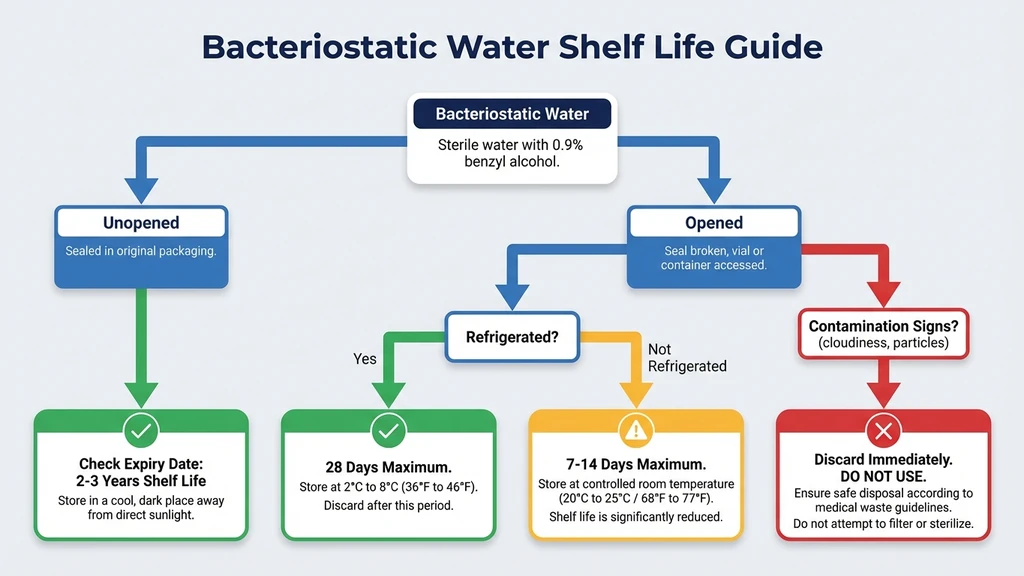
You are staring at a vial of bacteriostatic water in your fridge, trying to remember when you first stuck a needle through the stopper. Unopened bacteriostatic water lasts until the manufacturer's expiration date, typically 2 to 3 years from production. Once punctured, it lasts 28 days when refrigerated at 2-8°C (36-46°F). That 28-day limit comes from USP Chapter 797, the federal sterile compounding standard that governs every pharmacy in the United States.
The difference between "safe to use" and "time to discard" hinges on a single event: the first needle puncture. Before that moment, the sealed vial is a closed sterile system. After it, the clock starts.
| Condition | Shelf Life | Notes |
|---|---|---|
| Sealed, unopened, room temp (20-25°C) | 2-3 years (until expiry) | Store in a cool, dark location |
| Sealed, unopened, refrigerated (2-8°C) | 2-3 years (until expiry) | Acceptable but not required |
| Opened/punctured, refrigerated (2-8°C) | 28 days from first puncture | USP 797 beyond-use date |
| Opened/punctured, room temp (20-25°C) | Discard after 24-48 hours | Bacterial growth risk too high |
| Frozen | Do not freeze | Expansion cracks vials, disrupts preservative |
| Exposed to heat above 40°C | Discard immediately | Preservative effectiveness compromised |
If you are reconstituting a peptide and need exact water volumes, use the reconstitution calculator. For broader storage protocols, see the peptide storage guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
The USP 797 28-Day Rule Explained
USP Chapter 797 is the United States Pharmacopeia standard for compounding sterile preparations. It applies to hospitals, compounding pharmacies, and any facility that prepares injectable products. The chapter classifies any multi-dose vial, including bacteriostatic water, as having a beyond-use date (BUD) of 28 days after the first entry into the container, provided it is stored at 2-8°C.
This is not a suggestion. Compounding pharmacies that violate USP 797 face FDA enforcement action. The 28-day limit exists because controlled studies show that repeated needle punctures introduce cumulative microbial contamination that eventually overwhelms the preservative system (Motamedifar & Nehad, J Res Med Sci, 2014).
