You ordered a peptide kit and the listing says "reconstitution solution included." The vial arrives without a label identifying the liquid inside. You already have bacteriostatic water on your shelf. Are they the same thing?
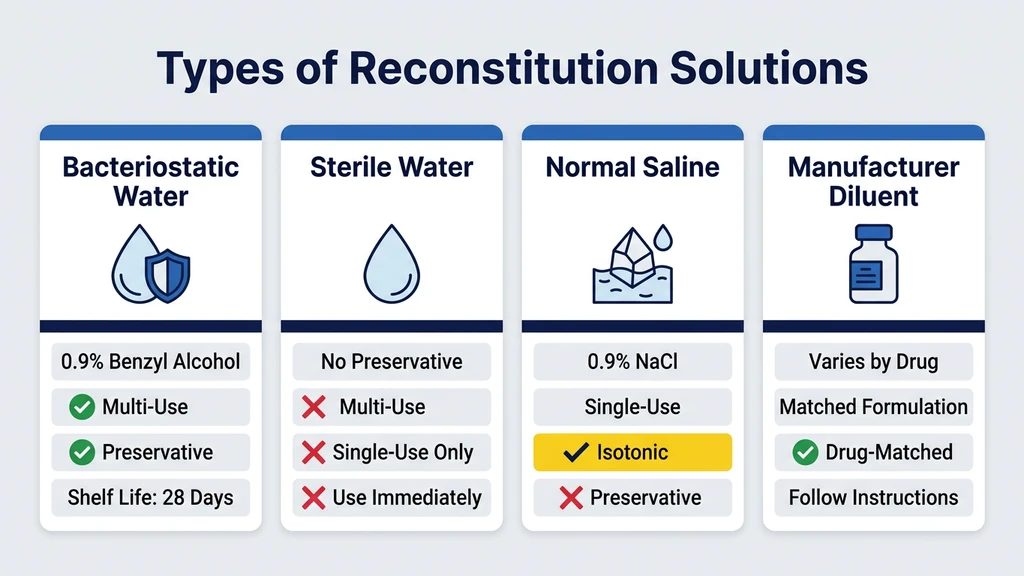
Not always. Bacteriostatic water is one type of reconstitution solution, but it is not the only one. The term "reconstitution solution" refers to any sterile liquid used to dissolve a lyophilized (freeze-dried) powder. Bacteriostatic water, sterile water for injection, 0.9% sodium chloride (normal saline), and manufacturer-supplied diluents all qualify. Each has a different composition, a different shelf life after opening, and a different set of peptides it pairs with best.
| Solution Type | Preservative | Multi-Dose Use | Shelf Life (Opened) | Best For |
|---|---|---|---|---|
| Bacteriostatic water (0.9% benzyl alcohol) | Yes | Yes (up to 28 days) | 28 days at 2-8°C | Peptides drawn from multiple times |
| Sterile water for injection (SWFI) | No | No (single use) | Use immediately, discard remainder | Single-dose vials, neonatal medications |
| 0.9% sodium chloride (normal saline) | No (unless bacteriostatic version) | Depends on formulation | 24 hours (preservative-free) or 28 days (bacteriostatic) | HGH, some manufacturer protocols |
| Manufacturer-supplied diluent | Varies (mannitol, glycerin, or preservatives) | Per manufacturer instructions | Varies by product | Specific branded peptides (Genotropin, Humatrope) |
If you need to calculate exact water volumes for your peptide vial, use the reconstitution calculator. For a full walkthrough of the reconstitution process, see the getting started guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Counts as a Reconstitution Solution
"Reconstitution solution" is a functional label, not a chemical name. It describes any sterile liquid whose purpose is to dissolve a powdered medication back into injectable form. The powder is the drug. The solution is the vehicle.
Think of it like the word "fuel." Gasoline, diesel, jet fuel, and ethanol are all fuels. They all combust. But you would not put diesel in a gasoline engine and expect it to run correctly. Reconstitution solutions work the same way. They all dissolve powders, but the wrong one can destabilize your peptide, introduce preservatives where none should be, or leave you with a solution that expires before you finish using it.
