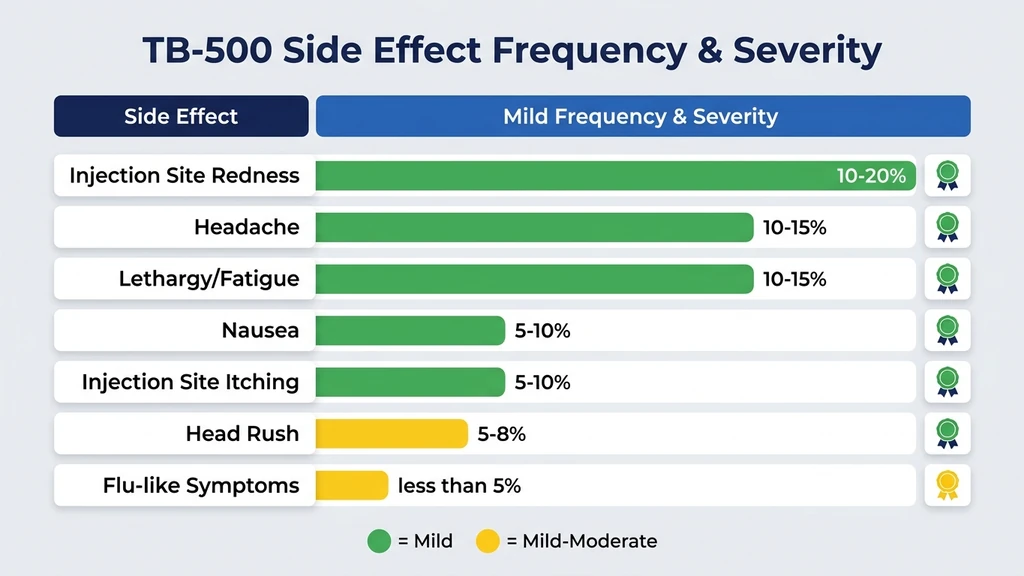
You are reading the label on a vial of TB-500, weighing whether the healing benefits justify the risks. The short answer: TB-500 side effects are mild for most users. Headache, temporary lethargy, and injection site redness are the most commonly reported issues, each affecting roughly 10 to 15% of users based on anecdotal reports and veterinary data. Serious adverse events are rare in published literature.
TB-500 is a synthetic fragment of thymosin beta-4 (TB-4), a 43-amino-acid protein your body produces naturally. TB-4 regulates cell migration, wound healing, and inflammation. TB-500 replicates the active region of this protein (amino acids 17 to 23), and it has been studied primarily in equine medicine and preclinical models. No human clinical trials have reached Phase 3. Every safety claim below comes from animal studies, in vitro research, or self-reported user data. Consult a healthcare provider before using any research peptide.
| Side Effect | Estimated Frequency | Severity | Typical Onset | What to Do |
|---|---|---|---|---|
| Headache | 10-15% | Mild | 1-4 hours post-injection | OTC pain relief, hydration |
| Lethargy / fatigue | 10-15% | Mild | 2-6 hours post-injection | Dose in the evening, rest |
| Nausea | 5-10% | Mild | 30-60 minutes post-injection | Take with food, reduce dose |
| Injection site redness | 10-20% | Mild | Immediate to 2 hours | Rotate sites, cool compress |
| Injection site itching | 5-10% | Mild | Immediate to 1 hour | Hydrocortisone cream |
| Head rush / lightheadedness | 5-8% | Mild | Minutes post-injection | Sit or lie down, slower injection |
| Temporary flu-like symptoms | <5% | Mild to moderate | 6-24 hours post-injection | Rest, hydration, dose reduction |
These frequency estimates come from aggregated veterinary reports and user surveys across peptide research communities. No controlled human trial has quantified TB-500 side effect rates with statistical precision. For dosing protocols that minimize side effects, see our TB-500 dosage calculator. For a full overview of what TB-500 does, see what does TB-500 do. Last updated: March 2026.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Are the Most Common TB-500 Side Effects?
