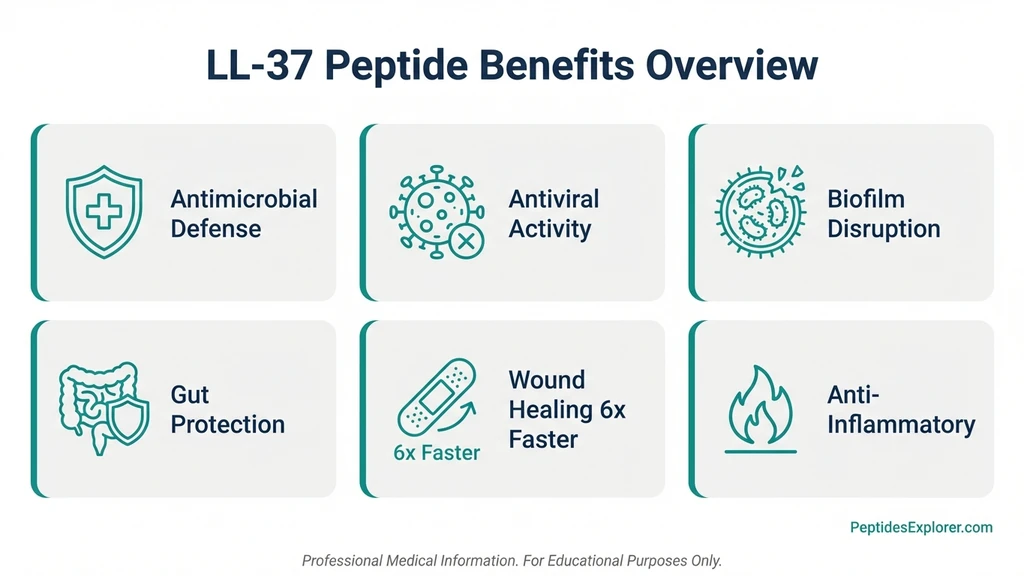
LL-37 is the only human cathelicidin antimicrobial peptide, a 37-amino-acid molecule produced by immune cells, skin, and mucosal surfaces. Research demonstrates benefits across eight domains: broad-spectrum antimicrobial activity, antiviral defense (including against SARS-CoV-2), wound healing acceleration, biofilm disruption, gut barrier protection, anti-inflammatory modulation, cancer immunotherapy potential, and cardiovascular protection.
Two randomized clinical trials confirm that topical LL-37 accelerates wound healing in venous leg ulcers. The lower dose produced healing rates roughly six times faster than placebo (Gronberg et al. 2014). Interest in LL-37 has surged 110% year-over-year as biohackers and clinicians explore its antimicrobial properties against antibiotic-resistant bacteria.
Every quantitative claim below links to PubMed. LL-37 is not FDA-approved for therapeutic use. Consult a healthcare provider before acting on any information.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
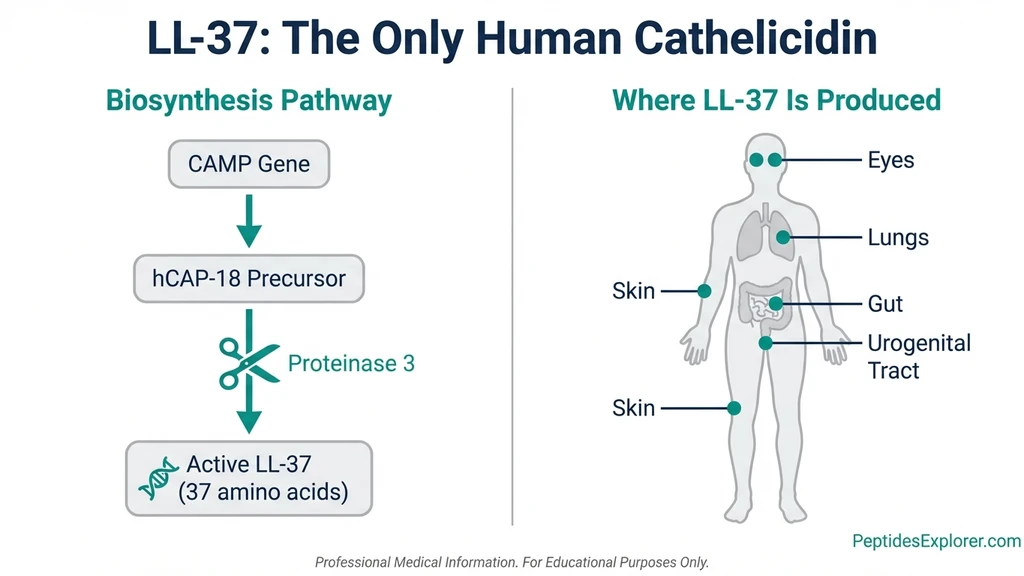
What Is LL-37? The Only Human Cathelicidin
LL-37 is cleaved from a precursor protein called hCAP-18 (human cationic antimicrobial peptide-18) by the enzyme proteinase 3. The name reflects its structure: it begins with two leucine residues and contains 37 amino acids total. It is the sole cathelicidin produced by the human body. Other mammals produce dozens.
