You are holding a vial of KPV lyophilized powder and weighing whether to take it orally for your gut symptoms or inject it for systemic inflammation. KPV is dosed at 200 to 500 mcg per day. Take it orally for gut inflammation, IBD, or colitis. Inject subcutaneously for systemic anti-inflammatory effects, joints, or skin conditions covering large areas. Most protocols run 4 to 8 weeks.
KPV is a tripeptide fragment of alpha-melanocyte-stimulating hormone (alpha-MSH, positions 11 through 13). It retains the anti-inflammatory activity of the full hormone without causing skin pigmentation. Unlike Melanotan II, KPV does not bind MC1R, MC3R, or MC5R. No tanning effect at any dose.
All dosage data below comes from animal studies and clinical observation. No human randomized controlled trials exist for KPV. Every claim links to its source. Consult a qualified healthcare provider before acting on any information.
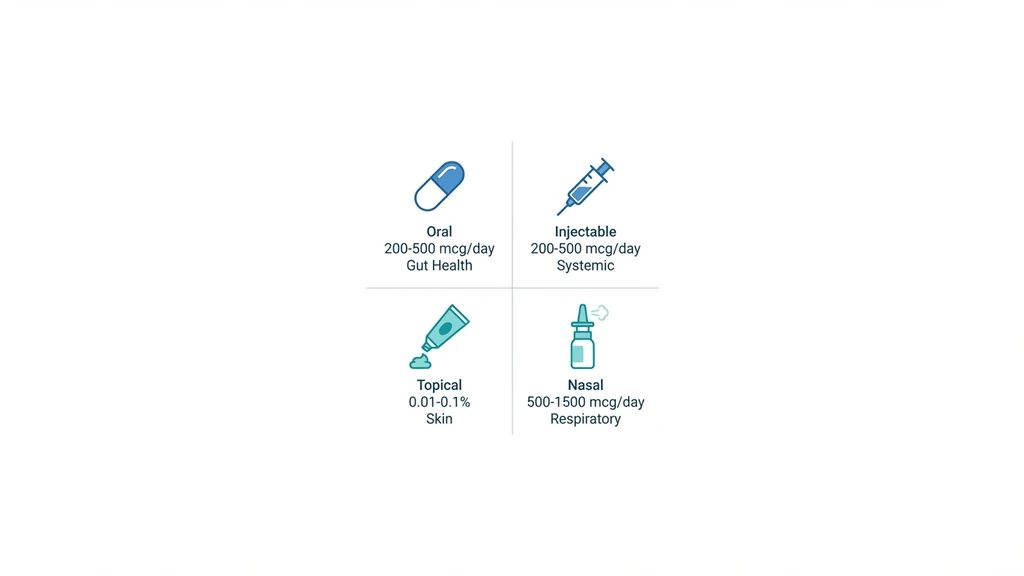
Quick reference:
| Route | Typical Dose | Best For | Onset |
|---|---|---|---|
| Oral | 200-500 mcg/day | Gut inflammation, IBD, colitis | 2-4 weeks |
| Subcutaneous | 200-500 mcg/day | Systemic inflammation, joints | 1-2 weeks |
| Topical | 0.01-0.1% cream | Skin inflammation, wounds, eczema | Variable |
| Nasal spray | 500-1,500 mcg/day | Sinus inflammation, respiratory | Days |
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is KPV and How Does It Work?
KPV is a three-amino-acid peptide (lysine-proline-valine) with a molecular weight of roughly 341 Da. It was first isolated from alpha-MSH, where it occupies positions 11 through 13, the minimum fragment required for anti-inflammatory signaling.
