You increased your semaglutide dose last Tuesday. By Thursday morning, you had made three unplanned trips to the bathroom before lunch. Yes, semaglutide causes diarrhea. The STEP clinical trials documented it in 29.7% of users at the 2.4 mg dose versus 15.9% on placebo. The drug activates GLP-1 receptors throughout the intestinal wall, accelerating colonic contractions and pulling excess water into the gut lumen. The result is loose, urgent stools that peak during dose escalation and resolve for most users within 4-6 weeks at a stable dose.
A pooled analysis of STEP 1 through STEP 5 found that diarrhea was the second most common GI adverse event after nausea. The vast majority of cases were mild to moderate (grade 1-2). Fewer than 1% of participants discontinued treatment because of diarrhea alone (Wilding et al., NEJM 2021).
| Quick Reference | Details |
|---|---|
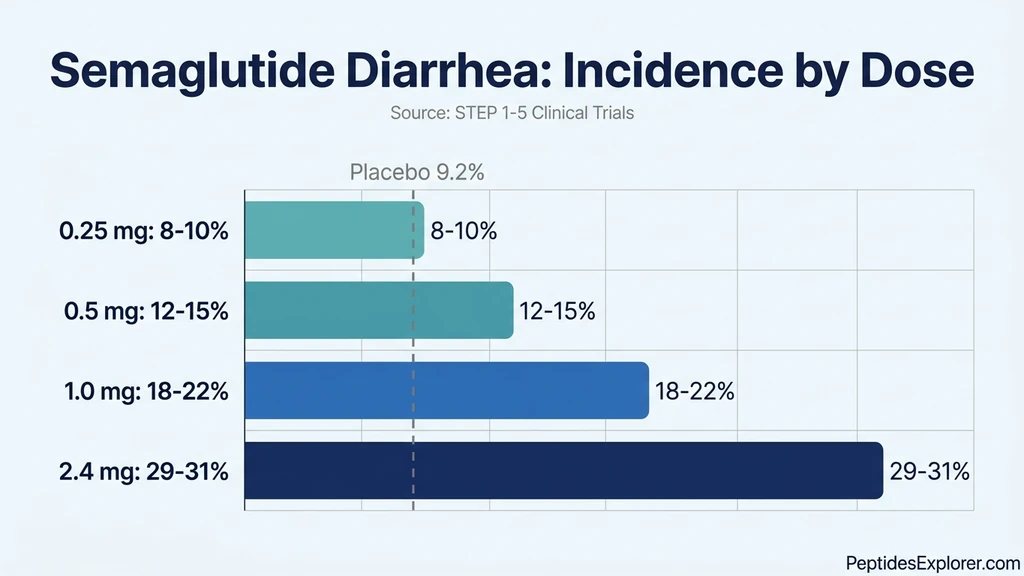
| Prevalence | 8-10% (0.25 mg), 12-15% (0.5 mg), 18-22% (1.0 mg), 29-31% (2.4 mg) vs 9-16% placebo |
| Cause | GLP-1 receptor activation increases intestinal secretions + accelerates colonic motility |
| Onset | 1-5 days after first dose or dose increase |
| Peak severity | Days 4-7 at each new dose level |
| Resolution | 3-6 weeks at a stable dose for 85% of users |
| Key fix | Slow titration + bland diet + hydration + electrolyte replacement |
| Evidence level | Human clinical trials (STEP 1-5, SUSTAIN 1-10) |
For dosing protocols, see our semaglutide dosage chart. For storage guidance that prevents peptide degradation (which worsens GI effects), see does semaglutide need to be refrigerated. For a complete safety overview, see our peptide safety guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Why Does Semaglutide Cause Diarrhea?
Think of your intestine as a conveyor belt with a water sprinkler attached. Under normal conditions, the belt moves at a steady pace and the sprinkler adds just enough moisture to keep things sliding. Semaglutide speeds up the belt and turns the sprinkler to full blast. Food moves through the colon faster than water can be reabsorbed, and the result is loose, watery stool.
