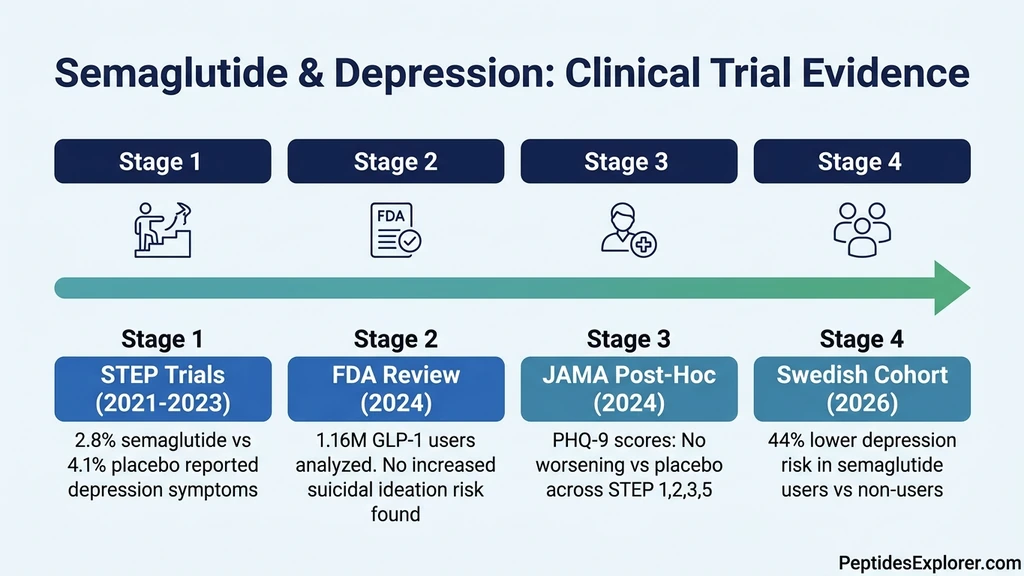
You started semaglutide six weeks ago. The weight is coming off. But the color has drained from your day: food no longer excites you, weekends feel hollow, and a heavy flatness has settled behind your eyes. The current clinical evidence does not support semaglutide as a cause of depression. Across the STEP 1, 2, 3, and 5 trials, only 2.8% of semaglutide-treated participants reported depressive symptoms requiring evaluation, compared to 4.1% on placebo. The FDA reviewed over 1.16 million GLP-1 receptor agonist users in 2024 and found no increased risk of suicidal ideation or behavior. In January 2025, the agency formally requested removal of the suicidality warning from all GLP-1 RA labeling.
That does not mean mood changes on semaglutide are imaginary. They are real, documented, and worth understanding. The distinction matters: semaglutide does not appear to cause depression through a direct pharmacological mechanism, but the metabolic, hormonal, and psychological upheaval it triggers can surface depressive symptoms in vulnerable individuals.
| Quick Reference | Details |
|---|---|
| Clinical trial depression rate | 2.8% semaglutide vs 4.1% placebo (STEP trials) |
| FDA verdict (Jan 2025) | Removed suicidality warning from all GLP-1 RA labels |
| Largest real-world study | 1.16M GLP-1 users: no increased self-harm risk vs comparator drugs |
| Swedish cohort (2026) | 44% lower depression risk in semaglutide users |
| Main mood triggers | Caloric deficit, dopamine modulation, sleep disruption, loss of food-based coping |
| When to worry | Persistent sadness >2 weeks, hopelessness, suicidal thoughts |
| Evidence level | Phase 3 RCTs, FDA safety review, real-world cohort studies |
For dosing protocols, see our semaglutide dosage chart. For a related GLP-1 mental health analysis, see can tirzepatide cause anxiety. For general safety information, consult our peptide safety guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What the Clinical Trials Actually Found
The strongest evidence on semaglutide and depression comes from the STEP program, four randomized controlled trials enrolling over 4,700 adults with obesity or overweight. A 2024 post-hoc analysis published in JAMA Internal Medicine specifically examined psychiatric safety across STEP 1, 2, 3, and 5 (O'Neil et al., JAMA Intern Med 2024).
Researchers tracked depression using the Patient Health Questionnaire-9 (PHQ-9), a validated screening tool that scores depressive symptom severity from 0 to 27. They also monitored suicidal ideation using the Columbia Suicide Severity Rating Scale (C-SSRS).
The findings were clear. Semaglutide 2.4 mg did not worsen PHQ-9 scores compared to placebo at any time point. In fact, PHQ-9 scores improved slightly more in the semaglutide group than in the placebo group, though the difference was not clinically significant. Suicidal ideation occurred in fewer than 1% of participants in both groups, with no difference between semaglutide and placebo.
| STEP Trial Data | Semaglutide 2.4 mg | Placebo |
|---|---|---|
| Depression requiring evaluation | 2.8% | 4.1% |
| PHQ-9 score change from baseline | Improved | Improved (less) |
| Suicidal ideation (C-SSRS) | <1% | <1% |
| Suicidal behavior | 0 events | 0 events |
| Psychiatric serious adverse events | 0.4% | 0.3% |
These numbers need context. The STEP trials excluded people with active major depressive disorder, recent psychiatric hospitalization, or current suicidal ideation. The participants were healthier than the general population of semaglutide users. Trial data tells us what happens in a selected population under close monitoring, not necessarily what happens in your living room.
