Your provider prescribed tirzepatide at 2.5 mg per week. Two days after your first injection, the nausea hit so hard you skipped dinner and spent the evening on the couch. Now you are scrolling forums at midnight wondering if you could take less and still see results.
Microdosing tirzepatide means using weekly doses of 0.5 to 2.0 mg, below the FDA-approved starting dose of 2.5 mg, to achieve partial appetite suppression and blood sugar control with fewer gastrointestinal side effects. The approach lacks dedicated clinical trial data at these specific doses, but it builds on sound pharmacology: tirzepatide activates GIP and GLP-1 receptors in a dose-dependent manner, and partial receptor occupancy still produces measurable metabolic effects.
| Quick Reference | Details |
|---|---|
| Definition | Weekly tirzepatide doses below the 2.5 mg starting point |
| Common microdoses | 0.5 mg, 1.0 mg, 1.25 mg, 2.0 mg weekly |
| Standard starting dose | 2.5 mg weekly (FDA-approved) |
| Full therapeutic range | 5 mg, 10 mg, 15 mg weekly |
| Primary goal | Reduce side effects while maintaining partial efficacy |
| Best candidates | GI-sensitive patients, post-weight-loss maintenance, cost reduction |
| Evidence level | Pharmacological rationale; no dedicated RCTs at sub-2.5 mg doses |
| Safety | Requires medical supervision and precise measurement |
This guide covers what microdosing tirzepatide means in clinical terms, who benefits from it, the protocols providers are using, how it compares to standard titration, what the evidence actually shows, and the risks involved. If you need a refresher on the standard dosing schedule first, start with the tirzepatide dosage chart in units. If you are considering a similar approach with semaglutide instead, see how to microdose semaglutide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Microdosing Tirzepatide Actually Means
The word "microdosing" has a specific meaning in pharmacology: taking roughly one-tenth of a pharmacologically active dose. In the tirzepatide context, the term is used more loosely. It refers to any weekly dose below the 2.5 mg starting point on the FDA-approved titration schedule, or to remaining at 2.5 mg indefinitely instead of escalating upward.
The standard tirzepatide titration for weight management follows four steps: 2.5 mg for 4 weeks, then 5 mg, then 10 mg, then 15 mg as the maximum dose. Each step lasts at least 4 weeks. The SURMOUNT-1 trial (Jastreboff et al., NEJM 2022) tested 5 mg, 10 mg, and 15 mg as treatment doses. The 2.5 mg dose was designed as a tolerability ramp, not a therapeutic endpoint.
Microdosing sits below that ramp. The most common sub-therapeutic protocols use 0.5 mg, 1.0 mg, 1.25 mg, or 2.0 mg per week. Some providers prescribe the standard 2.5 mg dose but extend the injection interval to every 10 or 14 days instead of every 7, achieving a similar reduction in weekly exposure. The goal is the same in every case: activate enough dual-receptor signaling to nudge appetite and glucose metabolism without triggering the dose-dependent GI effects that drive 5 to 7% of patients to discontinue treatment.
This is different from the standard titration. During titration, 2.5 mg is a stepping stone to higher doses. In microdosing, a sub-2.5 mg dose is the destination.
It is also different from maintenance dose reduction. Some providers step patients down from 15 mg to 5 or 7.5 mg after target weight is reached. That strategy uses clinically studied doses and is covered in detail in our tirzepatide maintenance dose after weight loss guide. Microdosing goes further, into territory where controlled trial data is sparse. For the comparable approach with semaglutide, see how to microdose semaglutide.
Why People Microdose Tirzepatide: Five Common Reasons
Five distinct patient populations gravitate toward sub-therapeutic tirzepatide doses, each with a different clinical rationale.
1. Severe GI sensitivity at the starting dose
Tirzepatide's gastrointestinal side effects are dose-dependent. SURMOUNT-1 documented nausea in 24.6% of participants at 5 mg, rising to 33.3% at 15 mg, versus 9.5% on placebo (Jastreboff et al., NEJM 2022). Even the 2.5 mg starting dose can cause nausea in 12 to 18% of patients based on pooled SURPASS trial data. For these individuals, starting at 1.0 mg and titrating upward in 0.5 mg increments over 8 to 12 weeks may allow the GI tract to adapt gradually. If constipation, diarrhea, or headaches are your primary concerns, microdosing may reduce their severity.
2. Maintenance after reaching goal weight
This is the most pharmacologically defensible use case. After reaching target weight on higher doses, some patients find that 5 mg produces continued unwanted weight loss or that the appetite suppression feels excessive. SURMOUNT-4 showed that stopping tirzepatide entirely leads to 50 to 67% weight regain within 12 months (Aronne et al., JAMA 2024). A microdose may provide enough dual-receptor activation to maintain the appetite set-point shift without the full pharmacological burden. Think of it as turning a thermostat to "hold" instead of "cool."