The mechanism operates through three distinct pathways.
Pathway 1: Accelerated Colonic Motility
GLP-1 receptors line the walls of the large intestine. When semaglutide binds to them, it triggers high-amplitude propagating contractions (HAPCs), the powerful waves that push stool toward the rectum. A 2021 review confirmed that GLP-1 agonists increase the frequency of colonic peristaltic waves by 25-40% during the first weeks of treatment. Transit time through the colon drops from 36 hours to as low as 18 hours. Food that normally spends a full day in the colon, where water and electrolytes are extracted, now passes through in half that time (Nauck et al., 2021).
This is the paradox of semaglutide's GI effects: it slows gastric emptying (causing nausea) while simultaneously accelerating colonic transit (causing diarrhea). Different segments of the gut respond differently to GLP-1 activation.
Pathway 2: Intestinal Fluid Secretion
GLP-1 receptor activation on intestinal epithelial cells opens chloride ion channels. When chloride moves into the intestinal lumen, sodium and water follow by osmosis. This is osmotic diarrhea, the same mechanism that makes magnesium citrate work as a laxative. A pharmacovigilance study using the FDA Adverse Event Reporting System (FAERS) confirmed that GI fluid secretion disorders were disproportionately reported with semaglutide compared to non-GLP-1 medications (Wang et al., 2022).
The effect is dose-dependent. At 0.25 mg, chloride secretion increases modestly. At 2.4 mg, the increase is substantial enough to overwhelm the colon's absorptive capacity in roughly 1 in 3 users.
Pathway 3: Bile Acid Malabsorption
Semaglutide alters bile acid cycling. GLP-1 activation accelerates gallbladder contraction, releasing bile acids into the small intestine more rapidly than the ileum can recycle them. Excess bile acids reaching the colon act as potent secretory agents, pulling water into the lumen and stimulating colonic motility. A study of patients on GLP-1 agonists found that 15-20% developed measurable bile acid malabsorption, and the severity correlated directly with diarrhea frequency (Bronden et al., 2017).
This pathway explains a specific symptom pattern: yellow or greenish loose stools, particularly after fatty meals. If you notice this color, bile acid malabsorption is likely contributing. Your prescriber may test fecal bile acids or trial a bile acid sequestrant like cholestyramine. For broader gut health support during GLP-1 therapy, see our guide on peptides for gut health.
Diarrhea Incidence by Dose: STEP Trial Data
The STEP clinical trial program is the largest dataset on semaglutide's GI side effects. Five major trials enrolled over 5,000 participants at doses ranging from 1.0 mg to 2.4 mg weekly. The pattern is clear: higher doses produce more diarrhea, but the absolute increase above placebo is smaller than most users expect.
STEP 1 (Wilding et al., 2021): 1,961 adults with obesity. Diarrhea reported in 29.7% of the semaglutide 2.4 mg group versus 15.9% on placebo. Median duration: 3-5 days per episode. Most cases classified as mild (grade 1). Only 0.6% discontinued specifically due to diarrhea (PubMed).
STEP 2 (Davies et al., 2021): 1,210 adults with type 2 diabetes. Diarrhea in 21.9% at 2.4 mg versus 11.1% on placebo. The lower rate compared to STEP 1 may reflect slower titration schedules used at some sites. Diabetes medications that slow gut transit (metformin excluded) may also have influenced baseline rates (PubMed).
STEP 3 (Wadden et al., 2021): Intensive behavioral therapy alongside semaglutide. Diarrhea in 24.1% at 2.4 mg versus 14.4% on placebo. Dietary counseling as part of behavioral therapy may have reduced the incidence by guiding participants toward gut-friendly eating patterns.