The side effects reported most frequently with TB-500 fall into two categories: systemic reactions (headache, lethargy, nausea) and local injection site reactions. Both categories are typically self-limiting, resolving within hours to days without intervention.
Headache
Headache is the single most frequently reported TB-500 side effect. Users describe it as a mild, diffuse tension headache appearing 1 to 4 hours after subcutaneous injection. It typically resolves within 4 to 8 hours.
The mechanism is likely related to TB-500's vasodilatory properties. Thymosin beta-4 promotes angiogenesis (new blood vessel formation) and modulates nitric oxide signaling, which can transiently lower blood pressure and trigger headache through the same pathway as nitroglycerin headaches (Crockford et al., Ann N Y Acad Sci, 2010). Standard over-the-counter analgesics like acetaminophen or ibuprofen are effective. If headaches persist beyond the first week of use, reduce the dose by 50% and reassess.
Lethargy and Fatigue
A noticeable drop in energy affects roughly 10 to 15% of TB-500 users, most commonly during the loading phase when doses are highest (typically 2 to 2.5 mg twice weekly). The fatigue is not debilitating. Users describe it as feeling slightly drained for 4 to 8 hours after injection.
Thymosin beta-4 modulates inflammatory cytokines including IL-1 and IL-6 (Goldstein et al., Expert Opin Biol Ther, 2012). This immune modulation may produce mild fatigue similar to the tiredness you feel when your immune system is actively fighting a cold. Dosing in the evening, before sleep, sidesteps this issue entirely for most users.
Nausea
Mild nausea occurs in an estimated 5 to 10% of users, typically within the first 30 to 60 minutes after injection. It is more common on an empty stomach and during the initial loading phase.
The fix is simple: eat a light meal 30 minutes before your injection. If nausea persists across multiple doses, reduce the per-injection amount. Splitting a 2.5 mg dose into two 1.25 mg injections 12 hours apart often eliminates nausea without reducing weekly intake.
Injection Site Reactions
Redness, mild swelling, and itching at the injection site are common with any subcutaneous peptide injection. TB-500 is no exception. These reactions affect 10 to 20% of users and resolve within 1 to 4 hours.
Rotate injection sites between the abdomen, thigh, and upper arm. Inject at least 2 inches from the navel and 2 inches from any previous injection site. Let the alcohol swab dry fully before inserting the needle. A cool compress reduces redness in under 30 minutes. Persistent itching responds to over-the-counter hydrocortisone cream. For detailed injection technique, see our peptide safety guide.
Head Rush and Lightheadedness
A brief lightheaded sensation immediately after injection affects 5 to 8% of users. This likely reflects a transient vasodilatory response. TB-500 upregulates endothelial nitric oxide synthase (eNOS), briefly widening blood vessels and reducing blood pressure (Qiu et al., J Cell Biochem, 2009).
Sit or lie down for 5 minutes after injection. Inject slowly over 10 to 15 seconds rather than all at once. Stay well hydrated on injection days. If lightheadedness is severe or persists beyond a few minutes, reduce the dose.
Does TB-500 Cause Cancer? The Thymosin Beta-4 Cell Migration Debate
This is the most serious concern people raise about TB-500, and it deserves a thorough, evidence-based answer. The worry stems from a real biological mechanism: thymosin beta-4 promotes cell migration, and cancer cells use migration to metastasize. The question is whether supplying extra TB-4 (via TB-500) accelerates that process.
Why the Concern Exists
Thymosin beta-4 is a key regulator of actin polymerization, the process that gives cells the ability to move. When you cut your skin, TB-4 signals keratinocytes and endothelial cells to migrate into the wound bed and close the gap. This same migration machinery is hijacked by cancer cells to spread from a primary tumor to distant organs.
Several in vitro studies have shown that thymosin beta-4 is overexpressed in certain tumor types, including melanoma, colorectal cancer, and non-small cell lung cancer (Wang et al., Cancer Res, 2006). Cancer cells appear to upregulate their own TB-4 production to enhance motility. This association between elevated TB-4 levels and tumor aggressiveness is the origin of the concern.