3. Cost reduction
Tirzepatide costs range from $300 to $1,100 per month at standard doses depending on insurance. A 30 mg compounded vial at 10 mg/mL costs roughly $150 to $350. At 15 mg per week, that vial lasts 2 weeks. At a 1.0 mg microdose, the same vial lasts 30 weeks. Even accounting for the 28-day beyond-use date after reconstitution, the per-month cost drops dramatically. Use the Peptide Cost Calculator to estimate your specific expense.
4. Transitioning from semaglutide
Patients switching from semaglutide to tirzepatide sometimes start with a tirzepatide microdose to assess tolerability before committing to the standard titration. GLP-1 receptor sensitivity varies between individuals, and cross-tolerance from prior semaglutide use does not guarantee smooth tolerance of tirzepatide's added GIP receptor activation.
5. Anxiety about starting a new injectable medication
Some patients, particularly those new to self-injection, want the smallest possible first exposure. Starting at 0.5 or 1.0 mg provides a low-stakes introduction to injecting tirzepatide. If they tolerate it well, confidence builds. If they react poorly, the pharmacological exposure was minimal. Tirzepatide's half-life is approximately 5 days (Coskun et al., Mol Metab 2018), so even a microdose lingers, but the absolute drug exposure is low.
Microdosing Protocols: What Providers Are Prescribing
No FDA-approved microdosing protocol exists for tirzepatide. The protocols below come from clinical practice patterns observed in obesity medicine settings and telehealth prescribing data. Your provider should determine the specific protocol for your situation.
Protocol A: Ultra-Low Start (0.5 mg to 1.25 mg)
This protocol is designed for patients who experienced severe GI side effects on prior GLP-1 agonist therapy or who have a history of gastroparesis or functional dyspepsia.
| Week | Dose | Frequency | Purpose |
|---|---|---|---|
| 1 to 2 | 0.5 mg | Once weekly | Assess baseline GI tolerance |
| 3 to 4 | 1.0 mg | Once weekly | Monitor for nausea, appetite changes |
| 5 to 8 | 1.25 mg | Once weekly | Evaluate weight trend, hunger scores |
| 9 to 12 | 2.0 mg | Once weekly | Bridge toward standard starting dose |
| 13 onward | 2.5 mg or stay at 2.0 mg | Once weekly | Based on response and tolerance |
The increments here are 50 to 100% increases per step, but the absolute dose increase is small: 0.5 mg per jump. Compare this to the standard schedule where the first escalation is from 2.5 mg to 5.0 mg, a 100% increase that doubles the absolute drug exposure overnight.
Protocol B: Extended Interval Dosing
Instead of reducing the dose, some providers extend the injection interval. Given tirzepatide's 5-day half-life (Thomas et al., Clin Pharmacokinet 2021), injecting 2.5 mg every 10 to 14 days maintains a lower average steady-state concentration than weekly dosing. This approach uses the standard 2.5 mg volume, which simplifies measurement.
| Interval | Effective Weekly Dose | Pros | Cons |
|---|---|---|---|
| Every 7 days | 2.5 mg/week | Stable levels, standard protocol | May still cause GI effects for sensitive patients |
| Every 10 days | ~1.75 mg/week | Simpler measurement, no dilution needed | Hunger returns before next dose |
| Every 14 days | ~1.25 mg/week | Longest interval between injections | Significant trough, inconsistent appetite control |
The downside is fluctuating drug levels. By day 10 after a 2.5 mg injection, plasma concentrations have dropped to roughly 25% of peak. Some patients report a "roller coaster" of good appetite control for the first 5 days followed by increasing hunger and cravings for the remaining days before the next injection. For context on how long the drug remains active, see how long tirzepatide stays in your system.
Protocol C: Post-Weight-Loss Taper to Microdose
This is the most conservative and clinically grounded approach. It uses standard doses during active weight loss, then tapers below the studied range for long-term maintenance.
| Phase | Dose | Duration | Goal |
|---|---|---|---|
| Titration | 2.5 mg to 15 mg | 16 weeks | Per FDA label |
| Weight loss | 10 or 15 mg | Until target weight | Typically 9 to 18 months |
| Taper | 7.5 mg, 5 mg, 2.5 mg | 4 weeks per step | Gradual step-down |
| Microdose maintenance | 1.0 to 2.0 mg | Indefinite | Prevent weight regain |
The taper phase uses clinically studied doses. Only the final maintenance step enters sub-therapeutic territory. This approach captures the proven benefits of higher doses during active weight loss while using microdosing principles only for the long-term phase where the goal shifts from losing weight to preventing regain. If your weight starts climbing during the taper, return to the previous dose immediately. See our guide on why you may not be losing weight on tirzepatide for troubleshooting stalls at any dose level.