The literal distinction: reconstitution solution is the category. Bacteriostatic water is a member of that category. So are sterile water for injection, bacteriostatic sodium chloride, preservative-free normal saline, and proprietary diluents that ship with branded medications like Genotropin or Humatrope.
Why the Confusion Exists
Peptide vendors and compounding pharmacies often use the term "reconstitution solution" interchangeably with "bacteriostatic water" because bacteriostatic water is the most common solvent for research peptides. When a seller includes "reconstitution solution" in a kit, they almost always mean bacteriostatic water (0.9% benzyl alcohol). But "almost always" is not "always."
Some vendors ship sterile water for injection instead, particularly with peptides sensitive to benzyl alcohol. Others include proprietary diluents with stabilizers like mannitol. The label on the included vial should identify the contents. If it does not, contact the vendor before using it. Injecting an unknown liquid defeats the purpose of careful peptide preparation.
For sourcing verified bacteriostatic water, see where to buy bacteriostatic water for injection.
Bacteriostatic Water: The Most Common Reconstitution Solution
Bacteriostatic water is sterile water with 0.9% benzyl alcohol added as a preservative. That single ingredient separates it from every other reconstitution solution and makes it the default choice for peptides intended for multi-dose use.
How Benzyl Alcohol Works as a Preservative
Benzyl alcohol disrupts bacterial cell membranes. At 0.9% concentration, it suppresses the growth of common contaminants: Staphylococcus epidermidis (skin flora), Pseudomonas aeruginosa (environmental bacteria), and Escherichia coli. It does not sterilize the solution on contact. It prevents bacteria from multiplying fast enough to reach dangerous colony counts.
This distinction matters. Bacteriostatic means "growth-inhibiting." Bactericidal means "bacteria-killing." Benzyl alcohol is bacteriostatic. It holds contamination in check rather than eliminating it. That is why refrigeration (2-8°C) and the 28-day discard rule from USP Chapter 797 work alongside the preservative. Cold temperature slows bacterial replication further, giving the benzyl alcohol a wider margin of safety (Akers et al., J Pharm Sci, 1984).
For complete storage rules, see does bacteriostatic water need to be refrigerated.
When to Use Bacteriostatic Water
Use bacteriostatic water when you will draw multiple doses from a single reconstituted peptide vial over days or weeks. This covers most research peptides: BPC-157, TB-500, semaglutide, tirzepatide, GHK-Cu, CJC-1295, ipamorelin, and PT-141, among others.
The preservative keeps the reconstituted solution usable for up to 28 days in the refrigerator. Without it, every needle puncture introduces bacteria into an unprotected solution, and the vial becomes a contamination risk within hours.
A 30 mL vial of bacteriostatic water costs $3-8 and provides enough solvent for 10-30 reconstitutions at typical 1-2 mL volumes. For mixing instructions specific to popular peptides, see how much bacteriostatic water for semaglutide or how to reconstitute tirzepatide.
When Not to Use Bacteriostatic Water
Benzyl alcohol is contraindicated in three situations.
Neonatal use. Benzyl alcohol toxicity in neonates causes gasping syndrome, a serious condition first documented in the 1980s. The FDA requires warning labels on all bacteriostatic water vials stating the product is not for use in neonates (Gershanik et al., N Engl J Med, 1982). This applies to compounded medications for infants, not to adult peptide use, but it illustrates why the preservative is not universally safe.
Intrathecal or epidural injection. Benzyl alcohol is neurotoxic when injected into the spinal canal. No reconstituted peptide should be administered by these routes, but the contraindication is absolute for bacteriostatic water specifically.
Peptide-specific incompatibility. Certain peptides degrade in the presence of benzyl alcohol. Some growth hormone formulations specify sterile water or a proprietary diluent. If the manufacturer instructions say "do not use bacteriostatic water," follow them. The preservative can alter the peptide's tertiary structure or accelerate aggregation in sensitive molecules.
Sterile Water for Injection: The Preservative-Free Alternative
Sterile water for injection (SWFI) is exactly what the name states: water that has been sterilized, typically by autoclaving, and packaged in a sealed container with no additives. No preservatives. No salts. No buffers. Pure H₂O in a sterile vial.
