The standard Selank dosage is 250-500 mcg per day administered intranasally, typically as 2-3 drops per nostril, 1-2 times daily. Selank (TP-7) is a synthetic heptapeptide analog of tuftsin approved in Russia since 2009 for generalized anxiety disorder. The Russian-approved protocol specifies 450 mcg/day via 0.15% nasal drops for 14 days. A 62-patient randomized trial found Selank comparable to medazepam (a benzodiazepine) for anxiety reduction, with additional cognitive and energizing benefits the benzodiazepine lacked (PMID: 18454096).
| Quick Reference | Details |
|---|---|
| Full name | Selank (TP-7) |
| Structure | Thr-Lys-Pro-Arg-Pro-Gly-Pro (tuftsin + Pro-Gly-Pro tail) |
| Molecular weight | 751.89 Da (CAS: 129954-34-3) |
| Mechanism | GABA modulation, BDNF upregulation, enkephalinase inhibition |
| Approved dosage (Russia) | 450 mcg/day nasal (0.15% solution, 3x daily) |
| Research dosage | 250-500 mcg/day nasal or subcutaneous |
| Cycle length | 2-4 weeks on, 1-2 weeks off |
| Half-life | 2-5 minutes (parent compound) |
| Duration of effect | 3-6 hours (sustained via gene expression changes) |
| Regulatory status | Approved in Russia/Ukraine; research chemical elsewhere |
Selank's short half-life (2-5 minutes) seems to contradict its lasting effects (3-6 hours). The explanation: Selank triggers downstream changes in gene expression across 36 GABA-related genes and alters receptor sensitivity that persist long after the peptide itself clears from plasma (PMC4757669). This is similar to how a match lights a fire that burns long after the match goes out.
For other anxiety-related peptides, see our peptides for anxiety guide. For the full peptide profile, see our Selank profile page. This is educational content about a research compound.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Selank Dosage Chart
Selank dosing varies by route of administration, experience level, and therapeutic goal.
Nasal Spray Dosage by Experience Level
| Level | Daily Dose | Frequency | Cycle Duration | Notes |
|---|---|---|---|---|
| Beginner | 200-300 mcg | Once daily (morning) | 14 days | Start here to assess tolerance |
| Intermediate | 400-500 mcg | 2x daily (morning + afternoon) | 14-21 days | Russian-approved range |
| Advanced | 600-900 mcg | 2-3x daily | 21-30 days | Research protocols only |
Weight-Based Dosing Reference
| Body Weight | Conservative Dose | Standard Dose | Upper Range |
|---|---|---|---|
| Under 130 lbs (59 kg) | 200 mcg/day | 300 mcg/day | 500 mcg/day |
| 130-180 lbs (59-82 kg) | 250 mcg/day | 450 mcg/day | 600 mcg/day |
| Over 180 lbs (82+ kg) | 300 mcg/day | 500 mcg/day | 750 mcg/day |
The Russian-approved protocol is specific: 0.15% nasal drops, 2 drops per nostril (approximately 75 mcg per drop), 3 times daily, for 14 days. This delivers 450 mcg daily. Research protocols at the V.V. Zakusov Institute of Pharmacology have tested doses up to 2,700 mcg/day for 21 days.
Use our peptide reconstitution calculator for preparation and our unit converter for dose calculations. See the peptide dosage chart for cross-compound comparisons.
Nasal Spray vs Subcutaneous Injection
Most users choose nasal administration. The data supports this preference.
| Parameter | Nasal Spray | Subcutaneous Injection |
|---|---|---|
| Bioavailability | 70-90% (estimated) | 50-70% (estimated) |
| Onset | 10-15 minutes | 15-30 minutes |
| Dose range | 200-900 mcg/day | 250-500 mcg/day |
| Convenience | High (non-invasive) | Lower (requires syringe) |
| CNS access | More direct (bypasses BBB via olfactory nerve) | Standard absorption pathway |
| Primary use | Anxiety, cognition | Research settings |
| Dose consistency | Moderate (technique-dependent) | High |
Nasal spray is preferred for two reasons. First, the olfactory nerve provides a more direct pathway to the brain, bypassing the blood-brain barrier partially. Second, the estimated bioavailability is actually higher for nasal administration than subcutaneous, which is unusual among peptides.
