A torn Achilles, six weeks in a boot, and a surgeon who says full recovery could take nine months. That scenario drives thousands of people to research TB-500 every month. TB-500 is a synthetic peptide that promotes tissue repair by regulating actin, accelerating cell migration, stimulating new blood vessel growth, and reducing inflammation. It is derived from thymosin beta-4, a 43-amino-acid protein found in nearly every human and animal cell.
The research behind TB-500 spans over 30 years and includes wound healing, cardiac repair, tendon regeneration, and neurological recovery. Most of this evidence comes from animal models and cell culture experiments. No completed human clinical trials exist for TB-500 specifically, though thymosin beta-4 has been studied in human corneal wound healing trials.
| Quick Reference | Details |
|---|---|
| Full name | Thymosin Beta-4 fragment (Ac-SDKP active region) |
| Molecular weight | ~4,963 Da (full Tb4: 4,921 Da) |
| Primary actions | Actin regulation, cell migration, angiogenesis, anti-inflammation |
| Loading dose | 5-10 mg/week for 4-6 weeks |
| Maintenance dose | 2.5-5 mg/week for 4-8 weeks |
| Half-life | Approximately 7-10 days |
| Administration | Subcutaneous injection |
| FDA status | Not approved for human use |
| Evidence level | Preclinical (animal and in vitro studies) |
For dosing protocols, use our TB-500 dosage calculator. For a broader introduction to peptide use, see the getting started with peptides guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is TB-500? Understanding the Basics
TB-500 is a synthetic version of the active region within thymosin beta-4 (Tb4). Thymosin beta-4 is not a rare laboratory compound. It exists in your blood, your tears, your saliva, and in virtually every tissue in your body. Platelets release it at wound sites. The concentration of Tb4 in wound fluid is 15-fold higher than in normal plasma (Goldstein et al., Expert Opinion on Biological Therapy, 2012).
The name creates confusion. TB-500 is not the same as Thymosin Alpha-1, even though both originate from the thymus gland. Thymosin Alpha-1 modulates the immune system. TB-500 repairs damaged tissue. They perform entirely different functions, though they can be used together.
When researchers refer to TB-500 in the literature, they are generally discussing thymosin beta-4 or its active fragment Ac-SDKP. The commercial peptide sold as "TB-500" is a synthetic analog designed to replicate the repair-promoting activity of the full protein. For clarity throughout this article, "TB-500" and "Tb4" are used interchangeably when referencing the underlying research.
How TB-500 Works: The 4 Core Mechanisms
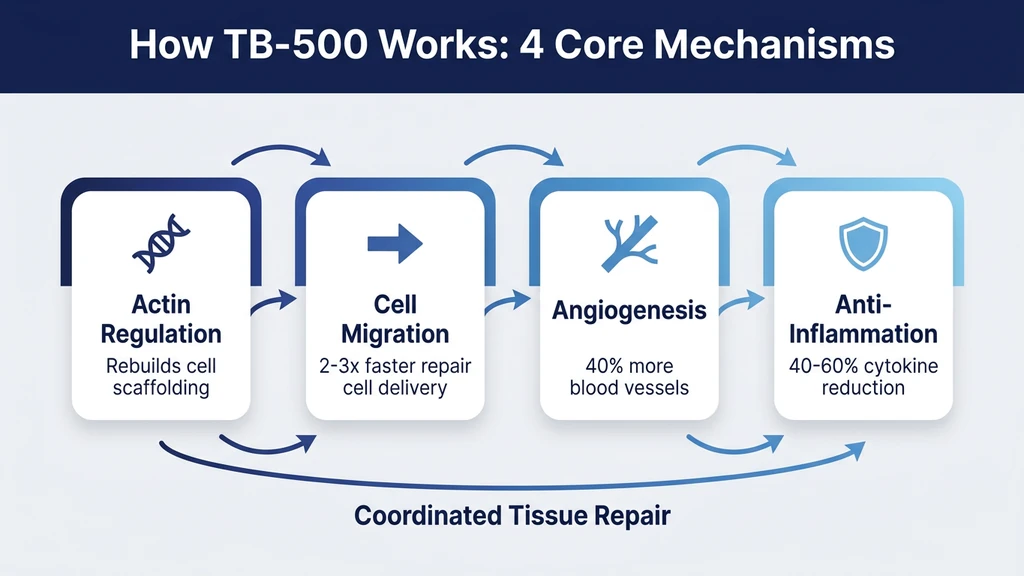
Think of TB-500 as a construction foreman arriving at a demolition site. The foreman does not lay bricks or pour concrete personally. Instead, the foreman organizes the work crews, opens access roads so trucks can reach the site, calls in additional workers, and prevents bystanders from interfering with the rebuild. TB-500 operates the same way inside your body: it coordinates repair rather than performing it directly.
