You lost 40 pounds on semaglutide. Your provider wants to stop the medication. You want to keep the appetite suppression without the nausea, the fatigue, and the $400 monthly bill. Or maybe you tried the standard 0.25 mg starting dose and the GI side effects knocked you flat for three days.
Either way, you have arrived at the same question: can you take less than the prescribed starting dose and still get meaningful results?
The short answer is yes, with caveats. Microdosing semaglutide means using doses between 0.05 mg and 0.25 mg per week, below the standard titration starting point, to achieve partial appetite suppression with reduced side effects. This approach is not FDA-approved at these doses, has limited clinical trial data, and requires precise measurement of very small injection volumes. But a growing number of clinicians are prescribing sub-therapeutic semaglutide doses for specific patient populations, and the pharmacological rationale is sound.
This guide covers what microdosing means in the semaglutide context, who benefits from it, the protocols clinicians are using, how to accurately measure doses this small with compounded vials, what the evidence actually says, and the risks you accept by going off the studied dose range. If you need a refresher on standard dosing first, start with the semaglutide dosage chart in mL.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Microdosing Semaglutide Actually Means
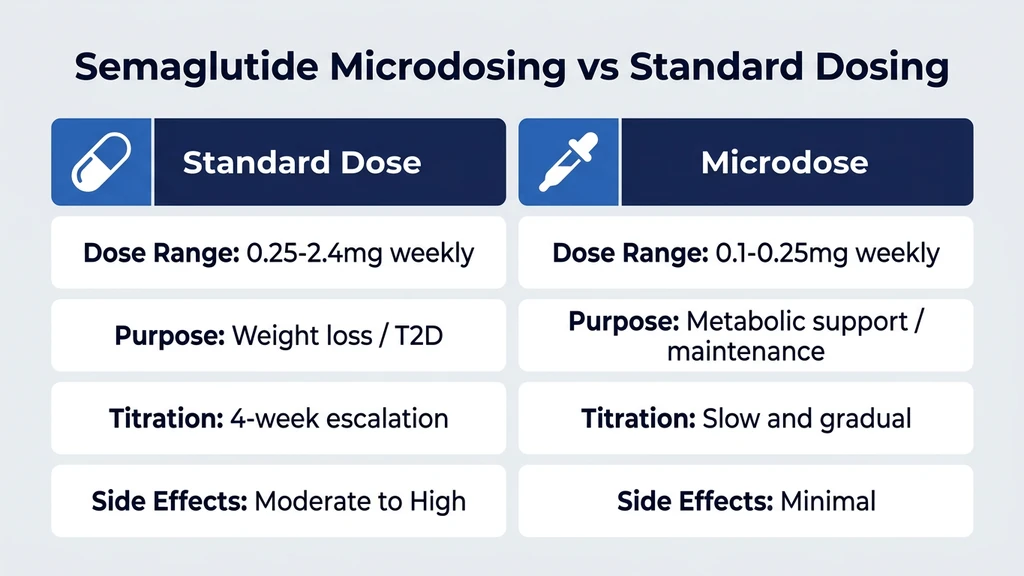
The term "microdosing" gets thrown around loosely online. In psychopharmacology, microdosing refers to taking roughly one-tenth of a pharmacologically active dose. Applied to semaglutide, microdosing refers to weekly doses below the 0.25 mg starting point from the FDA-approved titration schedule, or to staying at 0.25 mg long-term instead of titrating upward.
The standard semaglutide titration for weight management (Wegovy prescribing information, Novo Nordisk, 2021) follows a five-step escalation: 0.25 mg for 4 weeks, then 0.5 mg, 1.0 mg, 1.7 mg, and finally 2.4 mg as the maintenance dose. Each step lasts 4 weeks. The 0.25 mg dose was designed as a tolerability ramp, not a therapeutic endpoint. The clinical trials that produced the headline weight loss numbers all used 2.4 mg as the treatment dose.
Microdosing sits below that ramp. The most common microdose protocols use 0.0625 mg, 0.1 mg, 0.125 mg, or 0.15 mg per week. Some people use the standard 0.25 mg dose but inject every 10 to 14 days instead of every 7. The goal in every case is the same: activate enough GLP-1 receptor signaling to reduce appetite without triggering the dose-dependent side effects that make higher doses difficult to tolerate.
