
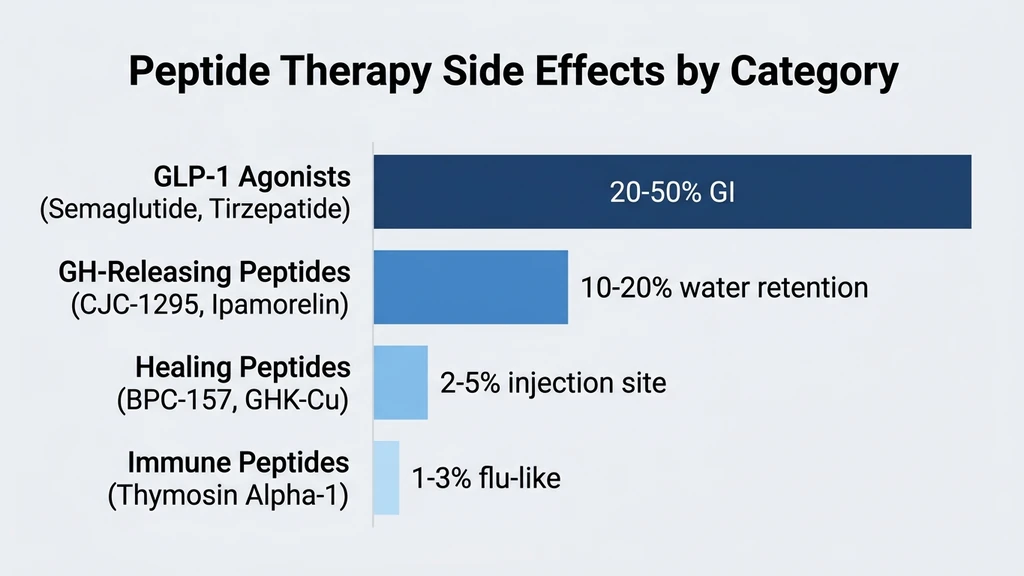
You are considering peptide therapy and want to know the real risks before starting. Peptide side effects vary dramatically by category: GLP-1 agonists (semaglutide, tirzepatide) cause GI symptoms in 20-50% of users, GH-releasing peptides (CJC-1295, Ipamorelin) may cause water retention and joint stiffness in 10-20%, and healing peptides (BPC-157, GHK-Cu) have the mildest profiles with mainly injection-site reactions. Most side effects are dose-dependent, temporary, and manageable with proper medical supervision (PMC5632578).
| Quick Reference | Details |
|---|---|
| Highest side effect rates | GLP-1 agonists (20-50% GI) |
| Moderate side effect rates | GH peptides (10-20%) |
| Lowest side effect rates | Healing peptides (BPC-157, GHK-Cu) |
| Most common across all | Injection site reactions |
| Universal rule | Start low, go slow |
| Monitoring required | Blood work before and during treatment |
| FDA-approved peptides | Semaglutide, tirzepatide, PT-141 |
| Research peptides | BPC-157, CJC-1295, GHK-Cu, TB-500 |
Use our peptide interaction checker before combining peptides. For dosing that minimizes side effects, see our peptide dosage calculator.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
How Common Are Peptide Side Effects?
The answer depends entirely on which peptide you are using. Grouping all peptides together is like asking "do medications have side effects?" The range spans from 50% GI symptom rates (semaglutide at maximum dose) to less than 5% for injection-site reactions with GHK-Cu.
| Peptide Category | Overall AE Rate | Serious AE Rate | Discontinuation Rate |
|---|---|---|---|
| GLP-1 agonists | 40-70% | 2-5% | 5-15% |
| GH-releasing | 15-30% | <1% | <3% |
| Healing (BPC-157, TB-500) | 5-15% | Very rare | <1% |
| Sexual (PT-141) | 40-60% | <1% | ~10% |
| Immune (Thymosin Alpha-1) | 5-10% | Very rare | <1% |
Dose-response is the dominant pattern. Higher doses produce more side effects across every category. Slow titration reduces incidence by 30-50% for GLP-1 agonists and GH peptides. This is why "start low, go slow" is the universal principle in peptide therapy.