Why Sterile Water Has a Short Shelf Life
Without benzyl alcohol, sterile water has no defense against microbial contamination once the vial is punctured. The USP classifies an opened SWFI vial as a single-use container. Draw what you need, reconstitute your peptide, and discard the remaining water.
In practice, this means a 30 mL vial of sterile water produces one reconstitution event. If you use 2 mL for a peptide vial, the other 28 mL go to waste. This is expensive and impractical for most home peptide users, which is why bacteriostatic water dominates the market. But in clinical settings where single-dose protocols are standard, SWFI is the preferred solvent.
When Sterile Water Is the Right Choice
Use sterile water for injection in these situations:
Single-dose reconstitution. You reconstitute a vial, draw the entire contents into a syringe, and inject it immediately. No leftover solution sits in the vial. This is common with some growth hormone protocols and certain clinical research applications.
Benzyl alcohol sensitivity. Patients with documented benzyl alcohol allergy or sensitivity require SWFI. The reaction is rare but real: local irritation, injection site pain, and in severe cases, systemic hypersensitivity.
Manufacturer specification. Some peptide and medication labels explicitly require SWFI. Genotropin cartridges, for instance, ship with a preservative-free diluent rather than bacteriostatic water. When a product specifies sterile water, use sterile water.
For information on how long your reconstituted peptide remains stable regardless of which solvent you use, see how long do reconstituted peptides last.
Normal Saline and Bacteriostatic Sodium Chloride
Normal saline (0.9% sodium chloride solution) adds another layer to the reconstitution landscape. It comes in two forms: preservative-free and bacteriostatic. The distinction is critical.
Preservative-Free Normal Saline
Preservative-free 0.9% sodium chloride is an isotonic solution, meaning its salt concentration matches that of human blood (approximately 308 mOsm/L). Some peptides and medications require an isotonic environment to maintain stability. Reconstituting these products in plain water (which is hypotonic) can cause osmotic stress that degrades the molecule.
HGH (human growth hormone) protocols occasionally specify normal saline. Certain lyophilized antibiotics require it. If the product label or compounding pharmacy instructions call for normal saline, do not substitute bacteriostatic water. The salt concentration matters for the peptide's structural integrity.
Like preservative-free sterile water, preservative-free normal saline is a single-use product. Once punctured, use it immediately and discard the remainder.
Bacteriostatic Sodium Chloride
Bacteriostatic sodium chloride combines 0.9% NaCl with 0.9% benzyl alcohol. It offers both isotonicity and preservative protection, making it suitable for multi-dose use with a 28-day refrigerated shelf life, the same window as bacteriostatic water.
This solution is less common in the peptide community but appears in clinical compounding. Some growth hormone and gonadotropin protocols specify it. If you see "bacteriostatic normal saline" or "bacteriostatic NaCl" in a protocol, it is this product.
The practical difference from bacteriostatic water: the 0.9% NaCl. For most research peptides, the salt does not matter. BPC-157, TB-500, and GLP-1 agonists dissolve equally well in bacteriostatic water or bacteriostatic saline. But for peptides that specify isotonic diluent, bacteriostatic sodium chloride provides the salt balance without sacrificing multi-dose convenience.
Manufacturer-Supplied Diluents: What Is in the Kit
Branded peptide medications often ship with a proprietary diluent rather than generic bacteriostatic water. These diluents are formulated specifically for the peptide they accompany.
Common Components in Proprietary Diluents
Mannitol. A sugar alcohol used as a stabilizer and tonicity agent. Genotropin (somatropin) diluent contains mannitol. It protects the growth hormone from aggregation during reconstitution and storage. You cannot replicate this stabilizing effect by substituting plain bacteriostatic water.
Glycerin. Used in some diluents as a stabilizer and viscosity modifier. It slows the degradation of sensitive peptides by maintaining a specific osmotic environment.
Metacresol. A preservative found in some multi-dose insulin and growth hormone formulations (Humatrope, Norditropin). It serves the same bacteriostatic function as benzyl alcohol but at a different concentration and with a slightly different antimicrobial spectrum. Some patients who react to benzyl alcohol tolerate metacresol, and vice versa.