For users who experienced nausea on semaglutide that disrupted daily life, mood consequences were not separately analyzed. Persistent nausea is itself a known depressogenic factor. For information on how long side effects typically persist, see how long do semaglutide side effects last.
The FDA Safety Review: 1.16 Million Users
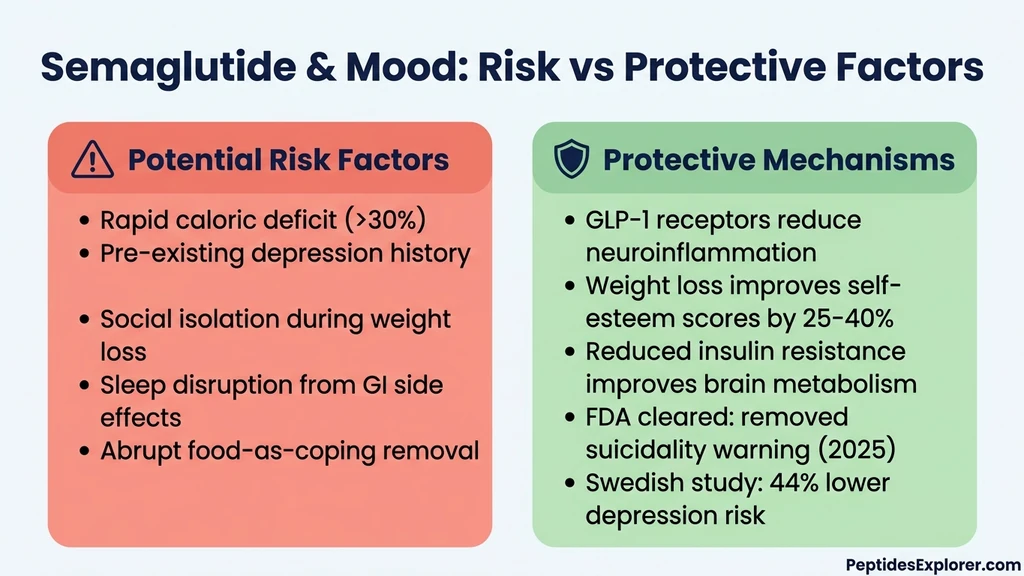
In January 2025, the FDA took an unusual step: it formally requested that drug manufacturers remove the warning about suicidal behavior and ideation from all GLP-1 receptor agonist labels, including Wegovy (semaglutide) and Ozempic (semaglutide) (FDA Safety Communication, Jan 2025).
This decision followed a retrospective cohort study comparing 1,161,983 GLP-1 RA users to 1,081,155 SGLT2 inhibitor users (a diabetes drug class with no known psychiatric effects). The study found no increased risk of intentional self-harm among GLP-1 RA users. The signal that had triggered the original investigation, a cluster of pharmacovigilance reports from European databases, did not hold up under rigorous epidemiological scrutiny.
The European Medicines Agency conducted its own parallel review and reached a similar conclusion in late 2024. The initial signal had been driven by reporting bias: as semaglutide prescriptions surged from 500,000 to over 9 million in the United States between 2020 and 2024, the raw number of psychiatric adverse event reports rose proportionally, but the rate per user did not increase.
This does not mean depression reports on semaglutide are fabricated. Individual case reports exist, including a published case series of two patients who developed depressive episodes temporally linked to semaglutide initiation (Bilal et al., PMC 2023). Case reports document real experiences. They cannot establish causation. The FDA review, with over a million participants, carries far greater statistical weight than individual reports.
For users weighing semaglutide against alternatives, tirzepatide showed comparable psychiatric safety profiles in head-to-head trials. See our best peptides for weight loss guide for a broader comparison.
The Swedish Cohort Study: Semaglutide May Protect Against Depression
A March 2026 national cohort study published in The Lancet Psychiatry examined GLP-1 receptor agonist use among people with pre-existing depression and anxiety in Sweden (Lancet Psychiatry, 2026). The findings surprised many clinicians.
People using GLP-1 receptor agonists were 42% less likely to experience worsening mental illness compared to matched controls not using these drugs. Semaglutide specifically was associated with a 44% lower risk of depression worsening and a 38% lower risk of anxiety disorder progression. The study followed patients for up to 3.5 years, long enough to capture sustained effects rather than short-term fluctuations.
A separate March 2026 analysis published in Science Daily found that semaglutide users had lower rates of depression, anxiety, and even substance use disorders compared to users of other anti-obesity and anti-diabetes medications (ScienceDaily, Mar 2026). The NIH reported concordant findings: people taking semaglutide had a lower risk of suicidal thoughts than those on other weight-loss or diabetes drugs (NIH Research Matters, 2024).
These results suggest that semaglutide may carry antidepressant properties through its central nervous system effects. But correlation is not causation. People who lose weight successfully may feel better because weight loss itself improves mood, reduces inflammation, and enhances social functioning. Separating the drug's direct brain effects from the downstream psychological benefits of weight loss remains an active research question.