GLP-1 Agonist Side Effects: Semaglutide, Tirzepatide, Retatrutide
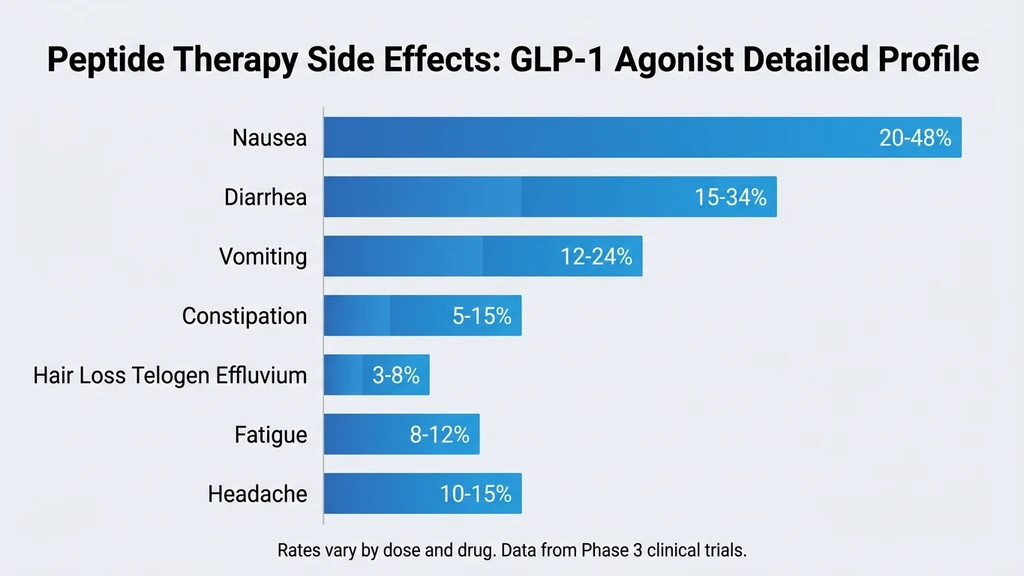
GLP-1 receptor agonists carry the highest side-effect burden of any peptide category. This is expected: they are the most pharmacologically potent peptides, producing 15-24% body weight loss in clinical trials. The side effects are the price of that efficacy (US Pharmacist, GI AEs of GLP-1 RAs).
Gastrointestinal: Nausea, Diarrhea, Vomiting, Constipation
GI symptoms are the signature side effect of GLP-1 drugs. They result from delayed gastric emptying (GLP-1 receptor activation) and, in retatrutide, additional glucagon receptor stimulation.
| Drug | Nausea | Diarrhea | Vomiting | Constipation |
|---|---|---|---|---|
| Semaglutide 2.4 mg | 44% | 30% | 24% | 24% |
| Tirzepatide 15 mg | 33% | 23% | 12% | 11% |
| Retatrutide 12 mg | 48% | 34% | 20% | 16% |
These symptoms peak during dose escalation (first 1-2 weeks at each new dose) and typically resolve within 3-4 weeks at a stable dose. Slow titration with 4-week holds between increases cuts GI adverse events by approximately 50%. See our individual side effect guides: semaglutide nausea, retatrutide diarrhea, and retatrutide nausea.
Hair Loss: Telogen Effluvium
Hair shedding is increasingly reported with GLP-1 drugs, though it is caused by rapid weight loss rather than the drug mechanism itself. Telogen effluvium occurs when caloric deficit shunts hair follicles from the growth phase into the resting phase. With 15-24% body weight loss, the risk is substantial.
Semaglutide and tirzepatide trials reported hair loss in approximately 3-5% of participants. Real-world rates appear higher. The shedding typically begins 2-4 months after significant weight loss and resolves 6-12 months after weight stabilizes. Read our semaglutide hair loss guide and retatrutide hair loss guide for prevention strategies.
