You start at 0.25 mg. You want to end at 2.4 mg. Between those two numbers sits 16 weeks of careful, non-negotiable escalation that most telehealth patients never actually complete.
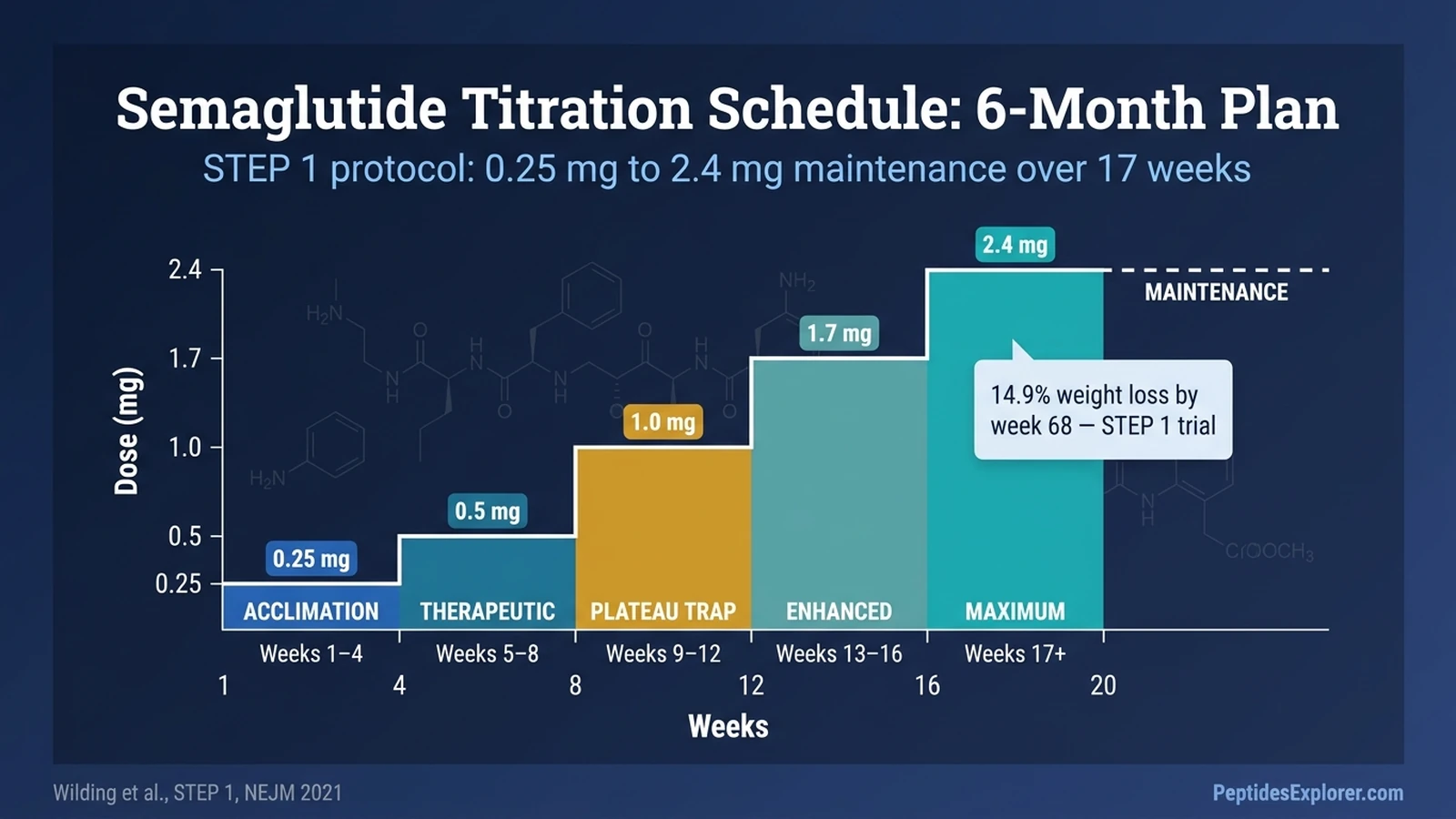
The standard semaglutide titration schedule climbs through five doses, 4 weeks each: 0.25 mg → 0.5 mg → 1.0 mg → 1.7 mg → 2.4 mg weekly. You reach the maintenance dose at week 17. You hold it for the rest of the year, and most likely, the rest of the treatment.
That is the official schedule. The real problem is the gap between what the Wegovy FDA label says and what your compounded prescription actually does. A large share of Ro, Hims, and Mochi patients get parked at 1.0 mg and never move. They lose weight. They plateau. They blame the drug. The drug is working exactly as designed at a dose below what earned the 14.9% weight-loss result in STEP 1.