For those tracking their weight loss journey, see semaglutide before and after for realistic timelines and outcomes.
How GLP-1 Receptors Affect Mood and Emotion
Understanding why some people feel emotionally flat on semaglutide while population-level data shows mood benefits requires examining the drug's central nervous system pharmacology. GLP-1 receptors are not confined to the gut and pancreas. They populate brain regions that directly regulate emotion, motivation, and reward.
GLP-1 Receptors in Emotional Brain Centers
GLP-1 receptors are expressed in the prefrontal cortex, amygdala, hippocampus, hypothalamus, and nucleus tractus solitarius. Each of these regions plays a role in mood regulation. The prefrontal cortex governs decision-making and emotional control. The amygdala processes fear and threat. The hippocampus consolidates memories and regulates the stress response. The hypothalamus coordinates hormonal cascades including cortisol release.
When semaglutide crosses the blood-brain barrier and activates these receptors, it modulates the release of serotonin, dopamine, and glutamate (Gunturu, Prim Care Companion 2024). This modulation is complex: serotonin may increase in some circuits while decreasing in others. The net effect appears to be mildly antidepressant in most people, but the transition period, while the brain recalibrates to new signaling patterns, can feel destabilizing.
Animal studies illuminate the timeline. GLP-1 agonists induced anxiety-like behavior immediately after treatment initiation, but this subsided with chronic treatment and was replaced by reduced depression-like behavior (Nature Mental Health, 2025). The parallel in humans: the first 2-6 weeks may feel emotionally rough before the brain adapts to its new neurochemical environment.
The Dopamine Reward System
Semaglutide's effect on the dopamine system explains the most common emotional complaint: anhedonia, the inability to feel pleasure. The ventral tegmental area (VTA) is the brain's dopamine factory, producing the neurotransmitter that makes food satisfying, achievements rewarding, and social connections enjoyable.
GLP-1 receptor stimulation in the VTA enhances dopaminergic neuron activity through a presynaptic mechanism while simultaneously increasing dopamine transporter expression in the limbic system and striatum. The result is more efficient dopamine reuptake, meaning dopamine signals are cleared from synapses faster. In practical terms, each reward experience produces a shorter, less intense pleasure signal.
This is the mechanism by which semaglutide reduces food cravings. But food is not the only thing that becomes less appealing. Users describe diminished interest in hobbies, sex, social gatherings, and creative pursuits. This blunted reward response is not clinical depression. It is a pharmacological side effect of altered dopamine kinetics. The distinction matters because it typically resolves as the brain adjusts to the new dopamine dynamics, whereas major depression often requires treatment escalation.
For users experiencing concurrent fatigue, see does semaglutide cause fatigue, which covers the overlapping metabolic mechanisms.
Neuroinflammation Reduction
Chronic low-grade inflammation is a well-established contributor to depression. Obesity elevates inflammatory markers (IL-6, TNF-alpha, CRP) that cross the blood-brain barrier and disrupt neurotransmitter synthesis. GLP-1 receptor activation reduces neuroinflammation through multiple pathways: suppressing microglial activation, reducing pro-inflammatory cytokine production, and enhancing blood-brain barrier integrity.
A 12-month propensity-matched cohort study published in eClinicalMedicine found that semaglutide use was associated with reduced risk of cognitive deficit and showed a protective association against adverse neuropsychiatric outcomes (eClinicalMedicine, 2024). The anti-inflammatory mechanism likely contributes to these protective effects.
This means semaglutide may simultaneously produce short-term emotional blunting (through dopamine modulation) and long-term mood protection (through neuroinflammation reduction). The timeline matters: the first weeks are the most vulnerable period, while the long-term trajectory favors improved mental health.
Five Reasons You Might Feel Depressed on Semaglutide
If clinical trials show no increased depression risk, why do some users genuinely feel depressed? Five mechanisms explain the disconnect between population data and individual experience. Most users encounter a combination of these factors rather than a single trigger.
1. Loss of Food as an Emotional Coping Mechanism
Food is the most accessible mood regulator humans have. A bowl of pasta triggers a serotonin and dopamine cascade within minutes. Ice cream after a hard day provides reliable comfort. Wine with dinner softens the edges of stress. Semaglutide removes this tool with pharmaceutical precision. Appetite suppression does not distinguish between hunger and emotional need.
For people who have used food to manage loneliness, boredom, grief, or anxiety for years or decades, semaglutide strips away a primary coping mechanism without replacing it. The underlying emotions surface, often for the first time in their unfiltered form. This is not a drug side effect. It is the unmasking of emotional patterns that food had been suppressing.
A psychological review of bariatric surgery outcomes found that 15-20% of patients develop or worsen depressive symptoms in the first year after weight loss surgery, driven primarily by the removal of food-based coping (Gill et al., 2019). Semaglutide produces a similar dynamic through pharmacological appetite suppression rather than surgical restriction. For broader context on peptides that may support mood, see peptides for anxiety.