Nasal administration tips: - Tilt head slightly forward (not backward). - Spray into nostril while gently inhaling. - Alternate nostrils between doses. - Wait 5 minutes before blowing nose. - Morning and early afternoon timing preferred. - Avoid evening dosing if sleep quality is affected (rare but reported).
For injection technique, see our how to inject peptides guide. For nasal spray technique comparison, see our BPC-157 nasal spray guide.
Goal-Specific Dosing Protocols
Selank's triple mechanism (anxiolytic + nootropic + immunomodulatory) means dosing should be tailored to the primary goal.
Protocol 1: Anxiety Relief (Primary Use)
| Parameter | Details |
|---|---|
| Daily dose | 250-500 mcg nasal |
| Frequency | 2-3x daily (morning, midday, early afternoon) |
| Cycle | 14-30 days on, 2 weeks off |
| Onset | Acute calming within 15 minutes; full anxiolytic stabilization at 3-5 days |
Selank enhances GABA-A receptor sensitivity without directly binding benzodiazepine sites. A 62-patient randomized trial comparing Selank to medazepam for GAD found comparable anxiolytic effects using the Hamilton Anxiety Rating Scale. Selank showed additional antiasthenic (anti-fatigue) and psychostimulant benefits the benzodiazepine lacked (PMID: 18454096). A separate trial against phenazepam confirmed anxiolytic effects persisted 1+ week after the last dose (PMID: 25176261).
Protocol 2: Cognitive Enhancement
| Parameter | Details |
|---|---|
| Daily dose | 300-500 mcg nasal |
| Frequency | Once daily (morning) |
| Cycle | 10-20 days on, 2 weeks off |
| Best paired with | Semax (300-600 mcg/day) for synergy |
Selank upregulates BDNF mRNA expression in the hippocampus and prefrontal cortex, supporting memory consolidation and neuroplasticity (PMID: 31625062). The nootropic effect is distinct from the anxiolytic mechanism. Where anxiety reduction comes from GABA modulation, cognitive enhancement comes from BDNF-driven neuroplasticity.
Animal studies showed Selank protected against ethanol-induced memory impairment through this BDNF pathway. Use our stack calculator for Selank+Semax combinations.
Protocol 3: Selank + Semax Stack
The most established nootropic peptide combination. Both were developed at the Institute of Molecular Genetics (Russian Academy of Sciences) and designed to complement each other.
| Component | Dose | Timing | Role |
|---|---|---|---|
| Semax | 300-600 mcg nasal | Morning | Focus, BDNF upregulation, dopamine |
| Selank | 250-500 mcg nasal | 30 min after Semax, or midday | Calm balance, GABA, stress buffer |
No dose reduction is needed when stacking because they act through entirely different pathways. Semax acts as an ACTH(4-10) analog targeting BDNF and NGF. Selank acts through GABA modulation and enkephalinase inhibition. The result: focused cognition (Semax) without the jitteriness or anxiety that stimulants can cause (Selank). See our peptides for cognitive function guide for more options.
Protocol 4: Immune Support
| Parameter | Details |
|---|---|
| Daily dose | 400-500 mcg nasal |
| Frequency | Once daily (morning) |
| Cycle | 21-30 days on, 2 weeks off |
Selank retains the immunomodulatory properties of its parent peptide tuftsin. It modulates IL-6 and interferon-gamma expression, enhances immunoglobulin production, and increases NK cell activity (PMID: 22017284). The effect is bidirectional: it boosts underactive immunity while calming overactive inflammatory responses (PMID: 19340573). Useful during periods of immune stress: travel, illness recovery, seasonal transitions.
For more on immune-supporting peptides, see our peptides for immune system guide.