Phosphate buffers. Some diluents include sodium phosphate or potassium phosphate to maintain a specific pH range. Peptides have an optimal pH for stability. Outside that range, the amino acid chain can unfold, aggregate, or hydrolyze. A buffered diluent holds the pH steady in a way that unbuffered water cannot.
Can You Substitute Bacteriostatic Water for a Manufacturer Diluent
Sometimes yes, sometimes no.
When substitution works: Generic somatropin vials that ship without a diluent can be reconstituted with bacteriostatic water. The peptide is stable in the 4.5-7.5 pH range of bacteriostatic water, and the benzyl alcohol provides adequate preservation for 28-day multi-dose use.
When substitution fails: Genotropin cartridges require their specific diluent because the delivery device and reconstitution mechanism are designed for the proprietary solution's viscosity and volume. Using bacteriostatic water in a Genotropin pen does not work mechanically.
When substitution degrades the peptide: If a manufacturer adds mannitol or a pH buffer to protect the peptide from aggregation, removing those stabilizers by substituting plain bacteriostatic water may reduce shelf life or potency. You might get 14 days of stability instead of 28.
The rule: follow the manufacturer's instructions. If the product specifies a particular diluent, use that diluent. If no diluent is specified and the product is a generic lyophilized peptide, bacteriostatic water is the standard choice. For complete peptide storage protocols, see how to store peptides.
Side-by-Side Comparison: Choosing the Right Solution for Your Peptide
The choice of reconstitution solution depends on three factors: how many doses you will draw from the vial, whether the peptide tolerates benzyl alcohol, and what the manufacturer specifies.
| Factor | Bacteriostatic Water | Sterile Water (SWFI) | Normal Saline (0.9% NaCl) | Bacteriostatic NaCl | Manufacturer Diluent |
|---|---|---|---|---|---|
| Preservative | 0.9% benzyl alcohol | None | None | 0.9% benzyl alcohol | Varies |
| Multi-dose use | Yes (28 days) | No (single use) | No (single use) | Yes (28 days) | Per label |
| Tonicity | Hypotonic | Hypotonic | Isotonic | Isotonic | Varies |
| pH | ~5.7 (unbuffered) | ~5.5 (unbuffered) | ~5.5 (unbuffered) | ~5.5 (unbuffered) | Often buffered (6.5-7.5) |
| Cost per vial (30 mL) | $3-8 | $2-5 | $2-5 | $5-10 | Included with product |
| Neonatal safe | No | Yes | Yes | No | Per label |
| Availability | OTC, online | Pharmacy, online | Pharmacy, online | Pharmacy | With product only |
For most research peptides drawn from multi-dose vials, bacteriostatic water is the correct choice. Use the reconstitution calculator to determine the exact volume for your vial size and desired concentration.
Decision Flowchart
Step 1: Check the manufacturer label. If the product specifies a diluent, use that diluent. Stop here.
Step 2: Will you draw multiple doses? If yes, you need a preservative. Choose bacteriostatic water (most peptides) or bacteriostatic sodium chloride (if isotonicity is required).
Step 3: Is benzyl alcohol contraindicated? If yes (neonatal use, known allergy, or manufacturer warning), use sterile water for injection and treat the vial as single-use.
Step 4: Does the peptide require isotonic solution? If yes (some HGH protocols, certain gonadotropins), use bacteriostatic sodium chloride or preservative-free normal saline. If no, bacteriostatic water is your default.
Most peptide users land on bacteriostatic water. It covers the widest range of use cases at the lowest cost with the longest shelf life after opening.
Common Mistakes When Choosing a Reconstitution Solution
These errors appear repeatedly in peptide forums and can waste expensive product or create safety risks.
Using Sterile Water and Expecting 28-Day Shelf Life
A user reconstitutes a 5 mg vial of BPC-157 with sterile water for injection, planning to draw 250 mcg daily for 20 days. By day 3, the solution has been punctured three times with no preservative protecting it. Bacterial contamination is accumulating silently.
By day 10, the user notices slight cloudiness. The remaining 12 days of peptide are unusable. The $50 vial is wasted.
The fix: use bacteriostatic water for any multi-dose vial. Sterile water is for single-dose reconstitution only. For BPC-157 dosing protocols, see how to take BPC-157.