This guide gives you the full 6-month plan, the three legitimate reasons to deviate from it, the quantified cost of doing it wrong, and the script to ask your provider to escalate past 1.0 mg. Run the numbers for your own vial with the semaglutide dosage calculator before each step.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Semaglutide Titration Schedule: Quick Reference Table
This is the 20-week schedule used in STEP 1, STEP 3, STEP 4, and copied into the Wegovy label. Hold each dose for a minimum of 4 weeks before advancing.
| Week Range | Dose (mg) | Purpose | What to Monitor | Do NOT Escalate If |
|---|---|---|---|---|
| 1–4 | 0.25 | Receptor acclimation; sub-therapeutic on purpose | Nausea, injection-site reactions, hydration | Vomiting more than twice a week, dehydration |
| 5–8 | 0.5 | First therapeutic dose; appetite suppression starts | Persistent nausea, early satiety, constipation | Nausea still severe or weight loss over 3 lbs/wk |
| 9–12 | 1.0 | Strong appetite control; plateau dose for many compounded patients | Muscle mass, gallbladder symptoms, energy | Unresolved GI symptoms from prior step |
| 13–16 | 1.7 | Enhanced efficacy; alternative maintenance if 2.4 not tolerated | Fatigue, mood, hair shedding | Vomiting or response already strong at 1.0 |
| 17–20 | 2.4 | Maximum approved weight-loss dose; ~15% loss by week 68 | All above plus HbA1c, lipids, kidney labs | GI intolerance; down-titrate to 1.7 permanently |
| 21+ | 2.4 (maintenance) | Indefinite; stopping causes ~7% regain per year | Plateau timing (weeks 60–68), bone density | Maintenance is the goal, not a stepping stone |
The core rule across every step: minimum 4 weeks per dose before advancing. If you cannot tolerate the next dose, stay at the current one for another 4 weeks. The FDA label explicitly permits this delay. A second extension is reasonable but warrants a conversation with your prescriber.
For the syringe-by-syringe conversion at each dose, bookmark the semaglutide dosage chart in mL. For any intermediate dose your pharmacy offers (like 0.75 mg on a compounded bridge), the peptide unit converter handles it instantly.
Why Every Step Is 4 Weeks (The Steady-State Rule)
Four weeks is not arbitrary. It is pharmacokinetics.
Semaglutide has a half-life of roughly 7 days. After one dose, blood levels peak, then fall to half by day 7. After two doses a week apart, the second dose stacks on top of residual drug from the first. After four doses, roughly 94% of steady-state concentration is reached.
Picture filling a bathtub where the drain is always open. The drug is the faucet. The drain is your metabolism clearing the peptide. At a given weekly dose, the tub fills to a specific water line: that's steady state. Increase the faucet rate (escalate the dose) and the water line rises over the next four weeks as the new flow overcomes the drain.
If you escalate before steady state, you are turning up the faucet while the tub is still filling from the last change. The water spikes higher than you planned. That spike is where severe nausea, vomiting, and dehydration come from. Wait the four weeks. Let the tub settle. Then adjust.
This is also why injections do not have to happen at the exact same hour each week. The drug's long half-life smooths out small timing variations. For the detailed pharmacokinetic timeline of what happens inside your bloodstream, see how long does semaglutide take to work.
Step-by-Step: What to Expect at Each Dose
Each dose has a specific therapeutic purpose, a predictable side-effect window, and a clear signal that tells you whether to advance or hold. Knowing what to expect prevents both premature escalation and unnecessary plateau at a sub-therapeutic dose.
Weeks 1–4: 0.25 mg (Receptor Acclimation)
This dose is not supposed to produce dramatic weight loss. That surprises most patients. Expect 2 to 4 pounds total across 4 weeks, maybe less. Some people lose nothing. That is fine.
The purpose is tolerance building. Your GLP-1 receptors, gastric emptying rate, and nausea pathways need to adapt to the drug before stronger doses arrive. Nausea, fatigue, and mild constipation are common. Most symptoms peak 48 to 72 hours after each injection and fade by day 4 or 5.
Advance at week 5 unless you have had vomiting more than twice in the last 7 days, signs of dehydration, or a resting heart rate jumping more than 15 bpm above baseline. For active management of the nausea window, read how to relieve nausea from semaglutide.
Weeks 5–8: 0.5 mg (First Therapeutic Dose)
This is where it becomes real. Appetite suppression arrives. Food noise quiets. Most patients lose 1 to 2 pounds a week during this window.