Why 28 Days and Not 30 or 60
The 28-day figure comes from challenge testing. Researchers inoculate multi-dose vials with known quantities of bacteria (Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans) and measure how long the preservative suppresses growth under refrigerated conditions. At 0.9% benzyl alcohol concentration and 2-8°C storage, bacterial counts remain below the infectious threshold for approximately 28 days after initial puncture. Beyond that point, the cumulative effect of preservative degradation and repeated microbial introductions pushes contamination risk above acceptable limits.
The USP chose 28 days as a conservative boundary with built-in safety margin. Some vials may remain sterile at day 35 or day 40. But "may remain sterile" is not a standard you want governing an injectable product. The 28-day cutoff replaces guesswork with a hard rule (USP General Chapter 797, Pharmaceutical Compounding: Sterile Preparations, 2023 revision).
Does the 28-Day Rule Apply to Home Use
USP 797 technically governs licensed compounding facilities, not individual users. No regulatory body inspects your refrigerator. But the science behind the rule does not change based on where the vial sits. Bacterial contamination follows the same kinetics in a home fridge as it does in a pharmacy refrigerator.
Home environments introduce additional risk factors that clinical settings control for: unfiltered air, higher ambient bacterial loads, less consistent refrigerator temperatures, and the absence of laminar flow hoods during draws. If anything, the 28-day limit is more important at home than in a pharmacy. Treat it as your hard stop. Write the date of first puncture on the vial with a permanent marker. When day 28 arrives, open a new vial.
For users reconstituting semaglutide or tirzepatide at home, this rule applies to every vial of bacteriostatic water you use.
Unopened Bacteriostatic Water: How Long It Really Lasts
A sealed vial of bacteriostatic water is a stable product with a long shelf life. No air enters, no bacteria enter, and the 0.9% benzyl alcohol preservative has nothing to fight. The water remains sterile from the manufacturing date until the printed expiration, typically 24 to 36 months later.
Manufacturer Expiration Dates
Most bacteriostatic water manufacturers stamp an expiration date 2 to 3 years from the date of production. Hospira (now Pfizer), a major producer, assigns 36-month shelf lives to its 30 mL multi-dose vials. Smaller compounding suppliers often use 24 months as a conservative estimate.
These dates assume storage at controlled room temperature: 20-25°C (68-77°F), with excursions permitted to 15-30°C per USP General Chapter 659 (Packaging and Storage Requirements). The expiration date printed on the vial is your maximum. There is no reason to use bacteriostatic water past this date, and no test you can run at home to verify it is still good.
You can store unopened vials in the refrigerator if you prefer. Cold storage does not harm the product and some users keep their entire supply in one location for convenience. Refrigeration is acceptable but not required for sealed vials.
Can Unopened Bacteriostatic Water Go Bad
Sealed bacteriostatic water can degrade under extreme conditions even before the printed expiration date. Three scenarios cause premature failure:
Sustained heat above 40°C. A vial stored in a hot garage, left in a car during summer, or placed near a radiator can experience temperatures that stress the rubber stopper seal. At 50-60°C, rubber elasticity decreases and micro-gaps can form. The water itself tolerates heat, but the closure system may not.
Direct UV exposure over months. Prolonged sunlight on a windowsill can degrade the rubber stopper and, over many weeks, catalyze trace photochemical reactions in the solution. The benzyl alcohol itself is not significantly light-sensitive, but the packaging system is.
Physical damage to the vial or seal. A cracked glass vial, a dislodged aluminum crimp cap, or a stopper pushed inward during shipping all compromise sterility immediately. Inspect every vial before first use. If the seal looks disturbed, return or discard it.
Under normal storage conditions (room temperature, away from light, intact packaging), unopened bacteriostatic water remains safe and effective until the manufacturer's printed expiry date.
Opened Bacteriostatic Water: The 28-Day Countdown
The moment you push a needle through the rubber stopper, two things change. First, you introduce a potential contamination pathway. Second, you start the 28-day beyond-use clock. Everything about how you handle the vial from this point forward determines whether it remains safe for the full 28 days.
What Happens Inside the Vial After Puncture
Each needle puncture creates a micro-channel through the rubber stopper. The stopper is designed to reseal after the needle withdraws, and it does, but not perfectly. Microscopic amounts of air, skin flora from the needle surface, and environmental particles enter with each draw.