How to Measure Tirzepatide Microdoses Accurately
Measuring 1.0 mg of tirzepatide from a compounded vial demands careful math and the right syringe. The margin for error gets tighter as the dose shrinks. One unit of overshoot on a small draw can mean a 20 to 50% dosing error.
Step 1: Identify your vial concentration.
Read the label. Common compounded tirzepatide concentrations are 5 mg/mL, 10 mg/mL, 20 mg/mL, and 40 mg/mL. If you reconstituted the vial yourself, your concentration depends on how much bacteriostatic water you added. The Peptide Reconstitution Calculator gives you the exact number.
Step 2: Calculate your draw volume.
Use this formula: mL = dose (mg) / concentration (mg/mL)
Then multiply by 100 to convert to syringe units.
| Dose | At 5 mg/mL | At 10 mg/mL | At 20 mg/mL | At 40 mg/mL |
|---|---|---|---|---|
| 0.5 mg | 10 units | 5 units | 2.5 units | 1.25 units |
| 1.0 mg | 20 units | 10 units | 5 units | 2.5 units |
| 1.25 mg | 25 units | 12.5 units | 6.25 units | 3.125 units |
| 2.0 mg | 40 units | 20 units | 10 units | 5 units |
| 2.5 mg | 50 units | 25 units | 12.5 units | 6.25 units |
Look at the 40 mg/mL column. A 1.0 mg dose requires drawing 2.5 units. That is nearly impossible to measure accurately on a standard insulin syringe where each line marks 1 unit. This is where concentration choice becomes critical.
Step 3: Pick the right concentration and syringe pairing.
For microdosing, you want the largest draw volume possible. That means using the lowest available concentration. At 5 mg/mL, a 1.0 mg dose requires 20 units, easily measurable on any insulin syringe. At 10 mg/mL, the same dose requires 10 units, still workable. Above 10 mg/mL, you need half-unit syringes for sub-2.5 mg doses.
If your pharmacy sent a 20 or 40 mg/mL vial, ask about exchanging it for a lower concentration, or request reconstitution instructions that produce 5 mg/mL. Alternatively, use 0.3 mL half-unit insulin syringes (BD Ultra-Fine or EasyTouch) that mark every 0.5 units. For an instant answer on your specific vial, use the Tirzepatide Dosage Calculator or the Peptide Unit Converter.
Step 4: Consider diluting for better accuracy.
Some providers instruct patients to add extra bacteriostatic water to an already-reconstituted vial. Adding 1 mL of bacteriostatic water to a 10 mg/mL vial containing 1 mL of solution produces 10 mg in 2 mL, or 5 mg/mL. This doubles your draw volume for any given dose. The tradeoff: larger total volume means more vial punctures and a higher contamination risk with each draw. Use aseptic technique throughout. For details on how to store peptides and reconstituted solutions, our storage guide covers best practices.
For a walkthrough of unit math at standard doses, see how many units is 2.5 mg of tirzepatide.
Microdosing Tirzepatide vs. Standard Titration: A Direct Comparison
The standard tirzepatide titration exists because clinical trials validated it. Understanding what you trade away at a microdose matters for informed decision-making.