This is distinct from the standard titration. During titration, 0.25 mg is a stepping stone. In microdosing, a sub-0.25 mg dose is the destination.
It is also distinct from dose reduction after reaching maintenance. Some providers taper patients from 2.4 mg down to 1.0 mg or 0.5 mg after target weight is achieved. That strategy uses clinically studied doses. Microdosing goes further, into territory where controlled trial data is sparse. For information on what happens when weight loss stalls at any dose, see not losing weight on semaglutide.
Why People Microdose Semaglutide: Four Common Reasons
Not everyone who explores microdosing is trying to cut corners. Four distinct patient populations gravitate toward sub-therapeutic doses, each with a different clinical rationale.
1. Side effect sensitivity
Semaglutide's GI side effects are dose-dependent. The STEP 1 trial (Wilding et al., N Engl J Med, 2021) documented nausea in 44.2% of participants at the 2.4 mg dose versus 17.4% on placebo. Vomiting occurred in 24.8% versus 6.2%. Even at the 0.25 mg starting dose, some individuals experience nausea severe enough to interfere with daily life. For these patients, starting at 0.1 mg and titrating upward in smaller increments (0.1 to 0.15 to 0.2 to 0.25 mg) over 8 to 12 weeks may allow the GI tract to adapt without the abrupt disruption. If nausea is your primary concern, read more about why semaglutide causes nausea and how long side effects last.
2. Maintenance after significant weight loss
This is the most pharmacologically defensible use case. After reaching goal weight on higher doses, some patients find that full-dose semaglutide causes continued unwanted weight loss or that the appetite suppression feels excessive. Rather than stopping entirely and risking the weight regain documented in the STEP 1 extension trial (Wilding et al., Diabetes Obes Metab, 2022), where participants regained two-thirds of lost weight within one year of stopping, a microdose may provide enough GLP-1 receptor activation to maintain the appetite set-point shift. This approach parallels how tirzepatide users handle the transition period; see tirzepatide maintenance dose after weight loss for the comparable strategy.
3. Cost reduction
A 5 mg vial of compounded semaglutide costs roughly $100 to $200 depending on the pharmacy. At the standard 2.4 mg weekly dose, that vial lasts about 2 weeks. At a 0.125 mg microdose, the same vial lasts 40 weeks. That is a 20-fold reduction in cost. For patients paying out of pocket, microdosing stretches the budget dramatically. One vial, properly stored and used within its expiration window, can last months at microdose levels (though the 28-day beyond-use date means you will still need fresh vials regularly).
4. Gradual introduction for anxiety about injections or medication
Some patients, particularly those new to peptide injections, want to start with the smallest possible dose to see how their body reacts before committing to the full titration. This is less about pharmacology and more about psychology. Starting at 0.1 mg gives them a low-stakes first experience. If they tolerate it well, confidence builds. If they react poorly, the exposure was minimal. Semaglutide's long half-life of approximately 7 days (Kapitza et al., J Clin Pharmacol, 2015) means even a microdose lingers, so this approach is not risk-free, but the absolute exposure is low.
Microdosing Protocols: What Clinicians Are Prescribing
No FDA-approved microdosing protocol exists. The protocols below come from clinical practice patterns reported in obesity medicine forums and telehealth prescribing data. Your provider should determine the specific protocol for your situation.
Protocol A: Ultra-Low Start (0.0625 mg to 0.125 mg)
| Week | Dose | Frequency | Notes |
|---|---|---|---|
| 1 to 2 | 0.0625 mg | Once weekly | Assess GI tolerance |
| 3 to 4 | 0.1 mg | Once weekly | Monitor appetite changes |
| 5 to 8 | 0.125 mg | Once weekly | Evaluate weight trend |
| 9 onward | Stay at 0.125 mg or titrate to 0.25 mg | Once weekly | Based on response |
This protocol is used for patients who experienced severe GI effects on prior GLP-1 agonist therapy or who have a history of gastroparesis. The increments are roughly 50 to 60 percent increases, smaller than the 100% jumps in the standard titration (0.25 to 0.5 mg).