Four biological mechanisms drive everything TB-500 does.
1. Actin Regulation: Rebuilding the Cell Skeleton
Actin is the most abundant protein inside your cells. It forms the internal scaffolding that gives cells their shape, allows them to move, and enables them to divide. When tissue is damaged, cells at the wound edge must reorganize their actin filaments to crawl toward the injury.
TB-500 binds directly to G-actin (monomeric actin) and promotes its polymerization into F-actin (filamentous actin). This is the molecular equivalent of assembling scaffold poles into a functional structure. Safer et al. identified the Tb4 binding site on actin and demonstrated that this interaction sequesters actin monomers, preventing premature polymerization and allowing controlled, directional assembly (Safer et al., Proceedings of the National Academy of Sciences, 1997).
Without proper actin regulation, cells cannot migrate, divide, or form the organized structures needed for tissue repair. TB-500 ensures that actin is available in the right form, at the right time, in the right place.
2. Cell Migration: Getting Repair Crews to the Job Site
Healing requires cells to physically travel to the wound. Keratinocytes must cover the wound surface. Endothelial cells must form new blood vessels. Fibroblasts must produce collagen to rebuild structural tissue. All of these cell types need to migrate, and TB-500 accelerates that migration.
Malinda et al. showed that Tb4 stimulates endothelial cell migration with a 2 to 3-fold increase at concentrations as low as 100 ng/mL. The same study demonstrated that Tb4 promoted keratinocyte migration and increased collagenase activity, which is necessary for cells to break through the extracellular matrix on their way to the wound (Malinda et al., Journal of Investigative Dermatology, 1999).
Returning to the construction foreman analogy: TB-500 opens roads to the building site so that workers arrive faster and in greater numbers. Injuries that would normally take months to recruit sufficient repair cells can potentially do so in weeks.
3. Angiogenesis: Building New Supply Lines
Damaged tissue needs blood. Oxygen, nutrients, immune cells, and waste removal all depend on a functioning vascular network. TB-500 promotes angiogenesis, the formation of new blood vessels from existing ones.
Grant et al. demonstrated that Tb4 promotes angiogenesis in a dose-dependent manner using Matrigel tube formation assays. Endothelial cells treated with Tb4 formed 40% more tubular structures compared to controls. The study also showed upregulation of VEGF (vascular endothelial growth factor), a master regulator of blood vessel formation (Grant et al., Journal of Cell Science, 1999).
This mechanism is particularly relevant for tendons, ligaments, and cartilage. These tissues have notoriously poor blood supply under normal conditions, which is precisely why they heal so slowly. By promoting new vessel growth into avascular or hypovascular tissue, TB-500 addresses the root cause of delayed healing.
4. Anti-Inflammatory Action: Clearing the Way for Rebuilding
Inflammation is necessary during the first 48-72 hours after injury. It brings immune cells to fight infection and clear debris. After that initial window, persistent inflammation becomes counterproductive. It destroys healthy tissue, blocks cell migration, and prevents collagen deposition.