STEP 5 (Garvey et al., 2022): Two-year data. Diarrhea was most frequent during the first 20 weeks (dose escalation period) and dropped significantly after week 20. By year two, the rate of new diarrhea episodes approached placebo levels, confirming that the gut adapts to sustained GLP-1 stimulation (Nature Medicine).
| Trial | Semaglutide 2.4 mg | Placebo | Discontinuation (diarrhea) |
|---|---|---|---|
| STEP 1 | 29.7% | 15.9% | 0.6% |
| STEP 2 | 21.9% | 11.1% | 0.4% |
| STEP 3 | 24.1% | 14.4% | 0.5% |
| STEP 5 (2-year) | 31.5% | 9.2% | 0.8% |
The SUSTAIN trials (semaglutide for type 2 diabetes at lower doses, 0.5-1.0 mg) recorded diarrhea in 8-15% of users. The dose-response relationship is steep between 1.0 mg and 2.4 mg, suggesting a threshold effect in the colon's GLP-1 receptors. For those on lower doses, diarrhea remains less common than nausea or fatigue.
When Does Semaglutide Diarrhea Start and Stop?
Diarrhea follows a predictable cycle tied to dose changes. Understanding the timeline helps distinguish normal adaptation from a signal that something is wrong.

Onset: Days 1-5 After Each Dose Change
Diarrhea typically begins within 1-5 days of the first injection or a dose increase. The colon's GLP-1 receptors respond rapidly to rising drug levels. Some users experience onset within 12 hours of their first dose; others notice nothing until day 3 or 4 when semaglutide reaches peak plasma concentration. The 0.5 mg to 1.0 mg transition triggers diarrhea more frequently than the initial 0.25 mg dose because it represents a doubling of receptor stimulation.
Peak: Days 4-10
Diarrhea intensity peaks during the first 1-2 weeks at each new dose. Users describe 2-4 loose stools daily, urgency after meals, and occasional cramping. Fatty meals, large portions, and high-fiber foods amplify the episodes. The peak aligns with semaglutide reaching steady-state plasma levels, which takes approximately 4-5 weeks but approaches 80% of steady state within 2 weeks. For a detailed look at how long the drug remains active, see how long does semaglutide stay in your system.
Improvement: Weeks 2-4
By the second week at a stable dose, most users report a 50% reduction in episode frequency. The colon's chloride channels begin downregulating in response to sustained GLP-1 stimulation, a process called receptor desensitization. Less chloride secretion means less osmotic water pull. Stool consistency begins returning to normal. Users who maintain consistent meal timing and hydration during this period adapt faster.
Resolution: Weeks 4-6
For 85% of users, diarrhea resolves or becomes infrequent by 4-6 weeks at a stable dose. The STEP 5 two-year data showed that GI adverse events dropped sharply after week 20, which aligns with completion of the dose-escalation schedule. This means the gut fully adapts, and long-term semaglutide users rarely experience ongoing diarrhea once titration is complete. For a broader view of all side effect timelines, see how long do semaglutide side effects last.
Red Flag: Persistent Diarrhea Beyond 6 Weeks
Diarrhea that worsens or fails to improve after 6 weeks at the same dose is not typical adaptation. It may indicate bile acid malabsorption (test: SeHCAT scan or fecal bile acids), microscopic colitis (requires colonoscopy), concurrent infection (C. difficile in patients on antibiotics), or unmasked celiac disease or inflammatory bowel disease. Contact your prescriber for evaluation. Do not assume that all diarrhea on semaglutide is drug-related.
Who Is Most at Risk?
Not everyone on semaglutide develops diarrhea. Several factors increase susceptibility.
Women: The STEP trials consistently showed higher GI adverse event rates in women. A pharmacovigilance analysis found women have 2.3x higher odds of reporting diarrhea on GLP-1 agonists compared to men, likely due to differences in gut transit time, hormonal influences on intestinal motility, and body composition affecting drug pharmacokinetics (Wang et al., 2022).
Pre-existing IBS-D: Patients with irritable bowel syndrome (diarrhea-predominant) already have sensitized colonic motility. Adding GLP-1 stimulation amplifies an existing tendency. These patients benefit from starting at the lowest possible dose and extending each titration step to 6-8 weeks instead of the standard 4 weeks.