Protocol B: Low Maintenance (0.25 mg indefinite)
| Phase | Dose | Duration | Notes |
|---|---|---|---|
| Standard titration | 0.25 mg to 2.4 mg | 16 weeks | Per Wegovy label |
| Weight loss phase | 2.4 mg | Until target weight | Typically 12 to 18 months |
| Taper | 1.0 mg, then 0.5 mg, then 0.25 mg | 4 weeks each step | Gradual step-down |
| Maintenance | 0.25 mg | Indefinite | Monitor weight quarterly |
This is the most conservative approach and stays within studied dose ranges during the taper. The 0.25 mg maintenance dose was not studied as a long-term monotherapy in STEP trials, but it is the dose with the mildest side effect profile and still activates GLP-1 receptors meaningfully.
Protocol C: Extended Interval Microdosing
Instead of reducing the dose, some providers extend the injection interval. Given semaglutide's 7-day half-life (Kapitza et al., J Clin Pharmacol, 2015), injecting 0.25 mg every 10 to 14 days maintains a lower average steady-state concentration than weekly dosing. This approach requires less precise measurement (you use the standard 0.25 mg volume) but produces fluctuating drug levels that may cause inconsistent appetite suppression.
| Interval | Effective Weekly Dose | Pros | Cons |
|---|---|---|---|
| Every 7 days | 0.25 mg/week | Stable levels, standard protocol | May still cause GI effects for sensitive patients |
| Every 10 days | ~0.175 mg/week | Simpler measurement | Hunger fluctuation mid-cycle |
| Every 14 days | ~0.125 mg/week | Longest stretch between injections | Significant trough before next dose |
For context on how long the drug remains active between doses, see how long does semaglutide stay in your system. If you are considering switching to tirzepatide instead of microdosing, the dual-agonist mechanism offers a different pharmacological profile that some patients tolerate better at lower doses.
How to Measure Microdoses with Compounded Semaglutide
Measuring 0.1 mg of semaglutide from a compounded vial requires careful math and the right syringe. The margin for error shrinks as the dose gets smaller. Here is how to do it accurately.
Step 1: Know your vial concentration.
Read the label. Common compounded concentrations are 2.5 mg/mL, 5 mg/mL, and 10 mg/mL. If you reconstituted the vial yourself, your concentration depends on how much bacteriostatic water you added. The Peptide Reconstitution Calculator gives you the exact number.
Step 2: Calculate your draw volume.
Use the formula: mL = dose (mg) / concentration (mg/mL)
Here is a microdose conversion table for common concentrations:
| Dose | At 2.5 mg/mL | At 5 mg/mL | At 10 mg/mL |
|---|---|---|---|
| 0.0625 mg | 0.025 mL (2.5 units) | 0.0125 mL (1.25 units) | 0.00625 mL (0.625 units) |
| 0.1 mg | 0.04 mL (4 units) | 0.02 mL (2 units) | 0.01 mL (1 unit) |
| 0.125 mg | 0.05 mL (5 units) | 0.025 mL (2.5 units) | 0.0125 mL (1.25 units) |
| 0.15 mg | 0.06 mL (6 units) | 0.03 mL (3 units) | 0.015 mL (1.5 units) |
| 0.25 mg | 0.10 mL (10 units) | 0.05 mL (5 units) | 0.025 mL (2.5 units) |
Notice the problem. At 5 mg/mL, a 0.1 mg dose requires drawing only 2 units on an insulin syringe. At 10 mg/mL, it requires 1 unit. These volumes are so small that standard insulin syringes cannot measure them reliably.
Step 3: Choose the right syringe and concentration pairing.
For microdosing, you want the largest draw volume possible. That means using the lowest available concentration. If you have a choice, pick 2.5 mg/mL over 5 mg/mL. At 2.5 mg/mL, a 0.1 mg dose requires 4 units, which is measurable on a 0.3 mL (30-unit) insulin syringe where each line represents 1 unit.
If you must use a 5 mg/mL or 10 mg/mL vial, invest in half-unit insulin syringes. These syringes have lines at every 0.5 units instead of every 1 unit, doubling your measurement precision. BD Ultra-Fine and EasyTouch both make 0.3 mL half-unit syringes that are widely available.
Step 4: Consider diluting for better accuracy.
Some providers instruct patients to add more bacteriostatic water to an already-reconstituted vial to lower the concentration. For example, adding 1 mL of bacteriostatic water to a 5 mg/mL vial (already containing 1 mL of solution) produces 5 mg in 2 mL, or 2.5 mg/mL. This doubles your draw volume for any given dose.
