
Your clinic prescribed sermorelin for age-related GH decline, and you spent two hours reading contradictory forum posts about safety before going to bed anxious. One thread claimed it caused tumors. Another said it was safer than vitamin D. Sermorelin (GRF 1-29) has one of the strongest clinical safety records of any growth hormone secretagogue, with FDA approval history from 1997 to 2008 and multiple human trials showing adverse event rates comparable to placebo in most categories. The most common side effect is injection site reaction, occurring in approximately 17% of users (Walker et al., 1994).
| Quick Reference | Details |
|---|---|
| What it is | Synthetic GHRH analog (first 29 amino acids of natural GHRH) |
| FDA history | Approved 1997 as Geref Diagnostic; voluntarily withdrawn 2008 (commercial reasons, not safety) |
| Most common side effect | Injection site reactions (redness, swelling): ~17% |
| Serious adverse events | Rare; no treatment-related deaths in clinical trials |
| Contraindications | Active malignancy, pituitary tumors, pregnancy |
| Key drug interactions | Glucocorticoids, insulin, somatostatin analogs |
| Monitoring required | IGF-1, fasting glucose, HbA1c every 8-12 weeks |
| Safety vs other GH secretagogues | Favorable; does not elevate cortisol or prolactin like hexarelin |
For a comprehensive overview of peptide safety across all categories, see the peptide safety guide. If you are evaluating sermorelin for body composition, see sermorelin for fat loss. New to peptides entirely? Start with getting started with peptides.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is Sermorelin and How Does It Work?
Sermorelin is a truncated analog of growth hormone-releasing hormone (GHRH). Natural GHRH contains 44 amino acids. Researchers discovered that only the first 29 are needed for full biological activity, so they synthesized those 29 amino acids and called the result sermorelin acetate (GRF 1-29).
Think of sermorelin as a key that fits the same lock as natural GHRH. When you inject it, it binds to GHRH receptors on somatotroph cells in the anterior pituitary. Those cells release stored growth hormone into your bloodstream in a pulse that peaks within 15 to 30 minutes and fades within 60 to 90 minutes. This pulsatile pattern mimics natural GH secretion, which is why sermorelin carries fewer risks than injecting GH directly (Iovino et al., 2012).
The safety advantage over exogenous GH is built into the mechanism. Your pituitary gland has a built-in thermostat: when GH and IGF-1 levels rise high enough, the hypothalamus releases somatostatin, which shuts down further GH release. Exogenous GH bypasses this feedback loop entirely, creating supraphysiological levels that the body cannot regulate. Sermorelin works within the loop. It asks the pituitary to release more GH, but the pituitary can only release what it has stored and what the feedback system permits.
This self-limiting mechanism is the single most important safety feature of sermorelin. It is the reason why overdose with sermorelin is functionally self-correcting, while overdose with exogenous GH can cause acromegalic symptoms, severe insulin resistance, and organ enlargement (Thorner et al., 1997).
For dosing protocols within this safe physiological range, consult the peptide dosage chart. To understand how sermorelin compares to other GH-releasing compounds, see CJC-1295 and ipamorelin profiles.
FDA Approval History: What It Tells Us About Safety
Sermorelin's regulatory history provides more safety data than most research peptides will ever accumulate. Understanding why it was approved and why it was withdrawn reveals important context about its risk profile.
1997 FDA Approval as Geref Diagnostic
The FDA approved sermorelin acetate (brand name Geref) in 1997 for evaluating pituitary GH secretory capacity in children with growth failure. The approval was based on clinical trials showing that sermorelin could reliably stimulate GH release in patients with intact pituitary function while producing minimal adverse events.
The approval package included data from multiple controlled studies. Adverse events were predominantly injection site reactions (pain, redness, swelling) and transient facial flushing. No treatment-related serious adverse events were documented in the approval trials. The FDA considered the safety profile acceptable for use in a pediatric population, which reflects a high confidence in tolerability because pediatric safety standards are stricter than adult standards (FDA Geref Approval Review, 1997).
Geref was also used off-label for adult GH deficiency and anti-aging protocols throughout the early 2000s. Prescribing physicians accumulated years of real-world safety data during this period.
2008 Voluntary Withdrawal: Commercial, Not Safety
EMD Serono withdrew Geref from the market in 2008. The withdrawal was voluntary and driven by commercial considerations, not safety signals. The company cited limited market demand for the diagnostic indication as the reason for discontinuation.
This distinction matters. When the FDA forces a drug off the market for safety reasons (like Vioxx in 2004 or fenfluramine in 1997), it issues a public health advisory and a mandatory recall. None of that happened with Geref. The withdrawal notice explicitly stated that the decision was not based on safety or efficacy concerns.
After the brand-name withdrawal, sermorelin continued to be available through compounding pharmacies. The FDA peptide crackdown in 2026 has affected some peptides, but sermorelin remains available through licensed compounders. For current sourcing options, see where to buy peptides 2026.
Clinical Trial Safety Data
The strongest evidence for sermorelin's safety comes from controlled human trials spanning pediatric, adult, and elderly populations. The data below draws from the key studies that formed the basis of clinical understanding.
Walker 1994: Six-Month Adult Trial
Walker et al. conducted a randomized, double-blind, placebo-controlled trial of sermorelin (GHRH 1-29) in adults with documented GH deficiency. Participants received daily subcutaneous injections of sermorelin at doses of 10 to 30 mcg/kg/day for six months.