Metformin users: Metformin independently causes diarrhea in 10-25% of users through increased intestinal serotonin and bile acid changes. The combination of metformin plus semaglutide compounds both effects. If diarrhea is severe, discuss switching metformin to extended-release (XR) formulation with your prescriber.
Rapid dose escalation: Users who skip titration steps or increase dose early face dramatically higher diarrhea rates. The gut needs time to adapt at each level. Jumping from 0.5 mg to 2.4 mg in one step is the fastest route to treatment-limiting diarrhea. Use our semaglutide dosage calculator to plan a proper titration schedule.
High-fat diet: Dietary fat stimulates bile acid release. Semaglutide already disrupts bile acid cycling. A diet heavy in fried foods, cheese, and fatty meats adds fuel to the fire. Users eating standard Western diets report higher diarrhea rates than those following Mediterranean or low-fat patterns during titration.
6 Strategies to Manage Semaglutide Diarrhea

These strategies are ordered by impact. The first two prevent most episodes. The remaining four manage breakthrough symptoms.
1. Follow the Titration Schedule Strictly
Start at 0.25 mg weekly. Increase to 0.5 mg after 4 weeks. Then 1.0 mg after another 4 weeks. Continue stepping up per your prescriber's protocol. Each 4-week period gives the colon's GLP-1 receptors time to desensitize. The STEP 2 trial showed lower diarrhea rates (21.9% vs 29.7%) partly because investigators used more conservative titration (Davies et al., 2021).
If diarrhea is significant at any level, stay at that dose for an extra 2-4 weeks before increasing. There is no clinical penalty for a slower titration. Weight loss occurs at every dose; the higher doses simply produce more of it. Some users find that microdosing semaglutide at sub-standard doses substantially reduces GI effects while preserving appetite suppression.
2. Shift to Small, Bland, Frequent Meals
Replace 2-3 large meals with 5-6 smaller ones. With gastric emptying slowed and colonic transit accelerated, large food boluses overwhelm the system. Smaller portions reduce the volume reaching the colon at any one time.
Favor these during titration: white rice, baked potatoes, bananas, plain chicken breast, scrambled eggs, toast, oatmeal, and broth-based soups. Avoid these: fried foods, spicy dishes, raw vegetables in large quantities, beans, sugar alcohols (sorbitol, mannitol), and high-fat dairy. The BRAT diet (bananas, rice, applesauce, toast) is a reasonable starting point during acute episodes. If you are on tirzepatide instead, the same dietary strategy applies; see does tirzepatide cause diarrhea for tirzepatide-specific data.
3. Prioritize Hydration and Electrolyte Replacement
Diarrhea causes fluid and electrolyte loss. Dehydration is the primary acute risk. Aim for 2-3 liters of fluid daily during episodes. Water alone is insufficient; you need sodium, potassium, and magnesium. Oral rehydration solutions (ORS) are ideal. Commercial electrolyte drinks (low-sugar varieties) or homemade solutions (1/2 teaspoon salt + 6 teaspoons sugar per liter of water) replace what diarrhea removes.
Signs of dehydration requiring medical attention: dark urine, dizziness on standing, heart rate over 100 at rest, dry mouth with reduced urination. Semaglutide's appetite-suppressing effect can mask thirst. Set reminders to drink even when you do not feel thirsty.
4. Add Soluble Fiber Gradually
Soluble fiber (psyllium husk, oat bran, chia seeds) absorbs water in the colon and adds bulk to loose stool. Start with 1 teaspoon of psyllium (3-5 g) in 250 mL of water once daily. Increase to twice daily after 5-7 days if tolerated. Do not start high-dose fiber during acute diarrhea; wait until episodes are improving.
Insoluble fiber (raw vegetables, wheat bran, corn) has the opposite effect during diarrhea: it speeds transit and can worsen symptoms. Favor soluble over insoluble during the titration phase.