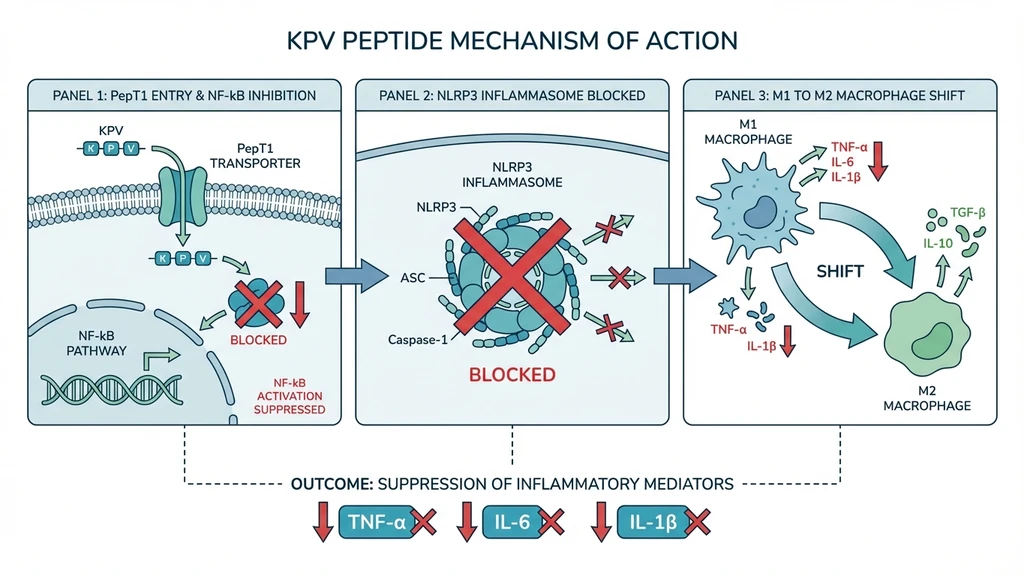
The peptide works through two primary pathways. First, it enters cells and blocks the nuclear translocation of NF-kB p65 by competing with importin-alpha-3 (Lam et al. 2012, PMC3403564). This shuts down transcription of TNF-alpha, IL-1beta, IL-6, IL-8, and other pro-inflammatory cytokines.
Second, KPV inhibits the NLRP3 inflammasome, breaking the self-perpetuating inflammatory cycle in conditions like colitis (PubMed 28426964).
KPV also shifts macrophage polarization from M1 (pro-inflammatory, destructive) to M2 (reparative) without suppressing overall immune function. The immune system stays capable of fighting infections. The overreaction calms down.
Why Does the Administration Route Matter?
Route selection changes everything about how KPV reaches its target. In the gut, KPV enters epithelial cells through PepT1 transporters with a Km of roughly 160 uM, among the highest affinities reported for this transporter (Dalmasso et al. 2008, PMC2431115).
During intestinal inflammation, PepT1 expression increases in the colon. The sicker the tissue, the more KPV it absorbs. This self-targeting mechanism makes oral delivery uniquely effective for gut conditions.
Subcutaneous injection bypasses the GI tract entirely, delivering KPV to the bloodstream for systemic distribution. This is the better choice for joint inflammation, whole-body inflammatory conditions, or any target outside the digestive system.
Topical application faces a skin barrier problem. KPV does not penetrate intact skin well by passive diffusion. Microneedle treatment or iontophoresis can increase permeation up to 35-fold (Kumar et al. 2017, J Pharm Sci), but standard topical creams deliver limited concentrations.
How Should You Dose Oral KPV? (Best for Gut Health)
Standard protocol: 200 to 500 mcg per day, taken once daily on an empty stomach.
Start at 200 mcg daily for the first week. Increase to 500 mcg if well tolerated. Take the dose 30 minutes before food to reduce competition with dietary peptides for PepT1 binding.
Dalmasso et al. demonstrated that oral KPV at 100 uM in drinking water reduced DSS-induced colitis severity by roughly 50% (measured by myeloperoxidase activity) in mice. The peptide also reduced IL-6, IL-12, IFN-gamma, and IL-1beta mRNA levels in colonic tissue (PMC2431115).
Duration: 4 to 8 weeks for active gut inflammation. Most protocols report improvement within 2 to 4 weeks. Reduced bloating, less abdominal pain, and improved stool consistency are the earliest signs.
For gut-specific conditions, oral delivery may outperform injection. Oral KPV places the peptide directly at the intestinal epithelium where inflammation occurs. The PepT1 upregulation during inflammation creates a self-targeting delivery system that injection cannot replicate.