Substituting Tap Water or Distilled Water
Neither tap water nor store-bought distilled water is sterile. Tap water contains chlorine, fluoride, minerals, and bacteria. Distilled water has been purified of minerals but is not sterile. It was packaged in a non-sterile facility, bottled in a non-sterile container, and has been sitting on a shelf exposed to whatever entered during bottling.
Injecting a solution made with non-sterile water introduces bacteria directly into your body. The peptide itself may dissolve fine, but the microbial payload makes it dangerous. This mistake sounds obvious, but online forums document it regularly.
The fix: only use USP-grade sterile or bacteriostatic water from a licensed pharmacy or verified vendor. For sourcing guidance, see where to buy bacteriostatic water.
Ignoring Manufacturer Diluent Instructions
A user receives a branded growth hormone kit with a proprietary diluent containing mannitol and metacresol. They discard the diluent and substitute bacteriostatic water because they read that bacteriostatic water "works for everything."
The growth hormone reconstitutes and appears to dissolve normally. But without the mannitol stabilizer, the protein begins aggregating within 7-10 days instead of the expected 21-28 days. The user injects partially degraded product for the second half of their vial, receiving reduced potency and potentially immunogenic protein aggregates.
The fix: use the included diluent. If the diluent vial breaks or is missing, contact the manufacturer for a replacement before substituting.
Mixing Solutions Together
Some users attempt to combine bacteriostatic water with normal saline to create a "custom" reconstitution solution with both isotonicity and preservation. This is unnecessary and potentially problematic. The resulting NaCl concentration is unknown, the benzyl alcohol concentration is diluted below 0.9%, and the solution is no longer validated for the 28-day USP shelf life.
The fix: use the correct solution as a standalone product. If you need isotonic + preserved, buy bacteriostatic sodium chloride. Do not improvise pharmaceutical solutions.
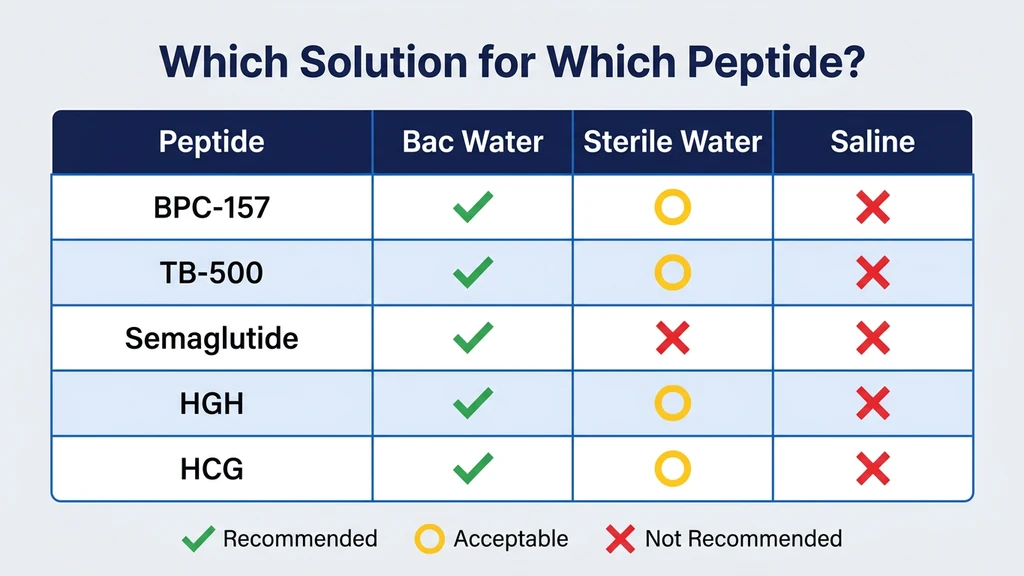
Which Peptides Need Which Solution
The table below maps common research peptides to their recommended reconstitution solution. Default is bacteriostatic water unless the peptide has specific requirements.