What the Evidence Actually Shows
The critical distinction: correlation between elevated TB-4 in tumors and causation of cancer by exogenous TB-500 are two different things. The current evidence does not support the claim that administering TB-500 causes cancer or accelerates tumor growth.
Three key findings:
In equine studies, TB-500 (marketed as Thymosin Beta-4 for veterinary use) has been administered to thousands of racehorses for tendon and ligament injuries since the early 2000s. No increase in cancer incidence has been reported in treated animals compared to untreated populations (Goldstein & Kleinman, Ann N Y Acad Sci, 2010).
A 2010 study in the Annals of the New York Academy of Sciences directly tested whether exogenous TB-4 promotes tumor growth in mice. The researchers found that systemic administration of TB-4 did not increase tumor size, tumor number, or metastatic spread in a melanoma mouse model (Goldstein & Kleinman, Ann N Y Acad Sci, 2010).
Thymosin beta-4 is present in every nucleated cell in your body at concentrations of 0.1 to 0.5 mM. It is the most abundant actin-sequestering protein in human cells. Adding a few milligrams weekly through TB-500 injection represents a small increment on top of a massive baseline (Huff et al., Int J Biochem Cell Biol, 2001).
The Precautionary Position
The absence of evidence for harm is not the same as evidence of safety. No long-term controlled human study has tracked cancer outcomes in TB-500 users over 5, 10, or 20 years. Such a study does not exist and may never be conducted for a research peptide.
The conservative clinical recommendation: if you have active cancer, a history of cancer within the last 5 years, or a known genetic predisposition to cancer (BRCA mutations, Lynch syndrome, familial adenomatous polyposis), avoid TB-500 until human data clarifies the relationship. The theoretical risk of promoting cell migration in someone with dormant micrometastases, even if unproven, is not worth taking when your body already produces TB-4 endogenously and the exogenous dose adds marginal supply.
For healthy individuals without cancer history, the available evidence from animal models and over two decades of veterinary use suggests a low risk profile. Discuss your individual situation with a healthcare provider.
Thymosin Beta-4 vs. TB-500: Does the Distinction Matter for Safety?
Online peptide communities often use "TB-500" and "thymosin beta-4" interchangeably. They are not the same molecule, and the difference matters for safety assessment.
Thymosin beta-4 is the full 43-amino-acid protein (molecular weight: 4,921 Da). TB-500 is a synthetic peptide fragment corresponding to the active region of TB-4, centered on the actin-binding domain at amino acids 17 to 23 (sequence: LKKTETQ). Some commercial TB-500 products contain the full-length TB-4 sequence; others contain only the active fragment. Labeling is inconsistent across suppliers (Crockford et al., Ann N Y Acad Sci, 2010).
This matters because the safety data from published studies uses full-length thymosin beta-4, not the truncated fragment marketed as TB-500. Extrapolating safety conclusions from TB-4 research to a potentially different molecule requires caution. The active fragment may have a narrower range of biological activity, which could mean fewer side effects, or it could mean unanticipated effects not seen with the full protein.
If you are sourcing TB-500, verify whether the product contains the full-length TB-4 sequence or the fragment. Third-party certificate of analysis (COA) testing confirms identity and purity. For guidance on evaluating peptide quality, see our peptide safety guide.
What Happens if You Take Too Much TB-500?
Overdose scenarios with TB-500 are poorly documented because no controlled human dosing studies exist. The information below comes from veterinary case reports and user accounts.
Acute Overdose Scenario
A user reconstitutes a 5 mg vial with 1 mL of bacteriostatic water and accidentally injects the entire vial instead of half. They receive 5 mg in a single injection, double the typical 2.5 mg loading dose. The expected result: more pronounced headache, significant lethargy lasting 8 to 12 hours, possible nausea and lightheadedness, and a larger injection site reaction.
No life-threatening overdose has been documented in veterinary or anecdotal literature. Equine doses of TB-4 range from 10 to 20 mg (for a 500 kg animal), providing a rough sense of the therapeutic window. The fix: skip your next scheduled dose, return to normal dosing, and use our TB-500 dosage calculator to double-check your reconstitution math.