GI symptoms tend to spike in the 72 hours after the first 0.5 mg injection, then settle. Early satiety becomes obvious at meals. Some patients report constipation for the first time. If that happens, see can semaglutide cause diarrhea and constipation for management.
Hold at 0.5 mg for another 4 weeks if nausea is still severe, vomiting is happening more than once a week, or you are losing weight faster than 3 pounds a week. Rapid loss at a low dose signals under-eating driven by nausea, not therapeutic appetite control, and risks muscle loss.
Weeks 9–12: 1.0 mg (The Compounded Plateau Trap)
1.0 mg is where roughly 30% of semaglutide patients find their personal maintenance dose. It is also the dose where most compounded telehealth patients silently stop escalating.
Ozempic maxes out at 2.0 mg for type-2 diabetes, so for many telehealth protocols, 1.0 mg becomes the default endpoint. Your provider may not tell you to keep climbing. Pay attention to muscle-mass loss, right-upper-quadrant pain (gallbladder), and energy levels. See also compounded semaglutide with B12, where B12 is added to offset the fatigue some patients report at this stage.
If you are still tolerating the dose at week 12 and your weight loss curve is still descending, you should advance to 1.7 mg. Full stop. Staying here by default is the single most common way patients fall short of trial-level results.
Weeks 13–16: 1.7 mg (Enhanced Efficacy)
The 1.7 mg step adds another layer of appetite control and pushes weight loss toward the 10% mark by week 16 for most responders. The FDA label recognizes 1.7 mg as an alternative permanent maintenance dose if 2.4 mg is not tolerated.
Two new side effects become more visible at this dose: hair shedding (telogen effluvium, typically onset 2 to 3 months after entering this range) and flat affect or low mood. Neither is universal, but both are common enough that the label flags them. Track protein intake; the semaglutide mixing chart includes guidance on B12 co-formulations often used at this stage.
Hold at 1.7 mg permanently if your response is strong, if you are experiencing fatigue you cannot offset with food and sleep, or if your provider recommends avoiding the 2.4 mg step for cardiovascular reasons. 1.7 mg is a legitimate destination.
Weeks 17–20: 2.4 mg (Maximum Approved Weight-Loss Dose)
This is the STEP 1 endpoint. At 2.4 mg maintenance, the average patient in the trial lost 14.9% of body weight by week 68. Eighty-six percent achieved at least 5% loss. A third achieved more than 15%.
Side-effect incidence at 2.4 mg from pooled STEP 1–3 data: nausea 43.9%, diarrhea 29.7%, vomiting 24.5%, constipation 24.2%. GI-event discontinuation was 4.5% on semaglutide versus 0.8% on placebo. Those percentages are the price of the peak result.
At week 21 you enter indefinite maintenance. Labs become important: HbA1c if diabetic, lipid panel, kidney function, and a thyroid check during the first year. If you are not losing weight despite a clean titration, see not losing weight on semaglutide for the full troubleshooting tree.
