Thymosin Alpha-1 (Ta1) is a 28-amino-acid peptide that strengthens the immune system by restoring T-cell function. It is approved in over 30 countries under the brand name Zadaxin for hepatitis B, hepatitis C, and as an immune support agent during cancer treatment. More than 11,000 patients have been studied in clinical trials.
Among immune-focused peptides, Ta1 stands apart. It has decades of clinical data behind it, not just animal studies or cell culture experiments. For people dealing with chronic infections, age-related immune decline, or weakened immunity from illness or treatment, Ta1 is one of the most evidence-backed options available.
This guide grades each benefit by the strength of the evidence: Strong (multiple randomized controlled trials), Moderate (smaller trials or consistent observational data), or Emerging (early-stage research with promising signals).
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is Thymosin Alpha-1?
Your thymus gland sits behind your breastbone, right above your heart. Think of it as a training camp for immune cells. During childhood, the thymus takes immature immune cells and turns them into T-cells: the soldiers that fight viruses, bacteria, and cancer cells.
The problem: your thymus starts shrinking after puberty. By age 50, it has lost most of its functional tissue. By 70, it produces very little. This process, called thymic involution, is a major reason older adults get sick more often and respond poorly to vaccines.
Thymosin Alpha-1 is one of the key peptides your thymus produces. Supplementing with a synthetic version replaces what your body can no longer make in adequate quantities. The synthetic form is identical to the natural peptide, and it has been manufactured and studied since the 1970s.
Ta1 is not the same as TB-500. Both come from the thymus, but they do completely different things. Ta1 (Thymosin Alpha-1) trains and activates immune cells. TB-500 (Thymosin Beta-4) repairs damaged tissue. They can be stacked together, but they serve different purposes. For details on TB-500, see the TB-500 profile page.
How Thymosin Alpha-1 Works
Ta1 operates on multiple levels of the immune system simultaneously. Here is a simplified breakdown of the key mechanisms.
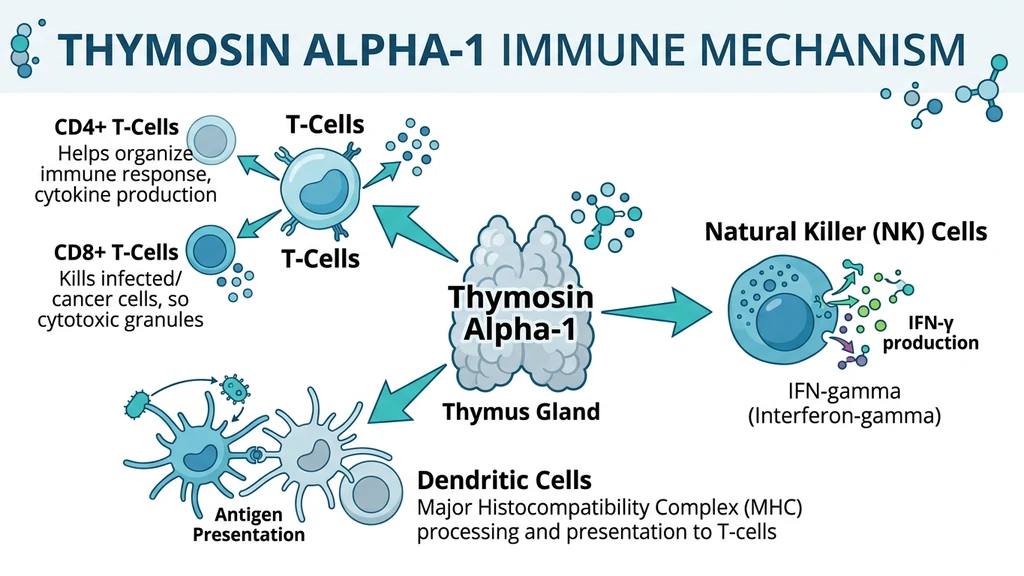
T-Cell Training and Activation
Ta1 pushes immature immune cells to mature into functional T-cells. Specifically, it increases the production of CD4+ helper T-cells (which coordinate immune responses) and CD8+ killer T-cells (which destroy infected or cancerous cells). It also boosts interleukin-2 (IL-2) receptors on T-cells, making them more responsive to activation signals.