Chronic Overuse Scenario
A user runs a loading phase at 5 mg twice weekly (double the standard protocol) for 8 weeks, then continues at 2.5 mg twice weekly indefinitely without cycling off. The risks: sustained immune modulation could theoretically blunt natural immune responses over time. Thymosin beta-4 modulates T-cell maturation and inflammatory signaling (Goldstein et al., Expert Opin Biol Ther, 2012). Continuous high-dose exogenous supply may downregulate endogenous production through negative feedback loops, though this has not been confirmed in research.
Standard protocols recommend a loading phase of 4 to 6 weeks followed by maintenance dosing at reduced frequency. Cycling (4 to 6 weeks on, 2 to 4 weeks off) is commonly practiced to avoid tolerance and allow endogenous production to normalize. See our guide on BPC-157 vs TB-500 for stacking and cycling strategies.
Drug Interactions and Contraindications
TB-500 drug interaction data is limited to mechanistic reasoning and theoretical pharmacology. No clinical drug interaction studies have been conducted in humans.
Potential Drug Interactions
Anticoagulants and antiplatelet agents (warfarin, heparin, aspirin, clopidogrel). Thymosin beta-4 promotes angiogenesis and modulates platelet function. Combining TB-500 with blood thinners could theoretically increase bleeding risk. If you take any anticoagulant, discuss TB-500 with your prescribing physician before use.
Immunosuppressants (prednisone, methotrexate, cyclosporine). TB-4 modulates immune function through T-cell differentiation and cytokine regulation. Adding an immune-modulating peptide on top of immunosuppressive therapy creates an unpredictable interaction. The effects could be additive (over-suppression) or antagonistic (reduced drug efficacy).
Anti-angiogenic cancer therapies (bevacizumab, sunitinib). TB-500 promotes blood vessel growth. Anti-angiogenic cancer drugs block blood vessel growth. These mechanisms directly oppose each other. Never combine TB-500 with any anti-angiogenic therapy. Use the peptide interaction checker to evaluate specific combinations.
Absolute Contraindications
Active cancer of any type. TB-500 promotes cell migration and angiogenesis. Both processes could theoretically benefit tumor growth and metastasis. Until human data proves otherwise, active cancer is an absolute contraindication.
Pregnancy and breastfeeding. Thymosin beta-4 plays a role in embryonic development, particularly in cardiac and vascular formation (Smart et al., Nature, 2007). Introducing exogenous TB-4 during pregnancy could interfere with fetal development. No reproductive toxicology studies exist for TB-500. The risk is unknown and unacceptable.
Age under 18. Children and adolescents have naturally high levels of thymosin beta-4 supporting growth and development. No safety data exists for pediatric use. The developing immune and vascular systems should not be exposed to exogenous immune-modulating peptides without clinical supervision.
Relative Contraindications
History of cancer (in remission). Discuss with your oncologist. The 5-year cancer-free benchmark is a commonly cited threshold, but this is a judgment call based on individual risk factors, cancer type, and your physician's assessment.
Autoimmune conditions (rheumatoid arthritis, lupus, MS). TB-4 modulates immune function. In some autoimmune conditions this modulation could be beneficial; in others it could trigger flares. Clinical guidance does not exist. Proceed only under medical supervision.
Severe cardiovascular disease. The vasodilatory and angiogenic effects of TB-500 could theoretically stress a compromised cardiovascular system. Patients with heart failure, recent MI, or unstable angina should avoid TB-500.
How to Monitor for Side Effects During a TB-500 Protocol
Because TB-500 lacks the structured safety monitoring of FDA-approved drugs, self-monitoring becomes your responsibility. A simple protocol reduces risk significantly.
Before Starting: Baseline Labs
Get blood work before your first injection. The minimum panel: complete blood count (CBC), comprehensive metabolic panel (CMP), C-reactive protein (CRP), and liver enzymes (ALT, AST). If you have any cancer risk factors, add a cancer-specific screening appropriate to your age and sex (PSA for men over 40, for example).
These baseline values give you a comparison point. Without them, you cannot detect changes that might indicate a problem. Store the results digitally so you can compare them at 6-week intervals.