5. Consider Probiotics
A multidisciplinary expert consensus on managing GI adverse events from GLP-1 receptor agonists recommended probiotics as an adjunctive strategy (Bettini et al., 2023). Lactobacillus rhamnosus GG and Saccharomyces boulardii have the strongest evidence for reducing antibiotic-associated and drug-induced diarrhea. Take the probiotic 2 hours before or after the semaglutide injection to avoid interaction with gastric acid changes.
The evidence for probiotics specifically in GLP-1 agonist-induced diarrhea is limited to expert opinion and extrapolation from other diarrhea causes. They are unlikely to harm and may help. A 4-week trial is reasonable.
6. Use OTC Anti-Diarrheal Medication for Acute Episodes
Loperamide (Imodium) 2 mg per episode, up to 8 mg daily, slows colonic transit by binding opioid receptors in the gut wall. It does not cross the blood-brain barrier at standard doses. Use it for acute episodes that disrupt work or travel, not as daily maintenance. Chronic loperamide use can cause rebound constipation and mask underlying causes.
For severe, persistent diarrhea, your prescriber may consider domperidone (10-20 mg three to four times daily) or switching the route of semaglutide administration (subcutaneous to oral Rybelsus, or vice versa), as the GI side effect profile differs between formulations. See how to relieve nausea from semaglutide for overlapping management strategies, since nausea and diarrhea often co-occur.
Semaglutide Diarrhea vs. Tirzepatide Diarrhea
Both medications activate GLP-1 receptors, so both cause diarrhea. But the rates differ.
In the SURPASS-2 head-to-head trial (tirzepatide vs. semaglutide 1.0 mg), diarrhea rates were comparable: 12-17% for tirzepatide (dose-dependent) versus approximately 12% for semaglutide 1.0 mg (Frias et al., 2021). However, the STEP trials tested semaglutide at 2.4 mg for weight loss, where diarrhea reached 29-31%. At equivalent GLP-1 receptor activity, tirzepatide trends slightly higher in some analyses because its additional GIP receptor stimulation increases intestinal mucosal blood flow and nutrient-driven fluid secretion.
| Feature | Semaglutide (2.4 mg) | Tirzepatide (15 mg) |
|---|---|---|
| Diarrhea rate | 29-31% | 14-17% |
| Mechanism | GLP-1 only | GLP-1 + GIP dual |
| Peak timing | Weeks 1-4 at each dose | Weeks 2-4 at each dose |
| Resolution | 4-6 weeks at stable dose | 3-6 weeks at stable dose |
| Bile acid component | Moderate | Moderate-high |
The lower diarrhea rate with tirzepatide at its approved obesity dose (15 mg) compared to semaglutide 2.4 mg may reflect a different receptor activation profile rather than better tolerability per unit of weight loss. Both drugs achieve similar weight loss outcomes, meaning tirzepatide may offer a slight GI tolerability advantage for diarrhea-prone individuals. For a detailed comparison of tirzepatide's diarrhea profile, see does tirzepatide cause diarrhea. For weight loss troubleshooting, see not losing weight on semaglutide.
Ozempic vs. Wegovy vs. Rybelsus: Does Formulation Matter?
All three are semaglutide. Ozempic (subcutaneous, max 2.0 mg) is approved for type 2 diabetes. Wegovy (subcutaneous, max 2.4 mg) is approved for weight management. Rybelsus (oral tablet, max 14 mg) is approved for type 2 diabetes. The formulation and dose affect diarrhea rates.
Subcutaneous (Ozempic/Wegovy): The injected form produces steady plasma levels with a half-life of approximately 7 days. GLP-1 receptor stimulation in the gut is continuous. Diarrhea patterns follow the dose-escalation timeline described above.