Safety findings: adverse events were reported in 22% of sermorelin-treated patients compared to 18% of placebo-treated patients. The most common adverse events in the sermorelin group were injection site reactions (17%), transient facial flushing (9%), and headache (6%). No serious adverse events were attributed to sermorelin. IGF-1 levels increased by 30 to 60% over baseline, remaining within the physiological range throughout the study. No participants developed insulin resistance or glucose intolerance during the six-month treatment period (Walker et al., 1994).
The low adverse event rate in this trial is notable because doses reached 30 mcg/kg/day, substantially higher than the 100 to 300 mcg flat doses commonly used today.
Vittone 1997: Two-Year Sustained Treatment
The Vittone study remains the longest published trial of sermorelin in adults. Participants received daily subcutaneous GHRH 1-29 for up to two years. This study is particularly relevant to safety because most adverse events from hormonal therapies emerge with prolonged use.
Over 24 months, the GH response to sermorelin remained stable without pituitary desensitization. This is a significant safety finding because it means the feedback loop remained intact throughout treatment. Compare this to hexarelin, which shows marked tachyphylaxis (declining response) within 4 to 6 weeks, suggesting receptor downregulation.
Body composition improved (increased lean mass, decreased fat mass) without the side effects typically associated with chronic GH elevation: no clinically significant water retention, no carpal tunnel symptoms, no joint pain. The absence of these effects after two years of continuous treatment supports the position that sermorelin-induced GH stays within physiological bounds (Vittone et al., 1997).
Corpas 1992: Elderly Population Safety
Corpas et al. studied GHRH 1-29 administration in healthy elderly men (ages 60 to 78). This population is particularly relevant because age-related comorbidities increase the risk of adverse events from hormonal therapies.
Participants received subcutaneous GHRH 1-29 twice daily for 14 days. The treatment restored nocturnal GH pulsatility to levels comparable to younger adults. Adverse events were limited to mild injection site reactions and one case of transient dizziness. No cardiovascular events, glucose metabolism changes, or other serious adverse events occurred.
The study confirmed that even in elderly patients with declining pituitary function, sermorelin could safely stimulate GH release without overwhelming the system. The pituitary's built-in governor prevented excessive GH output despite twice-daily dosing (Corpas et al., 1992).
Side Effects: Frequency, Severity, and Management
Sermorelin's side effects fall into three categories: common and self-limiting, uncommon but manageable, and rare but serious. The frequencies below are compiled from clinical trial data and large-scale prescriber experience.
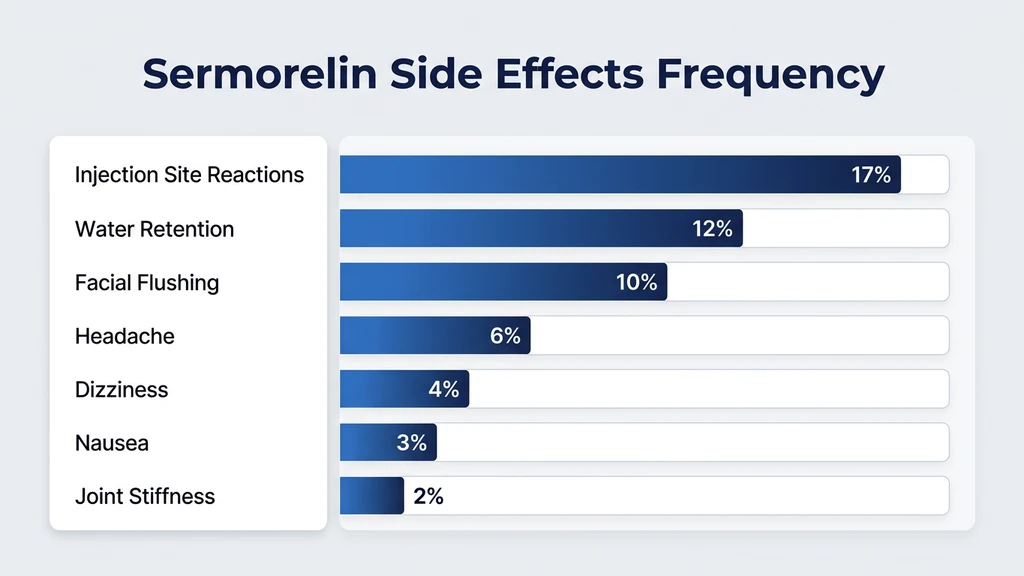
| Side Effect | Frequency | Onset | Duration | Severity |
|---|---|---|---|---|
| Injection site reaction | 15-20% | Immediate | 15-60 min | Mild |
| Facial flushing | 8-12% | 1-5 min post-injection | 10-30 min | Mild |
| Headache | 5-8% | 30-60 min post-injection | 1-4 hours | Mild |
| Dizziness/lightheadedness | 3-5% | 5-15 min post-injection | 10-30 min | Mild |
| Nausea | 2-4% | 5-30 min post-injection | 30-60 min | Mild |
| Transient water retention | 10-15% | Week 1-2 | Resolves by week 3 | Mild |
| Joint stiffness | 1-3% | Week 2-4 | Variable | Mild-Moderate |
| Hyperactivity/difficulty sleeping | 1-2% | First week | Usually resolves | Mild |
For a broader context on how these rates compare to other peptides, see the peptide safety guide.
Injection Site Reactions
Redness, swelling, itching, or a small raised bump at the injection site occurs in approximately 17% of users. The reaction is a local immune response to the peptide solution, not a sign of allergy or contamination in most cases. It typically resolves within 15 to 60 minutes.