Fatigue and Dizziness
Reduced caloric intake from appetite suppression can cause fatigue in the first 4-8 weeks. Dizziness may result from dehydration (especially when combined with GI fluid losses) or from blood pressure changes. Maintaining adequate protein intake (1.0-1.2 g/kg/day) and hydration (2-3 liters daily) reduces both symptoms.
See our guide on semaglutide fatigue for management.
Pancreatitis Risk
Acute pancreatitis has been reported in GLP-1 clinical trials at rates of 0.1-0.3%, compared to 0.1% on placebo. The FDA includes a boxed warning about pancreatitis risk on semaglutide labels. Symptoms include severe upper abdominal pain radiating to the back, nausea, and vomiting.
Stop the medication and seek emergency care if pancreatitis is suspected. Patients with a history of pancreatitis should avoid GLP-1 agonists.
Gallbladder Issues
Rapid weight loss increases gallstone formation. Semaglutide trials reported cholelithiasis in 1.5-2.6% of participants versus 0.7% on placebo. The risk correlates with the speed and magnitude of weight loss. Maintaining some dietary fat intake during weight loss may reduce risk.
Growth Hormone Peptide Side Effects: CJC-1295, Ipamorelin, Sermorelin
GH-releasing peptides have a milder side-effect profile than GLP-1 drugs. Most adverse events are mild and self-limiting. The primary concern is not acute toxicity but chronic elevation of IGF-1 and its theoretical long-term implications (PMC5632578).
Water Retention and Joint Stiffness
GH promotes sodium and water retention. Within the first 2-4 weeks of starting a GH-releasing peptide, many users notice mild edema in the hands, feet, or face, along with joint stiffness. This affects approximately 10-20% of users and resolves within 2-3 weeks at a stable dose.
Reducing sodium intake and starting at a lower dose minimizes this effect. If edema persists beyond 4 weeks, reduce the dose by 25-50%.
Tingling and Numbness (Paresthesia)
GH-mediated fluid shifts can compress peripheral nerves, producing tingling or numbness in the hands and feet. This mimics early carpal tunnel symptoms. The incidence is approximately 5-10% and is dose-dependent.
Lower the dose if paresthesia persists beyond 2 weeks. It resolves quickly once GH levels normalize.
Headaches
Headaches are reported in 5-15% of users during the first 1-2 weeks. They are typically mild and responsive to standard analgesics. The mechanism likely involves GH-mediated fluid shifts and intracranial pressure changes. Adequate hydration helps.
Elevated Blood Sugar and Insulin Resistance
GH is a counter-regulatory hormone to insulin. It raises blood glucose by promoting hepatic gluconeogenesis and reducing peripheral glucose uptake. In healthy adults, this effect is modest. In men with pre-existing insulin resistance or prediabetes, GH peptides can push fasting glucose above normal range.
Monitor fasting glucose and HbA1c at baseline and every 3 months. Men on metformin or sulfonylureas need closer monitoring. Sermorelin produces less glucose disruption than CJC-1295 due to its lower GH potency.
Potential Tissue Growth Concerns
Chronic GH elevation stimulates cell proliferation. Epidemiological studies associate elevated IGF-1 with increased cancer risk, though a causal link has not been established in GH peptide users specifically. This is a theoretical concern that demands monitoring, not a proven danger.
Keep IGF-1 within the age-adjusted normal range. Quarterly monitoring for anyone using GH-releasing peptides for more than 3 months. Men over 50 should also monitor PSA every 3-6 months.
Healing Peptide Side Effects: BPC-157, TB-500, GHK-Cu
Healing peptides have the mildest overall side-effect profile. No serious adverse events have been documented in the available literature, though human clinical trial data is limited for BPC-157 and TB-500 (PMC12446177).
Injection Site Reactions
The most common side effect across all healing peptides: redness, swelling, or mild pain at the injection site. Affects approximately 5-10% of users. Rotating injection sites and proper technique minimizes occurrence.
GHK-Cu topical application can cause skin redness and mild irritation in approximately 3-5% of users, especially at concentrations above 2%. This is not an allergic reaction; it resolves with lower concentration or less frequent application.