In simple terms: Ta1 turns recruits into trained soldiers and gives them better radios to hear commands.
Natural Killer Cell Boost
Natural killer (NK) cells are your immune system's first responders. They patrol the body and destroy virus-infected cells and early tumor cells without needing prior instructions. Ta1 increases NK cell activity and their production of interferon-gamma, a signaling molecule that amplifies the immune alarm.
Dendritic Cell Activation
Dendritic cells act as scouts. They capture foreign invaders, break them apart, and present the pieces to T-cells so they know what to attack. Ta1 makes dendritic cells better at this job by enhancing antigen presentation through both MHC class I and class II pathways. Better scouting means faster, more accurate immune responses.
Immune Balancing, Not Just Boosting
This is a critical distinction. Ta1 does not simply crank up immune activity across the board. It shifts the immune system toward a Th1-dominant response (cellular immunity: fighting infections and cancer) while modulating Th2 responses (which, when overactive, drive allergies and certain autoimmune conditions).
This balancing effect is why Ta1 can be useful in both immunodeficiency (where the immune system is too weak) and certain autoimmune contexts (where the immune system is misdirected). It restores proper function rather than amplifying everything indiscriminately.
8 Evidence-Backed Benefits of Thymosin Alpha-1
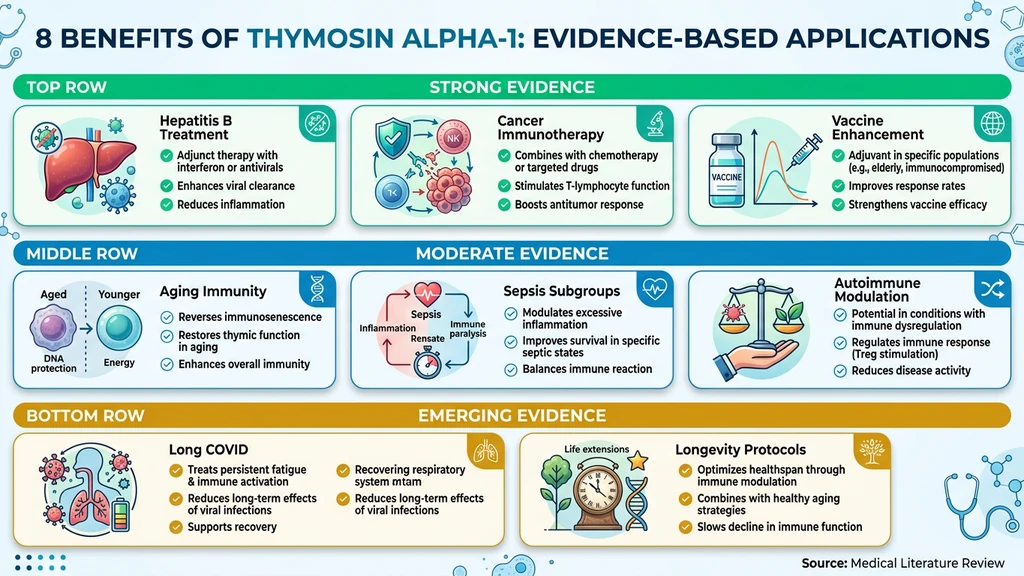
Each benefit below includes an evidence grade and the clinical data supporting it.
1. Chronic Hepatitis B Treatment (Strong)
This is Ta1's most established clinical application. A meta-analysis of 8 randomized controlled trials found that Ta1 monotherapy produces sustained viral clearance rates of 25-35% in chronic hepatitis B patients. When combined with interferon-alpha, response rates improve significantly over interferon alone. (Source: PubMed 18042265)
Ta1 is approved for hepatitis B treatment in more than 30 countries. The standard Zadaxin protocol (1.6 mg twice weekly) was developed from these trials.