2. Caloric Deficit and Cortisol Elevation
Semaglutide reduces caloric intake by 500-1,000 calories per day in most users. STEP 1 participants on semaglutide 2.4 mg lost 14.9% of body weight over 68 weeks, requiring a sustained daily deficit of approximately 600-800 calories. The body interprets this deficit as a threat.
The hypothalamic-pituitary-adrenal (HPA) axis responds by increasing cortisol production. Sustained cortisol elevation above baseline produces a specific mood profile: low motivation, emotional numbness, poor concentration, and a pervasive sense of joylessness. This cortisol-driven mood state looks and feels like depression but resolves when the body adapts to its new caloric equilibrium.
A controlled study on caloric restriction found that deficits exceeding 25% of maintenance calories increased cortisol by 10-20% and worsened self-reported mood within two weeks (Tomiyama et al., 2010). Users eating fewer than 1,000 calories daily on semaglutide are almost certainly triggering this cortisol-mood cascade. If your weight loss has plateaued despite severe restriction, see not losing weight on semaglutide before cutting calories further.
3. Sleep Disruption from Gastrointestinal Side Effects
Semaglutide's most common side effects are gastrointestinal: nausea, vomiting, diarrhea, and constipation. These symptoms worsen when lying down. Users report fragmented sleep with multiple awakenings during the first weeks of treatment, particularly during dose escalation.
Sleep deprivation is one of the most reliable predictors of depressive episodes. A single night of restricted sleep (4-5 hours) impairs prefrontal cortex function and increases amygdala reactivity by 60% (Yoo et al., 2007). Three consecutive nights of poor sleep produce measurable increases in depressive symptom scores. The cumulative sleep debt from weeks of GI-disrupted nights creates a substrate on which depressive symptoms grow.
The practical takeaway: aggressive management of GI side effects is not just about comfort. It is a mental health intervention. For strategies to manage nausea specifically, see how to relieve nausea from semaglutide.
4. Identity Disruption and Social Shifts
Losing 15-20% of body weight changes how the world interacts with you. Strangers are friendlier. Colleagues offer compliments. Romantic attention shifts. Clothes fit differently. The mirror shows a person you may not fully recognize. These changes, all nominally positive, trigger an identity crisis that manifests as depression in a significant minority of rapid weight-loss patients.
The psychological literature on post-weight-loss depression identifies several contributors. Fear of regain creates anticipatory grief. Loose skin replaces one body dissatisfaction with another. Relationships built around shared eating patterns strain or dissolve. The expectation that thinness would solve life problems collides with the reality that the same problems persist at a lower weight.
Users who have been overweight since childhood or adolescence face the deepest identity disruption. Their entire social, romantic, and professional identity was formed in a larger body. Semaglutide-driven weight loss can outpace their psychological adjustment capacity. For realistic outcome expectations, see semaglutide before and after.
5. Pre-Existing Depression Unrelated to Semaglutide
Depression affects roughly 8% of American adults in any given two-week period (CDC NHANES data). Among people with obesity, the prevalence is higher: approximately 20-25% of adults with BMI above 30 have clinically significant depressive symptoms. Starting semaglutide does not erase pre-existing depression. It may reveal it.
Many people with undiagnosed depression attribute their low mood to being overweight. "I will feel better when I lose the weight" is a common belief. When semaglutide produces significant weight loss and the depression persists, the loss of that explanatory framework itself becomes distressing. The drug did not cause the depression. It removed the explanation that had been masking it.
This distinction has practical consequences. Depression that existed before semaglutide requires treatment directed at the depression itself: psychotherapy, antidepressant medication, or both. Adjusting the semaglutide dose or discontinuing it will not address a condition that predated the prescription.
Pharmacovigilance Data: What Adverse Event Reports Show
Pharmacovigilance databases collect voluntary reports of adverse events from healthcare providers and patients. These databases (FAERS in the U.S., EudraVigilance in Europe, VigiBase globally) have accumulated thousands of psychiatric adverse event reports for semaglutide.
A 2024 EudraVigilance analysis of GLP-1 receptor agonists found that depression was the most commonly reported psychiatric adverse event for semaglutide at 50.3% of all psychiatric reports, followed by anxiety at 38.7% and suicidal ideation at 19.6% (PMC, 2024). A separate VigiBase study confirmed similar patterns across semaglutide, liraglutide, and dulaglutide.
These numbers require careful interpretation. Pharmacovigilance data cannot establish causation. The denominator is unknown (how many total users never reported any issue), reporting rates vary by drug popularity and media attention, and reports are unverified. When a drug receives intense media coverage, as semaglutide has since 2023, reporting rates spike regardless of actual adverse event frequency.
The FDA addressed this directly in its 2024-2025 review. When they compared GLP-1 RA users to SGLT2 inhibitor users using the same administrative healthcare databases (where the denominator is known), the apparent signal disappeared. The rate of intentional self-harm was not higher in the GLP-1 group. The lesson: raw pharmacovigilance counts can mislead. Controlled comparisons with known denominators tell the true story.
A 2025 VigiBase study examining psychiatric and psychological adverse effects across dulaglutide, semaglutide, and liraglutide found that while reports exist, the overall psychiatric adverse drug reaction reporting rate is not significantly elevated compared to expected background rates (ScienceDirect, 2025).