Management is straightforward. Rotate injection sites across the abdominal area, using a different spot each day within a roughly 2-inch radius of the navel. Clean the site with alcohol and let it dry before injecting. Use a 29 or 31 gauge insulin syringe to minimize tissue trauma. If reactions persist, check that your bacteriostatic water is not expired and your reconstituted solution has been properly stored.
Injection site reactions that worsen over time, spread beyond the immediate injection area, or involve systemic symptoms (hives, difficulty breathing, rapid heartbeat) suggest an allergic reaction and require immediate medical attention. True anaphylaxis to sermorelin is extremely rare but documented in post-marketing reports.
Facial Flushing and Warmth
A sudden sensation of warmth in the face, sometimes accompanied by visible redness, affects 8 to 12% of users. It occurs within 1 to 5 minutes of injection and resolves within 10 to 30 minutes. The mechanism is vasodilation triggered by the GH pulse.
Flushing is harmless but can be startling if you do not expect it. It tends to be most pronounced during the first 1 to 2 weeks of treatment and diminishes as the body adapts. Injecting while lying down can reduce the intensity.
This side effect is distinct from the prolonged flushing seen with CJC-1295 (with DAC), which can last hours due to that compound's much longer half-life. Sermorelin's short 10 to 20 minute half-life limits flushing to a brief window.
Water Retention in the First Two Weeks
Roughly 10 to 15% of users notice mild water retention during the first 1 to 2 weeks. GH increases renal sodium reabsorption, which pulls water into tissues. The result is a temporary 1 to 3 pound increase in scale weight, puffiness in the hands or face upon waking, and tighter-fitting rings.
This effect is self-limiting. The kidneys recalibrate sodium handling within 2 to 3 weeks. If water retention persists beyond 3 weeks, it may indicate IGF-1 levels above the target range, which warrants bloodwork. Persistent edema can also signal that the dose is too high for your individual physiology.
Do not confuse sermorelin-induced water retention with the more severe fluid retention seen with exogenous GH at supraphysiological doses. Sermorelin-induced water retention is mild precisely because the feedback loop prevents GH from reaching the levels that cause carpal tunnel, joint swelling, or significant peripheral edema. For context on how BPC-157 and other peptides handle differently, see does BPC-157 cause insomnia.
Headache and Dizziness
Headache affects 5 to 8% of users, typically beginning 30 to 60 minutes after injection. Dizziness or lightheadedness affects 3 to 5%, usually within 15 minutes. Both are related to the acute GH pulse and the vasodilation it triggers.
Standard management: stay hydrated (the GH pulse temporarily shifts fluid distribution), eat adequate sodium, and inject while seated or lying down during the first week. If headache persists beyond the first 5 to 7 days, reduce the dose by 50% for one week and then taper back up.
Persistent or severe headaches that do not respond to dose adjustment require medical evaluation. While unlikely, new-onset severe headaches during GHRH therapy should prompt imaging to rule out pituitary pathology.
Contraindications: Who Should Not Use Sermorelin
Contraindications are absolute reasons not to use sermorelin. These are not precautions or situations where you can proceed with monitoring. They represent scenarios where sermorelin use poses a clear, unacceptable risk.
Active Malignancy or Recent Cancer History
Growth hormone and IGF-1 are mitogenic. They promote cell proliferation across multiple tissue types without distinguishing between healthy and malignant cells. Epidemiological data from the Nurses' Health Study and European Prospective Investigation into Cancer and Nutrition (EPIC) show that individuals with IGF-1 levels in the top quartile of the normal range have a 40 to 80% increased risk of colorectal, breast, and prostate cancers compared to the bottom quartile (Renehan et al., 2004).
This does not mean sermorelin causes cancer. It means that in someone who already has a malignancy or occult pre-malignant lesion, increasing GH and IGF-1 could accelerate tumor growth. The standard recommendation is to avoid all GH secretagogues for at least 5 years after cancer remission.
The same caution applies to GHRP-6, GHRP-2, MK-677, and ipamorelin. Any compound that elevates GH and IGF-1 carries this theoretical risk in the setting of active malignancy.
Pituitary Tumors and Intracranial Lesions
Sermorelin works by stimulating the pituitary gland. If the pituitary harbors a tumor, stimulation could cause unpredictable hormone secretion, tumor growth, or hemorrhage (pituitary apoplexy). Patients with known pituitary adenomas, craniopharyngiomas, or a history of pituitary surgery or radiation should not use sermorelin without endocrinologist clearance.
Paradoxically, sermorelin was originally designed as a diagnostic tool to test pituitary function. In that clinical context, a single diagnostic dose under medical supervision is different from daily therapeutic dosing over months. The distinction is acute stimulation versus chronic exposure.
Pregnancy and Breastfeeding
No controlled studies of sermorelin have been conducted in pregnant or breastfeeding women. GH secretagogues alter the hormonal environment in ways that could theoretically affect fetal development. The precautionary principle applies: do not use sermorelin if you are pregnant, planning to become pregnant, or nursing.
Women of childbearing age should use reliable contraception during sermorelin treatment. If pregnancy is confirmed during a cycle, discontinue sermorelin immediately and notify your healthcare provider.
Hypersensitivity to Sermorelin or Mannitol
The lyophilized formulation of sermorelin typically contains mannitol as a bulking agent. True allergy to sermorelin itself is rare, but documented. Allergy to mannitol is more common and can mimic peptide allergy. If you have a known mannitol sensitivity, discuss alternative formulations with your compounding pharmacy.
Signs of allergic reaction include hives extending beyond the injection site, throat tightness, wheezing, rapid pulse, or facial swelling. Any of these requires epinephrine and emergency medical care.