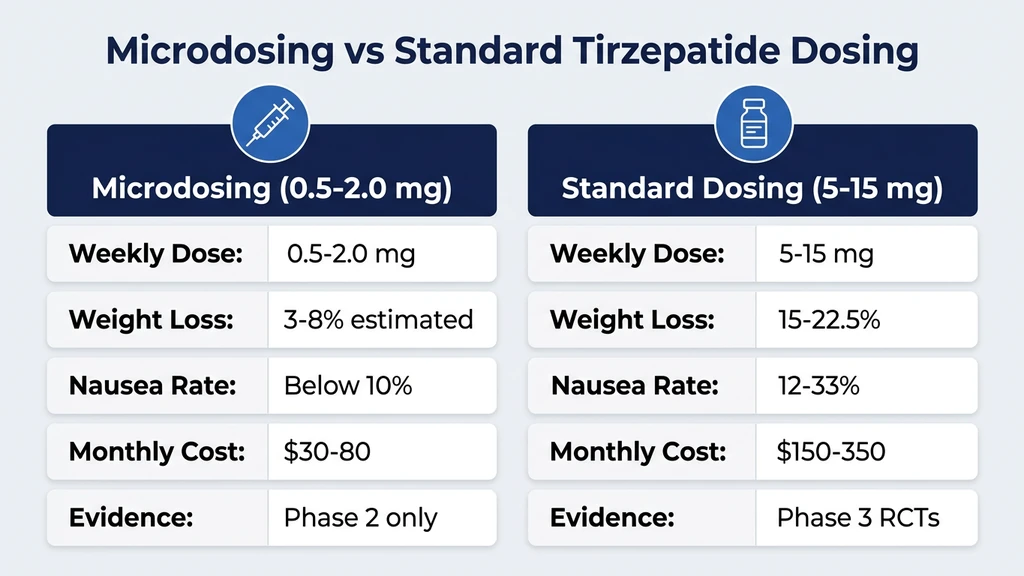
| Parameter | Standard Titration (2.5 to 15 mg) | Microdosing (0.5 to 2.0 mg) |
|---|---|---|
| Weight loss | 15 to 22.5% body weight at 72 weeks (SURMOUNT-1) | Unknown; estimated 3 to 8% based on dose-response extrapolation |
| HbA1c reduction | 1.9 to 2.4% at 5 to 15 mg (SURPASS-2) | Unknown at sub-2.5 mg doses |
| Nausea rate | 12 to 33% depending on dose | Estimated below 10% |
| Discontinuation due to GI effects | 4.3 to 7.1% | Expected below 2% |
| Cost (compounded) | $150 to $350/month | $30 to $80/month |
| Clinical trial evidence | Phase 3 RCTs (SURMOUNT, SURPASS programs) | None at sub-2.5 mg doses |
| FDA approval | Yes, at 5 to 15 mg for obesity and T2D | No |
The dose-response curve is not linear.
This is the key pharmacological point. The first milligrams of tirzepatide exposure produce a proportionally larger effect than subsequent milligrams. SURMOUNT-1 documented weight loss of 15.0% at 5 mg, 19.5% at 10 mg, and 20.9% at 15 mg (Jastreboff et al., NEJM 2022). Notice the diminishing returns: tripling the dose from 5 to 15 mg only increased weight loss by 39%. The jump from 0 to 5 mg produced the vast majority of the benefit.
This non-linear relationship suggests that sub-therapeutic doses capture a meaningful fraction of the metabolic effect. A 1.0 mg dose does not deliver one-fifth of the benefit of 5.0 mg. It likely delivers a higher proportion, perhaps 30 to 50%, though this remains an extrapolation without direct trial evidence.
Where standard titration wins clearly:
Standard doses are the proven path if you have a BMI above 35 with obesity-related comorbidities, or if you need significant HbA1c reduction for type 2 diabetes. The SURPASS-2 trial (Frias et al., NEJM 2021) showed tirzepatide 5 mg reduced HbA1c by 2.01% versus 1.86% with semaglutide 1 mg. At 15 mg, the reduction was 2.30%. If glycemic control is your primary goal, microdosing likely falls short.
Clinical Evidence: What the Research Shows About Low-Dose Tirzepatide
The honest assessment: no published randomized controlled trial has tested tirzepatide at doses below 2.5 mg weekly. The evidence for microdosing is indirect, drawn from dose-response pharmacology, receptor binding studies, and related trial data.
SURMOUNT-1 dose-response data
The SURMOUNT-1 trial tested 5 mg, 10 mg, and 15 mg against placebo in 2,539 adults with obesity (Jastreboff et al., NEJM 2022). The 5 mg arm, the lowest therapeutic dose, produced 15.0% mean body weight reduction at 72 weeks. Placebo produced 3.1%. The 5 mg dose was already dramatically effective, suggesting that receptor activation at even lower doses produces clinically meaningful effects.
Dual-receptor pharmacology
Tirzepatide is the only approved medication that activates both GIP and GLP-1 receptors simultaneously. The GIP receptor activation appears to amplify the weight loss effects of GLP-1 receptor agonism while potentially reducing GI intolerance (Nauck & D'Alessio, Lancet 2022). At microdose levels, this dual mechanism may produce more noticeable effects per milligram than a pure GLP-1 agonist like semaglutide at equivalent fractional doses.
SURPASS dose-finding studies
The Phase 2 dose-finding study for tirzepatide in type 2 diabetes (Frias et al., Lancet 2018) tested 1 mg, 5 mg, 10 mg, and 15 mg weekly. The 1 mg dose, the closest published data point to a microdose, produced statistically significant HbA1c reduction of 0.7% and 0.9% body weight loss at 26 weeks versus placebo. This is direct evidence that 1 mg tirzepatide weekly produces measurable metabolic effects, even though it was not advanced to Phase 3 trials.