Think of it like a screen door. A screen door keeps most insects out of your house, but a few get through every time you open it. Over a month of opening and closing, you will find insects inside. The rubber stopper works similarly. Each puncture lets a few organisms through. Benzyl alcohol kills most of them. But over 28 days and multiple punctures, the cumulative bacterial load can reach a threshold where the preservative is no longer sufficient.
The literal fact: benzyl alcohol is bacteriostatic (inhibits growth), not bactericidal (kills on contact). It slows bacterial replication enough to keep colony counts below dangerous levels, but it cannot sterilize a progressively contaminated solution indefinitely (Dempsey, Am J Health Syst Pharm, 2008).
Why Refrigeration Extends the Window to 28 Days
At room temperature (20-25°C), common contaminants like Staphylococcus epidermidis double their population every 20 to 30 minutes under ideal conditions. Benzyl alcohol at 0.9% suppresses this replication, but bacteria that survive the preservative still multiply, just slowly.
At refrigerator temperature (2-8°C), that doubling time extends dramatically. Cold slows enzymatic reactions inside bacterial cells, reducing their metabolic rate and reproductive capacity. The benzyl alcohol does not become more potent at lower temperatures. Instead, the bacteria become less active, giving the preservative a wider margin of safety.
This is why USP 797 specifies refrigeration as a condition for the 28-day BUD. At room temperature, the same vial carries unacceptable contamination risk after 24 to 48 hours. Refrigeration buys you 28 days. It is not optional for an opened vial.
If you have ever left an opened vial on the counter overnight, see does bacteriostatic water need to be refrigerated for guidance on whether it is still safe to use.
Factors That Shorten Bacteriostatic Water Shelf Life
The 28-day refrigerated window is a maximum, not a guarantee. Several common handling errors can shorten the usable life of an opened vial well below 28 days. Each factor increases contamination risk independently, and they compound when combined.
Temperature Abuse
Every hour a punctured vial spends above 8°C accelerates bacterial growth inside. The most common scenario: you pull the vial from the fridge, draw your bacteriostatic water, set the vial on the counter, and forget about it while you complete a reconstitution. Thirty minutes on the counter is harmless. Three hours raises concern. An overnight stay at 22°C effectively burns through days of your 28-day safety margin.
A punctured vial left in a warm room (above 25°C) for 48 hours or more should be discarded regardless of how many days remain on the calendar. The preservative was not designed to handle sustained room-temperature exposure in a compromised container.
Scenario: You open a vial on March 1 and refrigerate it. On March 10, you leave it out for 36 hours by accident. Even though only 10 days have passed, the effective remaining shelf life is unpredictable. The safe choice: discard and open a new $5 vial rather than risk contaminating a $50-100 peptide like BPC-157 or GHK-Cu.
Excessive Needle Punctures
Each puncture through the rubber stopper degrades its ability to reseal. After 10 to 15 punctures, the stopper surface becomes a patchwork of micro-channels. Even with proper swabbing technique, the cumulative breach area increases the odds that a contamination event occurs.
Clinical guidelines from the Centers for Disease Control (CDC) recommend discarding multi-dose vials when the stopper integrity appears compromised, regardless of the date (CDC Injection Safety Guidelines, 2023). For home users, a practical rule: if you have punctured the stopper more than 15 times, open a new vial.
A 30 mL vial provides enough bacteriostatic water for 10 to 30 peptide reconstitutions (at 1-3 mL per vial). If you are reconstituting multiple peptides simultaneously, you may reach 15 punctures well before day 28. Track both.
Contaminated Needles and Poor Technique
The single fastest way to ruin a vial of bacteriostatic water is to insert a used needle. A needle that has already touched a peptide vial stopper, your skin, or any non-sterile surface carries bacteria directly into the water.
Scenario: You draw 2 mL of bacteriostatic water with a needle, inject it into a peptide vial, then return the same needle to the bacteriostatic water vial for a second draw. That needle now carries organisms from the peptide vial stopper and potentially from the air. You have introduced a direct contamination event that no amount of refrigeration can undo.
The fix is simple: use a fresh, sealed needle for every draw from the bacteriostatic water vial. At $0.05 to $0.10 per needle, this is the cheapest insurance in peptide preparation. For step-by-step reconstitution technique, see how to reconstitute tirzepatide or how to reconstitute GHK-Cu.