During the Loading Phase: Weekly Check-ins
The loading phase (typically 2 to 2.5 mg twice weekly for 4 to 6 weeks) carries the highest side effect risk because doses are at their peak. Track three things weekly: injection site reactions (photograph any redness or swelling for comparison), energy levels on a simple 1 to 10 scale, and any new or unusual symptoms.
If headaches worsen rather than improve over the first 2 weeks, reduce the dose by 50%. If nausea persists beyond 3 doses, split injections into smaller amounts. If you develop any new lump or growth anywhere on your body, stop TB-500 and see a physician immediately.
During Maintenance: Monthly Awareness
Once you transition to maintenance dosing (2.5 mg once weekly or less), side effects typically diminish. Repeat blood work at 6 weeks and 12 weeks after starting. Compare against your baseline. Flag any significant changes in liver enzymes, inflammatory markers, or blood cell counts for your healthcare provider.
The practical reality: most users experience side effects only during the first 1 to 2 weeks. By the end of the loading phase, the body has adjusted. Maintenance dosing at lower frequency rarely produces noticeable adverse effects. For stacking protocols that include monitoring guidance, see our peptide stacking guide.
TB-500 Side Effects Compared to BPC-157
TB-500 and BPC-157 are frequently stacked together for injury recovery. Their side effect profiles differ in meaningful ways.
| Parameter | TB-500 | BPC-157 |
|---|---|---|
| Most common side effect | Headache (10-15%) | Nausea (5-10%) |
| Injection site reactions | Moderate (10-20%) | Mild (5-10%) |
| Lethargy | Common during loading | Uncommon |
| Cancer concern | Theoretical (promotes cell migration) | Minimal (no pro-migratory mechanism) |
| Systemic vasodilation | Yes (lightheadedness) | No |
| Key contraindication | Active cancer | Active GI bleeding |
| Human clinical trials | None completed | None completed (Phase 2 for IBD ongoing) |
BPC-157 has a gastric protective mechanism and primarily works through upregulation of growth hormone receptors and nitric oxide pathways in the gut. Its side effect profile is narrower because it does not share TB-500's strong pro-angiogenic and cell migration properties. Stacking both peptides does not appear to compound side effects based on user reports, though no controlled study has evaluated the combination. For recovery-focused protocols, see peptides for recovery.
When to Stop TB-500 and Seek Medical Attention
Most TB-500 side effects resolve on their own. The following situations require you to stop immediately and contact a healthcare provider:
Severe allergic reaction. Difficulty breathing, swelling of the face or throat, widespread hives, or rapid heartbeat within minutes of injection. This is anaphylaxis. Call emergency services.
Persistent chest pain or palpitations. TB-500's vasodilatory effects can unmask underlying cardiac issues. Any chest pain lasting more than a few minutes warrants evaluation.
New lump or growth. Any unexplained mass, swelling, or skin lesion that appears during TB-500 use should be evaluated by a physician. This is a precaution related to the theoretical cell migration concern, not evidence that TB-500 caused the growth.
Signs of infection at injection site. Redness that spreads rather than resolves, warmth, pus, red streaking from the site, or fever above 100.4 F (38 C). This indicates bacterial contamination, not a TB-500 side effect, but it requires prompt treatment with antibiotics.
Severe, persistent headache. A headache that does not respond to OTC analgesics and lasts more than 24 hours after injection is unusual and warrants medical evaluation.
Frequently Asked Questions
What are the most common TB-500 side effects?
Headache (10 to 15%), injection site redness (10 to 20%), lethargy (10 to 15%), and mild nausea (5 to 10%) are the most frequently reported TB-500 side effects. These are based on aggregated user reports and veterinary data, not controlled human trials. Most resolve within hours and diminish after the first 1 to 2 weeks of use.
Does TB-500 cause cancer?
No evidence from animal studies or veterinary use shows that exogenous TB-500 causes cancer. The concern stems from thymosin beta-4's role in promoting cell migration, a mechanism cancer cells exploit. A 2010 mouse study found no increased tumor growth with systemic TB-4 administration (Goldstein & Kleinman, Ann N Y Acad Sci, 2010). People with active cancer should still avoid TB-500 as a precaution.