2. Cancer Immunotherapy Support (Strong)
Ta1 is used alongside chemotherapy, radiation, and immunotherapy for several cancer types, including liver cancer (hepatocellular carcinoma), melanoma, and lung cancer. It reduces the immune suppression caused by chemotherapy and improves treatment response rates.
A 2024 study combining Ta1 with lenvatinib and the PD-1 inhibitor sintilimab in advanced liver cancer showed improved response rates and progression-free survival compared to the drug combination alone. (Source: Nature Scientific Reports)
For cancer patients, Ta1 helps maintain T-cell and NK cell counts during treatment, periods when the immune system is at its weakest.
3. Vaccine Response Enhancement (Strong)
Clinical studies show Ta1 significantly improves vaccine effectiveness in elderly and immunocompromised patients. The numbers are striking:
- Influenza vaccine: In elderly nursing home residents, Ta1 reduced influenza incidence from 19% to 6% and boosted antibody seroconversion rates. (Source: PubMed 17600281)
- Hepatitis B vaccine: Response rates jumped from 17% (placebo) to 64% (Ta1 group) in hemodialysis patients who previously failed standard vaccination.
- COVID-19 vaccine: A 2023 study found that higher plasma Ta1 levels correlated with stronger and more durable antibody responses to COVID-19 vaccination. (Source: Immunity & Ageing)
For anyone over 60 or with a weakened immune system, Ta1 before and during vaccination can make a measurable difference in antibody production.
4. Age-Related Immune Decline (Moderate)
Immunosenescence, the gradual weakening of the immune system with age, is one of the primary drivers of increased infection rates and cancer in older adults. Ta1 directly addresses this by restoring T-cell function to more youthful levels.
A 2025 review in the International Journal of Molecular Sciences confirmed Ta1's role in counteracting immunosenescence through multiple mechanisms: restoring thymic output, enhancing T-cell diversity, and reducing chronic low-grade inflammation ("inflammaging"). (Source: MDPI 2025)
This benefit is relevant for anyone over 50 experiencing more frequent colds, slower recovery from illness, or poor vaccine responses.
5. Sepsis Survival in Specific Populations (Moderate)
Sepsis is a life-threatening immune overreaction to infection. Ta1's role here is nuanced and requires honest reporting.
The TESTS Phase 3 trial (January 2025, 1,106 patients) tested Ta1 in general sepsis and found no significant survival benefit in the overall population. (Source: PubMed 39814420) This was a setback for Ta1's broadest clinical application.
However, subgroup analyses from earlier studies consistently show benefit in sepsis patients with severe lymphopenia (very low T-cell counts). In these patients, Ta1 restores immune function at a critical moment. The takeaway: Ta1 helps sepsis patients whose immune systems are failing, but does not provide a general benefit across all sepsis cases.
6. Autoimmune Disease Modulation (Moderate)
Because Ta1 balances rather than broadly stimulates the immune system, it has shown potential in certain autoimmune conditions. A 2016 study found altered serum Ta1 levels in patients with autoimmune diseases including Hashimoto's thyroiditis and lupus, suggesting Ta1 plays a regulatory role. (Source: PMC 5011367)
Important caveat: anyone with an active autoimmune condition should only use Ta1 under medical supervision. While the Th1/Th2 rebalancing effect is theoretically beneficial, enhancing T-cell function during an active flare could worsen symptoms. Work with a physician who understands peptide therapy.
7. Post-Viral Syndrome and Long COVID (Emerging)
A 2023 ex vivo study found that Ta1 restored T-cell function in blood samples from patients with post-acute sequelae of SARS-CoV-2 (long COVID). The peptide normalized T-cell exhaustion markers and improved cytokine production in cells that had become dysfunctional after COVID infection. (Source: PMC 10030336)
This is still early-stage research (laboratory, not clinical trials), but it provides a mechanistic basis for why some clinicians are using Ta1 in long COVID protocols. If you are considering Ta1 for post-viral symptoms, discuss it with your doctor.
8. Longevity and Anti-Aging Protocols (Emerging)
The connection between immune function and aging is well established. Chronic low-grade inflammation ("inflammaging") accelerates tissue damage, increases cancer risk, and reduces quality of life. By maintaining a functional immune system, Ta1 may slow these processes.