| Peptide | Recommended Solution | Notes |
|---|---|---|
| BPC-157 | Bacteriostatic water | Stable in bac water for 28 days at 2-8°C |
| TB-500 | Bacteriostatic water | No special requirements |
| Semaglutide | Bacteriostatic water | Compounded vials; prefilled pens use manufacturer diluent |
| Tirzepatide | Bacteriostatic water | Compounded vials; Mounjaro pens are pre-mixed |
| GHK-Cu | Bacteriostatic water | Copper peptide, avoid excessive agitation during mixing |
| Retatrutide | Bacteriostatic water | See how to reconstitute retatrutide |
| CJC-1295 | Bacteriostatic water | DAC variant stable for 14+ days |
| Ipamorelin | Bacteriostatic water | Standard reconstitution |
| MK-677 | Oral (liquid or capsule) | Not reconstituted; taken orally |
| PT-141 | Bacteriostatic water or sterile water | Short protocol, often single-dose use |
| Selank | Bacteriostatic water or sterile water | Nasal spray formulations may use different vehicles |
| MOTS-c | Bacteriostatic water | Light-sensitive; store in dark after reconstitution |
| HGH (generic somatropin) | Bacteriostatic water | Multi-dose vials |
| HGH (Genotropin) | Manufacturer diluent (contains mannitol) | Do not substitute |
| HGH (Humatrope) | Manufacturer diluent (contains metacresol) | Do not substitute |
| Gonadorelin | Bacteriostatic water or bacteriostatic NaCl | Some protocols specify saline |
For reconstitution instructions for specific peptides: - How to reconstitute GHK-Cu - How much bacteriostatic water for semaglutide - How to reconstitute tirzepatide - How to reconstitute retatrutide
Shelf Life Comparison After Opening
Shelf life is the practical differentiator between reconstitution solutions. It determines how long your reconstituted peptide remains safe to inject.
| Solution | Shelf Life (Unopened) | Shelf Life (After First Puncture) | Storage After Opening |
|---|---|---|---|
| Bacteriostatic water | 1-3 years (manufacturer date) | 28 days | 2-8°C (refrigerator) |
| Sterile water for injection | 1-3 years | Use immediately, discard remainder | N/A (single use) |
| Preservative-free normal saline | 1-2 years | Use immediately, discard remainder | N/A (single use) |
| Bacteriostatic sodium chloride | 1-3 years | 28 days | 2-8°C (refrigerator) |
| Manufacturer diluent | Per label (6 months to 2 years) | Per label (typically 14-28 days) | Per label (usually 2-8°C) |
The 28-day limit on bacteriostatic water comes from USP Chapter 797, the United States Pharmacopeia standard for sterile compounding. After 28 days, the benzyl alcohol preservative can no longer guarantee sterility in a multi-puncture vial, even under continuous refrigeration. The cumulative microbial load from repeated needle entries, combined with gradual preservative degradation, pushes the risk beyond acceptable limits.
Mark the date of first puncture on every vial. After 28 days, discard and open a new one. A 30 mL vial costs $3-8. A contaminated peptide injection costs far more in wasted product and potential infection. For detailed storage timelines by peptide, see how long do reconstituted peptides last and the peptide dosage chart for comprehensive shelf life data.
Reconstitution Solution FAQ
Frequently Asked Questions
Is reconstitution solution the same as bacteriostatic water?
Not always. Bacteriostatic water is one type of reconstitution solution. The category also includes sterile water for injection, 0.9% sodium chloride (normal saline), bacteriostatic sodium chloride, and manufacturer-supplied diluents. Each has different preservatives, shelf lives, and intended uses. Bacteriostatic water is the most common choice for multi-dose peptide vials.
Can I use sterile water instead of bacteriostatic water for peptides?
You can, but only for single-dose reconstitution. Sterile water contains no preservative, so a punctured vial must be used immediately and the remainder discarded. For multi-dose peptide vials where you draw daily or weekly injections over 2-4 weeks, bacteriostatic water with its 0.9% benzyl alcohol preservative is required for safe multi-puncture use.
What is the difference between bacteriostatic water and normal saline for reconstitution?
Bacteriostatic water contains 0.9% benzyl alcohol (preservative) but no salt. Normal saline contains 0.9% sodium chloride (salt) but typically no preservative. Bacteriostatic sodium chloride contains both. For most research peptides, bacteriostatic water is preferred. Saline is specified when isotonicity is required, as in some HGH and gonadotropin protocols.