Skipping the Alcohol Swab
The rubber stopper sits exposed to ambient air between uses. Dust, dead skin cells, and airborne bacteria settle on its surface. Without an alcohol swab before each needle insertion, you push surface contaminants through the stopper and into the water.
Ten seconds of wiping with a 70% isopropyl alcohol pad followed by five seconds of air drying eliminates the majority of surface organisms. This step is outlined in every pharmacy protocol and every injection guide on this site, including the getting started with peptides guide. It takes fifteen seconds. Skipping it can introduce enough bacteria to overwhelm the benzyl alcohol preservative by day 14 instead of day 28.
How to Check If Your Bacteriostatic Water Is Still Good
Visual inspection catches obvious contamination but misses the most dangerous kind: low-level bacterial growth that is invisible to the naked eye. Bacterial counts below 10,000 colony-forming units per milliliter (CFU/mL) produce no visible turbidity in a clear solution, yet they still pose an infection risk when injected subcutaneously (Cundell et al., PDA J Pharm Sci Technol, 2004). Your primary safety net is the 28-day rule. Visual inspection is your secondary check.
The Visual Inspection Protocol
Perform this check before every draw:
1. Hold the vial against a white background. A sheet of paper or a white countertop works. Bacteriostatic water should be perfectly clear and colorless, identical to tap water in appearance.
2. Look for cloudiness or haze. Any turbidity, even faint milkiness, indicates microbial growth or chemical contamination. Discard the vial.
3. Check for floating particles. Tilt the vial slowly and look for specks, fibers, or sediment moving through the liquid. These may be rubber fragments from a degraded stopper, fibers from a cotton swab, or bacterial colonies large enough to see. Any visible particulate means the vial is compromised.
4. Note the color. The water should be colorless. A yellow tint suggests chemical degradation. A green tint raises concern for Pseudomonas aeruginosa, a common environmental bacterium that produces pyocyanin pigment. Brown or amber discoloration indicates advanced degradation. Any color change warrants immediate disposal.
5. Inspect the stopper. Look at the rubber surface under light. Excessive puncture marks, visible tears, or rubber fragments floating inside the vial mean the seal can no longer be trusted.
When to Discard Even If It Looks Clear
Clear water is not proof of sterility. Discard under any of these conditions regardless of appearance:
- More than 28 days since first puncture
- More than 15 needle punctures through the stopper
- Left at room temperature for more than 48 hours after opening
- Exposed to temperatures above 40°C after opening
- Frozen and thawed (expansion may have compromised the seal)
- You cannot remember when you first opened it
- Past the manufacturer's printed expiration date
A 30 mL vial of bacteriostatic water costs $3 to $8. A single vial of research-grade semaglutide costs $50 to $150. A course of antibiotics for an injection site infection costs more still. The math favors replacing questionable water without hesitation.
Impact on Peptide Reconstitution and Potency
Bacteriostatic water is a vehicle: its job is to dissolve lyophilized peptide powder into an injectable solution while maintaining sterility across multiple doses. When the water is compromised, every peptide vial you reconstitute with it inherits the contamination.
What Contaminated Water Does to Your Peptides
Scenario 1: Water used on day 35, refrigerated the entire time. You reconstitute a 5 mg vial of TB-500 with 2 mL of this water. The TB-500 dissolves and the solution looks clear. But benzyl alcohol efficacy has declined past the 28-day threshold. Bacteria introduced during earlier punctures have had an extra week to replicate. By day 5 of drawing from the reconstituted peptide vial, you notice slight cloudiness. By day 8, the solution is visibly turbid. The entire vial, $40 to $80 worth of peptide, is wasted.
Scenario 2: Water left on the counter for 2 days, then used. You reconstitute BPC-157 with this water. Bacteria that multiplied during the 48 hours at room temperature transfer into the peptide vial. You inject 250 mcg subcutaneously. Three days later, the injection site is red, warm, and tender. A week later, you are on oral antibiotics for a localized cellulitis infection. Total cost: the peptide vial, the doctor visit, the antibiotic prescription, and a week of healing.
Neither scenario is hypothetical. Injection site infections from contaminated reconstitution water are documented in clinical literature (Kainer et al., N Engl J Med, 2012). The fix in both cases: verify the water is within its 28-day window and passed visual inspection before every reconstitution.