There is a limit. Over-diluting increases the total volume in the vial. If you add too much water, you may not be able to draw all doses before the 28-day beyond-use date. Also, adding water after initial reconstitution introduces a second opportunity for contamination. Use aseptic technique: swab both vial stoppers with alcohol, use a fresh needle, and work in a clean environment. For the difference between water types, see bacteriostatic water vs sterile water.
For a broader overview of concentration math, the semaglutide dosage chart in mL walks through the formula in detail. You can also reference our guide on how many mg is 40 units of semaglutide to understand the unit-to-mg relationship at various concentrations, and use the Semaglutide Dosage Calculator for instant math.

Evidence for Low-Dose Semaglutide Efficacy
The honest assessment: strong clinical trial data for microdosing semaglutide does not exist. The pivotal STEP trials tested 2.4 mg. The Ozempic diabetes trials tested 0.5 mg and 1.0 mg. Nobody ran a large, randomized, placebo-controlled trial on 0.1 mg weekly. But the existing data does provide indirect support for the concept.
Dose-response from STEP trials
The STEP 5 trial (Garvey et al., Nat Med, 2022) demonstrated that semaglutide 2.4 mg produced 15.2% mean body weight reduction at 104 weeks. The earlier SUSTAIN dose-finding studies for diabetes (Nauck et al., Diabetes Care, 2016) tested multiple dose levels including 0.05 mg, 0.1 mg, 0.2 mg, 0.3 mg, and 0.4 mg daily (note: daily, not weekly) of oral semaglutide and found dose-dependent reductions in HbA1c and body weight at every dose tested. The 0.1 mg daily oral dose (roughly equivalent to very low subcutaneous exposure due to oral semaglutide's approximately 1% bioavailability per the Rybelsus prescribing information) still produced statistically significant weight loss versus placebo.
GLP-1 receptor pharmacology
Semaglutide is a GLP-1 receptor agonist. GLP-1 receptors in the hypothalamus, brainstem, and gut respond in a dose-dependent manner. Even partial receptor occupancy reduces appetite signaling. Full receptor saturation, achieved at higher doses, produces maximal appetite suppression but also maximal GI effects. A microdose achieves partial occupancy: less suppression, but also less nausea and delayed gastric emptying.
The receptor binding kinetics support a non-linear dose-response curve. Blundell et al. (Diabetes Obes Metab, 2017) showed that semaglutide at 1.0 mg reduced ad libitum energy intake by approximately 24% versus placebo. The relationship between dose and appetite suppression is not linear; the first milligram of exposure produces a larger proportional effect than the second. This means microdoses capture a meaningful fraction of the appetite benefit despite using a small fraction of the standard dose.
Observational and real-world data
Published case series are limited, but the pharmacokinetic modeling from Kapitza et al. (J Clin Pharmacol, 2015) demonstrates that semaglutide at 0.25 mg weekly achieves steady-state plasma concentrations of approximately 12.5 nmol/L, which is well above the minimum effective concentration for GLP-1 receptor activation. At 0.1 mg weekly, steady-state concentrations would be proportionally lower (approximately 5 nmol/L) but still within a pharmacologically active range based on in vitro receptor binding data.
The bottom line: microdosing is not unsupported speculation. The pharmacology makes sense. But without dedicated trial data, the magnitude of benefit at specific microdose levels remains uncertain. If you are early in your semaglutide journey and wondering about timelines at any dose, see how long does semaglutide take to work.
Risks and Limitations of Microdosing
Microdosing is not a free lunch. The reduced side effects come with real tradeoffs that you need to understand before committing to this approach.
Insufficient weight loss
The STEP trials produced their impressive results at 2.4 mg. Lower doses produce less weight loss. If your BMI is above 35 and your clinical goal is substantial weight reduction, a microdose may not move the needle enough to improve metabolic markers like HbA1c, blood pressure, or triglycerides. Partial appetite suppression might produce 3 to 5 percent body weight loss instead of the 15 percent seen in trials. For some patients, that is meaningful. For others, it falls short of the threshold where cardiovascular and metabolic benefits are established.