Ta1 is commonly paired with Epitalon in longevity protocols. Epitalon activates telomerase (the enzyme that maintains chromosome caps), while Ta1 restores immune surveillance. Together, they address two pillars of biological aging. Use the Peptide Stack Calculator to explore combination options.
This remains an emerging application. No clinical trials have directly measured Ta1's effect on lifespan or biological age markers, but the immunological rationale is sound.
Thymosin Alpha-1 vs Other Immune Peptides
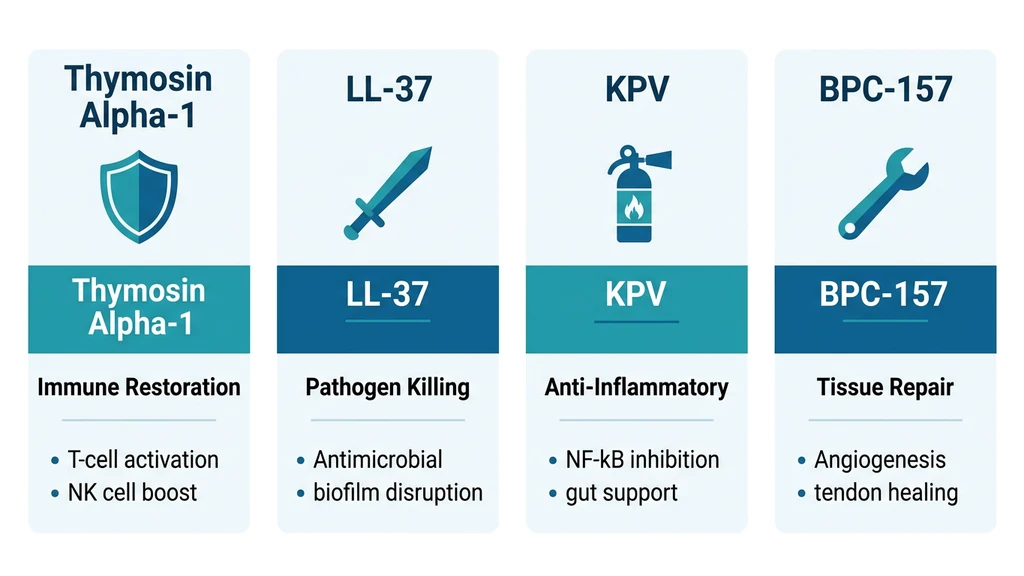
How does Ta1 compare to other immune-focused peptides? Here is a side-by-side comparison.
| Feature | Ta1 | LL-37 | KPV | BPC-157 |
|---|---|---|---|---|
| Primary action | T-cell activation, immune restoration | Antimicrobial, kills pathogens directly | Anti-inflammatory (gut/skin) | Tissue repair, gut healing |
| Immune mechanism | Adaptive immunity (T-cells, NK cells) | Innate immunity (direct pathogen killing) | Reduces inflammatory cytokines | Indirect: heals gut barrier |
| Clinical trials | 11,000+ patients | Limited human data | Limited human data | Limited human data |
| Approved anywhere? | Yes, 30+ countries (Zadaxin) | No | No | No |
| Best for | Chronic infections, cancer support, aging immunity | Acute infections, wound healing | IBD, skin inflammation | Injury repair, gut healing |
| Admin route | Subcutaneous injection | Injection or topical | Injection, oral, or topical | Injection or oral |
| Dosage frequency | Twice weekly | Daily | Daily | Daily |
When to choose Ta1: You need systemic immune restoration, not just local anti-inflammatory or antimicrobial effects. Ta1 is the peptide with the strongest clinical evidence for immune system support. For gut-specific immune issues, consider pairing Ta1 with KPV or BPC-157. Read our peptides for gut health guide for more on digestive applications.
Dosage Protocols
Three protocols cover most use cases. All use subcutaneous injection. See the Thymosin Alpha-1 profile page for complete dosage details and the reconstitution calculator for preparation.