Advanced context: Researchers are developing hyaluronic acid-functionalized nanoparticle delivery (HA-KPV-NP) that dramatically improves oral efficacy. Xiao et al. showed HA-KPV-NP at just 16 ug/kg/day protected mice from colitis, with tissue morphologically similar to healthy controls (PMC5498804). This represents the next generation of oral KPV delivery, though it remains in preclinical stages.
What Is the Injectable KPV Dosage? (Subcutaneous Protocol)
Standard protocol: 200 to 500 mcg per day, subcutaneous injection.
Choose injectable KPV when the target is outside the gut. Systemic inflammatory conditions, joint inflammation, and whole-body inflammation reduction all benefit from the higher bioavailability of subcutaneous delivery, which bypasses first-pass gut metabolism.
Reconstitution: Add bacteriostatic water to the lyophilized KPV vial. For a 10 mg vial, add 2 mL bacteriostatic water to yield 5,000 mcg/mL.
For a 500 mcg dose, draw 10 units (0.1 mL) on a U-100 insulin syringe. Use our Peptide Reconstitution Calculator for exact calculations.
Injection sites: Abdominal fat (two inches from the navel), outer thigh, or back of the upper arm. Rotate sites daily to prevent irritation.
Storage: Reconstituted KPV at 2 to 8 degrees Celsius (refrigerator). Use within 4 weeks. Unreconstituted vials remain stable at room temperature or refrigerated for 12 or more months.
Duration: 4 to 6 weeks for systemic anti-inflammatory effects. If you are new to peptide injections, see our getting started guide for step-by-step instructions.
What Is the Topical KPV Dosage? (Skin Protocol)
Standard protocol: 0.01 to 0.1% KPV cream or serum, applied twice daily to affected areas.
Topical KPV targets localized skin inflammation: eczema, rosacea, post-procedure redness, and wound healing. Apply a thin layer to clean, dry skin.
The bioavailability challenge is real. Kumar et al. found that KPV does not penetrate intact skin well by passive diffusion alone.
Microneedle treatment increased permeation to 4.4 ug/cm2/h. Combined iontophoresis with microneedles increased it 35-fold compared to passive diffusion (J Pharm Sci 2017). Standard topical creams deliver limited concentrations.
For widespread skin conditions (psoriasis, systemic dermatitis), subcutaneous injection at 200 to 500 mcg/day may be more effective than topical alone. A 2025 study showed KPV at 50 ug/mL restored keratinocyte viability and reduced IL-1beta production after fine dust exposure, with effects mediated through MAPK and NF-kB inhibition.
Compounded KPV creams from pharmacies are the most reliable option. DIY formulations risk inconsistent concentration.
Duration: 4 to 8 weeks.
What About Nasal KPV? (Spray Protocol)
Protocol: 500 to 1,500 mcg daily across multiple administrations (one spray of 50 mcg per nostril, up to 4 times daily).
Nasal delivery provides rapid absorption through the nasal mucosa, bypasses the GI tract, and requires no needles. It is particularly relevant for sinus inflammation and upper respiratory inflammation.
Evidence level: low. No published studies specifically examine nasal KPV delivery. Protocols are extrapolated from general peptide nasal delivery pharmacokinetics. Nasal administration is the least-studied route for KPV.
Use this route only when oral or injectable options are not suitable. For gut targets, oral is better. For systemic targets, injectable has more predictable dosing.
What Is the Right KPV Dosage for Specific Conditions?
The right KPV protocol depends on the condition being targeted and the tissue involved.
KPV for Gut Health and IBD
Route: Oral (PepT1 targeting). Dose: 200 to 500 mcg/day, empty stomach, 4 to 8 weeks.
Dalmasso et al. showed oral KPV reduced both DSS- and TNBS-induced colitis severity in mice. The peptide reduced body weight loss, colonic MPO activity by roughly 50%, and decreased IL-6, IL-12, IFN-gamma, and IL-1beta mRNA levels (PMC2431115).