When to Deviate From the Standard Schedule
The Wegovy label permits three deviations from the 4-week-per-step rule. Each has a specific trigger and a specific protocol. Improvising outside these is where titration goes wrong.
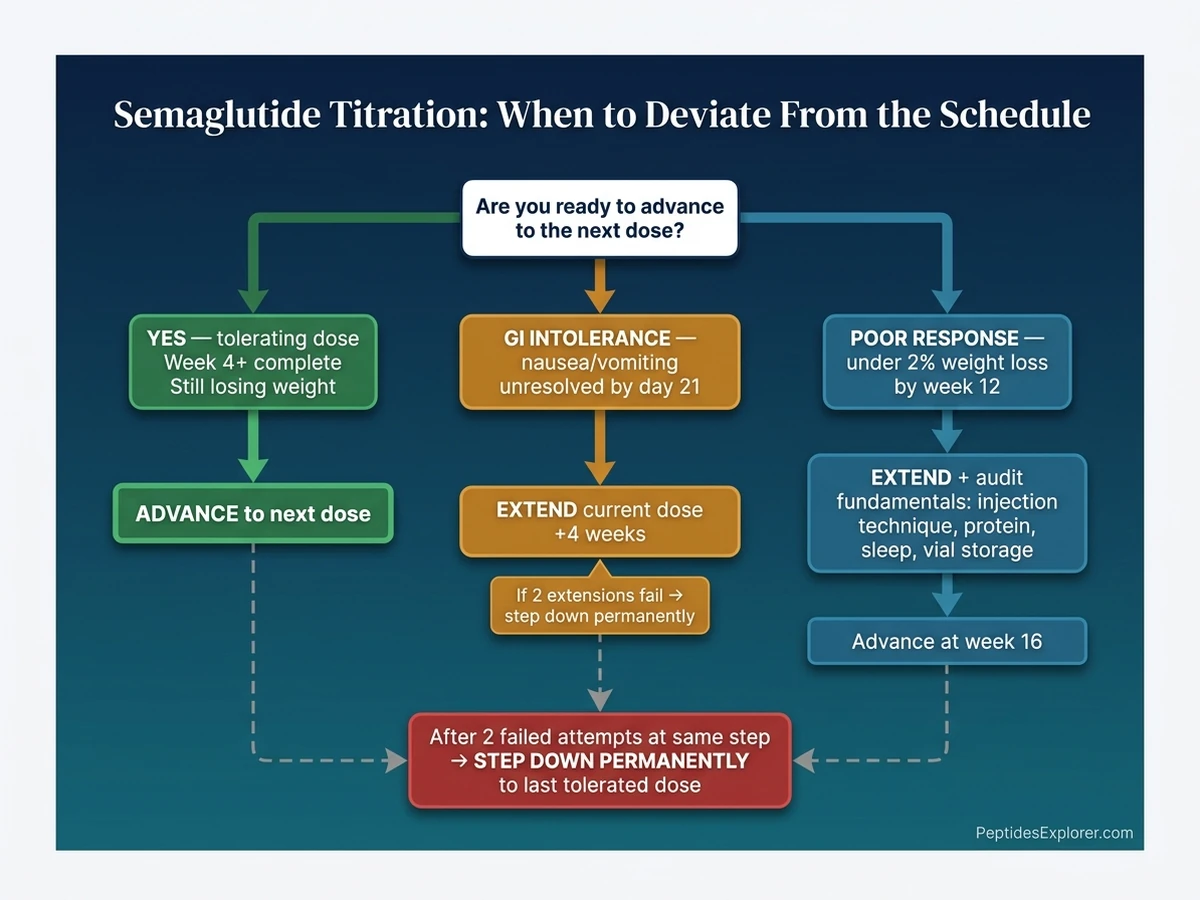
GI Intolerance: Extend the Current Dose 4 Weeks
The most common deviation. If nausea, vomiting, or diarrhea has not settled by day 21 of a dose, stay at that dose for another 4 weeks instead of advancing. The FDA label says this explicitly.
A second 4-week extension is reasonable if symptoms are improving but not resolved. A third extension without clinician review is not. At that point you are either responding to a dose below the standard schedule, or there is something else going on (medication interaction, food timing, delayed gastric emptying worse than expected).
If two consecutive escalation attempts at the same step fail, step down permanently. Example: a patient tolerates 0.5 mg for 8 weeks, then vomits after the first 1.0 mg injection, then vomits again after trying 1.0 mg on a second 4-week attempt. That patient's maintenance is 0.5 mg, or a compounded 0.75 mg bridge dose.
Slow Responder: Extend 1.0 or 1.7 mg Before Advancing
If weight loss at week 12 is under 2% of body weight, do not jump to the next dose expecting it to break the stall. Extend the current dose for 4 weeks first. Response often lags because true steady state takes 5 weeks per level, not 4.
Check the fundamentals before blaming the dose. Confirm injection technique, confirm the vial has not been light-exposed or frozen, confirm protein intake is at least 1 gram per kilogram target body weight, confirm sleep is adequate. A semaglutide dose escalation above 1.0 mg only works if the baseline lifestyle factors are not undermining it.
If you are a slow responder with clean fundamentals, the next step up is legitimate at week 16 instead of week 13.
Fast Responder: Do NOT Skip Steps
This is the deviation people ask about the most and the one with the weakest evidence. If you are at 0.5 mg and have already lost 10% of body weight by week 8, the temptation is to skip 1.0 mg and leap to 1.7 mg.
Do not. STEP trial data show that more than half of patients reach their optimal response below 2.4 mg. If you are responding hard at a low dose, hold there indefinitely. Patients who self-escalate faster than 4-week intervals discontinue 3 to 4 times more often because of GI events. Skipping steps upward is not supported by any published trial.
Holding at the dose that is working is the fast-responder's optimization, not climbing faster.
Three Titration Danger Scenarios (Quantified)
Titration goes wrong in three predictable ways. Each has a measurable cost. Knowing the numbers makes the guardrails easier to respect.
Scenario 1: Over-Escalation (Jumping to 1.0 mg at Week 5)
A patient on 0.25 mg for 4 weeks feels fine and skips directly to 1.0 mg to "speed things up."