This is the strongest piece of evidence supporting tirzepatide microdosing. The 1 mg dose in the Phase 2 study functioned as a proof-of-concept that sub-therapeutic doses are not pharmacologically inert. They are simply less potent.
Receptor binding kinetics
Tirzepatide binds GIP receptors with similar affinity to native GIP and GLP-1 receptors with approximately 5-fold lower affinity than native GLP-1 (Coskun et al., Mol Metab 2018). At microdose concentrations, partial receptor occupancy still triggers intracellular signaling cascades. The dose-response curve for receptor activation is typically sigmoidal: the steepest portion of the curve occurs at low receptor occupancy, meaning the first molecules of drug produce disproportionately large effects relative to full saturation.
What we do not know
No trial has compared microdose tirzepatide to microdose semaglutide. We do not know whether tirzepatide's GIP component provides additional benefit at sub-therapeutic GLP-1 receptor occupancy levels. We do not know the durability of weight loss at microdose maintenance. And we do not know whether cardiovascular risk reduction, demonstrated with semaglutide 2.4 mg in the SELECT trial (Lincoff et al., NEJM 2023), persists at microdose levels.
Managing Side Effects at Sub-Therapeutic Doses
One of the primary reasons people microdose tirzepatide is to avoid the GI side effects that make standard doses difficult. At sub-2.5 mg doses, side effects are generally milder, but they do not disappear entirely.
Expected side effect profile at microdose levels:
| Side Effect | Rate at 5 mg (SURMOUNT-1) | Estimated Rate at Microdose | Management |
|---|---|---|---|
| Nausea | 24.6% | Below 10% | Eat bland foods, small meals |
| Diarrhea | 18.7% | Below 8% | Stay hydrated, reduce fat intake |
| Constipation | 6.2% | Below 4% | Psyllium husk, 80+ oz water daily |
| Headache | 5 to 8% | Below 4% | Hydration, electrolytes |
| Fatigue | 3 to 5% | Below 3% | Adequate protein, sleep hygiene |
| Injection site reaction | 2 to 5% | 2 to 5% (dose-independent) | Rotate sites, warm solution to room temp |
For detailed management strategies for each side effect, see our guides on tirzepatide constipation, tirzepatide diarrhea, tirzepatide headaches, and tirzepatide fatigue.
The paradox of microdosing for side effect avoidance
Some patients microdose to avoid side effects, only to discover they tolerate 2.5 mg fine. The 2.5 mg dose was specifically designed as a tolerability ramp. The SURMOUNT trials documented that most GI side effects at 2.5 mg are mild and transient, peaking during weeks 1 to 3 and resolving by week 4 in the majority of cases. If your only reason for microdosing is fear of side effects rather than prior experience with them, consider that 2.5 mg may be tolerable after all.
When side effects at microdose levels signal a problem
Significant nausea at 0.5 or 1.0 mg is unusual. If it occurs, it may indicate gastroparesis, delayed gastric emptying from another cause, pregnancy (GLP-1 agonists are contraindicated in pregnancy), or a hypersensitivity to one of the excipients in the compounded formulation. Report any side effects at microdose levels to your provider. Review the peptide safety guide for a broader safety framework, and check is compound tirzepatide safe if you are using a compounding pharmacy formulation.
How to Store Tirzepatide for Extended Microdose Use
Microdosing creates a math problem with storage. A 30 mg vial at 1.0 mg per week contains 30 doses, enough for over 7 months. But reconstituted tirzepatide has a 28-day beyond-use date per USP <797> compounding standards. You cannot use one vial for 30 weeks.
Option 1: Request smaller vials
Some compounding pharmacies offer 5 mg or 10 mg vials. A 10 mg vial at 1.0 mg per week lasts 10 doses (10 weeks), still exceeding 28 days. A 5 mg vial at 1.0 mg per week lasts 5 weeks, closer to the limit but still over. Ask your pharmacy about 2.5 mg or 5 mg vials. Smaller vials waste less product.
Option 2: Accept some waste
Reconstitute the full vial, draw 4 microdoses over 28 days, and discard the remainder. At 1.0 mg weekly from a 30 mg vial, you use 4 mg and discard 26 mg. This sounds wasteful, but at compounded pricing of $150 to $350 per vial, you are paying $20 to $47 per month. That is dramatically cheaper than $300+ for standard-dose branded Mounjaro or Zepbound.
Option 3: Lyophilized powder with partial reconstitution
If your pharmacy ships lyophilized powder with separate bacteriostatic water, ask your provider whether partial reconstitution is appropriate. This is not standard practice and requires precise technique, but dissolving only the portion you will use within 28 days reduces waste.
Storage fundamentals regardless of dose:
Refrigerate at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Keep the vial upright. Never freeze. Discard if cloudy, discolored, or containing particles. For complete storage details, see how long tirzepatide lasts in the fridge and how to store peptides.