Semaglutide for Depression: Emerging Therapeutic Evidence
In a striking reversal of the initial safety concern, researchers are now investigating semaglutide as a treatment for depression. A 2025 randomized clinical trial examined semaglutide for cognitive dysfunction in patients with major depressive disorder (PubMed, 2025). The trial found that semaglutide improved cognitive symptoms in depressed patients without worsening depressive symptom severity.
A systematic review and meta-analysis of GLP-1 receptor agonist antidepressant effects concluded that GLP-1 RAs demonstrate significant antidepressant properties in both animal models and human observational data (Am J Geriatr Psychiatry, 2024). The proposed mechanisms include reduced neuroinflammation, improved insulin sensitivity in the brain, enhanced neuroplasticity through BDNF upregulation, and modulation of the gut-brain axis.
The American Psychological Association noted in a 2025 review that the mental health effects of GLP-1 drugs represent "a new era" in understanding the relationship between metabolic and psychiatric health (APA Monitor, 2025). Several clinical trials are now underway testing semaglutide specifically for treatment-resistant depression, bipolar disorder, and anxiety disorders.
This trajectory, from safety concern to potential therapeutic application, mirrors what happened with ketamine (initially an anesthetic, now an antidepressant) and lithium (initially a gout remedy, now the gold standard for bipolar disorder). The relationship between semaglutide and depression is more nuanced than "does it cause it or not."
For users interested in peptides with established mood benefits, Selank has clinical evidence for anxiety and depression reduction. See our selank dosage guide for protocols.
When to Seek Help: Warning Signs That Demand Action

Most mood changes on semaglutide are mild, temporary, and self-resolving. Certain patterns, however, demand professional evaluation. This section uses clear thresholds so you can distinguish normal adjustment from a problem that needs intervention.
Normal adjustment (no action required beyond self-care): Mild sadness or emotional flatness during the first 2-4 weeks. Reduced excitement about food. Occasional low-energy days. Brief periods of irritability during dose escalation. These symptoms should fluctuate, not worsen steadily, and they should not prevent you from functioning at work or in relationships.
Monitor closely (discuss at next appointment): Persistent low mood lasting more than 2 weeks at the same intensity. Noticeable social withdrawal that others comment on. Loss of interest in activities you previously enjoyed (beyond food). Difficulty concentrating at work. Sleeping significantly more or less than usual. These patterns suggest the mood disturbance is more than a transient adjustment.
Seek help within one week: Daily feelings of hopelessness or worthlessness. Crying spells that occur without clear trigger. Inability to perform work or home responsibilities. Complete loss of motivation. Significant appetite changes beyond what semaglutide explains (refusing all food, or binge eating despite the drug). These symptoms meet screening criteria for a major depressive episode and warrant professional evaluation.
Emergency (act now): Suicidal thoughts or plans. Self-harm ideation or behavior. Feeling that others would be better off without you. Giving away possessions. Sudden calmness after a period of severe depression (which can indicate a decision to act on suicidal thoughts). Contact the 988 Suicide and Crisis Lifeline (call or text 988) immediately. Do not wait for your next appointment.
| Severity | Symptoms | Action | Timeline |
|---|---|---|---|
| Normal | Mild sadness, flatness, first 2-4 weeks | Self-care, monitor | Resolves in 4-6 weeks |
| Monitor | Low mood >2 weeks, social withdrawal | Discuss with prescriber | Next scheduled visit |
| Urgent | Daily hopelessness, inability to function | Mental health evaluation | Within 1 week |
| Emergency | Suicidal thoughts, self-harm | 988 Lifeline or ER | Immediately |
For broader safety guidance on peptide use, see our peptide safety guide. If you are taking tirzepatide and experiencing similar concerns, see can tirzepatide cause anxiety for a parallel analysis.
Six Strategies to Protect Your Mental Health on Semaglutide
These strategies address the five mood-disrupting mechanisms identified above. Most users notice improvement within 1-2 weeks of implementing the first three.
1. Maintain a Moderate Caloric Deficit
Extreme caloric restriction is the most common preventable cause of mood deterioration on semaglutide. When appetite disappears, many users eat 600-800 calories daily without realizing it. At that deficit, cortisol rises by 20-30%, serotonin synthesis drops (tryptophan requires adequate caloric intake for conversion), and the brain enters a survival-mode state that mimics depression.
Aim for 1,200-1,600 calories daily depending on your body size. Track intake for at least the first month to calibrate your awareness. Prioritize protein (1.0-1.2 g/kg body weight daily) to protect muscle mass and provide tryptophan for serotonin production. A 180-pound person needs at least 82-98 grams of protein per day.
If you are struggling to eat enough, schedule 4-5 small meals rather than attempting 2-3 large ones. Protein shakes, Greek yogurt, and nuts are calorie-dense options that are easier to consume when appetite is suppressed. The weight loss will be marginally slower at 1,400 calories than at 800 calories, but your mood will be stable enough to sustain the treatment long-term. Use our semaglutide dosage calculator to ensure your dose matches your metabolic goals.