Best Practices for Reconstitution with Fresh Water
Check three things before every reconstitution:
1. Date on the vial. Confirm the first-puncture date is within 28 days. If there is no date written on the vial, discard it and open a new one.
2. Visual clarity. Hold against white background, inspect for cloudiness, particulates, or color.
3. Stopper condition. No excessive puncture marks, no rubber fragments inside.
Once the water passes all three checks, draw the required volume with a fresh needle and inject it slowly down the inner wall of the peptide vial. Never aim the stream directly onto the lyophilized powder cake. Let gravity and gentle swirling dissolve the peptide over 60 to 90 seconds. Never shake.
For exact water volumes by peptide and vial size, use the reconstitution calculator. For peptide-specific mixing guides, see how much bacteriostatic water for semaglutide or how to take BPC-157.
Multi-Puncture Vial Tracking System
A 30 mL vial of bacteriostatic water serves multiple reconstitutions across days or weeks. Without a tracking system, you lose the two most critical data points: when you first punctured it and how many times you have drawn from it.
The Two-Number Method
Write two things on the vial the first time you puncture it:
1. The date of first puncture. Example: "Opened 03/14/26." This starts your 28-day countdown.
2. A tally of punctures. Add a small tally mark each time you insert a needle. When the count reaches 15, open a new vial regardless of how many days remain.
Use a fine-tip permanent marker directly on the glass or plastic vial. Adhesive labels work too, but they can peel off in the humidity of a refrigerator. The marker is more reliable.
This dual-tracking method mirrors what compounding pharmacies use. It accounts for both time-based degradation (28 days) and mechanical degradation (stopper wear from repeated punctures). Whichever limit you hit first dictates when to discard.
Pre-Drawing Strategy for High-Volume Users
If you reconstitute multiple peptides in a month, you may draw from your bacteriostatic water vial 10 to 20 times. Each draw is a puncture, a contamination event, and a step toward stopper degradation. A pre-drawing strategy reduces all three risks.
How it works: On the day you open a new vial, draw all the water you will need for the next 28 days into individual sterile syringes. Cap each syringe and store them horizontally in the refrigerator.
Example: You plan to reconstitute four peptide vials over the next month, each requiring 2 mL of bacteriostatic water. On day one, draw four syringes of 2 mL each (8 mL total) from the vial. That is 4 punctures instead of the 4 you would have done anyway, but concentrated into a single sterile session where you control all variables. The remaining 22 mL stays sealed with only 4 puncture events for future use.
The pre-drawn syringes maintain sterility for the same 28-day period, provided they remain capped and refrigerated. This approach is especially useful when running a peptide stack that involves reconstituting BPC-157, TB-500, and GHK-Cu within the same month.
Storage Best Practices for Maximum Shelf Life
Proper storage is the difference between getting the full 28 days from an opened vial and losing it in a week. These rules apply to both opened and unopened bacteriostatic water.
Optimal Storage Conditions
Unopened vials: Store at controlled room temperature (20-25°C) in a cool, dark location. A medicine cabinet, a drawer, or a closet shelf all work. Avoid bathrooms where shower steam raises humidity and temperature cyclically. Avoid windowsills, the top of a refrigerator (warm air rises), and anywhere near a heat source.
Opened vials: Refrigerate at 2-8°C immediately after every draw. Place the vial on the back of the middle shelf, where temperature is most consistent. Avoid the refrigerator door, where temperature swings 5-10°C every time you open it. Keep the vial upright to minimize water contact with the stopper.
All vials: Keep away from direct sunlight. While benzyl alcohol is not highly photosensitive, UV exposure degrades rubber stoppers over time and can initiate trace chemical reactions. A refrigerator or closed cabinet provides adequate light protection. There is no need to wrap vials in aluminum foil the way you would for light-sensitive peptides like Selank or MOTS-c.
Common Storage Mistakes
Storing opened vials in the fridge door. The door is the warmest, most temperature-variable spot in any refrigerator. Every time you open the fridge, door-shelf items experience a brief temperature spike. Over 28 days, this cycling stresses the preservative system unnecessarily. Move the vial to the back of a middle shelf.