Mark the reconstitution date on every vial with a permanent marker. At microdose draw frequencies, you puncture the stopper less often but over a longer period. Each puncture introduces a contamination opportunity. Use a fresh needle for every draw and swab the stopper with alcohol every time.
Tracking Your Response to Microdose Tirzepatide
Microdosing produces subtler effects than standard dosing. You cannot rely on the dramatic appetite extinction that 10 or 15 mg produces. Instead, you need deliberate tracking to determine whether a sub-therapeutic dose is working for you.
Track these metrics weekly:
Body weight. Weigh yourself at the same time each morning, after using the bathroom, before eating. Record the 7-day average. Daily fluctuations of 2 to 4 pounds are normal. A downward trend in the weekly average is what matters. At microdose levels, expect 0.25 to 0.75% body weight loss per month, not the 1 to 2% monthly loss seen at standard doses.
Caloric intake. Use a food tracking app for at least the first 8 weeks. Microdose tirzepatide should reduce your average daily intake by 150 to 400 calories if it is working. Without tracking, you will not notice this shift.
Hunger scores. Rate your hunger before meals on a 1 to 10 scale. Rate how full you feel 20 minutes after eating. Even a 1 to 2 point shift toward lower hunger and earlier satiety is clinically relevant at these dose levels.
Fasting blood glucose. If you have access to a glucometer, check fasting glucose weekly. The Phase 2 trial showed that 1 mg tirzepatide reduced fasting glucose by 14 mg/dL versus placebo (Frias et al., Lancet 2018). Measuring glucose gives you an objective biomarker independent of subjective hunger perception.
Side effects. Log any GI symptoms, fatigue, injection site reactions, or other changes. At microdose levels, GI effects should be minimal. Significant symptoms warrant a conversation with your provider.
Decision point at 8 weeks:
If your weekly weight average has not decreased, caloric intake has not measurably dropped, hunger scores are unchanged, and fasting glucose is stable, the microdose is likely sub-therapeutic for you. Discuss titrating to 2.5 mg with your provider. Not everyone responds to sub-therapeutic doses, and the standard starting dose exists for a reason.
If you are losing 0.25 to 0.75% of body weight monthly with minimal side effects, the microdose is in your therapeutic window. Continue and reassess quarterly.
When Microdosing Tirzepatide Is Not Appropriate
Microdosing has a place, but it is not the right choice for every patient. These situations call for standard-dose treatment or a different approach entirely.
Active type 2 diabetes requiring aggressive glycemic control. Tirzepatide is FDA-approved for type 2 diabetes at 5 to 15 mg weekly (Mounjaro prescribing information, Eli Lilly, 2022). The SURPASS-2 trial demonstrated HbA1c reductions of 2.01% at 5 mg and 2.30% at 15 mg (Frias et al., NEJM 2021). At microdose levels, glycemic improvement is likely insufficient to achieve HbA1c targets below 7%. If your A1c is above 8%, standard dosing is the evidence-based path.
BMI above 40 with obesity-related comorbidities. Patients with severe obesity and comorbidities like obstructive sleep apnea, NAFLD, or osteoarthritis benefit most from the substantial weight loss achievable at standard doses. The SURMOUNT-1 trial showed 20.9% weight loss at 15 mg. A microdose cannot replicate this magnitude of effect.
History of medullary thyroid carcinoma or MEN2 syndrome. This is a contraindication at any dose. GLP-1 receptor agonists caused thyroid C-cell tumors in rodent studies at all dose levels tested (Mounjaro prescribing information, Eli Lilly, 2022). The risk is not dose-dependent in preclinical models.
Concurrent use of other GLP-1 receptor agonists. Combining a tirzepatide microdose with semaglutide, liraglutide, or dulaglutide stacks agonism on the same receptor. This amplifies GI effects without proportional clinical benefit and is medically inappropriate.
Self-prescribing without medical supervision. Microdosing requires the same monitoring as standard dosing: periodic blood work, weight tracking, and side effect assessment. The lower dose does not eliminate the need for a provider. Compounded tirzepatide from unverified sources carries additional risks. See is compound tirzepatide safe for a detailed safety analysis.