Expected outcome: severe nausea within 48 to 72 hours of the larger dose, 4 to 5 weeks of continuous symptoms while the drug reaches its new steady state, a 3 to 4 times higher probability of complete discontinuation within 60 days. Roughly 1 in 5 patients who self-escalate end up in the emergency department for IV fluids within the first 2 weeks. GI-event discontinuation at standard titration is 4.5%; for over-escalation, patterns from trial sub-analyses suggest 12 to 18%. See does semaglutide cause nausea for the symptom timeline and can semaglutide cause diarrhea for the counterpart risk.
The cost is not just discomfort. It is treatment failure, wasted money, and a psychological association between the drug and ER visits that makes a second attempt harder.
Scenario 2: Under-Escalation (Stuck at 0.25 mg Past Week 8)
A patient tolerates 0.25 mg beautifully. Their provider never calls. The patient does not ask to escalate. Six months pass.
STEP trial patients at 0.25 mg lost 2 to 4 pounds across 4 weeks. Patients who remained at this dose for 12+ weeks lost roughly 1 to 2% of body weight; standard-titration patients hit 5% by the same point. At 6 months the gap compounds to 8 to 10 percentage points less weight loss. A 250-pound patient on standard titration loses 25 pounds. The under-titrated version of the same patient loses 5 to 10 pounds.
The patient then concludes "semaglutide does not work for me" and discontinues. That is the danger. Not side effects. Not injury. Wasted therapeutic potential that was sitting one dose increase away.
Scenario 3: Abrupt Restart After Missed Doses
Life happens. Travel, illness, prescription gaps, and pharmacy delays cause real missed-dose situations. The restart protocol is specific and depends on gap length.
Missed 5 days or less: take the dose immediately, then resume the normal weekly schedule.
Missed 5 to 13 days (one full dose): skip the missed dose, take the next one on the scheduled day.
Missed 2 weeks to 3 months: restart at one dose level below the last one tolerated. If you were on 1.7 mg, restart at 1.0 mg for 4 weeks, then re-escalate.
Missed 3 months or more: full re-titration from 0.25 mg. Receptor sensitivity has returned to baseline. Restarting at your previous dose after a 3+ month gap produces GI severity equivalent to skipping two steps. High vomiting and dehydration risk.
Source: UCLA Health missed-dose guidance. A common restart mistake is assuming "I was tolerating 2.4 mg three months ago, so I can start there again." Your receptors disagree.

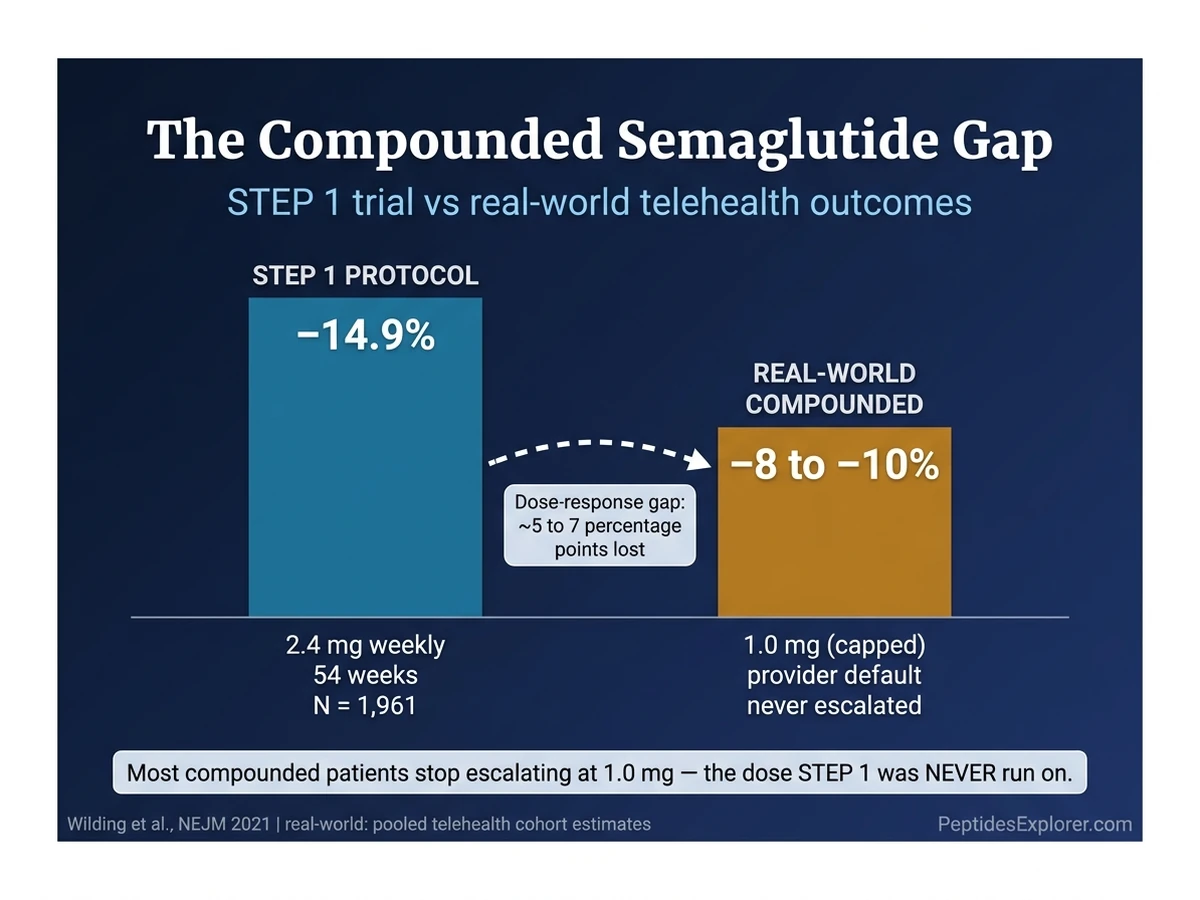
The Compounded Semaglutide Titration Gap
This is the angle most titration guides ignore, and it is the one that matters most to anyone using compounded semaglutide through a telehealth provider.