Drug Interactions: Medications That Affect Sermorelin Safety
Sermorelin interacts with several drug classes through pharmacodynamic mechanisms. These interactions do not necessarily prevent co-administration, but they require awareness and sometimes dose adjustments. Use the peptide interaction checker for a comprehensive review of your specific medications.
Glucocorticoids (Prednisone, Dexamethasone, Hydrocortisone)
Chronic glucocorticoid use suppresses GH secretion at the hypothalamic level by increasing somatostatin tone. This blunts sermorelin's effectiveness. More importantly, GH and cortisol have opposing effects on glucose metabolism: GH raises blood glucose while cortisol also raises blood glucose. The combination increases the risk of hyperglycemia and insulin resistance beyond what either agent produces alone.
If you require chronic glucocorticoid therapy, sermorelin may not produce meaningful GH elevation. Short courses of corticosteroids (less than 2 weeks) are less likely to interfere, but monitoring fasting glucose during the overlap period is recommended. Discuss timing with your prescriber.
Insulin and Oral Hypoglycemics
GH is a counter-regulatory hormone to insulin. It raises blood glucose by promoting hepatic glucose output and reducing peripheral glucose uptake. Sermorelin-induced GH elevations can worsen glycemic control in diabetic patients, potentially requiring insulin dose adjustments.
The clinical significance depends on GH magnitude. Sermorelin produces physiological GH pulses (typically 5 to 15 ng/mL), which have modest glucose effects compared to exogenous GH (which can produce sustained levels above 20 ng/mL). A study by Yuen et al. showed that low-dose GH replacement in GH-deficient adults required only minor insulin adjustments in the first 4 to 6 weeks, with glucose homeostasis normalizing thereafter (Yuen et al., 2007).
Diabetic patients considering sermorelin should test fasting glucose daily for the first 4 weeks and consult with their endocrinologist about potential insulin or metformin adjustments.
Somatostatin Analogs (Octreotide, Lanreotide)
Somatostatin analogs directly antagonize sermorelin. Somatostatin suppresses GH release from the pituitary, which is the opposite of what sermorelin is designed to do. Co-administration renders sermorelin ineffective.
If you take octreotide for a neuroendocrine tumor, acromegaly, or carcinoid syndrome, sermorelin is contraindicated. There is no dose adjustment that overcomes this pharmacological opposition.
Thyroid Hormones and Sermorelin
Hypothyroidism reduces pituitary GH secretory capacity. Untreated hypothyroidism blunts the GH response to sermorelin and may render treatment ineffective. Before starting sermorelin, confirm thyroid function with TSH and free T4 testing. If hypothyroid, optimize thyroid replacement before initiating sermorelin.
Conversely, excessive thyroid hormone (hyperthyroidism or over-replacement) can amplify GH effects and increase the risk of insulin resistance. Stable, euthyroid status is the prerequisite for predictable sermorelin responses.
Other GH Secretagogues and Exogenous GH
Combining sermorelin with exogenous GH (Humatrope, Genotropin, Norditropin) defeats the purpose of using a GHRH analog. Sermorelin's safety depends on the pituitary feedback loop. Exogenous GH bypasses that loop, and adding sermorelin on top creates unpredictable GH spikes.
Stacking sermorelin with other GH secretagogues like ipamorelin or GHRP-2 is common in peptide protocols and is pharmacologically rational because these compounds work through different receptors (GHRH receptor vs ghrelin receptor). The combined GH pulse is larger but remains subject to somatostatin feedback. For stacking guidance, see the peptide stacking guide. Use our peptide stack calculator to model combinations.
Sermorelin vs Other GH Secretagogues: Safety Comparison
Not all GH secretagogues share the same safety profile. The differences stem from receptor selectivity, half-life, and off-target hormonal effects. The table below compares sermorelin against the five most commonly used alternatives.
| Compound | Receptor | Half-Life | Elevates Cortisol? | Elevates Prolactin? | Tachyphylaxis | Safety Rating |
|---|---|---|---|---|---|---|
| Sermorelin | GHRH-R | 10-20 min | No | No | No | 4/5 |
| Ipamorelin | Ghrelin-R | ~2 hours | No | No | Minimal | 4/5 |
| CJC-1295 (DAC) | GHRH-R | 6-8 days | No | No | No | 3/5 |
| GHRP-6 | Ghrelin-R | 15-60 min | Yes (moderate) | Yes (mild) | Yes (6-8 wk) | 3/5 |
| GHRP-2 | Ghrelin-R | 15-60 min | Yes (mild) | Yes (mild) | Yes (6-8 wk) | 3/5 |
| MK-677 | Ghrelin-R | ~5 hours | Yes (initial) | Yes (initial) | No | 3/5 |
| Hexarelin | Ghrelin-R | 20-30 min | Yes (significant) | Yes (significant) | Yes (4-6 wk) | 2/5 |
For detailed dosing information on hexarelin, see hexarelin dosage.
Why Sermorelin Avoids Cortisol and Prolactin Elevation
Sermorelin is selective for the GHRH receptor. It does not interact with ghrelin receptors, which are the pathway through which GHRP-6, GHRP-2, and hexarelin elevate cortisol and prolactin. This receptor selectivity is sermorelin's primary safety advantage over ghrelin-receptor agonists.
Cortisol elevation from GHRP-6 and hexarelin can disrupt sleep architecture, increase blood pressure, promote visceral fat deposition, and suppress immune function when sustained. Arvat et al. documented that hexarelin elevated cortisol by 120% above baseline within 30 minutes of injection, an effect absent with GHRH analogs like sermorelin (Arvat et al., 1997).
