidental freezing, store pens and liquid vials on the middle shelf of the refrigerator, not against the back wall where the cooling element can create sub-zero pockets. This is the same guidance that applies to reconstituted peptides and bacteriostatic water.
Traveling with Semaglutide: Keeping It Cold on the Move
Semaglutide's weekly dosing schedule means most users will travel with it at some point. Maintaining the cold chain during travel is straightforward with the right equipment.
Short Trips (1-3 Days)
Ozempic pens: Room temperature is fine for trips under 3 days. The pen is rated for 56 days at up to 30°C. Place it in your carry-on bag (never checked luggage, where cargo holds can reach extreme temperatures or drop below freezing at altitude). Keep the cap on. If outdoor temperatures exceed 30°C, wrap the pen in a cool towel or place it in a small insulated pouch.
Compounded vials: Use a small insulated case with one gel ice pack. Pharmacy travel cases designed for insulin work well. Place a cloth or paper towel between the ice pack and the vial to prevent direct contact, which could freeze the solution. The vial should feel cool to the touch, not ice-cold.
Carry a letter from your prescriber. TSA and international security allow injectable medications with proper documentation. A copy of your prescription or a letter on clinic letterhead prevents delays at checkpoints. For unit-to-mg conversions your prescriber may reference, see how many mg is 40 units of semaglutide.
Longer Trips (4+ Days)
Hotel refrigerators are the simplest solution. Most hotel minibars or mini-fridges maintain 4-8°C, which is within range. Place the pen or vial on the middle shelf, not in the freezer compartment. If the fridge has no temperature display, test it by placing a glass of water inside for an hour. If the water is cold but not forming ice, the temperature is acceptable.
Dedicated medical coolers with phase-change gel packs (such as the FRIO wallet or MedActiv cooler) maintain 2-8°C for 24-48 hours without ice. These are reusable, TSA-compliant, and designed specifically for insulin and GLP-1 agonist pens.
Do not use dry ice. Dry ice (solid CO2 at -78.5°C) will freeze your semaglutide solution within minutes. Standard wet ice in a cooler is safer. Wrap the vial or pen in a waterproof bag to prevent water contact.
Crossing time zones does not affect dosing timing enough to matter. Semaglutide's half-life is approximately 168 hours (7 days). Shifting your injection day by 1-2 days to accommodate travel has minimal impact on steady-state plasma levels (Kapitza et al., Clin Pharmacokinet, 2015). For a full pharmacokinetic breakdown, see how long does semaglutide stay in your system.
Signs Your Semaglutide Has Degraded
Degraded semaglutide wastes your dose. You inject on schedule, wait for appetite suppression or blood glucose control, and nothing happens. Recognizing degradation before injection saves you a wasted week and a wasted dose.
Visual Inspection
Cloudiness or turbidity. Semaglutide solution should be clear and colorless. Any haziness indicates protein aggregation. Hold the pen or vial against a white surface under bright light. Even faint cloudiness is grounds for disposal. Do not confuse tiny air bubbles (which move when you tilt the container) with particulate matter (which stays suspended or sinks).
Visible particles. Fibers, specks, or flakes in the solution indicate contamination or advanced aggregation. This is more common in compounded vials than brand-name pens. Never inject a solution with visible particles.
Color change. Fresh semaglutide solution is colorless to very slightly yellow. A darker yellow, brown, or green tint signals chemical degradation. Discard the vial or pen immediately.
Frosted or cracked pen cartridge. If the glass cartridge inside an Ozempic pen appears frosted or has visible cracks, the pen froze at some point. Discard it.
Functional Signs of Lost Potency
Sometimes the solution looks perfect but the semaglutide has partially degraded. Partial degradation is harder to detect because it reduces potency by 20-50% rather than eliminating it entirely. Signs include:
Reduced appetite suppression. You notice increased hunger compared to previous weeks at the same dose. This is distinct from the natural adaptation that occurs during steady-state dosing. If appetite returns suddenly rather than gradually, storage failure is a likely cause.
Higher fasting blood glucose. Patients using semaglutide for type 2 diabetes may notice fasting glucose readings climbing 10-20 mg/dL above their established baseline on the same dose.
Increased injection site reactions. Aggregated protein triggers stronger local immune responses. If you experience unusual redness, swelling, or itching at the injection site with a vial or pen that has been in use for several weeks, the protein may have aggregated. This is distinct from normal injection site irritation, which typically occurs early in treatment and resolves. For guidance on expected side effects during titration, see does semaglutide cause nausea and does semaglutide cause fatigue.