The peptide adopts an alpha-helical shape that is amphipathic. One face carries a positive charge; the other is hydrophobic. This dual nature allows it to bind negatively charged bacterial membranes while inserting into the lipid bilayer. Human cell membranes, which are neutrally charged, are largely spared at therapeutic concentrations.
The CAMP gene on chromosome 3 encodes it. Neutrophils, macrophages, dendritic cells, natural killer cells, epithelial cells, and keratinocytes all produce LL-37. It appears in skin, airways, the gastrointestinal tract, the urogenital tract, and ocular surfaces: every tissue exposed to the outside world.
LL-37 is simultaneously an antimicrobial weapon and a signaling molecule. It recruits immune cells, modulates inflammation, promotes wound healing, and disrupts biofilms. It bridges innate and adaptive immunity (MDPI 2025).
How Does LL-37 Work? Mechanism of Action
LL-37 operates through three simultaneous pathways. These explain its broad range of benefits and why bacteria cannot easily develop resistance.
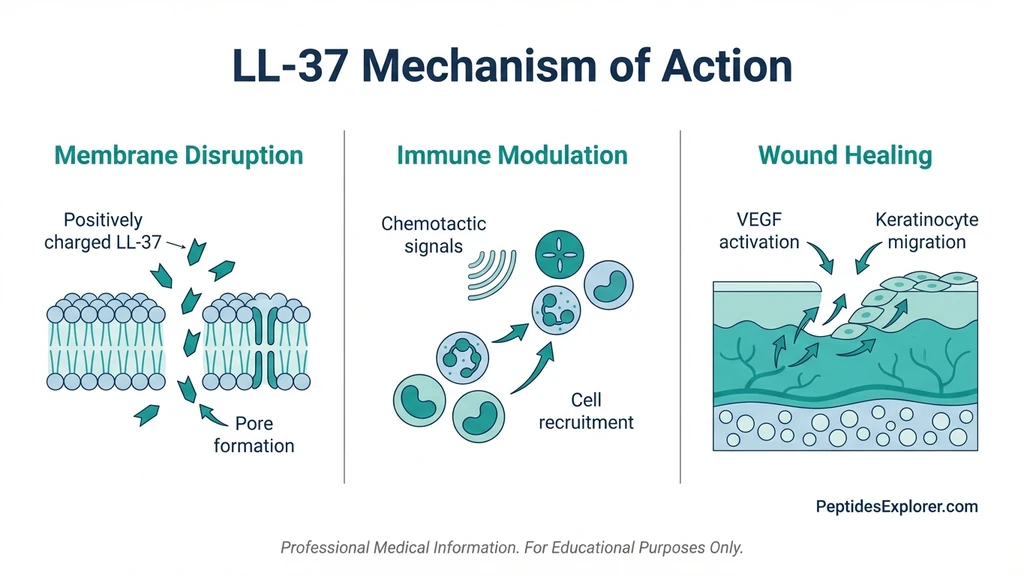
Membrane Disruption
The primary antimicrobial mechanism. LL-37 is attracted to negatively charged bacterial membranes through electrostatic interaction. Once bound, it oligomerizes into pore-forming structures that perforate the membrane, spilling the cell contents (Nature 2020).
LL-37 attacks multiple targets simultaneously: membranes, protein synthesis machinery, and genomic integrity. A bacterium would need to evolve defenses against all three attack vectors at once. Resistance development is extremely unlikely.
Immune Modulation
LL-37 recruits neutrophils, monocytes, and T cells to infection sites through chemotaxis (PubMed 12244186). In early infection, it amplifies the immune response. In later stages, it suppresses excessive production of TNF-alpha and IL-6, preventing collateral tissue damage (PubMed 20140902).