2. Protect Sleep at All Costs
Sleep deprivation is the fastest route to depressive symptoms on semaglutide. One night of restricted sleep impairs prefrontal cortex function. Three consecutive poor nights produce measurable increases in depressive symptom scores. GI side effects from semaglutide disrupt sleep precisely when your brain needs it most.
Three adjustments make a measurable difference. Inject in the morning so that peak drug levels and peak GI symptoms occur during waking hours rather than overnight. Eat your last meal 3-4 hours before bed so gastric contents have partially emptied before you lie down. Supplement magnesium glycinate (200-400 mg at bedtime), which promotes GABA-mediated relaxation and addresses the magnesium depletion that caloric restriction causes.
If reflux or nausea continue disrupting sleep despite these measures, elevate the head of your bed 6-8 inches using bed risers. This simple change reduces nocturnal reflux episodes by up to 70% in clinical studies.
3. Replace Food-Based Coping Before You Need To
Do not wait until you feel depressed to build new coping mechanisms. If food has been your primary stress relief, start developing alternatives during the first week of semaglutide treatment, before the appetite suppression strips away your old tools.
Exercise is the most evidence-based non-pharmacological antidepressant available. A 30-minute moderate walk reduces depressive symptom scores by 20-30%, with effects lasting 4-6 hours. Resistance training produces similar benefits through BDNF upregulation and endocannabinoid release. The minimum effective dose is 150 minutes per week of moderate activity.
Social connection serves as a second pillar. Depression thrives in isolation. Users who stop eating out, decline dinner invitations, and avoid food-centered social situations create a loneliness vacuum that deepens mood decline. Find non-food social activities: hiking groups, book clubs, volunteer work, gym classes.
Journaling, meditation, and cognitive behavioral therapy (CBT) address the thought patterns that contribute to depression. CBT is particularly effective for the catastrophic thinking ("I will never enjoy life without food") that some semaglutide users experience. If you used food to manage anxiety specifically, see peptides for anxiety for evidence-based peptide options.
4. Monitor Your Mood Systematically
Subjective assessment of your own mood is unreliable. Depression biases self-perception toward the negative, making it difficult to recognize gradual worsening or improvement. A structured monitoring approach removes this bias.
The PHQ-2 is a validated two-question screening tool you can use weekly. Question 1: "Over the past week, how often have you been bothered by little interest or pleasure in doing things?" Question 2: "Over the past week, how often have you been bothered by feeling down, depressed, or hopeless?" Rate each from 0 (not at all) to 3 (nearly every day). A total score of 3 or higher warrants the full PHQ-9 and a conversation with your healthcare provider.
Track your PHQ-2 score weekly in a simple spreadsheet or journal. Look for trends over 4-6 weeks rather than reacting to individual data points. A score of 1-2 that remains stable across weeks represents normal adjustment. A score that climbs from 1 to 3 to 5 over consecutive weeks signals a trajectory that needs intervention.
5. Communicate with Your Prescriber Proactively
Do not wait until you are in crisis to mention mood changes. Many prescribers ask about GI side effects at follow-up visits but do not specifically screen for mood disturbances. Bring it up yourself. Use your PHQ-2 tracking data to provide objective information rather than vague complaints.
Three interventions your prescriber may offer. First, slowing the titration schedule: spending 6-8 weeks at each dose instead of 4 gives the brain more time to adapt. Second, reducing the maintenance dose: many users achieve adequate weight loss at 1.7 mg rather than 2.4 mg, with fewer neuropsychiatric effects. Third, referral to a mental health professional, particularly one experienced with weight-loss-associated mood changes.
If you are already taking antidepressant medication, semaglutide may affect absorption. GLP-1 agonists slow gastric emptying, which can delay the absorption of oral medications. Your prescriber may need to adjust timing or dosing of psychiatric medications. Do not change psychiatric medication doses on your own.
6. Know When the Drug Is Not the Problem
If depression persists beyond 8-10 weeks at a stable semaglutide dose and you have implemented the strategies above, consider that semaglutide may not be the cause. Request comprehensive bloodwork: TSH and free T4 (thyroid dysfunction from rapid weight loss), vitamin D (deficiency prevalence in overweight individuals exceeds 40%), vitamin B12, folate, and iron (deficiencies impair serotonin synthesis), and AM cortisol (to evaluate HPA axis function).
If bloodwork is normal and depression persists, a mental health evaluation is the appropriate next step. The depression may be a pre-existing condition unmasked by the removal of food-based coping, a separate life stressor coinciding with semaglutide treatment, or a condition that requires targeted treatment regardless of what caused it.
Stopping semaglutide to "see if the depression improves" should be a last resort discussed with your prescriber, not a first-line response. Abrupt discontinuation leads to rapid weight regain in most patients, which itself is a potent depressogenic event.
Special Populations: Higher-Risk Groups
Certain populations face elevated risk for mood disturbances on semaglutide. Awareness allows proactive management rather than reactive crisis response.