Prolactin elevation, while less immediately dangerous, can cause gynecomastia in men, menstrual irregularities in women, decreased libido, and mood changes with chronic exposure. Sermorelin produces none of these effects because it does not touch the ghrelin receptor pathway that triggers prolactin release.
The CJC-1295 (DAC) Comparison: Duration Creates Risk
CJC-1295 with Drug Affinity Complex (DAC) produces sustained GH elevation lasting 6 to 8 days from a single injection. This prolonged action eliminates the pulsatile pattern that natural GHRH signaling uses. Constant GH elevation, even at physiological levels, differs from pulsatile release in its metabolic effects.
A clinical trial by Teichman et al. showed that CJC-1295 (DAC) raised mean GH levels 2 to 10-fold for up to 6 days. Adverse events included injection site reactions, transient warmth, and one case of a cardiac adverse event in the trial, though causality was not established (Teichman et al., 2006).
Sermorelin's short half-life means each GH pulse lasts 60 to 90 minutes, closely mimicking the 8 to 12 natural GH pulses the pituitary produces daily. This pulsatile pattern is important because GH receptors show differential signaling in response to pulsatile versus continuous exposure. Pulsatile GH preferentially activates lipolysis and protein synthesis. Continuous GH preferentially activates IGF-1 production, which carries greater long-term proliferative risk.
MK-677 (Ibutamoren): Oral Convenience, Different Risk Profile
MK-677 is an oral ghrelin receptor agonist with a 5-hour half-life. Its convenience (oral dosing, once daily) makes it popular, but the risk profile differs from sermorelin in three ways.
First, MK-677 raises cortisol and prolactin, particularly during the first 2 to 4 weeks. Second, MK-677 potently stimulates appetite through the ghrelin receptor, which can promote weight gain in users not prepared for increased hunger. Third, the longer half-life produces a more sustained (less pulsatile) GH elevation pattern.
Nass et al. published a one-year trial of MK-677 in elderly adults showing sustained GH and IGF-1 elevation without serious adverse events, but fasting glucose increased by approximately 7 mg/dL on average, and 2 of 65 participants developed diabetes during the study (Nass et al., 2008). This glucose effect is more pronounced than what sermorelin produces because MK-677's sustained action keeps GH elevated for longer periods, prolonging the counter-regulatory effect on insulin.
Long-Term Safety Considerations
Short-term safety data for sermorelin is reassuring. The critical questions are about years of use, not weeks.
IGF-1 and Cancer Risk: The Epidemiological Question
The association between elevated IGF-1 and cancer risk is the most frequently cited long-term concern for all GH secretagogues, including sermorelin. A meta-analysis by Renehan et al. in The Lancet examined 21 studies and found that individuals in the highest quintile of circulating IGF-1 had a 40% increased risk of prostate cancer, 49% increased risk of premenopausal breast cancer, and 65% increased risk of colorectal cancer compared to the lowest quintile (Renehan et al., 2004).
Two caveats temper this finding. First, these are observational associations, not demonstrations of causation. People with naturally high IGF-1 may share other genetic or lifestyle factors that contribute to cancer risk. Second, the highest quintile in these studies often represented IGF-1 levels above the normal range. Sermorelin, because it works within the pituitary feedback loop, raises IGF-1 to normal or high-normal levels, not supraphysiological levels.
The practical approach: monitor IGF-1 every 8 to 12 weeks. If your IGF-1 exceeds age-adjusted normal ranges (typically above 300 ng/mL for adults under 40, or above 250 ng/mL for adults over 50), reduce the sermorelin dose. If you have a first-degree relative with prostate, breast, or colorectal cancer, discuss the IGF-1 risk-benefit calculation with an oncologist before starting.
Pituitary Desensitization: Does the Response Fade?
One long-term concern with any pituitary-stimulating agent is receptor downregulation, where the target cells become less responsive over time. This is a documented problem with hexarelin (significant tachyphylaxis by 4 to 6 weeks) and to a lesser extent with GHRP-6.
Sermorelin does not appear to cause meaningful tachyphylaxis. The Vittone two-year trial showed sustained GH response throughout the 24-month treatment period. The likely explanation is that GHRH receptor signaling uses a different downstream cascade than the ghrelin receptor, one that does not trigger the same degree of receptor internalization and degradation (Vittone et al., 1997).
Standard cycling protocols (5 days on / 2 days off, or 12 to 16 weeks on / 4 to 8 weeks off) provide additional protection against any gradual decline in sensitivity. These off periods allow receptor recycling and restoration of baseline GHRH sensitivity.
Insulin Resistance: Monitoring Glucose Over Time
GH has anti-insulin effects. It promotes hepatic glucose output and reduces peripheral glucose uptake. During short-term sermorelin use (under 8 weeks), this effect is usually subclinical. With months of sustained use, cumulative GH exposure can shift fasting glucose upward by 5 to 10 mg/dL in some users.
For non-diabetic users, this shift is typically inconsequential. For pre-diabetic users (fasting glucose 100 to 125 mg/dL), it could push glucose into the diabetic range. For diabetic users, it necessitates medication adjustments.
Monitor fasting glucose and HbA1c at baseline, 8 weeks, and every 12 weeks thereafter. If fasting glucose rises above 100 mg/dL or HbA1c rises above 5.7% in a previously normal individual, reduce the sermorelin dose by 30 to 50% and recheck in 4 weeks. For broader context on metabolic monitoring during peptide use, see how to microdose semaglutide, which covers glucose monitoring in detail.