Pro-inflammatory when needed, anti-inflammatory when the threat subsides. This dual behavior distinguishes LL-37 from simple antimicrobial agents. It functions as an immune conductor.
Wound Healing Pathways
LL-37 stimulates angiogenesis through VEGF signaling, creating new blood vessels in damaged tissue. It promotes keratinocyte migration to close wounds and protects keratinocytes from apoptosis, keeping the repair cells alive longer (PubMed 21693141, PubMed 18923446).
These are not theoretical pathways. They are the mechanisms confirmed in two human clinical trials on venous leg ulcers.
What Are the 8 Evidence-Based Benefits of LL-37?
Each benefit below is graded by evidence strength. Two have human clinical trial data. The rest are supported by in vitro and animal studies.

1. Broad-Spectrum Antimicrobial Defense
LL-37 kills gram-positive bacteria (MRSA, Staphylococcus aureus, Streptococcus), gram-negative bacteria (E. coli, Pseudomonas aeruginosa, Salmonella), fungi (Candida albicans), and parasites. Activity against antibiotic-resistant strains is the primary reason it draws clinical interest (PubMed 9736536).
The mechanism is membrane disruption, not enzyme inhibition. Resistance develops far more slowly than with conventional antibiotics. Bacteria would need to fundamentally alter their membrane composition, which carries a steep fitness cost.
Evidence level: Strong. In vitro data confirmed across dozens of studies since 1998. Activity against MRSA and multidrug-resistant Pseudomonas verified independently by multiple research groups.
2. Antiviral Activity
LL-37 neutralizes multiple virus families through distinct mechanisms:
Influenza A: Dose-dependent neutralization via direct membrane disruption of the viral envelope. The mechanism is distinct from human defensins, which block viral attachment (PMC3542722).
SARS-CoV-2: Dual mechanism. LL-37 binds the spike protein receptor-binding domain with a dissociation constant (Kd) of 11.2 nM, blocking viral attachment. Simultaneously, it cloaks the ACE2 receptor (Kd = 25.5 nM), removing the docking site. Two barriers instead of one (PubMed 33849267).
RSV: Inhibits viral replication in polarized airway epithelial cells (PMC4700635).
HIV-1: Inhibits reverse transcriptase and protease.
Evidence level: Strong in vitro data. The SARS-CoV-2 binding affinities are precise and reproducible. No human antiviral trials completed.
3. Wound Healing Acceleration
This is the strongest evidence domain for LL-37. Two randomized controlled trials exist.
Gronberg et al. 2014 (n=34): Patients with venous leg ulcers received topical LL-37 at three concentrations (0.5, 1.6, and 3.2 mg/mL) or placebo. The 0.5 mg/mL group achieved healing rates roughly six times faster than placebo (p=0.003). The higher doses did not outperform the lower one. This inverted dose-response has practical dosing implications (PubMed 25041740).
Mahlapuu et al. 2021 (n=148): Phase IIb trial confirmed significant wound healing improvement, particularly in wounds measuring 10 cm2 or larger (PubMed 34687253).
The mechanism involves three simultaneous processes: angiogenesis (VEGF-driven new blood vessel formation), re-epithelialization (keratinocyte migration), and anti-apoptosis (keeping repair cells alive during the healing process).
Evidence level: HIGH. Two human RCTs with statistically significant results.
4. Biofilm Disruption
Biofilms are organized bacterial communities that resist both antibiotics and immune responses. LL-37 attacks biofilms through three mechanisms: preventing initial bacterial attachment to surfaces, disrupting quorum-sensing communication between bacteria, and working synergistically with polymyxin B to eradicate established biofilms of E. coli and Pseudomonas aeruginosa (ASM Journals, MDPI Antibiotics 2023).
This is directly relevant to chronic sinusitis, implant-associated infections, and chronic wound infections where biofilms prevent healing.
Evidence level: Moderate. Consistent in vitro data. No human biofilm-specific trials.
5. Gut Barrier Protection
Shih et al. (2023) demonstrated that LL-37 preserves intestinal goblet cells, increases mucin-2 expression, upregulates the antioxidant transcription factor Nrf2, and reduces cyclooxygenase-2 expression. The net result: less bacterial translocation across the gut lining and better intestinal homeostasis (PubMed 36958193).
A hybrid cecropin-LL37 peptide protected gut microbiota composition during EHEC (E. coli O157:H7) infection in animal models (Frontiers in Immunology 2020).