How Selank Works
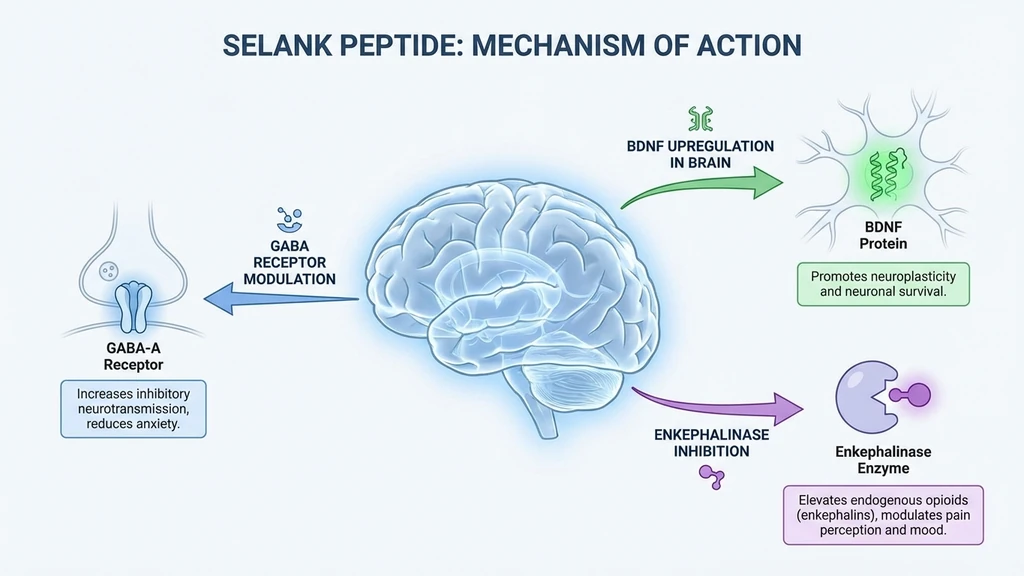
Selank modulates five systems simultaneously, which is why it produces both anxiolytic and cognitive effects without the trade-offs of traditional medications.
GABAergic modulation. Selank enhances GABA system activity by modulating the expression of 36 genes involved in GABAergic neurotransmission (PMC4757669). Unlike benzodiazepines, it does not directly bind GABA-A receptor benzodiazepine sites. This produces anxiolysis without sedation, tolerance, or dependence.
BDNF upregulation. Selank increases brain-derived neurotrophic factor expression in the hippocampus. BDNF supports neuroplasticity, learning, and memory consolidation. This mechanism drives cognitive enhancement and distinguishes Selank from anxiolytics that impair cognition (PMID: 31625062).
Enkephalinase inhibition. Selank inhibits the enzymes that break down enkephalins (endogenous opioid peptides). Higher enkephalin levels improve mood and stress resilience without euphoria or addiction risk. GAD patients show decreased leu-enkephalin levels; Selank treatment normalized these levels in clinical trials (PMID: 18577768).
Serotonin and dopamine regulation. Selank stabilizes serotonin turnover in the frontal cortex and hippocampus and modulates dopaminergic signaling. Unlike SSRIs, it does not produce emotional blunting (PMID: 18427834).
Immune modulation. Selank retains tuftsin's immunomodulatory properties, modulating IL-6 and interferon-gamma while enhancing immunoglobulin production and NK cell activity (PMID: 22017284).
Timeline of Effects
Selank's effects unfold in phases. Acute calming occurs within minutes; full therapeutic benefits build over days.
| Timeframe | What to Expect |
|---|---|
| 15-30 minutes | Acute calming effect, reduced mental noise |
| 1-3 days | Consistent mood stabilization beginning |
| 3-5 days | Full anxiolytic effect established |
| 1-2 weeks | Enhanced learning, memory consolidation improvements |
| 2-4 weeks | Peak cognitive and immune support benefits |
| Post-cycle (1+ week) | Anxiolytic effects persist after discontinuation |
The pharmacokinetic paradox: Selank has an ultra-short plasma half-life of 2-5 minutes (completely cleared from circulation within 10 minutes), yet effects persist for hours per dose and days cumulatively. The Pro-Gly-Pro tail enhances metabolic stability versus parent tuftsin, and the peptide triggers sustained downstream changes in gene expression, receptor sensitivity, and neurotransmitter metabolism that continue after clearance (PMID: 30255741).
This explains why once or twice daily dosing is sufficient despite the short half-life. Each administration reactivates the downstream cascade.