The Wegovy FDA schedule reaches 2.4 mg at week 17. STEP 1 patients held 2.4 mg for 54 weeks and lost 14.9%. Those numbers are only true at 2.4 mg. At 1.0 mg, the same patients would have lost roughly 60 to 70% as much. That is the dose-response curve.
Real-world compounded semaglutide patients rarely reach 2.4 mg. Telehealth protocols often cap at 1.0 mg because that matches the Ozempic evidence base the provider is most comfortable with. Pharmacy pricing scales with dose, so moving from 1.0 to 2.4 mg can double the monthly cost. Some patients stop escalating themselves because the side-effect step from 0.5 to 1.0 was unpleasant and they do not want to repeat it.
The result: many users settle at 1.0 mg, lose 7 to 10% of body weight, then plateau at a dose that is not the one STEP 1 was run on. They conclude the drug is weaker than advertised. The drug is not weaker. They are on a different dose.
The script to ask your provider to escalate past 1.0 mg:
"My current dose is 1.0 mg weekly. I've been tolerating it without significant GI symptoms for at least 4 weeks. My weight loss curve has flattened over the past 3 to 4 weeks. Based on the Wegovy FDA label, the next step is 1.7 mg, with 2.4 mg as the target maintenance. Can we move to 1.7 mg at my next refill, and plan to reach 2.4 mg if I continue to tolerate escalation?"
For specific telehealth protocol comparisons, see the current Ro weight loss reviews, Hims weight loss reviews, Mochi Health reviews, Henry Meds reviews, and Ivim Health reviews. Each has a different default ceiling and escalation philosophy. For total monthly cost at each dose level, read how much is semaglutide.
Ozempic vs Wegovy Titration: They Are Different
People talk about "semaglutide" as one drug. For the dose you inject, it is one molecule. For the titration schedule, it is two different prescriptions depending on the indication.
Wegovy (obesity, approved 2021): 5 escalation steps. 0.25 → 0.5 → 1.0 → 1.7 → 2.4 mg. 16 weeks minimum to maintenance. Maximum dose 2.4 mg, with a new 7.2 mg dose approved in March 2026 under the STEP UP protocol.
Ozempic (type-2 diabetes, approved 2017): 3 dose levels. 0.25 → 0.5 → 1.0 mg, with a jump to 2.0 mg available since 2022 if glycemic control requires it. Only 0.25 mg has a strict 4-week minimum; later levels require at least 4 weeks before increase but allow longer.
Rybelsus (oral, approved 2019): Different drug-delivery profile entirely. 3 mg → 7 mg → 14 mg daily, with 30-day minimums per step because absorption is so low.
If your compounded prescription came from a clinician most familiar with Ozempic, your titration may cap at 1.0 or 2.0 mg by habit, not by evidence. That is the compounded titration gap above. If your goal is weight loss, the Wegovy schedule is the relevant one, and 2.4 mg is the target.