How long does reconstitution solution last after opening?
It depends on the type. Bacteriostatic water and bacteriostatic sodium chloride last 28 days after first puncture when stored at 2-8°C (USP 797 standard). Sterile water for injection and preservative-free normal saline are single-use: draw what you need and discard the rest immediately. Manufacturer diluents follow their specific label instructions, typically 14-28 days.
Can I use bacteriostatic water for all peptides?
For most research peptides (BPC-157, TB-500, semaglutide, tirzepatide, GHK-Cu, CJC-1295, ipamorelin), yes. Exceptions exist: some branded growth hormones require their proprietary diluent containing stabilizers like mannitol. Benzyl alcohol is contraindicated for neonatal use and intrathecal injection. Always check the peptide's label or manufacturer instructions first.
Is the reconstitution solution included in peptide kits always bacteriostatic water?
Usually, but not always. Most peptide vendors include bacteriostatic water (0.9% benzyl alcohol) as the kit solvent. Some vendors ship sterile water for injection, particularly with benzyl-alcohol-sensitive peptides. Others include proprietary diluents. Check the vial label for the specific contents. If unlabeled, contact the vendor before using it.
Can I use distilled water to reconstitute peptides?
No. Store-bought distilled water is not sterile. It has been purified of minerals but was packaged in a non-sterile facility and may contain bacteria. Injecting a peptide reconstituted with non-sterile water risks infection. Only use USP-grade sterile water for injection or bacteriostatic water from a licensed pharmacy or verified vendor.
What happens if I use the wrong reconstitution solution?
The consequences depend on the mismatch. Sterile water in a multi-dose vial leads to bacterial contamination by day 3-5. Bacteriostatic water in place of a manufacturer diluent may cause faster peptide degradation (10-14 days instead of 28). Tap or distilled water introduces bacteria directly. The safest approach: use bacteriostatic water for generic peptides and manufacturer diluents for branded products.
The Bottom Line
Reconstitution solution is the category. Bacteriostatic water is the most common member. They overlap frequently, but they are not identical.
For multi-dose peptide vials drawn from over days or weeks, bacteriostatic water (0.9% benzyl alcohol) is the standard. It provides 28 days of preservative protection at 2-8°C. Sterile water works for single-dose reconstitution only. Normal saline is specified when isotonicity matters. Manufacturer diluents contain proprietary stabilizers that generic solvents cannot replicate.
The decision path is straightforward. Check the manufacturer label first. If no diluent is specified, choose bacteriostatic water for multi-dose use or sterile water for single-dose use. Mark the date on every opened vial. Discard bacteriostatic water after 28 days and sterile water after immediate use. Use the reconstitution calculator for exact volumes. For complete storage protocols, see how to store peptides.
Related articles: - Bacteriostatic Water vs Sterile Water: head-to-head comparison with shelf life and safety data - How Long Does Bacteriostatic Water Last?: complete shelf life guide with USP 797 rules - Does Bacteriostatic Water Need to Be Refrigerated?: full storage guide with 28-day protocol - Where to Buy Bacteriostatic Water for Injection: sourcing guide with price comparison and vendor red flags - How to Inject BPC-157: injection technique guide covering reconstitution through disposal - Getting Started with Peptides: complete beginner guide covering supplies, reconstitution, and injection technique
Related Articles
How Long Does Bac Water Last?
Unopened bacteriostatic water lasts until manufacturer expiry (2-3 years). After puncture, it lasts 28 days refrigerated at 2-8°C per USP 797.
Bacteriostatic vs Sterile Water
Bacteriostatic water contains 0.9% benzyl alcohol for 28-day multi-use. Sterile water has no preservative and is single-use only.
Bacteriostatic Water Refrigeration
Unopened bacteriostatic water stores at room temperature. Once opened or punctured, refrigerate at 2-8°C and use within 28 days.
Where to Buy Bacteriostatic Water
Where to buy bacteriostatic water for injection from pharmacies, medical suppliers, and online retailers. USP grade requirements and prices.