Stacking: Combine with BPC-157 at 250 to 500 mcg/day for synergistic gut healing. KPV controls inflammation. BPC-157 drives tissue repair and angiogenesis. This is the most widely recommended gut healing stack in the peptide community.
Cancer prevention data (unique): Viennois et al. demonstrated that KPV reduced colitis-associated tumorigenesis in mice through a PepT1-dependent mechanism. KPV-treated mice developed fewer tumors, smaller tumor sizes, and lower overall tumor burden. The effect was abolished in PepT1-knockout mice, confirming the transport pathway (PMC4957955).
KPV for Skin Inflammation and Wound Healing
Route: Topical (localized) or subcutaneous (systemic skin conditions). Topical dose: 0.01 to 0.1% cream twice daily. Injectable dose: 200 to 500 mcg/day subcutaneous for widespread conditions.
KPV inhibits inflammatory responses in keratinocytes. In diabetic mouse wound models, KPV improved full-thickness wound repair rate through inflammatory inhibition, angiogenesis, and collagen deposition.
Antimicrobial bonus: KPV has direct antimicrobial activity against Staphylococcus aureus and Candida albicans at picomolar concentrations (Cutuli et al. 2000, PubMed 10670585). For infected wounds, this provides dual anti-inflammatory and antimicrobial action in a single peptide.
KPV for Joint Inflammation and Arthritis
Route: Subcutaneous injection (systemic anti-inflammatory). Dose: 200 to 500 mcg/day, 4 to 8 weeks.
Getting et al. administered alpha-MSH fragments including KPV to rats with adjuvant-induced arthritis. Treatment significantly attenuated both clinical and histological signs of arthritis. Effects were comparable to prednisolone, without the weight gain, metabolic disruption, or bone density loss associated with corticosteroids (PMC2095288).
Stack option: Combine with TB-500 at 2 to 2.5 mg twice per week for whole-body inflammatory conditions where multiple tissues need repair. See our complete guide to peptides for joint pain for detailed stacking protocols.
How Does KPV Compare to BPC-157?
KPV and BPC-157 work through different mechanisms and serve different roles in a healing protocol. Neither replaces the other. They are complementary.
| Factor | KPV | BPC-157 |
|---|---|---|
| Primary action | Anti-inflammatory (NF-kB inhibition) | Tissue repair (angiogenesis, growth factors) |
| Mechanism | Calms the inflammatory environment | Rebuilds damaged tissue |
| Best for | Active inflammation, immune overreaction | Tissue damage, wound healing, recovery |
| Oral route | Via PepT1 transporters | Gastric acid stable, direct absorption |
| Gut target | Reduces inflammatory signaling in epithelial cells | Drives mucosal repair, blood vessel formation |
| Onset (gut) | 2-4 weeks | 1-3 weeks |
| Typical dose | 200-500 mcg/day | 250-500 mcg/day |
| Unique property | Antimicrobial activity at picomolar concentrations | VEGF upregulation for tissue rebuilding |
| Used together? | Yes: gold standard gut healing stack | Yes: KPV calms, BPC-157 rebuilds |
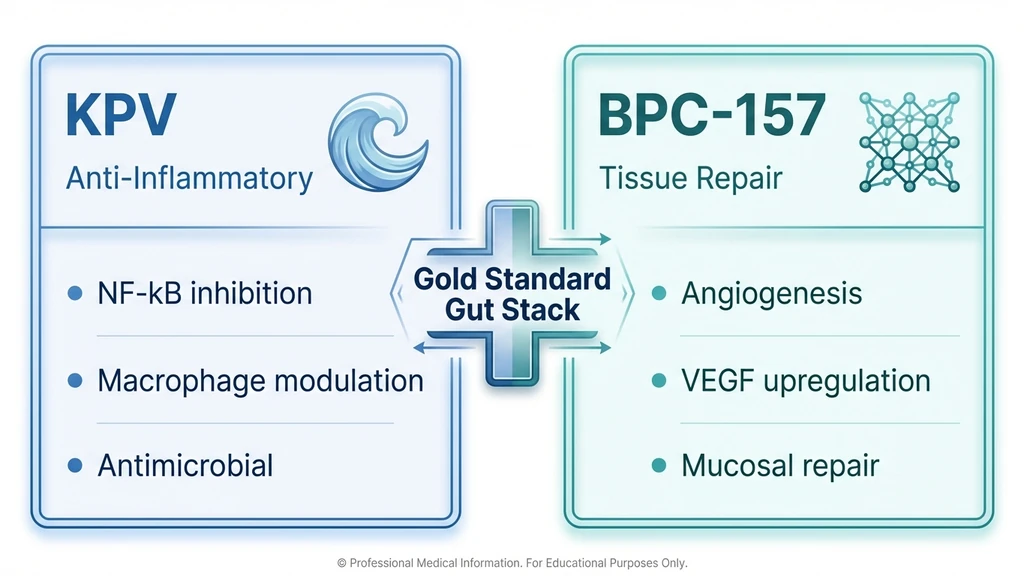
Combined protocol: KPV 200 to 500 mcg/day oral plus BPC-157 250 to 500 mcg/day oral or injected. Run 4 to 8 weeks. KPV acts as the fire extinguisher. BPC-157 acts as the construction crew. For active inflammation, start both simultaneously. For tissue damage after inflammation is controlled, BPC-157 alone may suffice.