False sense of security about dose precision
Drawing 2 units on an insulin syringe involves significant measurement uncertainty. A half-unit error at this scale represents a 25% dosing variation. If you draw 1.5 units instead of 2, you injected 0.075 mg instead of 0.1 mg. If you draw 2.5 units, you injected 0.125 mg. The biological effects at these doses are subtle enough that you may not notice the inconsistency, but the variable exposure undermines the purpose of a controlled protocol. For details on syringe precision, see the peptide dosage chart.
No long-term safety data at sub-therapeutic doses
The FDA approved semaglutide based on trials using 0.5 mg, 1.0 mg, and 2.4 mg. Safety monitoring at those doses is extensive. At 0.1 mg weekly, the safety profile is extrapolated, not proven. While lower doses generally produce fewer adverse effects, the long-term consequences of chronic low-level GLP-1 receptor activation have not been studied in isolation. Theoretical concerns include subtle changes in gallbladder motility, pancreatic enzyme secretion, and gastric emptying that might not manifest as obvious symptoms but could have cumulative effects over years.
Weight regain risk during maintenance microdosing
If you taper from 2.4 mg to a microdose for maintenance, you may cross below the effective threshold for appetite control at some point during the taper. The STEP 1 extension data showed that weight regain begins within weeks of dose reduction. A microdose might slow but not prevent this regain. Without regular monitoring and willingness to adjust the dose upward if weight trends reverse, maintenance microdosing can fail silently.
Fatigue and hair loss can still occur
Some semaglutide side effects are not purely dose-dependent. Hair thinning (telogen effluvium) is related to rapid weight loss and caloric deficit, not directly to the drug dose. If a microdose produces enough appetite suppression to significantly reduce caloric intake, hair loss can still occur. Similarly, fatigue during early treatment may relate to metabolic adjustment rather than dose magnitude.
Provider skepticism
Many providers are unfamiliar with microdosing protocols. If you request a sub-0.25 mg prescription, your provider may decline, not because the approach is dangerous, but because it falls outside their training and comfort zone. This can push patients toward self-dosing from existing prescriptions, which introduces medication management risks. Always discuss your dosing plan with your prescriber.
Microdosing vs. Standard Titration: Side-by-Side Comparison
This comparison helps you decide which approach fits your clinical situation. Neither is universally superior. The right choice depends on your goals, tolerance, and medical history.
| Factor | Microdosing (0.1 to 0.25 mg) | Standard Titration (0.25 to 2.4 mg) |
|---|---|---|
| Expected weight loss | 2 to 5% body weight (estimated) | 12 to 17% body weight (STEP trial data) |
| Nausea incidence | Minimal to none at sub-0.25 mg | 44% at 2.4 mg (STEP 1) |
| Time to meaningful results | 4 to 8 weeks for appetite change | 4 to 8 weeks for appetite change; 12 to 16 weeks for visible weight loss |
| Monthly cost (compounded) | $5 to $25 (vial lasts months) | $100 to $200+ per month |
| Clinical trial support | Indirect/pharmacological inference | Extensive (STEP 1 through 5, SUSTAIN, SELECT) |
| FDA-approved | No | Yes (Wegovy 2.4 mg, Ozempic 0.5/1.0 mg) |
| Best for | Maintenance, side-effect-sensitive patients, cost-constrained | Primary weight loss, type 2 diabetes management |
| Provider acceptance | Variable | Standard of care |
The most practical approach for many patients is a hybrid: use standard titration to reach goal weight, then taper to the lowest effective dose for maintenance. This captures the proven benefits of higher doses during active weight loss while using microdosing principles for the long-term phase where the goal shifts from losing weight to not regaining it.
For patients considering alternatives altogether, the peptide safety guide covers the broader landscape. If tirzepatide's dual-agonist approach interests you, check does tirzepatide burn fat for a mechanistic comparison.
How to Store Semaglutide for Extended Microdose Use
Microdosing creates a unique storage challenge. A 5 mg vial at 0.125 mg per week contains 40 doses, enough for 40 weeks. But reconstituted semaglutide has a 28-day beyond-use date per USP <797> compounding standards. You cannot use one vial for 40 weeks.
Option 1: Smaller vials
Some compounding pharmacies offer 1 mg or 2 mg vials. A 2 mg vial at 0.125 mg per week lasts 16 weeks of doses, but you still hit the 28-day limit. Request 1 mg vials reconstituted at 1 mg/mL. At 0.125 mg per week, that vial lasts 8 doses (8 weeks), still exceeding 28 days. The math is unforgiving at microdose levels.