N-Acetyl Selank Amidate
N-Acetyl Selank Amidate is a modified version designed for improved stability and potency.
| Feature | Standard Selank | N-Acetyl Selank Amidate |
|---|---|---|
| Half-life | 2-5 minutes | Estimated 10-20 minutes |
| Stability | Degrades quickly (aminopeptidases) | Resistant to enzymatic breakdown |
| Potency | Standard | Reported as 2-3x more potent |
| Dosage adjustment | Standard doses | Reduce to 150-300 mcg daily |
| Cost | Lower | Higher (premium product) |
| Clinical data | Russian clinical trials | Limited (community-derived protocols) |
The N-acetyl group protects the N-terminus from aminopeptidases. The C-terminal amide protects from carboxypeptidases. Together, these modifications extend active time and may improve blood-brain barrier penetration.
The trade-off: standard Selank has published clinical trial data from randomized controlled trials. N-Acetyl Selank Amidate does not. The modification makes pharmacological sense but has no independent clinical validation. If using this variant, start at the lower end of the dose range (150 mcg) and titrate up.
Selank vs Semax
Both are Russian-developed nootropic peptides from the same laboratory, but they target different primary goals.
| Feature | Selank | Semax |
|---|---|---|
| Primary effect | Anxiolytic (anti-anxiety) | Nootropic (pro-focus) |
| Derived from | Tuftsin (immunoglobulin G fragment) | ACTH(4-10) fragment |
| Key mechanism | GABA modulation, enkephalin stabilization | BDNF/NGF upregulation, dopamine modulation |
| Best for | Anxiety, stress resilience, calm focus | Focus, memory, motivation, learning |
| Russian indication | GAD, neurasthenia | Stroke recovery, cognitive decline |
| Typical dose | 250-500 mcg/day nasal | 300-600 mcg/day nasal |
| Onset | 10-15 minutes | 5-10 minutes |
| Duration | 3-6 hours | 3-4 hours |
| Immune effects | Strong immunomodulatory | Minimal |
| Stimulating? | Mildly calming | Mildly stimulating |
Choosing between them: If your primary concern is anxiety or stress, choose Selank. If your primary concern is focus and cognitive performance, choose Semax. For both, stack them together (see Protocol 3 above). Verify combinations with our interaction checker.
Side Effects and Safety
Selank has one of the mildest side effect profiles among research peptides, backed by Russian clinical use since 2009.
| Side Effect | Frequency | Notes |
|---|---|---|
| Nasal irritation (spray) | Common | Mild, temporary, technique-dependent |
| Light headache | Occasional | Usually first 1-3 days only |
| Mild fatigue | Rare | Paradoxical; resolve by reducing dose |
| Vivid dreams | Rare | Reported anecdotally, not confirmed in trials |
| Allergic reaction | Very rare | Discontinue if rash or swelling |
What Selank does NOT cause (based on clinical data):
- No cognitive impairment (unlike benzodiazepines)
- No tolerance with proper cycling (2-4 weeks on, 1-2 weeks off)
- No withdrawal symptoms (anxiolytic effects persist 1+ week after discontinuation per PMID: 25176261)
- No dependence potential (not a controlled substance, does not bind benzodiazepine sites)
- No hormonal effects
- No significant cardiovascular effects
Contraindications: Pregnancy, breastfeeding, active autoimmune conditions (Selank's immune modulation could aggravate flares), known allergy to tuftsin-related peptides. Use caution with immunosuppressant medications.
Cycling is recommended. While no tolerance has been documented in clinical trials of 2-6 weeks, cycling (2-4 weeks on, 1-2 weeks off) maintains optimal GABAergic receptor sensitivity. No taper is required: abrupt discontinuation is safe.
For general peptide safety, see our peptide safety guide and are peptides legal.
Regulatory Status
Russia and Ukraine. Approved prescription medication since 2009 for generalized anxiety disorder and neurasthenia. Available as 0.15% nasal drops.
United States. Not FDA-approved. Not a recognized prescription medication or dietary supplement. Available through compounding pharmacies with a valid prescription or as a research chemical. The FDA has flagged immunogenicity concerns for compounded peptide formulations.
Europe, Australia, Canada. Not approved. Generally unregulated but not specifically banned. Classified as a research compound. Access is primarily through research chemical vendors or telehealth clinics with compounding pharmacy partnerships.
For the broader regulatory landscape, see our peptide legality guide and FDA peptide crackdown analysis.
Frequently Asked Questions
What is the standard Selank dosage for anxiety?