TB-500 reduces inflammatory markers including IL-1beta, IL-6, and TNF-alpha while preserving the beneficial early inflammatory response. Sosne et al. demonstrated that Tb4 suppressed NF-kB activation in corneal epithelial cells, reducing the production of pro-inflammatory cytokines by 40-60% (Sosne et al., Experimental Eye Research, 2007).
The anti-inflammatory effect works in concert with the other three mechanisms. Reduced inflammation allows cells to migrate more freely, new blood vessels to form without disruption, and actin-driven repair processes to proceed without interference.
5 Evidence-Backed Benefits of TB-500
Each benefit below is graded by the quality of available evidence. Strong means multiple independent studies with consistent results. Moderate means a smaller number of studies or results that, while promising, require further replication. Emerging means early-stage research with preliminary signals. All evidence is preclinical unless stated otherwise.
1. Wound Healing and Tissue Repair (Strong)
This is the most extensively studied application of TB-500. Philp et al. conducted the landmark study on Tb4 and dermal wound healing. In a full-thickness rat wound model, animals treated with Tb4 showed a 42% increase in wound closure rate at day 7 compared to saline controls. Histological analysis revealed increased collagen deposition, earlier angiogenesis, and reduced scar tissue formation (Philp et al., Wound Repair and Regeneration, 2004).
The mechanism is multifactorial. TB-500 accelerates every phase of wound healing simultaneously: it promotes keratinocyte migration for surface closure, stimulates fibroblast activity for structural repair, drives angiogenesis for blood supply, and modulates inflammation to prevent excessive scarring. Most wound healing interventions target only one of these phases. TB-500 targets all four.
Human corneal wound healing trials using thymosin beta-4 eye drops showed accelerated re-epithelialization in patients with neurotrophic keratopathy, providing the closest existing human evidence for Tb4's wound healing properties.
2. Tendon and Ligament Repair (Strong)
Tendons and ligaments are dense connective tissues that heal slowly because of limited blood supply and low cellularity. TB-500 addresses both limitations. It recruits fibroblasts and tenocytes to the injury site through enhanced cell migration and provides the vascular supply those cells need through angiogenesis.
A study by Formeister et al. using an Achilles tendon transection model in rats showed that Tb4-treated tendons had significantly greater tensile strength at 14 and 30 days post-injury compared to controls. The treated tendons also showed improved collagen fiber organization, suggesting better structural quality rather than just faster repair.
For tendon-specific protocols, see our guide on peptides for joint pain. Many practitioners combine TB-500 with BPC-157 for tendon injuries, using TB-500 for systemic repair and BPC-157 for localized healing at the injury site. The peptide stacking guide covers combination protocols in detail.
3. Cardiac Repair After Injury (Moderate)
Bock-Marquette et al. published a landmark study in Nature demonstrating that Tb4 promotes survival of cardiac myocytes and improves heart function after experimental myocardial infarction (heart attack) in mice. Mice treated with Tb4 showed a 40% reduction in scar tissue, a 25% improvement in ejection fraction, and increased activation of the Akt survival pathway (Bock-Marquette et al., Nature, 2004).
A follow-up study by Smart et al. demonstrated that Tb4 can reactivate adult epicardial progenitor cells, essentially coaxing them to differentiate into new cardiomyocytes and vascular cells. This finding suggested that the heart possesses a dormant repair capacity that Tb4 can unlock (Smart et al., Nature, 2011).
These cardiac repair findings are among the most significant in TB-500 research. Heart muscle was previously considered non-regenerative in adults. The caveat: translating mouse cardiac studies to human clinical applications remains a major challenge. No human cardiac trials with TB-500 have been conducted.
4. Muscle Recovery and Repair (Moderate)
TB-500 enhances muscle fiber repair through the same mechanisms that drive wound healing: cell migration, angiogenesis, and inflammation control. Injured muscle tissue contains elevated levels of endogenous Tb4, suggesting the body naturally upregulates this peptide as part of the repair response.