Common Semaglutide Storage Mistakes
These errors account for preventable potency loss and wasted medication. Each is easy to avoid once you know the risk.
Storing the Pen with the Needle Attached
Leaving a needle screwed onto an Ozempic pen between injections creates a continuous air channel between the solution and the outside environment. Air enters through the needle tip. Bacteria follow. Pressure changes from temperature fluctuations can push solution out through the needle or draw air bubbles in.
Novo Nordisk instructs users to remove and discard the needle after every injection. Attach a new needle only when you are ready to inject. This takes 10 seconds and eliminates the primary contamination vector for pen-based semaglutide. The getting started with peptides guide covers needle handling in detail.
Placing the Vial Against the Fridge Back Wall
The back wall of most refrigerators sits directly against the cooling coil. Temperatures there can drop to -2°C to 0°C, below the freezing point of semaglutide solution. A compounded vial placed against the back wall may partially freeze overnight, denaturing the protein along the glass-solution interface without forming a visible ice block.
Place vials on the middle shelf, 5-10 centimeters from the back wall. Use the door shelves only for items that tolerate temperature swings. Semaglutide is not one of them.
Using a Vial Past 28 Days Because It Looks Clear
Clarity does not equal sterility or potency. A vial of reconstituted semaglutide at day 35 may look identical to day 1 under visual inspection. But cumulative deamidation, oxidation, and microbial growth below the visible threshold have been progressing since reconstitution.
The 28-day limit for compounded vials is not conservative. It is the validated window within which preservative efficacy and peptide stability overlap. Beyond that point, every injection carries increasing risk of reduced potency and contamination. At semaglutide's cost per vial, the financial incentive to stretch a vial is real. Resist it. A partial dose from degraded semaglutide delivers less weight loss or glucose control than a fresh dose at the correct potency.
Skipping the Alcohol Swab on Compounded Vials
The rubber stopper on a compounded semaglutide vial collects dust, skin cells, and airborne bacteria between draws. Each unswabbed puncture pushes surface contaminants into the solution. Over 4 weeks of twice-weekly draws, that adds up to 8 contamination opportunities.
Swab the stopper with a fresh alcohol prep pad for 10 seconds before every draw. Let it air dry for 5 seconds. This single step prevents the most common contamination pathway in home peptide use. For a complete sterile technique guide, see how to take BPC-157, which covers the same injection hygiene principles that apply to semaglutide.
Drawing Multiple Doses Into Syringes in Advance
Some users pre-draw several weeks of semaglutide doses into individual syringes to avoid repeated vial punctures. This practice introduces more problems than it solves. Semaglutide adsorbs to syringe barrel surfaces (particularly polypropylene) over days to weeks, reducing the actual dose delivered. Insulin syringes are designed for immediate use, not long-term storage. The silicone lubricant on the plunger can leach into the solution over extended contact.
Draw each dose immediately before injection. Use a fresh needle for each draw. Return the vial to the fridge within 60 seconds. This protocol, combined with the semaglutide dosage chart in mL, ensures accurate dosing every week.
Semaglutide Storage FAQ
Frequently Asked Questions
Does semaglutide need to be refrigerated before first use?
Yes. All forms of semaglutide require refrigeration at 2-8°C (36-46°F) before first use. This includes Ozempic pens, Wegovy pens, compounded lyophilized vials, and compounded liquid vials. Lyophilized (freeze-dried) vials can also be stored in the freezer at -20°C for extended shelf life. Never store liquid semaglutide in the freezer.
How long can semaglutide be out of the fridge?
Ozempic pens: up to 56 days at room temperature (below 30°C) after first use. Wegovy pens: up to 28 days at room temperature after first use. Compounded liquid vials should not be stored at room temperature. A brief overnight excursion at 20-25°C is unlikely to cause significant degradation for any form, but return to the fridge promptly.
What happens if semaglutide is not refrigerated?
Sustained storage above 30°C accelerates deamidation, oxidation, and aggregation of the semaglutide protein. Potency decreases over time. A pen or vial stored at room temperature (20-25°C) for a few hours is fine. A pen left in a hot car (50-70°C) for 30 minutes or more should be discarded. Degraded semaglutide delivers reduced appetite suppression and glucose control.