For a deeper look at BPC-157 protocols, including interactions with alcohol, see our BPC-157 and alcohol guide. Use our Peptide Reconstitution Calculator to dial in exact amounts.
How Do You Prepare and Reconstitute KPV?
Equipment needed: KPV vial (typically 5 mg or 10 mg), bacteriostatic water, U-100 insulin syringes (29 to 31 gauge), alcohol swabs, sharps container.
Reconstitution math: 10 mg vial plus 2 mL bacteriostatic water equals 5,000 mcg/mL. For a 250 mcg dose, draw 5 units. For a 500 mcg dose, draw 10 units.
Step-by-Step Reconstitution
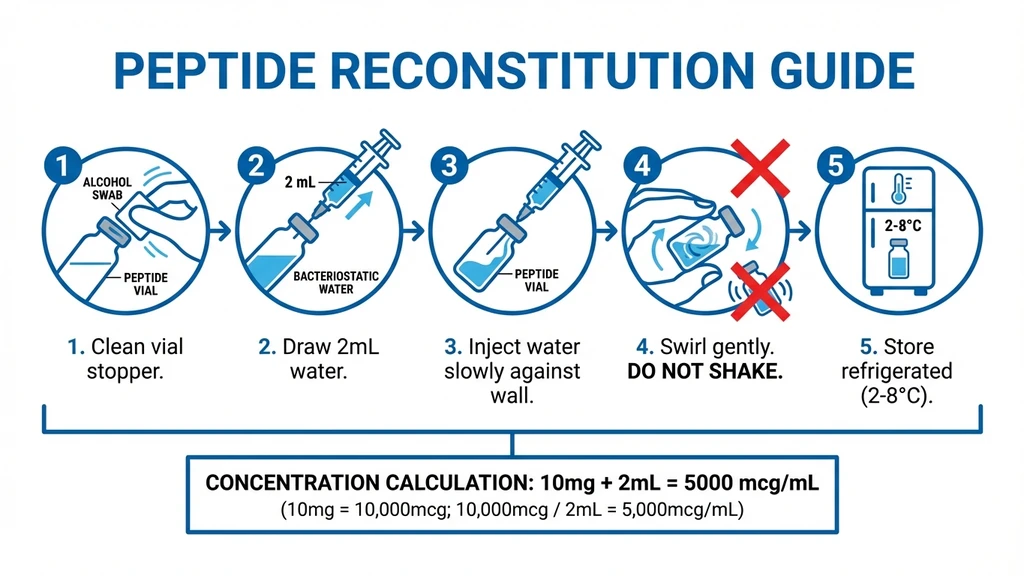
- 1.Clean the tops of both the KPV vial and bacteriostatic water vial with alcohol swabs.
- 2.Draw 2 mL of bacteriostatic water into a syringe.
- 3.Inject the water slowly against the inside wall of the KPV vial. Do not spray directly onto the powder.