People with a history of depression or anxiety: The STEP trials excluded people with active major depression. Real-world prescribing does not. A person with recurrent depression who starts semaglutide faces compounding risk factors: neurochemical vulnerability plus caloric restriction plus sleep disruption plus coping mechanism removal. The Lancet Psychiatry Swedish study (2026) actually showed that GLP-1 RA users with pre-existing depression fared better than expected, but that study population was medically managed. If you have a depression history, ensure your mental health provider is aware of your semaglutide use.
People on psychiatric medications: SSRIs (sertraline, fluoxetine, escitalopram), SNRIs (venlafaxine, duloxetine), and other oral psychiatric medications may have altered absorption due to semaglutide-induced delayed gastric emptying. This does not mean they are contraindicated. It means blood levels may fluctuate during early semaglutide treatment, potentially reducing efficacy. If depression symptoms worsen after starting semaglutide and you take psychiatric medication, altered absorption is a likely contributor.
People with eating disorder history: Semaglutide fundamentally alters the relationship with food. For individuals recovering from anorexia, bulimia, or binge eating disorder, this alteration can destabilize recovery. The appetite suppression may feel like a "sanctioned" version of restriction. The loss of food-related pleasure may trigger grief. A specialized eating disorder therapist should be part of the treatment team.
Adolescents and young adults: Semaglutide was FDA-approved for adolescents aged 12 and older with obesity in 2023. Adolescent brains are still developing, with incomplete prefrontal cortex myelination and heightened emotional reactivity. The safety data in this population is limited. Parents and prescribers should monitor mood weekly during treatment.
For general safety across all populations, see our peptide safety guide.
Important Warnings and Disclaimers
This article synthesizes clinical trial data, FDA safety reviews, and peer-reviewed observational studies. It is not a substitute for medical advice from your prescriber or mental health professional.
Depression is a serious medical condition. If you are experiencing depressive symptoms on semaglutide, do not rely on internet articles to guide your treatment decisions. Discuss your symptoms with your healthcare provider. If you are experiencing suicidal thoughts, contact the 988 Suicide and Crisis Lifeline (call or text 988) or go to the nearest emergency department.
Do not stop semaglutide abruptly without medical guidance. Discontinuation can lead to rapid weight regain, rebound appetite, and its own mood consequences. Work with your prescriber to develop a plan if medication changes are warranted.
Do not self-medicate depression with alcohol. Alcohol worsens sleep quality, depletes serotonin precursors, elevates cortisol, and interacts with the metabolic changes semaglutide produces.
Do not adjust psychiatric medication doses based on information in this article. Medication changes require prescriber oversight, particularly when semaglutide may be affecting drug absorption.
This article reflects evidence available as of April 2026. The science of GLP-1 receptor agonists and mental health is evolving rapidly. New data may alter current recommendations.
For broader context on semaglutide side effects, see how long do semaglutide side effects last. For storage information, see does semaglutide expire. For dosing guidance, see our semaglutide dosage chart and semaglutide dosage calculator.
Frequently Asked Questions
Does semaglutide cause depression?
The current clinical evidence does not support semaglutide as a direct cause of depression. Across the STEP 1, 2, 3, and 5 trials, only 2.8% of semaglutide-treated participants reported depressive symptoms compared to 4.1% on placebo. The FDA reviewed over 1.16 million GLP-1 RA users and found no increased risk, leading them to remove the suicidality warning from all GLP-1 labels in January 2025. However, the metabolic and psychological changes semaglutide triggers can surface mood disturbances in vulnerable individuals. For full trial data, see our analysis of the semaglutide STEP trial results.
Can Ozempic make you feel sad or emotionally numb?
Yes, some users report emotional flatness or reduced pleasure (anhedonia) on Ozempic (semaglutide). This is likely related to dopamine modulation in reward pathways and the removal of food as an emotional coping tool, not clinical depression caused by the drug itself. The feeling typically peaks during weeks 2-6 of treatment or dose escalation and resolves within 4-8 weeks at a stable dose. If sadness persists beyond 8 weeks, discuss it with your prescriber. For related side effects, see does semaglutide cause fatigue.
Did the FDA find a link between semaglutide and suicidal thoughts?
No. The FDA conducted a retrospective cohort study comparing 1,161,983 GLP-1 RA users to 1,081,155 SGLT2 inhibitor users and found no increased risk of intentional self-harm. In January 2025, the FDA formally requested that manufacturers remove the suicidal behavior and ideation warning from all GLP-1 receptor agonist labels, including Wegovy and Ozempic. The NIH separately confirmed that semaglutide users had a lower risk of suicidal thoughts than users of other weight-loss drugs. For dosing protocols, see our semaglutide dosage calculator.
Should I stop semaglutide if I feel depressed?
Do not stop semaglutide abruptly without consulting your prescriber. Abrupt discontinuation causes rapid weight regain in most patients, which itself is a potent trigger for depressive episodes. Instead, implement the management strategies first: maintain adequate caloric intake (1,200-1,600 calories), protect sleep, replace food-based coping mechanisms, and monitor your mood weekly with the PHQ-2 tool. If depression persists beyond 8 weeks at a stable dose despite these strategies, discuss dose reduction or discontinuation with your prescriber. See our peptide safety guide for general guidance.