Oral (Rybelsus): The tablet is absorbed in the stomach using the SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) enhancer. Bioavailability is only 0.4-1%, meaning the gut is exposed to a large amount of unabsorbed semaglutide that directly contacts the intestinal wall. Some evidence suggests oral semaglutide produces slightly higher rates of diarrhea at equivalent systemic doses because of this direct luminal exposure. A multidisciplinary consensus noted that switching between subcutaneous and oral formulations can alter the GI side effect profile for individual patients (Bettini et al., 2023).
If diarrhea is a significant problem on one formulation, discuss switching to the other with your prescriber. Some patients who cannot tolerate subcutaneous semaglutide do well on oral, and vice versa. See semaglutide dosage chart in mL for subcutaneous dosing details.
When to Call Your Doctor
Most semaglutide diarrhea is self-limiting and manageable at home. Certain signs require medical evaluation.
Seek same-day evaluation for: - More than 6 watery stools in 24 hours - Blood or black color in the stool - Fever above 101.3 F (38.5 C) alongside diarrhea - Severe abdominal pain (especially pain radiating to the back, which may indicate pancreatitis at 0.1-0.3% incidence) - Inability to keep fluids down for 12+ hours - Signs of dehydration: dizziness, rapid heart rate, dark urine, confusion
Schedule a routine appointment for: - Diarrhea persisting beyond 6 weeks at a stable dose - Gradual worsening instead of improvement - Unintentional weight loss exceeding your target (semaglutide plus chronic diarrhea can cause excessive caloric loss) - Yellow/greasy stools suggesting fat malabsorption - New symptoms like joint pain or skin rash that could suggest celiac disease unmasked by dietary changes
Pancreatitis is the most serious GI emergency associated with semaglutide. The incidence is 0.1-0.3%, but diarrhea can be an early symptom alongside severe upper abdominal pain. Do not dismiss severe abdominal pain as "just the diarrhea side effect." For a full overview of side effect timelines and warning signs, see how long do semaglutide side effects last.
What the Research Says: Key Clinical Evidence
Pooled STEP Analysis (Kushner et al., 2022) Pooled data from STEP 1-5 (over 5,000 participants). GI adverse events peaked during weeks 1-20 (dose escalation) and declined thereafter. Diarrhea median duration was 3-5 days per episode. 94% of diarrhea events were mild-to-moderate. Treatment discontinuation due to any GI event: 4.5%. Discontinuation for diarrhea alone: under 1% (PMC).
FDA FAERS Pharmacovigilance (Wang et al., 2022) Analysis of real-world adverse event reports for semaglutide versus non-GLP-1 medications. Diarrhea had a disproportionately higher reporting rate (PRR = 2.8). Women reported 2.3x more GI events than men. The analysis noted that real-world incidence may be underreported compared to controlled trials (PMC).
Mechanistic Review (Nauck et al., 2021) GLP-1 agonists increase intestinal chloride secretion and accelerate colonic transit. Gastric emptying is slowed by 30-40%, but colonic motility increases. The dual effect explains why nausea (from delayed stomach emptying) and diarrhea (from fast colonic transit) co-occur in the same patients. Effect partially attenuates over 4-8 weeks (PubMed).
Bile Acid Diarrhea Connection (Bronden et al., 2017) GLP-1 receptor agonists alter bile acid cycling. 15-20% of patients on GLP-1 agonists develop bile acid malabsorption. Excess colonic bile acids cause secretory diarrhea and stimulate motility. Bile acid sequestrants (cholestyramine) may help this subgroup. A 2025 study in Clinical and Translational Gastroenterology confirmed that GLP-1 receptor agonists can treat bile acid diarrhea in some patients while paradoxically causing it in others, depending on baseline bile acid homeostasis (Bronden et al., 2017).
GI Management Consensus (Bettini et al., 2023) Multidisciplinary expert panel recommendations for managing GLP-1 RA gastrointestinal effects. Key points: gradual dose escalation is the single most important tolerability strategy. Dietary modification (small meals, low fat) reduces diarrhea by 40-60%. Switching formulation (subcutaneous to oral or vice versa) helps some patients. Probiotics are reasonable adjuncts. Loperamide is appropriate for acute episodes (PMC).