- 4.Gently swirl the vial until the powder dissolves completely. Never shake.
- 5.Refrigerate immediately after reconstitution.
Use our Peptide Reconstitution Calculator for exact calculations with any vial size.
Storage and Stability
Unreconstituted: Room temperature or refrigerated. Stable for 12 or more months when kept dry. Reconstituted: Refrigerate at 2 to 8 degrees Celsius. Use within 4 weeks. Do not freeze reconstituted solutions. Do not expose to direct sunlight or heat.
KPV is a small tripeptide, which makes it generally more stable than larger peptides. Proper storage still matters for maintaining potency.
How Long Should You Cycle KPV?
Standard cycle: 4 to 8 weeks on, followed by 2 to 4 weeks off.
No long-term human safety data exists. Cycling provides recovery periods and allows assessment of whether benefits persist after discontinuation.
For chronic gut conditions (ulcerative colitis, Crohn's), some protocols extend to 12 weeks before cycling off for 4 weeks. After an initial cycle, some users reduce to 200 mcg/day on 3 to 5 days per week as maintenance. This is anecdotal, not evidence-based.
Timeline of effects: Most users report improvement within 2 to 4 weeks for gut symptoms. Full mucosal healing takes 6 to 8 or more weeks. Stacking with BPC-157 often accelerates the timeline.
Is KPV Safe? Side Effects and Warnings
KPV has a favorable preclinical safety profile. The common and rare side effects reported in the peptide community are mild.
Common Side Effects
Mild injection site redness that resolves quickly. Minor GI discomfort when starting oral dosing, usually limited to the first 2 to 3 days. Slight fatigue during the initial days of use. Mild headache (uncommon).
What KPV Does Not Cause
No skin tanning. Unlike Melanotan I/II or full-length alpha-MSH, KPV does not bind melanocortin receptors (MC1R, MC3R, MC5R). Zero pigmentation effect at any dose.
No immunosuppression. KPV modulates the immune response (M1 to M2 macrophage shift) without impairing immune defense. It turns down overactive inflammation. It does not shut off the immune system.
Important Safety Limitations
No human clinical trials. All dosage data is extrapolated from animal studies and clinical observation. No FDA- or EMA-approved KPV product exists.
Not for active infections. Reducing inflammation during an acute infection may be counterproductive. Address infections before starting anti-inflammatory protocols.
Pregnancy and breastfeeding: Contraindicated. No safety data available.
Drug interactions: People on immunosuppressive medications should consult a doctor. Combining immune modulators without monitoring carries risk.
Quality control: As a research peptide, KPV sourcing quality varies dramatically. Third-party Certificate of Analysis (COA) verification is essential. Do not purchase without independent purity testing.
Frequently Asked Questions
What is the standard KPV peptide dosage?
The standard KPV dosage is 200 to 500 mcg per day, administered orally or by subcutaneous injection. Start at 200 mcg daily and increase to 500 mcg after the first week if well tolerated. Protocols run 4 to 8 weeks. Confirm syringe units with the peptide reconstitution calculator before drawing your first dose.
Is oral KPV effective or do I need to inject it?
Oral KPV is effective, especially for gut inflammation. KPV is absorbed through PepT1 transporters in the intestinal lining with high affinity (Km roughly 160 uM), giving it direct access to inflamed gut epithelial cells. For gut conditions, oral may be preferable to injection. For systemic targets, see the peptide injections complete guide.
How does KPV compare to BPC-157 for gut healing?
They work through different mechanisms and are best used together. KPV suppresses NF-kB signaling (anti-inflammatory). BPC-157 drives angiogenesis and tissue rebuilding (reparative). Most gut healing protocols combine KPV at 200 to 500 mcg/day with BPC-157 at 250 to 500 mcg/day for 4 to 8 weeks. See the full peptides for gut health breakdown.
Will KPV cause skin tanning like Melanotan?
No. KPV is derived from alpha-MSH but does not bind melanocortin receptors (MC1R, MC3R, MC5R). It preserves only the anti-inflammatory signaling fragment. No pigmentation change occurs at any dose. If tanning is the goal instead, see the Melanotan II profile, not KPV.