LL-37 is naturally expressed throughout the gastrointestinal tract. When gut LL-37 levels drop, often due to vitamin D deficiency, barrier function deteriorates.
Evidence level: Moderate. Animal and in vitro data with clear mechanistic pathways. No human gut-specific trials. For dedicated gut anti-inflammatory support, see our KPV peptide dosage guide. For a comprehensive ranking of all gut-healing peptides, see Peptides for Gut Health.
6. Anti-Inflammatory Effects
LL-37 neutralizes lipopolysaccharide (LPS), the bacterial endotoxin that triggers septic inflammatory cascades. It also decreases pro-inflammatory cytokine release from stimulated neutrophils and modulates inflammatory responses in keratinocytes challenged with TNF-alpha and IFN-gamma (PubMed 20140902, PubMed 23524263).
The anti-inflammatory action is context-dependent. LL-37 does not simply suppress inflammation. It amplifies it during active infection, then resolves it once the threat passes. This makes it fundamentally different from corticosteroids or NSAIDs, which suppress inflammation indiscriminately.
Evidence level: Moderate. Well-characterized mechanism across multiple in vitro and animal studies.
7. Cancer Immunotherapy Potential
A completed phase 1 clinical trial (NCT02225366) tested intra-tumoral LL-37 injections in melanoma patients. The peptide activates plasmacytoid dendritic cells, enhances CD8+ T cell proliferation, and directs immune-mediated tumor destruction (PubMed 19272013, PubMed 36559241).
Separately, LL-37 inhibits pancreatic cancer cell growth by suppressing autophagy, the process cancer cells use to survive nutrient deprivation (Frontiers in Pharmacology 2022).
Evidence level: Early clinical. Phase 1 trial completed. Mechanistic data from multiple cancer types. No phase 2/3 data published.
8. Cardiovascular Protection
LL-37 induces endothelium-dependent vasorelaxation in human blood vessels (PubMed 18397922). Observational data shows that higher circulating LL-37 levels predict lower ischemic recurrence risk after myocardial infarction. The peptide may also modulate lipid metabolism in atherosclerotic plaques (AHA Journals).
Evidence level: Low. Observational and mechanistic data only. No interventional cardiovascular trials.
How to Naturally Boost Your LL-37 Levels
Before reaching for a syringe, consider that your body already produces LL-37. The question is whether it produces enough. Several strategies increase endogenous cathelicidin expression without exogenous peptide administration.

Vitamin D: The Master LL-37 Regulator
The CAMP gene contains a vitamin D response element (VDRE). When 1,25-dihydroxyvitamin D3 binds the vitamin D receptor (VDR), it directly upregulates LL-37 transcription in myeloid cells and keratinocytes (PMC3346901).
A study of healthy adults found a positive correlation between circulating 25(OH)D levels and plasma LL-37 concentrations (BMC Research Notes 2012). Low vitamin D means low cathelicidin.
Practical application: Supplement with 2,000 to 5,000 IU vitamin D3 daily, calibrated to blood levels. Target serum 25(OH)D of 40 to 60 ng/mL. Adding phenylbutyrate (500 mg twice daily) may synergistically enhance LL-37 expression through a complementary histone deacetylase inhibition pathway (Frontiers in Immunology 2022).
This is the single most impactful intervention for boosting innate LL-37 production.
Other Upregulation Strategies
Exercise: Physical activity increases cathelicidin expression in circulating immune cells. The effect is dose-dependent: moderate exercise upregulates production; overtraining suppresses it.
Butyrate: This short-chain fatty acid, produced by gut bacteria fermenting dietary fiber, upregulates CAMP gene expression in colonic epithelial cells. High-fiber diets increase butyrate production. Butyrate supplements (300 to 600 mg daily) may also work.
Sunlight exposure: Activates the vitamin D pathway in skin, stimulating local LL-37 production in keratinocytes. UVB exposure is the relevant wavelength.
Adequate sleep: Immune cell cathelicidin production depends on proper circadian signaling. Chronic sleep deprivation impairs innate immune peptide expression.