Mild Nausea and Dizziness
BPC-157 produces mild nausea in approximately 3-5% of users, typically only at higher doses (500+ mcg). Dizziness is reported rarely. Both symptoms are transient, lasting minutes to hours, and resolve with continued use.
TB-500 has similar rates. GHK-Cu injections rarely produce systemic symptoms.
Headaches
Mild headaches are reported in approximately 2-5% of BPC-157 and TB-500 users. They are dose-dependent and usually self-limiting. Adequate hydration and starting at the lower end of the dose range prevents most cases.
Theoretical Cancer Growth Concerns
BPC-157 and TB-500 promote angiogenesis (blood vessel formation) and cell proliferation as part of their healing mechanism. The theoretical concern: these same processes could feed a pre-existing tumor. USADA has flagged this risk (USADA, 2024).
No clinical evidence supports this concern in practice. No tumor growth has been reported in any BPC-157 or TB-500 study, animal or human. The risk remains theoretical but warrants caution in anyone with active or recent cancer. See our BPC-157 side effects guide and TB-500 side effects guide.
Sexual Function Peptide Side Effects: PT-141
PT-141 (bremelanotide) has a distinct side-effect profile driven by melanocortin receptor activation. It is the only peptide category where nausea is both common and expected as part of the mechanism.
Nausea: 40% of Users
Nausea is the most common PT-141 side effect, affecting approximately 40% of users. It results from melanocortin receptor activation in the brainstem area postrema. The nausea is typically mild to moderate, onset within 15-30 minutes of injection, and resolves within 1-2 hours.
First-dose nausea is more severe than subsequent doses. Taking 25 mg ondansetron (Zofran) 30 minutes before PT-141 can reduce nausea significantly. See our peptide therapy side effects guide for cross-peptide management.
Flushing: 20% of Users
Facial and upper body flushing affects approximately 20% of users. It results from melanocortin-mediated vasodilation. The flushing is temporary (30-60 minutes) and cosmetic rather than medically concerning.
Blood Pressure Changes
PT-141 causes a transient increase in blood pressure: approximately +6 mmHg systolic and +3 mmHg diastolic. In healthy adults, this is clinically insignificant. In men with uncontrolled hypertension or cardiovascular disease, it adds risk.
Blood pressure monitoring is required before the first dose. Those on antihypertensive medications should coordinate with their prescriber.
Headaches: 11% of Users
Headaches affect approximately 11% of PT-141 users. They typically onset within 30-60 minutes of injection and resolve within 2-4 hours. Standard analgesics are effective. The mechanism involves melanocortin-mediated cerebrovascular changes.
Immune Peptide Side Effects: Thymosin Alpha-1, LL-37, KPV
Immune-modulating peptides have favorable safety profiles overall. Thymosin Alpha-1 has the most clinical data, having been approved in over 35 countries.
Injection Site Reactions
Mild redness and soreness at injection sites affect 3-8% of Thymosin Alpha-1 users. LL-37 injections may produce slightly more local inflammation due to its antimicrobial mechanism. KPV injections are well tolerated with minimal site reactions.
Flu-Like Symptoms
Thymosin Alpha-1 can produce mild flu-like symptoms (low-grade fever, fatigue, body aches) in the first 1-3 days of use. This reflects immune activation and typically resolves spontaneously. It affects approximately 5-10% of users and is more common with the first dose.
Autoimmune Considerations
Immune-stimulating peptides carry theoretical risk for people with autoimmune conditions. Thymosin Alpha-1 modulates rather than simply stimulates the immune system, which may actually benefit autoimmune conditions. Studies in hepatitis patients showed no autoimmune flares (Romani et al., Immunol Lett, 2007).
LL-37 has a stronger pro-inflammatory profile and may trigger autoimmune flares in susceptible individuals. Use with caution in anyone with lupus, rheumatoid arthritis, or inflammatory bowel disease.