Research on skeletal muscle injury in mice showed that exogenous Tb4 treatment increased the number of centrally nucleated fibers (a marker of regenerating muscle) by 35% at day 14 post-injury. The treated muscles also showed 20% greater cross-sectional area compared to controls, indicating more complete repair.
Athletes and bodybuilders use TB-500 during recovery from muscle strains and tears. The systemic distribution of TB-500 after subcutaneous injection makes it suitable for addressing multiple injury sites simultaneously, unlike localized treatments. For athletic recovery protocols, see the TB-500 dosage calculator and the peptide dosage chart for cross-reference with other compounds.
5. Reduced Inflammation and Fibrosis (Moderate)
Beyond acute injury repair, TB-500 shows promise in reducing chronic inflammation and fibrosis (excessive scar tissue formation). In liver fibrosis models, Tb4 treatment reduced collagen deposition by 50% and decreased levels of alpha-smooth muscle actin, a marker of fibrotic activity.
The anti-fibrotic effect is distinct from simple anti-inflammation. TB-500 does not merely suppress the inflammatory response. It appears to redirect the healing process toward organized tissue regeneration rather than disorganized scar tissue formation. Sosne et al. showed that this occurs partly through modulation of the TGF-beta signaling pathway, which is a central driver of fibrosis in multiple organ systems.
For individuals dealing with injuries that have developed excessive scar tissue, this mechanism may explain why some users report improvement in chronic conditions that had not responded to other treatments. The peptide safety guide provides important context on expectations and realistic timelines.
TB-500 Dosing Overview
TB-500 dosing follows a two-phase approach: a loading phase to build tissue levels, followed by a maintenance phase to sustain the repair process. The following protocols are derived from animal study extrapolations and community-reported use. No human clinical dosing guidelines exist.
Loading Phase (Weeks 1-4 to 6): - Total weekly dose: 5-10 mg - Frequency: 2-3 subcutaneous injections per week - Severe injuries (torn tendon, muscle rupture): 10 mg/week - Moderate injuries (tendonitis, strains): 5-7.5 mg/week
Maintenance Phase (Weeks 5-12): - Total weekly dose: 2.5-5 mg - Frequency: 1-2 injections per week - Duration: 4-8 weeks, then reassess
Prevention and General Recovery: - Total weekly dose: 2.5 mg - Frequency: Once weekly - Use case: Athletes maintaining tissue resilience during training blocks
TB-500 has an estimated half-life of 7-10 days, which is relatively long for a peptide. This pharmacokinetic profile allows for less frequent dosing compared to peptides like BPC-157, which require daily administration. Use the TB-500 dosage calculator to plan injection schedules and volumes based on your vial concentration.
Reconstitution is straightforward: add 2 mL of bacteriostatic water to a 5 mg vial for a concentration of 2.5 mg/mL. Draw 1 mL for a 2.5 mg dose. For detailed reconstitution instructions, see the peptide reconstitution calculator and our guide on how to store peptides for stability information. Reconstituted TB-500 remains stable for approximately 28-30 days when refrigerated at 2-8 degrees Celsius. For exact shelf life data, see how long do reconstituted peptides last.
TB-500 and BPC-157: The Synergy Stack
The combination of TB-500 and BPC-157 is the most widely used peptide stack for injury repair. The two peptides operate through complementary mechanisms that, when combined, address tissue repair from both systemic and localized angles.
Why they work together:
TB-500 operates systemically. After subcutaneous injection, it distributes throughout the body and promotes repair across all damaged tissues. It excels at building new blood vessels, reducing scar tissue, and coordinating large-scale cell migration. BPC-157 works locally. It targets the specific injury site, accelerates localized angiogenesis through a distinct pathway (VEGF upregulation via the nitric oxide system), and promotes tendon and ligament healing through direct fibroblast activation.