Option 2: Reconstitute only what you will use in 28 days
If you receive lyophilized powder and a separate vial of bacteriostatic water, you can partially reconstitute. This is not standard practice and requires precise technique, but some providers instruct patients to dissolve only a portion of the powder. This approach is difficult to execute accurately with typical home equipment and is not recommended without provider guidance.
Option 3: Accept some waste
The most practical approach: reconstitute the full 5 mg vial, use 4 microdoses over 28 days, and discard the remainder. At 0.125 mg per week from a 5 mg vial, you use 0.5 mg (4 doses) and discard 4.5 mg. This is wasteful but safe. At $100 to $200 per vial, you are paying $25 to $50 per month. That is still dramatically cheaper than full-dose treatment.
Storage fundamentals apply regardless of dose:
Refrigerate at 2 to 8 degrees Celsius. Keep the vial upright with the cap on. Never freeze. Discard if cloudy, discolored, or containing particles. For the complete storage guide, see does semaglutide need to be refrigerated and how to store peptides.
Mark the reconstitution date on every vial with a permanent marker. At microdose levels, you are drawing from the vial infrequently enough that losing track of the date is a real risk. The bacteriostatic water preservative (0.9% benzyl alcohol) does inhibit bacterial growth, but it does not make the solution sterile indefinitely.
Step-by-Step: Your First Semaglutide Microdose
Assuming your provider has prescribed a sub-therapeutic dose and you have a compounded semaglutide vial, here is the exact procedure. This example uses a 0.1 mg dose from a 2.5 mg/mL vial.
Gather your supplies: - Reconstituted semaglutide vial (2.5 mg/mL, refrigerated) - 0.3 mL (30-unit) insulin syringe, half-unit markings preferred - Alcohol swabs - Sharps container
Calculate your draw volume: 0.1 mg / 2.5 mg/mL = 0.04 mL = 4 units
Procedure:
- 1.Wash your hands thoroughly. Remove the vial from the refrigerator and let it reach room temperature for 5 minutes. Cold solution is more viscous and harder to draw accurately.
- 1.Swab the vial stopper with an alcohol pad. Let it dry for 10 seconds.
- 1.Remove the cap from your insulin syringe. Pull the plunger back to the 4-unit mark to draw air into the syringe.
- 1.Insert the needle through the vial stopper. Push the plunger to inject the air into the vial. This equalizes pressure and makes drawing easier.
- 1.Invert the vial so the stopper faces down. Position the needle tip in the liquid, away from the stopper.
- 1.Pull the plunger slowly to the 4-unit mark. At this small volume, go slowly. One unit of overshoot is a 25% dosing error.
- 1.Check for air bubbles. Tap the syringe barrel gently to move bubbles to the top. Push the plunger slightly to expel them. Re-draw to the 4-unit mark if needed.
- 1.Remove the needle from the vial. Do not recap.
- 1.Clean your injection site (abdomen, thigh, or upper arm) with an alcohol swab. Let it dry completely.
- 1.Pinch a fold of skin. Insert the needle at a 45 to 90 degree angle (90 degrees for most insulin syringes). Depress the plunger slowly and steadily. Count to 5 after the plunger is fully depressed before withdrawing the needle.
- 1.Dispose of the syringe in your sharps container. Return the vial to the refrigerator.
For a complete injection tutorial covering technique, site rotation, and troubleshooting, see the getting started with peptides guide. If you are using a different reconstitution setup, the Peptide Reconstitution Calculator confirms your concentration before you draw.
When Microdosing Is Not Appropriate
Microdosing has a place, but it is not the right choice for every patient. The following situations call for standard-dose treatment or a different medication entirely.
Active type 2 diabetes requiring glycemic control. Semaglutide is FDA-approved for type 2 diabetes at 0.5 mg and 1.0 mg weekly (Ozempic prescribing information, Novo Nordisk, 2023). The glycemic benefits at microdose levels are likely insufficient to achieve HbA1c targets. If your primary indication is diabetes management, microdosing substitutes hope for proven efficacy.