Bloodwork Monitoring Protocol
Safe sermorelin use requires periodic bloodwork. The table below outlines the minimum monitoring schedule. Think of it like checking the oil in your car: sermorelin is the engine, and bloodwork confirms it is running within safe parameters.
| Test | Baseline | 8 Weeks | 12 Weeks | Ongoing |
|---|---|---|---|---|
| IGF-1 | Yes | Yes | Yes | Every 12 weeks |
| Fasting Glucose | Yes | Yes | Yes | Every 12 weeks |
| HbA1c | Yes | No | Yes | Every 12-24 weeks |
| TSH + Free T4 | Yes | No | Yes | Every 24 weeks |
| Comprehensive Metabolic Panel | Yes | No | Yes | Every 24 weeks |
| Complete Blood Count | Yes | No | No | Annually |
| PSA (men over 40) | Yes | No | Yes | Every 24 weeks |
IGF-1 is the primary marker. It reflects the cumulative GH output over the preceding 24 hours and serves as the best proxy for whether sermorelin is producing the intended effect within safe bounds. Target range: upper third of the age-adjusted normal range, not above it.
Fasting glucose and HbA1c catch early insulin resistance. A rising trend matters more than any single value.
TSH and free T4 matter because hypothyroidism blunts the GH response and can develop independently during sermorelin treatment (not caused by sermorelin, but it should be ruled out if GH response suddenly declines).
PSA in men over 40 establishes a baseline and tracks for any prostate-related changes during IGF-1 elevation. This is a precautionary measure given the epidemiological association between IGF-1 and prostate cancer risk.
For tracking how sermorelin clears your system between cycles, use the peptide half-life tracker. For cost planning of regular bloodwork alongside peptide purchases, try the peptide cost calculator.
Reconstitution and Storage: Safety Beyond the Molecule
The sermorelin molecule itself may be safe, but a contaminated or degraded product is not. Improper reconstitution and storage are the most common sources of preventable adverse events in peptide protocols.
Sermorelin arrives as a lyophilized (freeze-dried) powder in a sealed vial. Reconstitute with bacteriostatic water, not sterile water, because the 0.9% benzyl alcohol in bacteriostatic water prevents bacterial growth in multi-dose vials. Add the water slowly down the inside wall of the vial and swirl gently. Never shake. Shaking denatures the peptide by creating air-liquid interfaces that unfold the protein structure.
Typical reconstitution: 2 mL of bacteriostatic water into a 5 mg vial produces a concentration of 2,500 mcg/mL. At a 300 mcg dose, each injection draws 0.12 mL (12 units on a U-100 insulin syringe). Use the peptide reconstitution calculator for precise volumes.
After reconstitution, store the vial upright in the refrigerator at 36 to 46 degrees F. Reconstituted sermorelin remains stable for approximately 2 to 3 weeks under proper refrigeration. Do not freeze reconstituted solution. Do not leave the vial at room temperature for more than 30 minutes during use. For detailed shelf life data, see how long do reconstituted peptides last.
Signs of degradation: cloudiness, particulate matter floating in solution, or color change from clear to yellow or brown. Discard any vial showing these signs. A degraded peptide may not cause acute harm, but it will not produce the intended GH response and may contain breakdown products with unknown effects. For comprehensive storage guidance, see how to store peptides. For injection technique and sterility, review how to inject BPC-157, which covers universal subcutaneous injection principles applicable to all peptides.
Quantified Danger Scenarios: When Sermorelin Becomes Unsafe
Most sermorelin adverse events are mild and self-limiting. The following scenarios represent situations where sermorelin use becomes genuinely dangerous. Understanding the numbers behind each scenario helps you distinguish real risks from theoretical ones.
Scenario 1: Using Sermorelin with Undiagnosed Cancer
A 52-year-old man starts sermorelin for anti-aging without recent cancer screening. He has an undetected early-stage prostate tumor. Over 6 months of daily sermorelin, his IGF-1 rises from 180 to 280 ng/mL. The elevated IGF-1 could accelerate tumor growth by promoting angiogenesis and inhibiting apoptosis. Prostate cancer doubling time, normally measured in years, could shorten by 20 to 40% under sustained IGF-1 stimulation based on in vitro data (Renehan et al., 2004).
Prevention: cancer screening before starting sermorelin. PSA test for men over 40. Mammogram for women over 40. Colonoscopy if over 45 or with family history. These screens are standard preventive care, and they become essential before any GH-elevating protocol.
Scenario 2: Contaminated Product from Unverified Source
A user purchases sermorelin from an unverified online supplier to save 40% on cost. The product contains bacterial endotoxins from non-sterile manufacturing. After 3 days of injections, the user develops fever, chills, and injection site abscess. Endotoxin contamination in injectable peptides can cause sepsis, which carries a mortality rate of 15 to 30% depending on the causative organism and treatment delay.
Prevention: purchase only from suppliers that provide third-party certificate of analysis (CoA) showing purity above 98% via HPLC and endotoxin levels below 0.25 EU/mL. For sourcing guidance, see where to buy peptides 2026 and understand the FDA peptide crackdown 2026 regulatory landscape.
Scenario 3: Diabetic Patient Without Glucose Monitoring
A type 2 diabetic starts sermorelin at 300 mcg/day without informing their endocrinologist. Over 4 weeks, the GH-mediated increase in hepatic glucose output raises fasting glucose from 140 to 185 mg/dL. HbA1c climbs from 7.1% to 8.3% over 3 months. The patient attributes increased thirst and fatigue to aging. Sustained hyperglycemia at this level increases the 10-year risk of cardiovascular events, neuropathy, and nephropathy.