Leaving the vial out during reconstitution. You draw 2 mL, set the vial on the counter, spend 10 minutes reconstituting and mixing your peptide, then remember to put the water back. That 10 minutes is not dangerous. But if this happens every few days and occasionally extends to an hour or two, cumulative room-temperature exposure adds up. Draw, cap, refrigerate, then continue your reconstitution.
Storing near food with strong odors. This does not affect sterility or chemical stability, but rubber stoppers can absorb volatile organic compounds over weeks. While functionally harmless, it is a reminder that bacteriostatic water vials belong in a dedicated space, ideally alongside your reconstituted peptide vials. For peptide-specific storage timelines, see how long do reconstituted peptides last and how long does tirzepatide last in the fridge.
Bulk Purchasing and Stock Rotation
Bacteriostatic water is inexpensive enough to buy in bulk. A pack of ten 30 mL vials typically costs $25 to $50. At that price, there is no financial reason to stretch a single vial beyond its safe window.
When you buy in bulk, practice first-in, first-out (FIFO) stock rotation. Place new vials behind older ones. Use the vial with the earliest expiration date first. Check expiration dates when your shipment arrives and discard any vial that will expire within the next 3 months if you are unlikely to use it by then.
Store your bulk supply at room temperature in a dark cabinet. Open one vial at a time. Keep the rest sealed until needed. This system ensures you always have fresh bacteriostatic water available and never face the temptation of using a vial past its 28-day window because you "ran out." For sourcing guidance, see where to buy bacteriostatic water for injection.
Signs of Expired or Compromised Bacteriostatic Water
Recognizing compromised water is your last line of defense before a contamination event becomes an injection. Some signs are obvious. Others require knowing what to look for.
Visible Signs of Expiration
Turbidity. Any cloudiness, haziness, or loss of crystal clarity. Fresh bacteriostatic water is as transparent as distilled water. Hold the vial at eye level in front of a bright light source. Even faint milkiness indicates microbial growth.
Floating matter. Specks, fibers, flakes, or sediment. These may be rubber particles from stopper degradation, cotton fibers from a non-sterile swab, or visible microbial colonies. Any particulate contamination disqualifies the vial.
Color change. Bacteriostatic water is colorless. Yellow suggests oxidative degradation. Green suggests bacterial pigmentation (Pseudomonas species produce distinctive green pigments). Any departure from colorless requires disposal.
Stopper damage. A stopper that appears chewed up, shows visible holes rather than clean puncture points, or has shed rubber fragments into the solution can no longer maintain a seal. This is more common with thin-gauge needles (18G) that core the rubber rather than parting it. Use 25G to 29G needles to minimize stopper damage.
Hidden Signs You Cannot See
The most dangerous contamination is invisible. At bacterial loads below 10,000 CFU/mL, the solution looks perfectly clear. You have no way to detect this contamination without laboratory culture testing, which is impractical for home use.
This is precisely why the 28-day rule and the hygiene protocols exist. They replace laboratory testing with behavior-based safety margins. Following the rules means you never rely on visual inspection alone.
Three non-visual indicators that should prompt disposal:
1. Loss of date tracking. If you cannot confirm when the vial was first punctured, treat it as expired. "I think it was about three weeks ago" is not good enough. Write the date on the vial at first puncture, every time.
2. Lapsed refrigeration. If you know or suspect the vial sat at room temperature for more than a few hours, assume the safety margin has shrunk. For a vial left out overnight (8-12 hours), you might still use it. For 24 hours or more, discard.
3. Changed handling conditions. If someone else used your vial (a partner, a roommate), you cannot verify their sterile technique. Assume the worst and open a fresh vial. The peptide safety guide covers shared-use protocols in detail.
Bacteriostatic Water Shelf Life FAQ
Frequently Asked Questions
How long does bacteriostatic water last unopened?
Unopened bacteriostatic water lasts until the manufacturer's printed expiration date, typically 2 to 3 years from production. Store at controlled room temperature (20-25°C) in a cool, dark location. Refrigeration is acceptable but not required. Discard if the seal appears compromised or if the vial has been exposed to sustained heat above 40°C.
How long does bacteriostatic water last after opening?
Bacteriostatic water lasts 28 days after the first needle puncture when stored at 2-8°C (refrigerated). This beyond-use date comes from USP Chapter 797, the federal standard for sterile compounding. At room temperature, a punctured vial should be discarded after 24 to 48 hours due to accelerated bacterial growth risk.