Side Effect Severity Matrix
This table summarizes the expected side effects by peptide category, severity, and management approach.
| Side Effect | GLP-1 Agonists | GH Peptides | Healing Peptides | PT-141 | Immune Peptides |
|---|---|---|---|---|---|
| Nausea | Moderate-Severe (20-48%) | Rare (<5%) | Mild (3-5%) | Moderate (40%) | Rare (<3%) |
| Diarrhea | Moderate (15-34%) | Rare (<3%) | Rare (<2%) | Rare (<5%) | Rare (<2%) |
| Headache | Mild (10-15%) | Mild (5-15%) | Mild (2-5%) | Mild (11%) | Rare (<5%) |
| Injection site | Mild (5%) | Mild (5-10%) | Mild (5-10%) | Mild (5%) | Mild (3-8%) |
| Water retention | N/A | Moderate (10-20%) | N/A | N/A | N/A |
| Blood sugar | Mild risk | Moderate risk | N/A | N/A | N/A |
| Blood pressure | Minimal | Minimal | N/A | Mild (+6 mmHg) | N/A |
| Hair loss | Moderate (3-5%) | N/A | N/A | N/A | N/A |
| Flushing | Rare | Mild (5%) | N/A | Moderate (20%) | Rare |
Severity scale: Mild (does not limit daily activity), Moderate (may limit some activities), Severe (requires medical intervention or treatment discontinuation).
How to Minimize Peptide Side Effects
Five principles apply across every peptide category. Following all five reduces side-effect incidence by an estimated 40-60%.
Start Low, Go Slow
Begin at 50-75% of the standard dose for any new peptide. Hold for 2-4 weeks before increasing. This is the single most effective strategy for reducing side effects across all categories. GLP-1 trials proved it: slow escalation cut GI adverse events by approximately 50% (Rosenstock et al., Lancet, 2023).
Proper Reconstitution and Storage
Improperly reconstituted peptides can cause increased injection-site reactions and reduced efficacy. Use bacteriostatic water for multi-dose vials. Refrigerate at 36-46F (2-8C). Never freeze. Never shake. Read our how to reconstitute peptides guide and how to store peptides guide.
Injection Technique and Site Rotation
Rotate injection sites to prevent lipodystrophy and reduce local reactions. Abdomen, thigh, upper arm, and buttock are all appropriate subcutaneous sites. Use a new needle for each injection. Clean the site with alcohol before injecting. See our how to inject peptides guide.
Medical Monitoring and Blood Work
Baseline labs before starting any peptide: metabolic panel, CBC, fasting glucose. Add IGF-1 for GH peptides. Add PSA for men over 40. Add hormonal panel for women. Recheck at 4-6 weeks and quarterly thereafter. Full protocol in our peptide safety guide.
This catches problems early. Elevated IGF-1, rising fasting glucose, or changing PSA values trigger dose adjustment before symptoms appear.
Knowing When to Stop
Discontinue and contact your prescriber if you experience: severe abdominal pain (pancreatitis risk with GLP-1 drugs), persistent vomiting or inability to eat for 48+ hours, bloody stools, signs of dehydration, or any symptom that does not improve with dose reduction.
In GLP-1 clinical trials, 5-15% of participants discontinued due to adverse events. Discontinuation is a valid choice when the side-effect burden exceeds the benefit.
The Regulatory Landscape: FDA Peptide Oversight 2024-2026

The FDA has increased enforcement against unapproved peptide products since 2024. An FDA survey found that 23% of tested research peptide products were mislabeled: wrong peptide, wrong concentration, or contaminated with bacterial endotoxins. This contamination adds side-effect risk beyond the peptide itself.
Compounded semaglutide came under particular scrutiny. The FDA issued safety communications about salt form issues and purity concerns. Contaminated peptides can cause injection-site infections, sterile abscesses, and systemic inflammatory reactions that mimic drug side effects but are actually caused by impurities.
How to protect yourself: request third-party certificates of analysis (COAs) for every peptide you purchase. Look for purity above 98%. Use only sterile bacteriostatic water for reconstitution. Buy from suppliers that test for endotoxin content. Our peptide dosage chart includes sourcing recommendations.
Frequently Asked Questions
What are the most common peptide therapy side effects?