Standard Immune Support - Dose: 1.6 mg - Frequency: Twice per week (e.g., Monday and Thursday) - Duration: 8-12 weeks, then 4 weeks off - Who it's for: General immune support, age-related decline, infection prevention
Acute Immune Protocol - Dose: 1.6 mg - Frequency: Daily for 5-7 days, then twice weekly - Duration: 1 week loading, then 4-8 weeks maintenance - Who it's for: Active infections, post-chemotherapy recovery, acute immune challenges
Longevity and Prevention - Dose: 1.6 mg - Frequency: Once or twice weekly - Duration: 8-12 weeks, cycled 2-3 times per year - Who it's for: Healthy adults over 50 looking to maintain immune function
The dose of 1.6 mg comes directly from the Zadaxin prescribing protocol and the majority of clinical trials. There is little reason to deviate from it. Higher doses have not shown proportionally better results. Store reconstituted Ta1 in the refrigerator and use within 2-3 weeks. For storage best practices, see our how to store peptides guide.
Side Effects and Safety
Ta1 has one of the strongest safety profiles of any peptide in research use. Data from over 11,000 patients across clinical trials shows a consistent pattern.
Common (mild, usually temporary): - Injection site redness or soreness (most frequently reported) - Mild fatigue in the first few days - Low-grade flu-like symptoms as the immune system activates (resolves within 48 hours)
Uncommon: - Low-grade fever - Muscle aches - Headache
Serious adverse events: Rare across all clinical trials. The safety data from decades of Zadaxin use in 30+ countries supports this. (Source: PubMed 38308608)
Who should NOT use Ta1 without medical supervision: - Organ transplant recipients on immunosuppressants (boosting immunity could trigger rejection) - People with active autoimmune flares - Pregnant or breastfeeding women (insufficient safety data)
For a broader overview of what to expect when starting peptides, see our getting started with peptides guide.
Regulatory Status: Where Things Stand in 2025-2026
Ta1's regulatory history is unusual. It is approved in over 30 countries but not in the United States.
Zadaxin (brand name Ta1) has been prescribed in China, South America, and parts of Asia since the 1990s for hepatitis B/C and cancer immune support. It has orphan drug designation in the US for hepatocellular carcinoma.
December 2024: The FDA's Pharmacy Compounding Advisory Committee (PCAC) reviewed Ta1 as part of a broader evaluation of compounded peptides. The committee did not recommend Ta1 for the FDA's "demonstrably difficult to compound" list, which would have protected compounding pharmacies' ability to produce it. (Source: FDA PCAC Meeting)
Current access: Ta1 remains available through compounding pharmacies, research peptide suppliers, and in countries where Zadaxin is approved. The regulatory landscape is evolving, and availability may change. For guidance on navigating peptide access, see our retatrutide access guide (the regulatory principles apply broadly).
Who Should Consider Thymosin Alpha-1?
Ta1 is not for everyone. Here is a framework to determine whether it fits your situation.
Strong candidates: - Adults over 50 with noticeably declining immune function (frequent colds, slow recovery, poor vaccine responses) - People undergoing chemotherapy or radiation who want to maintain immune function during treatment (with oncologist approval) - Chronic hepatitis B or C patients (clinical evidence is strongest here) - Anyone preparing for vaccination who has historically poor immune responses
Moderate candidates: - Adults 40-50 interested in proactive immune maintenance - People with recurrent infections who have not responded to standard treatments - Longevity-focused individuals building a comprehensive anti-aging protocol alongside Epitalon
Not recommended without medical guidance: - Active autoimmune disease - Organ transplant recipients - Anyone under 30 with a healthy immune system (your thymus is still functional)
Use the Peptide Interaction Checker to verify Ta1 is compatible with any other peptides or supplements you are taking. For broader peptide selection guidance, see the complete peptide dosage chart.
Frequently Asked Questions
How long does it take for Thymosin Alpha-1 to work?
Blood markers of immune function (T-cell counts, NK cell activity) typically show measurable changes within 2-4 weeks of starting the standard twice-weekly protocol. Subjective improvements like fewer colds and faster recovery usually take 4-8 weeks. The acute loading protocol (daily for one week) can produce faster initial results.