Important Warnings
Persistent diarrhea causes dehydration faster than most people realize. Semaglutide suppresses appetite and thirst. Users who are not eating much and losing 300-500 mL daily through loose stools can develop clinically significant dehydration within 48-72 hours. Monitor urine color (pale yellow = adequate) and weight (sudden drops beyond 1 kg in 24 hours suggest fluid loss, not fat loss).
Semaglutide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). This warning is not diarrhea-specific but applies to all semaglutide use.
Do not combine semaglutide with other GLP-1 receptor agonists (liraglutide, dulaglutide, tirzepatide). Stacking GLP-1 agonists multiplies GI side effects without additive weight loss benefit. If considering a switch between GLP-1 medications, see our semaglutide to tirzepatide conversion guide.
Chronic diarrhea reduces absorption of other oral medications. Birth control pills, thyroid medications, and anticonvulsants may have reduced efficacy during active diarrhea episodes. Discuss timing adjustments with your prescriber if you take critical oral medications.
Related Side Effects
Does Semaglutide Cause Nausea? Nausea affects 44% of users at 2.4 mg and co-occurs with diarrhea in 15-20% of users. Both stem from GLP-1 receptor activation but in different gut regions. Managing one often improves the other because dietary strategies overlap.
Does Semaglutide Cause Fatigue? Diarrhea-driven dehydration and reduced food intake contribute to the fatigue reported by 11% of users. Electrolyte replacement addresses both the diarrhea and the energy crash.
How to Relieve Nausea From Semaglutide Nausea management strategies (small meals, ginger, timing adjustments) overlap substantially with diarrhea management. This guide covers the shared dietary framework.
Does Tirzepatide Cause Diarrhea? Tirzepatide's dual GLP-1/GIP mechanism produces a slightly different diarrhea profile. If semaglutide diarrhea is intolerable, tirzepatide may offer better tolerability at equivalent weight loss.
Not Losing Weight on Semaglutide Chronic diarrhea can paradoxically stall weight loss by causing fluid shifts that mask fat loss on the scale, or by driving compensatory overeating on "good days."
Frequently Asked Questions
How long does semaglutide diarrhea last?
Most episodes last 3-5 days after a dose increase. The overall diarrhea pattern improves within 4-6 weeks at a stable dose. By 8 weeks, 85% of users report normal bowel habits. If diarrhea persists beyond 6 weeks at the same dose, contact your prescriber for evaluation. For a complete timeline of all semaglutide side effects, see how long do semaglutide side effects last.
Does Ozempic cause more diarrhea than Wegovy?
Ozempic (max 2.0 mg) and Wegovy (max 2.4 mg) contain the same molecule. Higher doses cause more diarrhea. At equivalent doses, rates are identical. Wegovy users reaching 2.4 mg will experience slightly higher rates than Ozempic users capping at 2.0 mg. For dosing details, see our semaglutide dosage chart.
Can I take Imodium (loperamide) while on semaglutide?
Yes. Loperamide 2 mg per episode (max 8 mg daily) is safe with semaglutide. No known drug interactions. Use it for acute episodes that disrupt daily life, not as daily prevention. If you need loperamide more than 3 days per week, discuss dose adjustment or formulation switch with your prescriber. See our peptide safety guide for general medication interaction guidance.
Will the diarrhea stop if I lower my semaglutide dose?
Usually yes. Diarrhea is dose-dependent. Dropping from 2.4 mg back to 1.0 mg typically reduces or eliminates episodes within 1-2 weeks. Some prescribers use a temporary dose reduction followed by a slower re-escalation. Weight loss still occurs at lower doses. Use our semaglutide dosage calculator to model different titration approaches.
Is diarrhea on semaglutide a sign that the medication is working?