How long does KPV take to work for gut issues?
Most users report improvement within 2 to 4 weeks for active gut inflammation. Reduced bloating, less abdominal pain, and improved stool consistency are the earliest signs. Full mucosal healing takes 6 to 8 weeks or more. Stacking with BPC-157 often accelerates the timeline. The peptide stacking guide covers exact protocols.
Can I take KPV as a nasal spray?
KPV nasal spray protocols range from 500 to 1,500 mcg/day across multiple administrations. Nasal delivery provides rapid absorption and may help sinus inflammation. This is the least-studied route with no published research specific to nasal KPV. For comparison, see the better-studied BPC-157 nasal spray guide for nasal delivery principles.
Does KPV suppress the immune system?
No. KPV modulates the immune response rather than suppressing it. It shifts macrophages from M1 (pro-inflammatory) to M2 (reparative) phenotype and inhibits excessive NF-kB activation. The immune system retains its ability to fight infections. For broader immune support context, see peptides for immune system.
Is KPV safe to take with probiotics?
Yes. KPV and probiotics work through complementary pathways. KPV reduces inflammation and restores barrier integrity, creating a better environment for beneficial bacteria. Many gut protocols combine KPV with BPC-157 and a quality probiotic. The peptide interaction checker verifies any combination.
What supplies do I need for KPV injections?
You need a KPV lyophilized vial (5 mg or 10 mg), bacteriostatic water, U-100 insulin syringes (29 to 31 gauge), alcohol swabs, and a sharps container. Use the peptide reconstitution calculator for exact mixing ratios, and how to reconstitute peptides for step-by-step technique.
The Bottom Line
KPV is a tripeptide with strong preclinical evidence for anti-inflammatory activity across gut, skin, joint, and systemic applications. The dosage is consistent across routes: 200 to 500 mcg per day. The route selection is what matters.
Choose oral for gut inflammation. PepT1-mediated transport delivers KPV directly to inflamed intestinal tissue. Choose subcutaneous injection for systemic effects outside the GI tract. Consider topical only for localized skin conditions, and recognize its bioavailability limitations.
For gut healing, the KPV plus BPC-157 stack remains the most recommended combination. KPV controls the inflammatory environment. BPC-157 drives the repair. Used together for 4 to 8 weeks, they address both sides of the healing equation. For a complete ranking of all gut-healing peptides including LL-37 and collagen peptides, see Peptides for Gut Health. For LL-37's antimicrobial gut barrier protection data, see the LL-37 Benefits Guide.
Every dosage protocol in this guide derives from preclinical research. No human clinical trials exist. Work with a qualified healthcare provider to determine whether KPV is appropriate for your situation. For all dosage references, see the peptide dosage chart. For proper handling, see how to store peptides and how long reconstituted peptides last. For sourcing, see where to buy peptides in 2026. For reconstitution instructions, see how to reconstitute peptides. For a broader safety overview, see our peptide safety guide.
Related Articles: - Peptides for Gut Health - LL-37 Peptide Benefits - Peptide Stacking Guide - Peptide Dosage Chart - How to Store Peptides
Helpful Tools
Related Articles
BPC-157 Dosage Per Body Weight (Chart)
BPC-157 dosage by body weight: 1.6-10 mcg/kg. Full chart for 120-280 lbs, injection vs oral doses, and why most use flat 250-500 mcg.
GHK-Cu + BPC-157 + TB-500 Blend Dosage
GHK-Cu + BPC-157 + TB-500 blend dosage chart for 70 mg and 42 mg vials. Per-injection breakdown in mcg, reconstitution steps, and cycling protocol.
Peptides for Gut Health: 5 Best
BPC-157, KPV, LL-37, collagen peptides, and larazotide for gut healing. Condition-specific protocols for IBS, leaky gut, Crohn's, and bloating.
BPC-157 TB-500 Dosage: Combined Protocol
BPC-157 TB-500 dosage: 250-500 mcg BPC-157 + 2-2.5 mg TB-500 daily. Weight-adjusted charts, injury-type matrix, blend vial math, and cycle timing.