Tirzepatide Microdosing vs. Semaglutide Microdosing
If you are considering microdosing a GLP-1 agonist, you have two main options: tirzepatide and semaglutide. The comparison at sub-therapeutic doses involves important pharmacological differences.
| Parameter | Tirzepatide Microdose | Semaglutide Microdose |
|---|---|---|
| Receptor targets | GIP + GLP-1 (dual agonist) | GLP-1 only |
| Common microdose range | 0.5 to 2.0 mg weekly | 0.05 to 0.25 mg weekly |
| Half-life | ~5 days | ~7 days |
| Evidence at low doses | Phase 2 data at 1 mg | SUSTAIN dose-finding data |
| GI tolerability | May be better due to GIP buffering | Dose-dependent nausea |
| Cost (compounded) | $30 to $80/month at microdose | $20 to $50/month at microdose |
| Available concentrations | 5, 10, 20, 40 mg/mL | 2.5, 5, 10 mg/mL |
Tirzepatide's potential advantage at microdose levels: GIP receptor activation appears to partially counteract the nausea and vomiting driven by GLP-1 receptor stimulation in the area postrema of the brainstem (Samms et al., J Clin Invest 2023). This means tirzepatide may produce a more favorable side-effect-to-efficacy ratio at low doses than semaglutide, where the only active mechanism also drives the GI effects.
Semaglutide's advantage: Longer half-life (7 versus 5 days) means more stable plasma levels between weekly injections, potentially more consistent appetite control. Also, semaglutide has more published real-world data on sub-therapeutic dosing from the years it has been available before tirzepatide's approval.
For detailed guidance on the semaglutide approach, see how to microdose semaglutide. For the standard dosing comparison, see the semaglutide dosage chart in mL. And if you are weighing a switch between the two medications, the semaglutide to tirzepatide switching guide covers cross-titration protocols. You can also explore dosing math for both with the Semaglutide Dosage Calculator and the Tirzepatide Dosage Calculator.
Step-by-Step: Your First Tirzepatide Microdose Injection
Assuming your provider has prescribed a sub-therapeutic dose and you have a compounded tirzepatide vial, here is the exact procedure. This example uses a 1.0 mg dose from a 5 mg/mL vial.
Gather your supplies: - Reconstituted tirzepatide vial (5 mg/mL, refrigerated) - 0.5 mL (50-unit) or 0.3 mL (30-unit) insulin syringe - Alcohol swabs - Sharps container
Calculate your draw volume: 1.0 mg / 5 mg/mL = 0.2 mL = 20 units
Procedure:
- 1.Wash your hands. Remove the vial from the refrigerator and let it sit at room temperature for 5 minutes. Cold solution is more viscous and harder to draw accurately.
- 1.Swab the vial stopper with an alcohol pad. Let it dry for 10 seconds.
- 1.Pull the plunger back to the 20-unit mark to draw air into the syringe.
- 1.Insert the needle through the stopper. Push the plunger to inject air into the vial. This equalizes pressure.
- 1.Invert the vial. Position the needle tip in the liquid, away from the stopper.
- 1.Pull the plunger slowly to the 20-unit mark. At this volume, accuracy matters. One unit of overshoot is a 5% dosing error; at smaller doses the error percentage climbs higher.
- 1.Check for air bubbles. Tap the syringe barrel gently to move bubbles to the top. Push the plunger slightly to expel them. Re-draw to the 20-unit mark if needed.
- 1.Remove the needle from the vial.
- 1.Clean your injection site (abdomen, thigh, or upper arm) with an alcohol swab. Let it dry.
- 1.Pinch a fold of skin. Insert the needle at a 90-degree angle. Depress the plunger slowly. Count to 5 after the plunger is fully depressed before withdrawing.
- 1.Dispose of the syringe in your sharps container. Return the vial to the refrigerator.
For a complete injection tutorial with site rotation guidance and troubleshooting, see how to inject tirzepatide. If your vial was shipped as lyophilized powder, see how to reconstitute tirzepatide before attempting any draw. For broader guidance on injection technique, consult the peptide dosage chart.
Frequently Asked Questions
What is the lowest effective dose of tirzepatide for weight loss?
The lowest FDA-studied therapeutic dose is 5 mg weekly, which produced 15.0% body weight loss in SURMOUNT-1. The Phase 2 trial showed 1 mg weekly produced 0.9% weight loss at 26 weeks. Microdoses of 0.5 to 2.0 mg likely produce partial appetite suppression, but no Phase 3 trial has confirmed their efficacy for weight management.
Can I split my tirzepatide dose without my doctor knowing?
Self-adjusting doses without provider oversight is strongly discouraged. Even dose reductions require monitoring of metabolic markers and side effects. Your prescriber needs accurate dosing information to make safe clinical decisions. Unmonitored dose changes introduce uncontrolled variables that can compromise 100% of your care plan.
How long does it take to feel the effects of microdose tirzepatide?
Tirzepatide reaches steady-state plasma concentration after approximately 4 weeks of consistent weekly dosing. At microdose levels, appetite changes are subtle and may take 4 to 8 weeks to become noticeable. Track caloric intake and hunger scores rather than relying on how you feel. The Phase 2 trial measured outcomes at 26 weeks.