For drug-to-drug switching, see semaglutide to tirzepatide switching.
After 2.4 mg: Maintenance and the Plateau Window
Reaching 2.4 mg is not the end of titration. It is the start of the maintenance phase. For most patients, that phase lasts indefinitely.
Weight loss typically continues for 60 to 68 weeks after reaching 2.4 mg, then flattens. The plateau is not a dosing problem. It is your body reaching a new set point where intake, metabolism, and appetite regulation settle into balance.
The most important STEP trial data for maintenance comes from STEP 4. Patients who reached 2.4 mg and then switched to placebo regained 6.9% of body weight over the next year. Patients who continued 2.4 mg lost an additional 7.9%. Net difference: 14.8 percentage points. Stopping the maintenance dose reverses the result.
Options at the plateau, in order of evidence:
- 1.Maintain 2.4 mg indefinitely. Prevents regain. Most patients' long-term answer.
- 2.Intensify lifestyle. Resistance training and protein targets of 1.2 to 1.6 g/kg often produce another 2 to 4% of loss.
- 3.Switch to tirzepatide. SURMOUNT-1 data show roughly 22.5% average loss, versus 14.9% for semaglutide at 2.4 mg. See the semaglutide to tirzepatide switching guide for conversion schedule.
- 4.Move to the 7.2 mg Wegovy dose (FDA-approved March 2026). STEP UP data show 20.7% loss at 7.2 mg versus 17.5% at 2.4 mg. Requires a fresh titration from 2.4 mg upward under physician supervision.
For reconstitution math at higher concentrations, see how much bacteriostatic water for semaglutide and the semaglutide mixing chart.
Common Titration Mistakes
1. Advancing at 3 weeks instead of 4. You have not reached steady state. The next dose will hit harder than it is supposed to. Hold a full 4 weeks every time.
2. Skipping the 0.25 mg step. Patients sometimes convince themselves they can start at 0.5 mg because "0.25 is sub-therapeutic anyway." The 0.25 mg step is receptor acclimation, not weight loss. Skipping it produces a nausea window severe enough to end treatment for 15 to 20% of people who try.
3. Treating 1.0 mg as the destination. It is a waypoint for most patients, not a maintenance dose. If you are tolerating it and still losing, advance.
4. Confusing "extended" with "permanent." Staying at 0.5 mg for 8 weeks because of GI symptoms is extending. Staying at 0.5 mg for 9 months without attempting escalation is drift. Revisit the question every 8 weeks.
5. Missing the re-titration protocol. If you take a 3-month break and restart at your old dose, you will get the worst nausea of your life. Receptor sensitivity resets. Restart from 0.25 mg.
6. Ignoring labs during maintenance. At 2.4 mg, annual kidney function, lipid panel, and thyroid check catch issues early. Rapid weight loss stresses the system; monitoring costs nothing and prevents expensive surprises.
7. Microdosing when the goal is weight loss. Microdosing semaglutide has a legitimate use case for maintenance of loss at sub-therapeutic doses. It is not a substitute for proper titration during active weight loss. Mixing the two approaches produces the worst of both: side effects without the efficacy.
Frequently Asked Questions
How long does it take to reach the 2.4 mg maintenance dose of semaglutide?
At the standard FDA-approved schedule, you reach 2.4 mg at week 17 (the start of the fifth 4-week block). That assumes clean tolerance at every step. If you extend a single step by 4 weeks for GI symptoms, the timeline shifts to week 21. See the full semaglutide dosage chart in mL for volume-per-dose conversions along the way.
Can I skip dose escalation steps if I tolerate semaglutide well?
No. No published trial supports skipping. Patients who self-escalate faster than 4-week intervals discontinue 3 to 4 times more often for GI events. The 4-week rule exists because semaglutide has a 7-day half-life, and steady state requires ~4 half-lives. If you are tolerating a dose, the right next step is advancing on schedule, not leaping. Read how long does semaglutide take to work for the pharmacokinetic rationale.
What should I do if I cannot tolerate the next dose?
Stay at the current dose for another 4 weeks. The Wegovy FDA label explicitly permits this delay. If the next attempt at escalation still fails, step down to the last dose you tolerated and stay there. Manage symptoms in the meantime with the protocol in how to relieve nausea from semaglutide.
Is 1.7 mg a real maintenance dose or just a stepping stone?
Both. The FDA label lists 1.7 mg as an alternative permanent maintenance dose if 2.4 mg is not tolerated. If you reach 1.7 mg with strong response and mild side effects, holding there is legitimate and evidence-based. Many compounded patients do exactly this. Track your progress against the troubleshooting tree in not losing weight on semaglutide before assuming 2.4 mg is required.