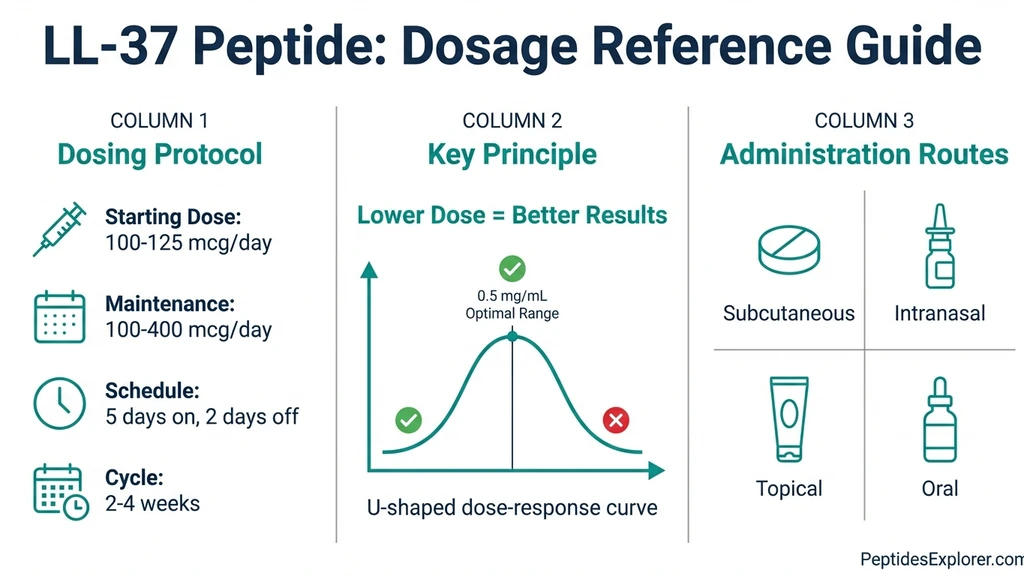
What Is the Right LL-37 Dosage?
All dosage information below comes from published research protocols and clinical observation. No regulatory body has established official LL-37 dosing guidelines.
Subcutaneous Injection Protocol
Starting dose: 100 to 125 mcg daily. Maintenance range: 100 to 400 mcg daily. Frequency: Daily injection, 5 days on, 2 days off. Cycle length: 2 to 4 weeks on, equal time off. Injection sites: Abdominal fat, outer thigh, or upper arm. Rotate sites daily, spacing injections 2 to 3 cm apart.
Start at the lower end. The clinical trial data contains an important counterintuitive finding: the 0.5 mg/mL dose outperformed both 1.6 and 3.2 mg/mL doses for wound healing. More is not better with LL-37. The dose-response curve is inverted.
For step-by-step injection instructions, see our peptide injection guide. If you are new to peptides, start with our getting started guide.
Reconstitution
Standard preparation: Add 2 to 3 mL of bacteriostatic water to a 5 mg lyophilized vial. This yields 1,667 to 2,500 mcg/mL.
For a 200 mcg dose from a 5 mg/2 mL reconstitution (2,500 mcg/mL): draw 8 units on a U-100 insulin syringe.
Storage: Refrigerate at 2 to 8 degrees Celsius after reconstitution. Use within 4 weeks. Unreconstituted vials are stable at room temperature for 12 or more months.
Use our Peptide Reconstitution Calculator for precise calculations with any vial size. See our complete reconstitution guide for step-by-step instructions.
Nasal Spray Protocol
Some practitioners use intranasal LL-37 for sinus-related biofilm infections. Doses range from 100 to 200 mcg per nostril, once or twice daily.
Caution: LL-37 can induce inflammation in olfactory epithelium at high concentrations (PMC5319384). Start with the minimum effective dose. This route has less published support than subcutaneous injection.
The Dose-Response Paradox
The Gronberg et al. wound healing trial revealed an inverted U-shaped dose-response curve. The lowest dose (0.5 mg/mL) produced the best outcomes. The middle dose (1.6 mg/mL) performed worse. The highest dose (3.2 mg/mL) performed worst of the three active groups.
Animal toxicology data supports this pattern. At 100 mcg/kg, LL-37 was safe. At 3,000 mcg/kg, organ toxicity appeared. The therapeutic window is narrow, and more peptide does not produce more benefit.
This is the most important dosing concept for LL-37: conservative dosing outperforms aggressive dosing.
How Does LL-37 Compare to Other Immune Peptides?