Is depression more common on semaglutide or tirzepatide?
Head-to-head data from the SURPASS-2 trial found comparable psychiatric adverse event rates between semaglutide and tirzepatide. Both drugs affect GLP-1 receptors in brain regions that regulate mood. Tirzepatide's additional GIP receptor activation may theoretically buffer some mood effects, but this has not been confirmed in clinical trials. Switching medications for mood symptoms alone is unlikely to help because the underlying mechanisms (caloric restriction, dopamine modulation, sleep disruption) are shared across the GLP-1 drug class. See can tirzepatide cause anxiety for a detailed comparison.
Can semaglutide help with depression?
Emerging evidence suggests it might. A 2025 randomized trial tested semaglutide for cognitive dysfunction in major depressive disorder and found cognitive improvement without mood worsening. A 2026 Swedish cohort study found 44% lower depression risk in semaglutide users. Multiple mechanisms support antidepressant effects: reduced neuroinflammation, improved brain insulin sensitivity, and enhanced neuroplasticity. Clinical trials are now underway testing semaglutide specifically as a depression treatment. For peptides with established anxiolytic effects, see peptides for anxiety.
How long do mood changes last on semaglutide?
Most mood disturbances on semaglutide peak during weeks 2-6 of treatment or dose escalation and resolve within 4-8 weeks at a stable dose. The timeline mirrors the adaptation curve for GI side effects: the brain needs 6-8 weeks to recalibrate neurotransmitter signaling, cortisol levels, and dopamine kinetics to the new pharmacological environment. Mood changes that worsen steadily or persist beyond 8-10 weeks at a stable dose warrant medical evaluation. For details on side effect duration across all symptoms, see how long do semaglutide side effects last.
What should I do if I have suicidal thoughts on semaglutide?
Contact the 988 Suicide and Crisis Lifeline immediately by calling or texting 988. You can also go to the nearest emergency department. Do not wait for your next scheduled appointment. Do not assume the thoughts are a medication side effect that will pass on its own. While the FDA found no causal link between semaglutide and suicidal ideation, suicidal thoughts from any cause require immediate professional intervention. After stabilization, your prescriber can evaluate whether semaglutide contributed and adjust your treatment plan accordingly. See our peptide safety guide for emergency resources.
The Bottom Line
The clinical evidence is reassuring: semaglutide does not cause depression through a direct pharmacological mechanism. Across four Phase 3 trials, the FDA's 1.16-million-person safety review, and a 2026 Swedish national cohort study, the drug either showed no increased depression risk or showed a protective association. The FDA removed the suicidality warning from all GLP-1 RA labels in January 2025.
That does not mean your low mood is imaginary. Five real mechanisms can surface depressive symptoms during semaglutide treatment: the loss of food-based emotional coping, cortisol elevation from caloric deficit, sleep disruption from GI side effects, identity disruption from rapid weight change, and unmasking of pre-existing depression. Each of these is addressable.
Maintain a moderate caloric deficit of 1,200-1,600 calories daily. Protect sleep by injecting in the morning and managing GI symptoms aggressively. Build non-food coping mechanisms before you need them. Monitor your mood weekly using the PHQ-2. Communicate with your prescriber at the first sign of persistent low mood, not after weeks of suffering.
Red flags requiring immediate action: suicidal thoughts or self-harm (call 988), daily hopelessness lasting more than 2 weeks, or complete inability to function at work or home.
Use our semaglutide dosage calculator to plan your dosing schedule. For side effect management across all symptoms, see how long do semaglutide side effects last and does semaglutide cause fatigue.
Related semaglutide articles: - Does Semaglutide Cause Nausea?: GI side effects that disrupt sleep and compound mood issues - Does Semaglutide Cause Fatigue?: overlapping caloric restriction and cortisol mechanisms - Not Losing Weight on Semaglutide: when frustration drives mood decline - Semaglutide Before and After: realistic expectations reduce disappointment-driven depression - How Long Does Semaglutide Side Effects Last?: timeline for all side effects including mood changes - Can Tirzepatide Cause Anxiety?: parallel analysis for the other major GLP-1 drug - Peptides for Anxiety: evidence-ranked anxiolytic peptides including Selank and BPC-157 - Peptide Safety Guide: comprehensive safety framework
Explore all peptide profiles and tools at PeptidesExplorer.
Related Articles
Does Semaglutide Cause Heartburn?
Does semaglutide cause heartburn? Yes, in 1.9-5% of users. Delayed gastric emptying mechanism, GERD differences, and 7 ways to manage it.
Can Semaglutide Cause Diarrhea?
Diarrhea affects 8-30% of semaglutide users depending on dose. Learn the GLP-1 mechanism, STEP trial incidence data, timeline, and 6 management strategies.
Semaglutide Nausea Relief: 9 Proven Tips
Relieve semaglutide nausea with 9 actionable strategies: ondansetron dosing, ginger protocols, dietary timing, acupressure, and when to call your doctor.
Semaglutide Side Effects Duration
Most semaglutide side effects resolve within 2-4 weeks as your body adjusts. See clinical timelines for nausea, diarrhea, constipation, and fatigue.