The Russian-approved dose is 450 mcg/day nasal (0.15% solution, 2 drops per nostril, 3 times daily) for 14 days. Research protocols use 250-500 mcg/day via nasal spray in 2-4 week cycles. A 62-patient randomized trial found this dose range comparable to medazepam for anxiety reduction (PMID: 18454096).
How fast does Selank work?
Nasal administration produces acute calming effects within 10-15 minutes. Subcutaneous injection takes 15-30 minutes. Full anxiolytic stabilization occurs after 3-5 days of consistent use. Despite Selank's 2-5 minute plasma half-life, effects last 3-6 hours because the peptide triggers sustained gene expression changes.
Is Selank nasal spray better than injection?
For most users, nasal spray is preferred. It offers higher estimated bioavailability (70-90% vs 50-70% subcutaneous), faster onset (10-15 vs 15-30 minutes), and non-invasive administration. The olfactory nerve provides more direct CNS access, partially bypassing the blood-brain barrier.
Can you take Selank and Semax together?
Yes. The Selank-Semax stack is the most established nootropic peptide combination. They act through entirely different mechanisms: Selank modulates GABA and enkephalins; Semax activates BDNF and NGF via the ACTH pathway. No dose reduction is needed. Use Semax (300-600 mcg) in the morning, Selank (250-500 mcg) 30 minutes later.
Does Selank cause dependence or withdrawal?
No dependence has been reported in clinical studies. Selank does not directly bind benzodiazepine receptor sites. Russian trial data shows anxiolytic effects persisted for 1+ week after discontinuation with no withdrawal symptoms (PMID: 25176261). No taper is required when stopping.
What is N-Acetyl Selank Amidate?
A modified version with N-acetyl and C-amide groups that protect against enzymatic degradation. Estimated 2-3x more potent with an extended half-life of 10-20 minutes versus 2-5 minutes for standard Selank. Reduce dose to 150-300 mcg daily. It lacks independent clinical trial data.
Is Selank better than benzodiazepines for anxiety?
A 62-patient Russian trial showed Selank produced comparable anxiolytic effects to medazepam with additional antiasthenic and psychostimulant benefits the benzodiazepine lacked. Selank does not cause sedation, tolerance, cognitive impairment, or dependence. It is not FDA-approved and has less clinical data than established benzodiazepines.
Is Selank FDA approved?
No. Selank is approved in Russia and Ukraine for generalized anxiety disorder since 2009 but is not FDA-approved in the United States. It is available through compounding pharmacies with a valid prescription or as a research chemical. The FDA has flagged immunogenicity concerns with compounded formulations.
The Bottom Line
Selank is one of the few peptides with government-approved clinical use (Russia, since 2009). Its defining advantage: anxiolytic effects comparable to benzodiazepines without sedation, tolerance, cognitive impairment, or dependence.
Standard dosing is 250-500 mcg daily, nasal or subcutaneous, in 2-4 week cycles. The modified N-Acetyl Selank Amidate variant offers improved stability at lower doses but lacks independent clinical validation.
For preparation guidance, use our reconstitution calculator. For storage information, see how to store peptides. For alternative anxiety peptides, see our peptides for anxiety guide.
This is educational content about a research compound. Consult a healthcare provider before using any peptide.
Related Articles
BPC-157 Dosage Per Body Weight (Chart)
BPC-157 dosage by body weight: 1.6-10 mcg/kg. Full chart for 120-280 lbs, injection vs oral doses, and why most use flat 250-500 mcg.
How Much Sermorelin Per Day: Complete Dosage Guide
Sermorelin dosage: 200-500 mcg/day subcutaneous before bedtime. Starting dose, titration, gender-specific protocols, and IGF-1 monitoring.
BPC-157 TB-500 Dosage: Combined Protocol
BPC-157 TB-500 dosage: 250-500 mcg BPC-157 + 2-2.5 mg TB-500 daily. Weight-adjusted charts, injury-type matrix, blend vial math, and cycle timing.
Semaglutide Mixing Chart: Every Vial Size (2026)
Semaglutide mixing chart for 3mg, 5mg, 10mg, 15mg vials. BAC water ratios, unit conversion, step-by-step reconstitution, titration schedule, and storage guide.