BMI above 40 with obesity-related comorbidities. Patients in this category benefit most from the maximum weight loss achievable through standard titration to 2.4 mg. The SELECT trial (Lincoff et al., N Engl J Med, 2023) demonstrated a 20% reduction in major adverse cardiovascular events with semaglutide 2.4 mg in overweight/obese patients with established cardiovascular disease. A microdose is unlikely to replicate this level of cardiovascular risk reduction.
History of medullary thyroid carcinoma or MEN2 syndrome. This is a contraindication at any semaglutide dose, including microdoses. GLP-1 receptor agonists caused thyroid C-cell tumors in rodent studies at all dose levels tested (Wegovy prescribing information, Novo Nordisk, 2021). The risk is not dose-dependent in the preclinical models.
Concurrent use of other GLP-1 receptor agonists. Combining a semaglutide microdose with another GLP-1 agonist (dulaglutide, liraglutide, or tirzepatide) is not appropriate. The receptor is the same. Stacking agonists amplifies GI side effects without proportional clinical benefit.
Self-prescribing without medical supervision. Microdosing requires the same medical oversight as standard dosing. Blood work, weight monitoring, and side effect assessment still matter. The lower dose does not eliminate the need for a provider relationship. If you are exploring peptides independently, at minimum review the peptide safety guide for risk mitigation fundamentals.
Tracking Your Response to Microdose Semaglutide
Because microdosing produces subtler effects than standard dosing, you need more deliberate tracking to determine whether it is working. Relying on how you "feel" is insufficient at doses where the appetite suppression may register as a mild reduction in cravings rather than the dramatic appetite extinction reported at higher doses.
Track these metrics weekly:
- Body weight. Weigh yourself at the same time each day (morning, after using the bathroom, before eating). Record the 7-day average, not individual days. Daily fluctuations of 1 to 3 pounds are normal and meaningless. A downward trend in the weekly average is what matters.
- Caloric intake. Use a food tracking app for at least the first 8 weeks. Microdose semaglutide should reduce your average daily intake by 100 to 300 calories if it is working. Without tracking, you will not notice this shift because the reduction is gradual, not dramatic.
- Hunger and satiety scores. Rate your hunger at meals on a 1 to 10 scale. Rate how full you feel after eating. Look for a trend toward lower pre-meal hunger and earlier satiety. Even a 1 to 2 point shift is clinically relevant at microdose levels.
- Side effects. Log any nausea, constipation, diarrhea, injection site reactions, fatigue, or other changes. At microdose levels, GI effects should be minimal. If you experience significant nausea at 0.1 mg, that is useful data suggesting unusual sensitivity.
- Waist circumference. Measure weekly. The tape measure catches visceral fat changes that the scale may miss, especially if you are exercising and gaining muscle simultaneously.
Decision points:
At 8 weeks, evaluate. If your weekly weight average has not decreased by at least 1%, your caloric intake has not measurably dropped, and your hunger scores are unchanged, the microdose is likely sub-therapeutic for you. Discuss titrating upward with your provider. The standard 0.25 mg dose may be the minimum effective dose for your metabolism, and there is no shame in that. Every person's GLP-1 receptor sensitivity is different.
If you are losing 0.5 to 1% of body weight per month and experiencing no side effects, the microdose is likely in your therapeutic window. Continue and reassess quarterly.
Frequently Asked Questions
What is the lowest effective dose of semaglutide for weight loss?
The lowest FDA-studied dose is 0.25 mg weekly, used as a titration starting point. Clinical evidence suggests 0.5 mg is the minimum dose producing statistically significant weight loss in trials. Microdoses of 0.1 to 0.125 mg may provide partial appetite suppression based on pharmacological models, but no controlled trials confirm efficacy at these levels.
Can I cut my semaglutide dose in half without telling my doctor?
This is strongly discouraged. Even dose reductions should involve your prescriber. They need to monitor your metabolic markers, adjust expectations, and document your treatment plan. Self-adjusting doses introduces uncontrolled variables into your care and may void your pharmacy's follow-up protocols.
How long does it take to feel the effects of microdose semaglutide?
Semaglutide reaches steady-state plasma concentration after approximately 4 to 5 weeks of consistent weekly dosing at any dose level. Appetite changes at microdose levels are subtle and may take 4 to 8 weeks to become noticeable. Track caloric intake and hunger scores rather than relying on subjective feelings.