Is TB-500 safe to inject?
TB-500 has a mild side effect profile based on available data. No serious adverse events have been documented in published veterinary or preclinical literature. It is not FDA-approved for human use, and no Phase 3 human trials exist. Use sterile injection technique, source from third-party tested suppliers, and consult a healthcare provider before starting.
How long do TB-500 side effects last?
Most TB-500 side effects resolve within 4 to 8 hours after injection. Headache and lethargy are the longest-lasting, occasionally persisting for 12 to 24 hours during the loading phase. Injection site redness fades within 1 to 4 hours. By the second week of use, most users report that side effects have diminished significantly or stopped entirely.
Can you take TB-500 with BPC-157?
TB-500 and BPC-157 are commonly stacked for injury recovery. User reports do not indicate compounded side effects from the combination. The two peptides work through different mechanisms: TB-500 promotes cell migration and angiogenesis, while BPC-157 upregulates growth hormone receptors and protects the GI tract. No controlled study has evaluated the combination's safety.
Who should not take TB-500?
People with active cancer, pregnant or breastfeeding women, and anyone under 18 should avoid TB-500. Those taking anticoagulants, immunosuppressants, or anti-angiogenic cancer therapies should consult their physician first. People with a cancer history within the last 5 years should discuss the theoretical cell migration risk with their oncologist before use.
Does TB-500 affect the immune system?
Yes. Thymosin beta-4 modulates T-cell maturation and inflammatory cytokines including IL-1 and IL-6 (Goldstein et al., Expert Opin Biol Ther, 2012). This immune modulation is generally mild and may contribute to the fatigue some users experience during the loading phase. People with autoimmune conditions should use TB-500 only under medical supervision.
What blood work should you get before starting TB-500?
Get a complete blood count (CBC), comprehensive metabolic panel (CMP), C-reactive protein (CRP), and liver enzymes (ALT, AST) before your first injection. Repeat at 6 and 12 weeks. These baselines allow you to detect changes in immune markers, liver function, or inflammatory status that might indicate a problem.
The Bottom Line
TB-500's side effect profile is mild compared to most injectable peptides. Headache, lethargy, and injection site reactions are the norm. They peak during the loading phase and fade within 1 to 2 weeks for most users.
The cancer question has a nuanced answer: thymosin beta-4 promotes cell migration, cancer cells exploit cell migration, but direct evidence that exogenous TB-500 promotes tumor growth does not exist in published research. If you have active cancer or recent cancer history, avoid it. For everyone else, the available animal and veterinary data suggests low risk.
The biggest safety gap is the absence of controlled human trials. Every frequency estimate and safety claim above carries that caveat. Monitor your own response with baseline blood work, track side effects during the loading phase, and work with a healthcare provider who understands peptide protocols. Use the TB-500 dosage calculator to dial in your dose. For stacking with BPC-157, see BPC-157 vs TB-500. For broader safety principles, see the peptide safety guide. Explore dosage tools and peptide profiles on our homepage.
Related Articles: - What Does TB-500 Do? - BPC-157 vs TB-500 - Peptides for Recovery - Peptide Stacking Guide - Peptide Safety Guide
Related Articles
BPC-157 TB-500 Side Effects: What the Research Shows About Stack Safety
BPC-157 TB-500 stack side effects: headaches, fatigue weeks 1-2, TB-500 anaphylaxis risk, cancer screening recommendation. Week-by-week timeline and when to stop.
Peptide Therapy Side Effects: Complete Guide (2026)
Peptide therapy side effects by category: GLP-1, GH-releasing, healing, and immune. Severity matrix, incidence rates, and management strategies.
Can Tirzepatide Cause Joint Pain? Clinical Data and Relief Strategies
Tirzepatide does not directly cause joint pain. SURMOUNT trial arthralgia rates match placebo. Learn 5 indirect causes, uric acid risks, and 8 relief strategies backed by clinical evidence.
Is Sermorelin Safe? Risks & Data
Is sermorelin safe? Review clinical trial data, side effect rates (injection site reactions in 17%), contraindications, and monitoring protocols.