LL-37 occupies a specific niche: antimicrobial defense combined with immune modulation. Other peptides serve different primary functions. The table below maps their strengths for protocol design.
| Feature | LL-37 | Thymosin Alpha-1 | KPV | BPC-157 |
|---|---|---|---|---|
| Primary action | Antimicrobial + immune modulation | T-cell maturation + NK cell activation | Anti-inflammatory (NF-kB inhibition) | Tissue repair + angiogenesis |
| Targets | Bacteria, viruses, fungi, biofilms | Adaptive immune system | Gut inflammation, skin inflammation | Tendons, gut, muscles, bones |
| Route | Subcutaneous, nasal | Subcutaneous | Oral, subcutaneous, topical | Subcutaneous, oral |
| Best for | Infections, wound healing, immunity | Chronic viral conditions, immunodeficiency | IBD, autoimmune inflammation | Injury healing, gut repair |
| Human clinical trials | Yes (wound healing RCTs) | Yes (hepatitis B/C) | No (preclinical only) | No (preclinical only) |
| Synergy with LL-37 | N/A | Excellent (innate + adaptive) | Complementary anti-inflammatory | Excellent (antimicrobial + repair) |
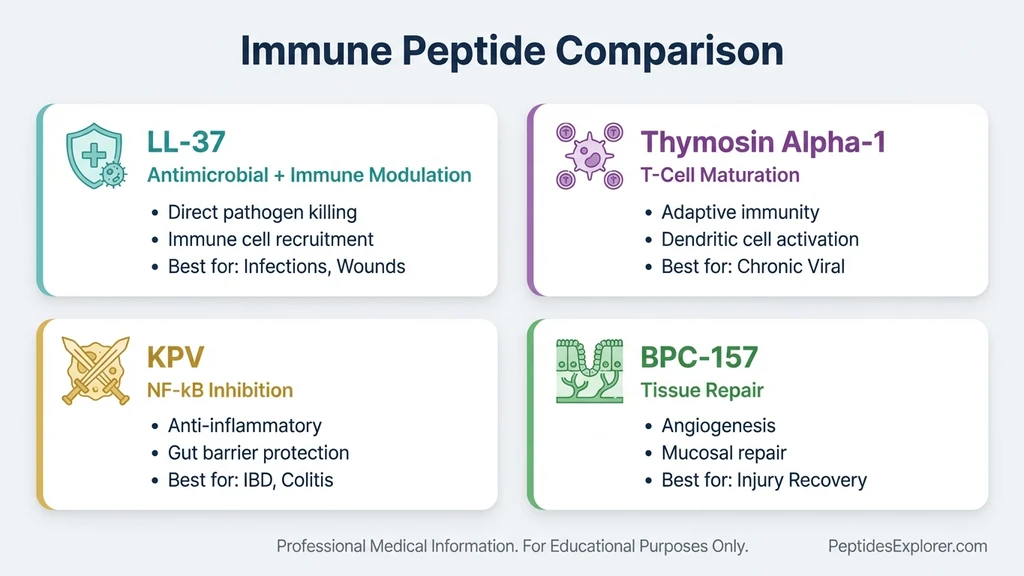
No single peptide covers all bases. LL-37 handles the antimicrobial and immune signaling role. BPC-157 handles tissue reconstruction. Thymosin Alpha-1 drives adaptive immune maturation. KPV provides targeted anti-inflammatory action. Stacking combines their strengths.
How Should You Stack LL-37 With Other Peptides?
Three stacking protocols appear most frequently in peptide community discussions and clinical observation. None have been tested in formal combination trials.
LL-37 + BPC-157: Wound Healing and Gut Repair
LL-37 provides antimicrobial coverage and immune modulation. BPC-157 drives tissue regeneration through angiogenesis and growth factor upregulation. The mechanisms are complementary with no known antagonistic interactions.
Protocol: LL-37 at 100 to 200 mcg/day subcutaneous plus BPC-157 at 250 to 500 mcg/day subcutaneous or oral. Run 2 to 4 weeks. Best when infection risk accompanies tissue damage: post-surgical wounds, contaminated injuries, or gut conditions with microbial imbalance.
For BPC-157 safety information, see our BPC-157 side effects guide.
LL-37 + TB-500: Recovery Stack
TB-500 promotes cell migration, reduces inflammation through actin regulation, and accelerates recovery from musculoskeletal injuries. LL-37 adds antimicrobial protection during the healing window when damaged tissue is vulnerable to infection.
Protocol: LL-37 at 100 to 200 mcg/day plus TB-500 at 2 to 2.5 mg twice per week. Run 4 weeks. Common for post-surgical or post-injury recovery. See our TB-500 dosage chart for detailed protocols.
LL-37 + Thymosin Alpha-1: Immune Defense Stack
Thymosin Alpha-1 enhances adaptive immunity: T cell maturation, NK cell activation, and dendritic cell function. LL-37 provides innate immune defense and direct pathogen killing. Together, they cover both arms of the immune system.