Is microdosing tirzepatide safer than taking the full dose?
Lower doses produce fewer dose-dependent side effects like nausea (24.6% at 5 mg versus an estimated sub-10% at microdoses). However, dose-independent risks like thyroid C-cell tumor concerns and pancreatitis remain theoretical at any dose. Microdosing is not a workaround for contraindications. Medical supervision is still required.
Will microdose tirzepatide show up on a drug test?
Standard workplace drug tests do not screen for tirzepatide or GLP-1 agonists. These tests target substances like amphetamines, opioids, and cannabinoids. However, WADA added GLP-1 receptor agonists to its 2024 monitoring program. Competitive athletes should check their sport's anti-doping rules before using any dose.
Can I microdose tirzepatide and semaglutide at the same time?
No. Combining two GLP-1 receptor agonists stacks stimulation on the same receptor pathway. This amplifies gastrointestinal side effects without proportional clinical benefit. The SURPASS-2 trial compared tirzepatide and semaglutide as alternatives, not as combination therapy. Choose one medication, not both.
How much does microdosing tirzepatide cost per month?
A 30 mg compounded vial costs $150 to $350. At a 1.0 mg weekly microdose, you use approximately 4 mg per 28-day period, putting your monthly cost at $20 to $47 depending on waste. Standard-dose branded Mounjaro costs $1,000+ monthly without insurance. Microdosing compounded tirzepatide costs roughly 95% less.
Should I microdose tirzepatide or switch to semaglutide instead?
The answer depends on your reason for considering lower doses. If GI side effects drove you away from tirzepatide, semaglutide may trigger similar issues since both activate GLP-1 receptors. If cost is the driver, microdosing compounded tirzepatide is typically cheaper than switching. Discuss both options with your provider using the data from your first 4 weeks.
The Bottom Line
Microdosing tirzepatide means using weekly doses of 0.5 to 2.0 mg, below the FDA-approved 2.5 mg starting point, to achieve partial appetite suppression and metabolic benefits with fewer side effects. The Phase 2 dose-finding trial provides direct evidence that 1 mg tirzepatide weekly produces measurable HbA1c and weight reductions versus placebo (Frias et al., Lancet 2018), and the non-linear dose-response curve from SURMOUNT-1 supports the premise that sub-therapeutic doses capture a meaningful fraction of the metabolic benefit.
The approach works best for two groups: patients tapering to maintenance after reaching goal weight, and patients whose GI sensitivity prevents tolerating the standard 2.5 mg starting dose. For primary weight loss in patients with BMI above 30, standard titration to 5 to 15 mg remains the evidence-based choice per the SURMOUNT program (Jastreboff et al., NEJM 2022).
If you pursue microdosing, precision matters. Use the lowest available vial concentration (5 mg/mL) with a 0.3 mL insulin syringe. Track your weight, caloric intake, hunger scores, and fasting glucose weekly. Reassess at 8 weeks. And keep your provider fully informed.
For dose calculations, use the Tirzepatide Dosage Calculator. For reconstitution planning, use the Peptide Reconstitution Calculator. For unit conversions, the Peptide Unit Converter handles the math instantly.
Related Articles: - Tirzepatide Maintenance Dose After Weight Loss - stepping down after reaching goal weight - How to Microdose Semaglutide - the semaglutide-specific approach - Tirzepatide Dosage Chart in Units - standard dose conversion reference - How Long Does Tirzepatide Take to Work? - timeline expectations - Semaglutide to Tirzepatide Switching Guide - cross-titration protocols - Peptide Safety Guide - comprehensive safety reference - Compound Semaglutide with B12 - alternative compounded formulations - How Long Does Tirzepatide Stay in Your System? - pharmacokinetics
Helpful Tools
Related Articles
How to Microdose Semaglutide: Guide
Learn how to microdose semaglutide at 0.1-0.25 mg for appetite control with fewer side effects. Covers protocols, measuring small doses, evidence, and risks.
Tirzepatide Maintenance Dose Guide
Most patients maintain on 5-10 mg tirzepatide weekly after reaching goal weight. SURMOUNT-4 data shows stopping leads to 50-67% weight regain within 12 months.
Tirzepatide Injection Site Reactions
Injection site reactions affect 3-7% of tirzepatide users. SURMOUNT trial rates, 5 reaction types, severity guide, and 9 prevention strategies.
Does Tirzepatide Cause Insomnia?
Does tirzepatide cause insomnia? Clinical trials say no, but 5 indirect pathways explain why some users lose sleep. GI disruption, blood sugar, caloric deficit fixes inside.