Prevention: baseline glucose testing, weekly fasting glucose checks for the first month, endocrinologist consultation before starting any GH secretagogue if diabetic or pre-diabetic.
Safety During Peptide Stacking
Sermorelin is frequently combined with other peptides, particularly ipamorelin, CJC-1295, and various healing or anti-inflammatory compounds. Stacking introduces additive effects that alter the risk profile.
For stacking with other GHRH-class peptides such as tesamorelin, the same feedback-loop safety applies.
The sermorelin plus ipamorelin combination is considered the safest GH secretagogue stack because neither compound elevates cortisol or prolactin. The combined GH pulse is 3 to 5 times larger than either alone, but it remains subject to somatostatin feedback. Monitor IGF-1 more frequently (every 8 weeks rather than 12) when stacking because the larger GH pulse produces higher IGF-1 levels. See the peptide stacking guide for protocol details, and use the peptide stack calculator to model your specific combination.
Adding sermorelin to a weight loss peptide like semaglutide or tirzepatide is pharmacologically plausible but creates a metabolic tension. Sermorelin raises GH, which raises blood glucose. GLP-1 agonists lower blood glucose. The net metabolic effect depends on dose ratios and individual physiology. If you combine these classes, glucose monitoring becomes even more critical.
Stacking sermorelin with healing peptides like BPC-157 or thymosin alpha-1 does not introduce significant interaction concerns. These peptides work through different pathways (angiogenesis, immune modulation) that do not overlap with GH signaling. The main risk is cumulative injection site reactions from multiple daily injections.
For musculoskeletal recovery stacks, see peptides for joint pain and BPC-157 muscle growth. For immune support combinations with sermorelin, see peptides for immune system. For anxiety and sleep quality considerations during GH secretagogue use, see peptides for anxiety.
Special Populations: Additional Safety Considerations
Certain populations require modified approaches to sermorelin use. Standard protocols assume a healthy adult between 30 and 60 years old. Deviations from that profile change the risk calculation.
Adults Over 65
The Corpas study demonstrated safety in men up to age 78. However, older adults are more likely to have comorbidities that alter sermorelin's risk profile: impaired glucose tolerance, cardiovascular disease, occult malignancy, and polypharmacy.
Start at 50% of the standard dose (50 to 100 mcg/day) and titrate upward over 4 weeks while monitoring blood glucose and blood pressure. The pituitary's GH reserve declines with age, so lower doses may produce proportionally larger effects than expected. Use the CJC-1295 ipamorelin dosage calculator to model age-adjusted protocols for the sermorelin-ipamorelin stack.
Women of Reproductive Age
Women have naturally higher GH secretion than men during reproductive years, driven by estrogen's potentiating effect on GHRH-stimulated GH release. Standard sermorelin doses may produce larger GH pulses in premenopausal women than in men of the same age.
Start conservatively (100 mcg/day) and assess tolerance for 2 weeks before increasing. Monitor for excessive water retention, breast tenderness (from GH-mediated effects on mammary tissue), and menstrual irregularities. Discontinue if menstrual cycle disruption occurs.
Oral contraceptives increase GH-binding protein levels, which can alter the free-to-bound GH ratio. This interaction is not dangerous, but it may blunt the clinical response to sermorelin.
Patients with Liver or Kidney Disease
The liver converts GH to IGF-1. Impaired hepatic function reduces IGF-1 production, which could blunt sermorelin's downstream effects. More importantly, liver disease alters the metabolism and clearance of both sermorelin and the GH it releases, making dose-response relationships unpredictable.
Kidney disease impairs the clearance of IGF-1 and sermorelin metabolites. Elevated IGF-1 levels may accumulate in patients with reduced glomerular filtration rate, increasing the risk of prolonged GH effects.
In both populations, use sermorelin only under specialist supervision with more frequent bloodwork (every 4 weeks initially). Check the effects of liver health on peptide metabolism in does BPC-157 cause liver damage and BPC-157 and alcohol.
Important Warnings
Sermorelin is available through compounding pharmacies with a prescription and through research peptide suppliers. While it has a legitimate FDA approval history (Geref, 1997 to 2008), its current use for anti-aging and body composition is off-label. Medical supervision is recommended.
Do not self-diagnose GH deficiency. Symptoms like fatigue, weight gain, and poor recovery overlap with dozens of other conditions. Baseline bloodwork (IGF-1, metabolic panel, thyroid panel, testosterone) establishes whether GH decline is actually contributing to your symptoms before committing to treatment.
Do not exceed 300 mcg/day without medical guidance. Higher doses can push IGF-1 above age-adjusted normal ranges, negating the safety advantage of the pituitary feedback loop.
Report any new or worsening symptoms to your healthcare provider. Persistent headaches, visual field changes, new skin lesions or mole changes, unintended weight gain, or joint swelling warrant medical evaluation.
Sermorelin does not replace foundational health behaviors. Sleep, nutrition, exercise, and stress management affect GH secretion more profoundly than any peptide. A 2019 systematic review found that 7 to 9 hours of quality sleep increases overnight GH output by 300 to 400% compared to sleep deprivation (Van Cauter et al., 2000). Fix those foundations first.
This article is educational content for research purposes. Consult a qualified healthcare provider before starting, adjusting, or discontinuing any peptide protocol.
Frequently Asked Questions
What is the most common side effect of sermorelin injections?