Protocol: LL-37 at 100 to 200 mcg/day plus Thymosin Alpha-1 at 1.6 mg twice per week. Run 2 to 4 weeks. Relevant for chronic infections, post-viral syndromes, and immune reconstitution.
What Are the Side Effects and Safety Risks of LL-37?
LL-37 showed a favorable safety profile in the two wound healing RCTs. The safety picture at higher doses and with systemic administration is less clear.

Common Side Effects
Injection site reactions: redness, swelling, and mild pain at the injection site. These resolve within hours. Flu-like symptoms or a transient inflammatory response during the first days of use. Headache (uncommon).
These reactions may reflect LL-37 doing its job: recruiting immune cells and modulating the inflammatory response.
Serious Risks at High Doses
LL-37 is cytotoxic to human cells at high concentrations. It damages plasma membranes through the same pore-forming mechanism that kills bacteria. At therapeutic doses, this effect is minimal because human membranes carry a neutral charge. At supraphysiological doses, selectivity breaks down.
Documented cytotoxic effects include apoptosis of osteoblasts, vascular smooth muscle cells, neutrophils, airway epithelial cells, and T lymphocytes. Animal toxicology studies showed organ damage at 3,000 mcg/kg, while 100 mcg/kg was safe (PMC3836506, PMC8227053).
The inverted dose-response curve from the wound healing trial is not an anomaly. It reflects real toxicity at higher concentrations. Dose conservatively.
Autoimmune Considerations
Elevated LL-37 is associated with several autoimmune conditions. In psoriasis, LL-37 forms complexes with self-DNA that activate plasmacytoid dendritic cells, driving the inflammatory cascade. Similar mechanisms are implicated in rosacea, contact dermatitis, and lupus (SLE) (PMC7388365).
If you have an active autoimmune condition, particularly psoriasis, rosacea, or lupus, exogenous LL-37 may worsen symptoms. This is a hard contraindication.
Thrombosis Risk
LL-37 promotes NETosis, the formation of neutrophil extracellular traps. NETs are part of antimicrobial defense, but excessive NETosis contributes to hypercoagulation. This was studied specifically in COVID-19-associated thrombosis (PMC9123294).
Individuals with a history of blood clots or active clotting disorders should exercise caution. No direct causal link between therapeutic LL-37 doses and thrombotic events has been established, but the mechanistic pathway warrants awareness.
Regulatory Status
LL-37 is not FDA-approved for any therapeutic indication. It is available as a research chemical in the United States. It is not a controlled or scheduled substance. It cannot be legally marketed or sold as a drug or dietary supplement.
Always source from vendors who provide third-party Certificates of Analysis (COA) with verified purity testing.
Frequently Asked Questions
What is LL-37 peptide used for?
LL-37 is researched for antimicrobial defense (including against MRSA and antibiotic-resistant bacteria), wound healing acceleration, antiviral activity against influenza and SARS-CoV-2, immune modulation, biofilm disruption, and gut barrier protection. Two randomized clinical trials have demonstrated wound healing benefits in venous leg ulcers.
Is LL-37 the same as cathelicidin?
LL-37 is the active form of human cathelicidin. The CAMP gene encodes a precursor protein called hCAP-18, which is cleaved by the enzyme proteinase 3 to produce the 37-amino-acid active peptide LL-37. Humans produce only one cathelicidin. Other mammals produce multiple cathelicidins.
How does vitamin D affect LL-37 levels?
Vitamin D directly regulates LL-37 production. The CAMP gene contains a vitamin D response element, so 1,25-dihydroxyvitamin D3 upregulates LL-37 transcription in immune cells and keratinocytes. Supplementing with 2,000 to 5,000 IU vitamin D3 daily to achieve serum 25(OH)D levels of 40 to 60 ng/mL supports optimal cathelicidin expression.
What is the typical LL-37 dosage for subcutaneous injection?
Research protocols use 100 to 200 mcg daily via subcutaneous injection, administered 5 days on and 2 days off, in cycles of 2 to 4 weeks. Starting at 100 mcg is recommended. Clinical trial data shows an inverted dose-response curve: the lowest dose (0.5 mg/mL) produced healing rates six times faster than placebo, outperforming higher doses.
Can LL-37 help with gut health?