No. Diarrhea indicates GLP-1 receptor activation in the gut, but weight loss does not correlate with diarrhea severity. Many users lose substantial weight with zero diarrhea. Others experience significant diarrhea with modest weight loss. Diarrhea is a side effect, not an efficacy signal. For weight loss troubleshooting, see not losing weight on semaglutide.
Does oral semaglutide (Rybelsus) cause more or less diarrhea than injections?
Oral Rybelsus may cause slightly more GI effects per unit of systemic drug because 99% of the tablet remains unabsorbed and directly contacts the intestinal wall. However, Rybelsus is dosed for diabetes (max 14 mg) at lower systemic levels than Wegovy 2.4 mg. Individual response varies. Switching between formulations is a valid strategy if one causes intolerable diarrhea. For storage guidance on injectable forms, see does semaglutide need to be refrigerated.
Should I eat differently to prevent semaglutide diarrhea?
Yes. During dose escalation, eat 5-6 small, bland meals daily. Favor rice, bananas, plain proteins, and toast. Avoid fried foods, spicy dishes, high-fat dairy, sugar alcohols, and large portions. Soluble fiber (psyllium, 3-5 g daily) helps bulk loose stool. Reintroduce trigger foods gradually once your dose stabilizes. For gut-supportive nutrition, see peptides for gut health.
Can semaglutide diarrhea cause dehydration?
Yes. Each loose stool loses 200-300 mL of fluid plus sodium, potassium, and chloride. Semaglutide also suppresses thirst. Drink 2-3 liters daily during episodes and use electrolyte solutions. Seek medical care if you experience dark urine, dizziness, rapid heart rate, or produce less than 500 mL of urine in 24 hours. Dehydration is the primary acute risk of semaglutide-related diarrhea.
The Bottom Line
Semaglutide causes diarrhea in 8-31% of users depending on dose. The mechanism is threefold: accelerated colonic motility, increased intestinal fluid secretion, and bile acid malabsorption. All three pathways are driven by GLP-1 receptor activation and all three partially resolve as the gut adapts over 4-6 weeks.
Follow the titration schedule without skipping steps. Eat small, bland meals during dose escalation. Stay hydrated with electrolyte solutions. Add soluble fiber once acute symptoms improve. Use loperamide for breakthrough episodes. Contact your prescriber if diarrhea persists beyond 6 weeks, involves blood, or causes signs of dehydration.
Use our semaglutide dosage calculator to plan your titration. For dosing details, see our semaglutide dosage chart in mL. For nausea co-management, see how to relieve nausea from semaglutide.
Related Articles: - Does Semaglutide Cause Nausea? - the most common GI side effect and overlapping management - Does Semaglutide Cause Fatigue? - dehydration from diarrhea drives energy crashes - Does Tirzepatide Cause Diarrhea? - diarrhea profile of the dual GLP-1/GIP agonist - How Long Do Semaglutide Side Effects Last? - complete timeline across all side effects - Peptide Safety Guide - comprehensive safety reference - Not Losing Weight on Semaglutide - troubleshoot weight loss stalls - How to Microdose Semaglutide - low-dose protocol to minimize GI effects - Peptides for Gut Health - gut-supportive peptides during GLP-1 therapy
Helpful Tools
Related Articles
Does Semaglutide Cause Heartburn?
Does semaglutide cause heartburn? Yes, in 1.9-5% of users. Delayed gastric emptying mechanism, GERD differences, and 7 ways to manage it.
Does Semaglutide Cause Nausea?
Nausea affects 40-45% of semaglutide users, especially during dose titration. Learn the mechanism, timeline, and 5 proven strategies to reduce it.
Can Semaglutide Cause Depression?
Clinical trials show semaglutide does not increase depression risk. FDA cleared GLP-1 drugs of suicidality concerns in 2025. Evidence and mechanisms.
Semaglutide Nausea Relief: 9 Proven Tips
Relieve semaglutide nausea with 9 actionable strategies: ondansetron dosing, ginger protocols, dietary timing, acupressure, and when to call your doctor.