Can I take Thymosin Alpha-1 if I have an autoimmune disease?
Only under medical supervision. Ta1 modulates rather than broadly stimulates the immune system, and it can help rebalance Th1/Th2 responses. However, enhancing T-cell function during an active autoimmune flare could worsen symptoms. Work with a physician who has experience with peptide therapy to assess your specific condition. Hashimoto's thyroiditis and lupus patients have shown altered Ta1 levels, suggesting a regulatory role.
What is the difference between Thymosin Alpha-1 and TB-500?
They come from the same organ (thymus) but do completely different things. Thymosin Alpha-1 is a 28-amino-acid immune peptide that activates T-cells and NK cells. TB-500 (Thymosin Beta-4) is a 43-amino-acid repair peptide that heals damaged tissue, reduces inflammation, and promotes wound healing. Ta1 strengthens your immune system; TB-500 repairs your body. They can be stacked safely for combined immune and healing support.
Is Thymosin Alpha-1 FDA approved?
Not in the United States, but it is approved in over 30 countries under the brand name Zadaxin. It has FDA orphan drug designation for hepatocellular carcinoma and has been used in compassionate use settings in the US. The clinical trial data is extensive, with over 11,000 patients studied. Current US access is through compounding pharmacies and research peptide suppliers.
Does Thymosin Alpha-1 help with bodybuilding?
Ta1 is not a performance peptide. It does not directly build muscle, burn fat, or increase growth hormone. Its value for athletes and bodybuilders is indirect: a stronger immune system means fewer training days lost to illness, better recovery from the immune stress of intense training, and improved vaccine responses. If your primary goal is muscle growth, peptides like Ipamorelin or CJC-1295 are more relevant. Ta1 is best viewed as an immune insurance policy.
What happens if I take too much Thymosin Alpha-1?
Clinical trials have not identified serious toxicity at standard doses. The 1.6 mg twice-weekly dose has been used safely in thousands of patients over decades. Taking more than the recommended dose does not produce proportionally better results and may increase the likelihood of flu-like side effects (fatigue, low-grade fever, muscle aches). Stick to 1.6 mg per injection.
The Bottom Line
Thymosin Alpha-1 occupies a unique position among peptides. It has real clinical evidence, real regulatory approvals, and real safety data from thousands of patients. For immune support, it is the most thoroughly studied option available.
Whether you are managing a chronic infection, supporting your immune system during cancer treatment, counteracting age-related immune decline, or building a longevity protocol, Ta1 delivers measurable immunological improvements at a well-established dose.
Explore the full Thymosin Alpha-1 profile on PeptidesExplorer for detailed pharmacology, stacking protocols, and the complete reference list. Use our Peptide Stack Calculator to build a protocol that fits your goals. For all dosage references, see the peptide dosage chart. For reconstitution, see how to reconstitute peptides and how long reconstituted peptides last. For safe handling, see our peptide safety guide and where to buy peptides in 2026. For the regulatory landscape, see FDA peptide crackdown 2026. For stacking strategies, see the peptide stacking guide.
Related Articles: - LL-37 Peptide Benefits - KPV Peptide Dosage - Peptides for Gut Health - Peptide Stacking Guide - What Does TB-500 Do?
Related Articles
Best Peptides for Men Over 40: Ranked Guide (2026)
Top 10 peptides for men over 40: CJC-1295, BPC-157, PT-141, Tesamorelin ranked by evidence. Stacking protocols, dosing, and safety monitoring guide.
Best Peptides for Men Over 50: Protocols
Top 8 peptides for men over 50 ranked by safety. Sermorelin, BPC-157, Tesamorelin protocols with adjusted dosing and blood work requirements.
Best Peptides for Women Over 40: Guide
Top 8 peptides for women over 40: GHK-Cu, collagen peptides, BPC-157, PT-141. Perimenopause vs menopause protocols, dosing, and safety guide.
How Much Sermorelin Per Day: Complete Dosage Guide
Sermorelin dosage: 200-500 mcg/day subcutaneous before bedtime. Starting dose, titration, gender-specific protocols, and IGF-1 monitoring.