Injection site reactions (redness, mild swelling, itching) occur in approximately 17% of users based on clinical trial data. The reaction resolves within 15 to 60 minutes and diminishes in frequency after the first 1 to 2 weeks of treatment. Rotating injection sites and using a 29 or 31 gauge needle reduces the incidence.
Can sermorelin cause cancer?
No direct causal link between sermorelin and cancer has been established. The concern is that sermorelin raises IGF-1, and epidemiological data shows a 40 to 80% higher cancer risk in people with naturally elevated IGF-1. Sermorelin's safety advantage is that pituitary feedback limits IGF-1 to physiological levels, unlike exogenous GH which can push IGF-1 far above normal ranges.
How long can you safely use sermorelin?
The longest published clinical trial ran for 2 years with sustained efficacy and no serious safety signals. Standard practice is 12 to 16 weeks on followed by 4 to 8 weeks off, repeated in cycles. Bloodwork monitoring every 8 to 12 weeks (IGF-1, fasting glucose, HbA1c) confirms the protocol remains safe during extended use.
Is sermorelin safer than HGH injections?
Yes, by design. Sermorelin stimulates the pituitary to release GH within its natural feedback loop, limiting output to physiological levels. Exogenous HGH bypasses that loop entirely, creating supraphysiological levels that increase the risk of insulin resistance, carpal tunnel, joint pain, and organ enlargement. Clinical trials show adverse event rates of 22% with sermorelin versus 30 to 50% with therapeutic HGH doses.
Does sermorelin affect blood sugar levels?
GH has anti-insulin effects that can raise fasting glucose by 5 to 10 mg/dL in some users. For non-diabetics, this shift is clinically insignificant. For diabetics or pre-diabetics, it may require medication adjustments. The glucose effect is less pronounced with sermorelin than with exogenous GH or long-acting secretagogues like MK-677, which raised fasting glucose by 7 mg/dL on average in a 1-year trial.
What bloodwork should I get before starting sermorelin?
At minimum: IGF-1 (establishes GH baseline), fasting glucose and HbA1c (metabolic safety), TSH and free T4 (thyroid function affects GH response), comprehensive metabolic panel (liver and kidney function), and PSA for men over 40. Total cost for these labs is typically $150 to $300 through direct-to-consumer lab services.
Can sermorelin interact with my current medications?
Glucocorticoids blunt sermorelin's effect and compound its glucose-raising properties. Somatostatin analogs like octreotide directly antagonize sermorelin, rendering it ineffective. Insulin and oral hypoglycemics may require dose adjustments due to GH's counter-regulatory effects. Thyroid medications interact indirectly: hypothyroidism reduces GH response, so thyroid levels should be optimized first.
Why was Geref (sermorelin) taken off the market?
EMD Serono voluntarily withdrew Geref in 2008 for commercial reasons, citing limited market demand for the diagnostic indication. The FDA did not issue a safety recall, public health advisory, or mandatory withdrawal. This contrasts with drugs removed for safety reasons (like Vioxx or fenfluramine), which receive formal FDA-mandated recalls accompanied by public warnings.
The Bottom Line
Sermorelin has one of the best-documented safety profiles among GH secretagogues. FDA approval for a pediatric diagnostic application in 1997, a voluntary (not safety-driven) market withdrawal in 2008, and clinical trials spanning up to 2 years with adverse event rates near placebo levels provide a foundation of evidence that most research peptides lack entirely.
The mechanism itself is the safety feature. By working through the pituitary feedback loop rather than bypassing it, sermorelin keeps GH and IGF-1 within physiological bounds. It does not elevate cortisol or prolactin. It does not cause the tachyphylaxis seen with hexarelin. It does not produce the sustained, non-pulsatile GH elevation that makes CJC-1295 (DAC) and MK-677 riskier for long-term use.
The real dangers are not the molecule. They are contaminated products from unverified sources, use in the setting of undiagnosed malignancy, and failure to monitor bloodwork during chronic use. Address those three factors, and sermorelin becomes a low-risk intervention for age-related GH decline.
Use the peptide interaction checker to screen for drug interactions before starting. Calculate your preparation volumes with the peptide reconstitution calculator. Plan your monitoring costs with the peptide cost calculator.
Related articles: - Sermorelin for Fat Loss - dosing, cycling, and body composition evidence - Peptide Safety Guide - safety profiles for 15 peptides - Peptide Dosage Chart - dosing reference across all categories - Hexarelin Dosage - another GH secretagogue with different risk dynamics - Peptide Stacking Guide - safe combination protocols - FDA Peptide Crackdown 2026 - regulatory landscape for peptide access - GHK-Cu Injection Dosage - copper peptide dosing and safety - MOTS-c Peptide Dosage - mitochondrial peptide protocols
Explore all peptide profiles and tools at PeptidesExplorer.
Helpful Tools
Related Articles
TB-500 Side Effects: What Research Shows
TB-500 side effects: headache (10-15%), lethargy, nausea, and injection site reactions. Cancer risk evidence, contraindications, and monitoring.
Sermorelin for Fat Loss: Dosing & Data
Sermorelin stimulates natural GH release to enhance fat oxidation. Dosing protocols (100-300mcg/day), cycling, and CJC-1295 comparison.
Does Sermorelin Increase Testosterone?
Sermorelin does not directly increase testosterone. It stimulates GH release, which supports testosterone indirectly via IGF-1 and improved sleep. Clinical evidence reviewed.
How Much Sermorelin Per Day: Complete Dosage Guide
Sermorelin dosage: 200-500 mcg/day subcutaneous before bedtime. Starting dose, titration, gender-specific protocols, and IGF-1 monitoring.