Preclinical research shows LL-37 preserves intestinal barrier function by protecting goblet cells, increasing mucin-2 production, upregulating the antioxidant factor Nrf2, and reducing bacterial translocation across the gut lining. LL-37 is naturally expressed throughout the gastrointestinal tract, and its production is directly linked to vitamin D status.
Is LL-37 safe?
Topical LL-37 showed a favorable safety profile in two randomized clinical trials for venous leg ulcers. However, LL-37 is cytotoxic to human cells at high concentrations, and elevated levels are associated with psoriasis, rosacea, and lupus. Common side effects include injection site reactions and transient flu-like symptoms. It is not FDA-approved for therapeutic use.
Can you stack LL-37 with BPC-157?
LL-37 and BPC-157 are commonly combined. They work through complementary mechanisms: LL-37 provides antimicrobial defense and immune modulation, while BPC-157 drives tissue regeneration through angiogenesis. A typical protocol uses LL-37 at 100 to 200 mcg/day with BPC-157 at 250 to 500 mcg/day for 2 to 4 weeks.
Does LL-37 work against antibiotic-resistant bacteria?
Yes. LL-37 uses membrane disruption rather than targeting specific bacterial enzymes, making resistance development extremely difficult. It has demonstrated activity against MRSA, vancomycin-resistant enterococci, and multidrug-resistant Pseudomonas aeruginosa. Bacteria would need to fundamentally alter their membrane composition to resist it.
How long does it take for LL-37 to work?
In the venous leg ulcer clinical trial, measurable wound healing improvements appeared within 4 weeks of twice-weekly topical application. For subcutaneous protocols targeting immune support, anecdotal reports suggest effects within 1 to 2 weeks. Full protocol duration is typically 2 to 4 weeks.
Is LL-37 legal?
LL-37 is legal to purchase and possess in the United States as a research chemical. It is not a controlled or scheduled substance. However, it is not FDA-approved for human therapeutic use and cannot be marketed or sold as a drug or dietary supplement.
The Bottom Line
LL-37 is the body's own antimicrobial weapon, backed by two human clinical trials confirming its wound healing potential. The evidence for antimicrobial defense, antiviral activity, and immune modulation is substantial. The evidence for gut protection, cancer immunotherapy, and cardiovascular effects is early but mechanistically sound.
Three practical takeaways. First, dose conservatively. The clinical data proves that lower doses outperform higher ones. Start at 100 mcg daily and resist the impulse to escalate. Second, boost your natural production. Vitamin D at 2,000 to 5,000 IU daily upregulates LL-37 expression through the CAMP gene, a free intervention that supports every other benefit. Third, respect the contraindications. Active autoimmune conditions, particularly psoriasis and lupus, are incompatible with exogenous LL-37.
For those exploring peptide protocols, LL-37 stacks effectively with BPC-157 for tissue repair, TB-500 for recovery, and Thymosin Alpha-1 for adaptive immune support. Use our Peptide Stack Calculator to design a protocol, and our Peptide Interaction Checker to screen for conflicts.
Every claim in this guide links to its source. Where the evidence is strong, we say so. Where it is preliminary, we say that too. This is how peptide information should work. For all dosage references, see the peptide dosage chart. For proper handling, see how to store peptides and how long do reconstituted peptides last. For sourcing, see where to buy peptides in 2026. For overall safety, see the peptide safety guide. New to peptides? Start with our getting started with peptides guide.
Related Articles: - Thymosin Alpha-1 Benefits - KPV Peptide Dosage - Peptides for Gut Health - Peptide Stacking Guide - BPC-157 Side Effects
Helpful Tools
Related Articles
Peptides for Inflammation: 7 Best Ranked
Top 7 anti-inflammatory peptides ranked: BPC-157, KPV, LL-37, thymosin alpha-1, GHK-Cu, TB-500, Selank. Mechanisms, dosage protocols, and 10+ PubMed citations.
Peptides for Immune System: 7 Best
Best peptides for immune system support ranked by clinical evidence. Thymosin Alpha-1, LL-37, KPV, BPC-157, Selank dosages, stacking protocols, and safety data.
Peptides for Recovery: 7 Best Ranked by Evidence
Top 7 peptides for recovery ranked: BPC-157, TB-500, GHK-Cu, ipamorelin, CJC-1295, MOTS-c, thymosin alpha-1. Post-workout and injury protocols.
GHK-Cu Dosage: Protocols & Charts
GHK-Cu dosage guide with injection protocols by goal (skin, hair, healing), reconstitution steps, dosage charts, side effects, and before/after timeline.