Why does Ozempic only go to 2.0 mg while Wegovy goes to 2.4 mg?
Different FDA indications. Ozempic is approved for type-2 diabetes, where the evidence base capped at 2.0 mg. Wegovy is approved for obesity, where the evidence base went to 2.4 mg in STEP 1 through STEP 4. Same molecule, different trials, different labels. For cost implications at each dose, see how much is semaglutide.
Do I need to restart titration from 0.25 mg if I missed 3 months of doses?
Yes. GLP-1 receptor sensitivity returns to baseline after 3 or more months without drug exposure. Restarting at your previous dose produces GI severity equivalent to skipping two titration steps. Full re-titration from 0.25 mg is the safe protocol. Source: UCLA Health missed-dose guidance. If you need help calculating draw volumes during restart, use the semaglutide dosage calculator.
What happens if I stay on 0.25 mg semaglutide too long?
0.25 mg is designed to be sub-therapeutic. Trial patients lost 2 to 4 pounds across 4 weeks at this dose. Staying at 0.25 mg for 12+ weeks produces roughly 1 to 2% body-weight loss by that point, versus 5% on standard titration. At 6 months the lag compounds to 8 to 10 percentage points less loss. If you want sub-therapeutic dosing for maintenance rather than active loss, read how to microdose semaglutide for the distinct protocol.
Is compounded semaglutide titrated the same as Wegovy?
On paper, yes. Most 503A and 503B pharmacies mirror the 5-step Wegovy schedule. In practice, many telehealth protocols cap at 1.0 mg and add B12 (cyanocobalamin) for fatigue management rather than continuing escalation. See compound semaglutide with B12 for the rationale. If your goal is matching STEP 1 outcomes, you need to reach 2.4 mg, which may require asking your provider to escalate past their default ceiling.
The Bottom Line
The standard semaglutide titration schedule is 5 doses, 4 weeks each, 16 weeks to maintenance: 0.25 → 0.5 → 1.0 → 1.7 → 2.4 mg weekly. Advance on schedule unless GI symptoms force a 4-week extension. Do not skip steps. Do not stop at 1.0 mg by default. If you miss 3+ months, re-titrate from 0.25 mg.
The single most common failure mode is not side effects. It is settling at 1.0 mg because that is where the compounded prescription capped, then concluding the drug underperformed. STEP 1's 14.9% weight loss was measured at 2.4 mg maintained for 54 weeks. That is the dose and duration the evidence base actually describes.
Before every step, confirm the volume you need to draw with the semaglutide dosage calculator. For dose-to-mL conversions across every concentration, reference the semaglutide dosage chart in mL. For access routes to legitimate semaglutide, read how to get semaglutide.
For personalized protocols, dosing tools, and side-by-side telehealth comparisons, visit peptidesexplorer.com.
Related Articles: - Semaglutide Dosage Chart in mL — draw volumes for every dose and concentration - Semaglutide Mixing Chart — reconstitution math by vial size - How Long Does Semaglutide Take to Work — pharmacokinetic timeline - Not Losing Weight on Semaglutide — plateau troubleshooting - How to Microdose Semaglutide — sub-therapeutic maintenance protocol - Semaglutide to Tirzepatide Switching — conversion schedule - How Much Is Semaglutide — cost by dose and source
Sources: - STEP 1 Trial (Wilding et al., NEJM 2021) — PMID 33567185 - STEP 4 Trial (Rubino et al., JAMA 2021) — PMID 33755728 - Wegovy FDA Label 2025 - Ozempic FDA Label 2025 - GI Tolerability at 2.4 mg (Wharton et al., 2022) - UCLA Health — Missed Dose Guidance
Related Articles
How Many mg Is 40 Units Semaglutide?
40 units of semaglutide = 0.8 mg at 2 mg/mL, 1 mg at 2.5 mg/mL, 2 mg at 5 mg/mL, or 4 mg at 10 mg/mL. Full conversion table with syringe guide.
Semaglutide Dosage Chart in mL
Semaglutide dosage chart in mL for compounded 5mg and 10mg vials. Covers 0.25 to 2.4 mg doses with unit conversions and syringe markings.
Semaglutide to Tirzepatide Switching
Switching semaglutide to tirzepatide? Clinical dose equivalence table, step-by-step protocol, titration schedule, and common mistakes to avoid.
How Much Is Semaglutide? 2026 Cost Breakdown
Semaglutide costs $149-$1,349/month depending on formulation. Compare Wegovy, Ozempic, compounded, and oral prices with savings strategies.