Can you freeze semaglutide?
Do not freeze liquid semaglutide (pens or reconstituted vials). Ice crystals mechanically shear the protein structure, causing irreversible denaturation and aggregation (Bhatnagar et al., J Pharm Sci, 2007). Lyophilized (freeze-dried) semaglutide powder can be stored in the freezer at -20°C because it contains no water and therefore forms no ice crystals.
How long does compounded semaglutide last in the fridge?
Reconstituted compounded semaglutide lasts 28 days in the fridge at 2-8°C from the date of reconstitution or first needle puncture. Unreconstituted lyophilized powder lasts until the pharmacy's expiration date, typically 6-24 months at 2-8°C. Pre-mixed liquid vials follow the pharmacy's beyond-use date, commonly 30-90 days, but only 28 days after first puncture.
Does Ozempic go bad if left out overnight?
No. A single overnight at room temperature (8-12 hours at 20-25°C) will not degrade Ozempic significantly. The pen is rated for 56 continuous days at up to 30°C after first use. Return it to the fridge in the morning if you prefer refrigerated storage, or continue room-temperature storage and track the 56-day window from first injection.
How should you store semaglutide while traveling?
For trips under 3 days, Ozempic pens need no cold packing below 30°C. For longer trips or compounded vials, use an insulated case with a gel ice pack. Place a cloth between the ice pack and the vial to prevent freezing. Never pack semaglutide in checked luggage. Carry it in your carry-on with a prescription letter. Hotel refrigerators (4-8°C) work for extended stays.
What does degraded semaglutide look like?
Degraded semaglutide solution may appear cloudy, contain visible particles or fibers, or show a yellow-to-brown color change. However, partial degradation (20-50% potency loss) produces no visible changes. If appetite suppression weakens suddenly at a stable dose, or injection site reactions increase after weeks of use, temperature-related degradation is a likely cause.
The Bottom Line
Semaglutide storage reduces to three rules matched to your product form. Refrigerate at 2-8°C before first use, regardless of form. After first use, Ozempic pens tolerate room temperature for 56 days, Wegovy pens for 28 days, and compounded vials must stay refrigerated for a maximum of 28 days. Never freeze liquid semaglutide.
The molecule is more resilient than shorter peptides like BPC-157 or Selank, thanks to its fatty acid side chain and commercial formulation excipients. But resilience has limits. Heat above 30°C, freezing, and time beyond the validated window all cause irreversible protein damage that no amount of re-refrigeration can fix.
Mark the date of first use on the pen or vial. Check the solution for clarity before every injection. Discard at the manufacturer or USP limit, not when it "looks bad." For dosing calculations, use the semaglutide dosage calculator. For reconstitution volumes, see how much bacteriostatic water for semaglutide. For complete peptide storage protocols across all 29 peptides, see the peptide storage guide.
Related articles: - Does Semaglutide Expire? — shelf life data for Ozempic pens, compounded vials, and lyophilized powder - How Long Does Bacteriostatic Water Last? — 28-day shelf life rule for your reconstitution solvent - Bacteriostatic Water vs Sterile Water — choosing the right diluent for semaglutide reconstitution - How Long Does Semaglutide Take to Work? — week-by-week timeline to set expectations during storage-sensitive titration - Not Losing Weight on Semaglutide? - troubleshooting guide including storage-related potency loss - How Long Do Reconstituted Peptides Last? - shelf life data for all reconstituted peptides - Compound Semaglutide With B12 — storage requirements for B12-enhanced compounded semaglutide
Related Articles
Does Semaglutide Expire? Storage
Semaglutide expires: Ozempic pens last until the label date (typically 2 years), compounded vials 6 months, reconstituted vials 28 days refrigerated.
Tirzepatide Fridge Storage Guide
Tirzepatide lasts until its expiry date unreconstituted at 2-8°C and 28 days once reconstituted. Refrigeration guide for Mounjaro pens and compounded vials.
Why Is My Semaglutide Red? Color Guide
Red or pink semaglutide means vitamin B12 was added by a compounding pharmacy. Brand Ozempic/Wegovy is always clear. Learn when color signals degradation.
How to Travel With Semaglutide: TSA Rules, Cold Chain, Packing & International Tips
Complete guide to traveling with semaglutide. TSA rules for Ozempic pens, cold chain packing, international customs regulations, hotel storage, and time zone dosing adjustments.