Will microdose semaglutide cause nausea?
Nausea is dose-dependent with semaglutide. At 2.4 mg, 44% of patients report nausea. At 0.25 mg, the rate is much lower. At true microdoses of 0.1 to 0.125 mg, clinically significant nausea is uncommon. If nausea occurs at these levels, you may have unusual GLP-1 receptor sensitivity, and starting even lower may help.
Is microdosing semaglutide the same as compounded semaglutide?
No. Compounded semaglutide refers to how the medication is prepared (by a compounding pharmacy rather than the brand manufacturer). Microdosing refers to the dose amount. You can microdose with compounded semaglutide or with branded Ozempic or Wegovy, though the pre-filled pens make sub-0.25 mg doses impractical.
Can I microdose semaglutide while breastfeeding or pregnant?
Semaglutide is contraindicated in pregnancy at any dose. The Wegovy prescribing information recommends discontinuing semaglutide at least 2 months before a planned pregnancy due to its long washout period. There is insufficient data on semaglutide in breast milk. Do not microdose as a workaround for these contraindications.
How do I switch from microdosing back to standard dosing?
Resume the standard titration from your current microdose level. If you were on 0.125 mg, increase to 0.25 mg for 4 weeks, then 0.5 mg, and so on. Do not jump from a microdose directly to a high therapeutic dose. The GI tract needs the same gradual adaptation regardless of your starting point.
Does insurance cover semaglutide for microdosing?
Insurance coverage for semaglutide is tied to approved indications (type 2 diabetes for Ozempic, obesity for Wegovy), not to specific dose levels. If you have coverage, your provider can prescribe 0.25 mg and instruct you on microdose administration. Most microdosers use compounded semaglutide, which is typically not covered by insurance.
The Bottom Line
Microdosing semaglutide means using weekly doses of 0.05 to 0.25 mg to achieve partial appetite suppression with fewer side effects and lower cost than standard treatment. The pharmacological rationale is supported by GLP-1 receptor dose-response data and semaglutide's pharmacokinetic profile (Kapitza et al., J Clin Pharmacol, 2015), though no dedicated randomized controlled trial has validated a specific microdose protocol.
The approach works best for two populations: patients tapering to maintenance after reaching goal weight, and patients whose GI sensitivity prevents tolerating the standard 0.25 mg starting dose. For primary weight loss in patients with BMI above 30, standard titration to 2.4 mg remains the evidence-based choice per the STEP trial program (Wilding et al., N Engl J Med, 2021).
If you pursue microdosing, precision matters. Use the lowest available concentration (2.5 mg/mL) with half-unit insulin syringes. Track your weight, caloric intake, and hunger scores weekly. Reassess at 8 weeks. And keep your provider in the loop.
For concentration math and draw volumes, use the Semaglutide Dosage Calculator. For reconstitution planning, use the Peptide Reconstitution Calculator. And for the complete dosing reference at standard levels, bookmark the semaglutide dosage chart in mL.
Related Articles: - Not Losing Weight on Semaglutide? — troubleshooting at any dose - How Long Does Semaglutide Take to Work? — timelines for results - Semaglutide to Tirzepatide Switching Guide — if microdosing is not enough - How Long Does Semaglutide Stay in Your System? — pharmacokinetics - Peptide Safety Guide — comprehensive safety reference - Getting Started with Peptides — beginner's guide to peptide use
Related Articles
Microdosing Tirzepatide Explained
Microdosing tirzepatide means using 0.5-2.5 mg weekly instead of 5-15 mg. Protocols, clinical evidence, and side effect management.
Semaglutide Titration Schedule: 6-Month Plan
Week-by-week semaglutide titration from 0.25 to 2.4 mg. Based on STEP trial data. When to extend, down-titrate, or restart after missing doses.
How Many mg Is 40 Units Semaglutide?
40 units of semaglutide = 0.8 mg at 2 mg/mL, 1 mg at 2.5 mg/mL, 2 mg at 5 mg/mL, or 4 mg at 10 mg/mL. Full conversion table with syringe guide.
Semaglutide Dosage Chart in mL
Semaglutide dosage chart in mL for compounded 5mg and 10mg vials. Covers 0.25 to 2.4 mg doses with unit conversions and syringe markings.