Injection site redness and swelling are the most universal across all peptide categories. For GLP-1 agonists specifically, nausea (20-48%), diarrhea (15-34%), and vomiting (12-24%) dominate. Healing peptides like BPC-157 and GHK-Cu have the mildest profiles, with mainly injection-site reactions at 5-10% incidence.
Are peptides safe long-term?
FDA-approved peptides (semaglutide, tirzepatide, PT-141) have established safety profiles from multi-year clinical trials. Research peptides (BPC-157, CJC-1295, GHK-Cu) lack long-term safety data because extended human trials have not been conducted. Medical supervision and regular blood monitoring every 3 months are essential for any long-term protocol.
Can peptides cause cancer?
This is a theoretical concern, not a proven risk. GH-releasing peptides and healing peptides promote cell proliferation as part of their mechanism. No clinical trial has demonstrated increased cancer incidence from peptide therapy. Chronic IGF-1 elevation is associated with cancer risk epidemiologically. Keep IGF-1 within age-adjusted normal range.
What peptide has the least side effects?
GHK-Cu and BPC-157 consistently report the mildest side-effect profiles. GHK-Cu (topical) has virtually no systemic side effects. BPC-157 produces mainly injection-site reactions in 5-10% of users. Sermorelin has the mildest profile among GH-releasing peptides due to its self-limiting mechanism.
How long do peptide side effects last?
GLP-1 GI symptoms peak during dose escalation (days 1-5 at each new dose) and resolve within 3-4 weeks at stable dosing. GH peptide water retention resolves in 2-3 weeks. PT-141 nausea lasts 1-2 hours per dose. BPC-157 injection-site reactions resolve within 24-48 hours. Most side effects are temporary.
Do I need to stop peptides if I get side effects?
Not necessarily. Most peptide side effects are mild, temporary, and manageable with dose adjustment. Reduce the dose by 25-50% and reassess after 1-2 weeks. Stop immediately for severe symptoms: persistent vomiting, bloody stools, severe abdominal pain, or dehydration. Consult your prescriber before restarting.
Are research peptides riskier than FDA-approved ones?
Research peptides carry additional risk from variable purity and contamination. An FDA survey found 23% of research peptides were mislabeled. FDA-approved peptides (semaglutide, tirzepatide) undergo strict manufacturing controls. Request certificates of analysis showing purity above 98% for any research peptide.
Can I take multiple peptides at once?
Yes, but start with one peptide at a time. Add a second only after 4-8 weeks of confirmed tolerability. Running multiple peptides from day one makes it impossible to identify which compound causes any side effect. Use our peptide interaction checker before combining compounds.
The Bottom Line
Peptide therapy side effects range from the significant (GLP-1 GI symptoms in 20-50% of users) to the negligible (GHK-Cu topical reactions in 3-5%). The severity depends on the peptide category, the dose, the speed of escalation, and individual tolerance.
Start low, go slow. Get baseline blood work. Monitor regularly. Rotate injection sites. Know when to reduce the dose and when to stop. These five principles reduce side-effect incidence by 40-60% across all peptide categories.
Use our peptide interaction checker before combining peptides. For individual side-effect profiles, see our guides on retatrutide side effects, BPC-157 side effects, GHK-Cu side effects, and CJC-1295/Ipamorelin side effects. Consult your physician before starting any peptide protocol.
Helpful Tools
Related Articles
Can Tirzepatide Cause Joint Pain? Clinical Data
Can tirzepatide cause joint pain? SURMOUNT arthralgia rates match placebo. Review 5 indirect causes, uric acid risks, and 8 relief tactics.
BPC-157 TB-500 Side Effects: Stack Safety Data
BPC-157 TB-500 stack side effects: headaches, fatigue weeks 1-2, anaphylaxis risk, cancer screening tips. Week-by-week timeline inside.
Does Semaglutide Cause Heartburn?
Does semaglutide cause heartburn? Yes, in 1.9-5% of users. Delayed gastric emptying mechanism, GERD differences, and 7 ways to manage it.
Does Tirzepatide Cause Insomnia?
Does tirzepatide cause insomnia? Trials say no, but 5 indirect pathways explain lost sleep. GI, blood sugar, and caloric deficit fixes.