Can you use bacteriostatic water after 28 days?
Using bacteriostatic water beyond 28 days post-puncture is not recommended. The 0.9% benzyl alcohol preservative loses effectiveness against cumulative contamination over time. Even if the water looks clear, bacterial counts may exceed safe thresholds for injection. A replacement 30 mL vial costs $3 to $8, far less than the peptide it protects.
Does bacteriostatic water expire if never opened?
Yes, sealed bacteriostatic water has a manufacturer-assigned expiration date, usually 2 to 3 years from production. After this date, the preservative system and packaging integrity are no longer guaranteed. Check the printed date on the label or box before use. Store unopened vials in a cool, dark location at 20-25°C.
How do you know if bacteriostatic water is bad?
Inspect for cloudiness, floating particles, or any color change (yellow, green, or brown). Check the stopper for excessive puncture damage or rubber fragments in the solution. Also discard if the vial is past 28 days from first puncture, has more than 15 needle entries, or was left unrefrigerated for over 48 hours after opening.
Does bacteriostatic water need to be refrigerated before opening?
No. Sealed, unopened bacteriostatic water is stable at controlled room temperature (20-25°C). Refrigeration before opening is acceptable but unnecessary. After the first needle puncture, refrigeration at 2-8°C becomes mandatory to maintain sterility for the full 28-day beyond-use window per USP 797 guidelines.
Can you freeze bacteriostatic water to make it last longer?
Do not freeze bacteriostatic water. Water expands approximately 9% when frozen, which can crack glass vials and compromise seals on plastic vials. Freezing also drives benzyl alcohol out of uniform distribution, leaving some portions under-preserved after thawing. If accidentally frozen, inspect for cracks and cloudiness before considering use.
How many times can you puncture bacteriostatic water?
Limit needle punctures to 15 per vial. Each puncture degrades the rubber stopper's ability to reseal and introduces a potential contamination pathway. After 15 punctures, stopper integrity becomes unreliable regardless of how many days remain in the 28-day window. Use a fresh 25-29G needle for each draw to minimize stopper coring.
The Bottom Line
Bacteriostatic water shelf life follows one rule: unopened vials last until the manufacturer's expiration date (2-3 years). Opened vials last 28 days at 2-8°C. That 28-day limit is set by USP 797 and reflects the point where cumulative contamination risk exceeds the preservative's capacity.
Three actions protect every vial you open. Write the date of first puncture on the glass. Refrigerate immediately after every draw. Discard at 28 days or 15 punctures, whichever comes first. The cost of a fresh vial ($3-8) is trivial compared to the cost of contaminated water ruining a $50-100 peptide or causing an injection site infection.
For exact reconstitution volumes, use the reconstitution calculator. For detailed storage protocols across all peptides, see the peptide storage guide. For sourcing, see where to buy bacteriostatic water for injection.
Related articles: - Bacteriostatic Water vs Sterile Water: head-to-head comparison of the two diluent types - Is Reconstitution Solution the Same as Bacteriostatic Water?: comparing all diluent categories - Does Bacteriostatic Water Need to Be Refrigerated?: full refrigeration guide with USP rules - Does Semaglutide Expire?: expiration timelines for Ozempic, Wegovy, and compounded vials - Does Semaglutide Need to Be Refrigerated?: storage rules for all semaglutide forms - How to Inject BPC-157: injection guide covering sterile technique from reconstitution to disposal
Related Articles
Bacteriostatic Water Refrigeration
Unopened bacteriostatic water stores at room temperature. Once opened or punctured, refrigerate at 2-8°C and use within 28 days.
Reconstitution Solution vs Bac Water
Reconstitution solution is a broad category. Bacteriostatic water is one type. Compare bac water, sterile water, and saline for peptide mixing.
Bacteriostatic vs Sterile Water
Bacteriostatic water contains 0.9% benzyl alcohol for 28-day multi-use. Sterile water has no preservative and is single-use only.
Semaglutide Refrigeration Guide
Yes, semaglutide must be refrigerated at 2-8°C before first use. Ozempic pens last 56 days at room temp after opening. Compounded vials: 28 days.