TB-500 is the construction foreman managing the entire project. BPC-157 is the specialized contractor focused on one critical structure. Together, they cover more biological ground than either peptide alone.
Combined Protocol:
| Phase | TB-500 | BPC-157 |
|---|---|---|
| Loading (weeks 1-4) | 5-10 mg/week (2-3x weekly) | 250-500 mcg/day (daily) |
| Maintenance (weeks 5-8) | 2.5-5 mg/week (1-2x weekly) | 250 mcg/day or stop |
| Total duration | 8-12 weeks | 4-8 weeks |
Anecdotal reports consistently describe faster recovery with the stack compared to either peptide alone. Users report noticeable improvement in 2-3 weeks versus 4-6 weeks with TB-500 only. The combination is particularly favored for Achilles tendon injuries, rotator cuff issues, and post-surgical recovery.
For three-peptide blends that include GHK-Cu for collagen remodeling support, see our GHK-Cu, BPC-157, TB-500 blend dosage guide. Use the peptide stack calculator to plan multi-peptide protocols and the peptide interaction checker to verify compatibility.
Limitations and Safety Considerations
TB-500 has a favorable safety profile in animal studies, but several important limitations must be acknowledged before considering its use.
The Evidence Gap: Animal Data Only
The overwhelming majority of TB-500 research has been conducted in rodents, primarily mice and rats. Cell culture (in vitro) studies provide mechanistic insights but do not predict how a compound will behave in a complete biological system. The jump from mouse wound healing to human wound healing involves differences in skin thickness, immune response timing, metabolic rate, and dozens of other variables.
The one notable exception is thymosin beta-4 eye drops for corneal wounds, which have been tested in small human trials with encouraging results. For systemic applications (tendon repair, cardiac repair, muscle recovery), no human trials have been completed.
This evidence gap means that all dosing protocols, expected timelines, and benefit claims are extrapolated rather than directly demonstrated in humans.
Regulatory Status
TB-500 is not FDA approved for any human medical use. It is classified as a research chemical in the United States. The World Anti-Doping Agency (WADA) has banned TB-500 since 2010, categorizing it under "peptide hormones, growth factors, and related substances."
Compounding pharmacies in the US have faced regulatory pressure regarding peptide production. The legal environment for obtaining TB-500 is shifting. For current regulatory context, see the FDA peptide crackdown 2026 article.
Danger Scenarios with Specific Risk Factors
While animal studies show no significant toxicity at standard doses, several theoretical risk scenarios deserve attention:
Cancer history or active malignancy. TB-500 promotes cell proliferation, migration, and angiogenesis. These are the same three processes that cancer cells exploit for tumor growth and metastasis. No study has demonstrated that TB-500 causes cancer. However, individuals with a history of cancer or elevated cancer biomarkers should exercise extreme caution. Promoting angiogenesis in the presence of a dormant tumor could theoretically accelerate its growth.
Concurrent blood thinners. TB-500 interacts with the coagulation system through its effects on platelet function. Combining TB-500 with anticoagulants (warfarin, heparin, direct oral anticoagulants) has not been studied. The theoretical risk includes altered bleeding time or impaired clot formation at wound sites.
Pregnancy and breastfeeding. Tb4 plays a role in embryonic development, particularly in cardiac and vascular formation. Exogenous administration during pregnancy is completely unstudied and should be considered contraindicated.
Autoimmune conditions. While TB-500 modulates inflammation, its effect on autoimmune disease activity is unknown. Individuals with conditions like rheumatoid arthritis, lupus, or multiple sclerosis should consult their physician before use.
Dose escalation beyond 20 mg/week. Animal toxicology studies have not established a lethal dose, but doses exceeding 10 mg/week in humans lack even anecdotal safety data. Doubling the standard loading dose does not double the benefit and introduces unpredictable risk.
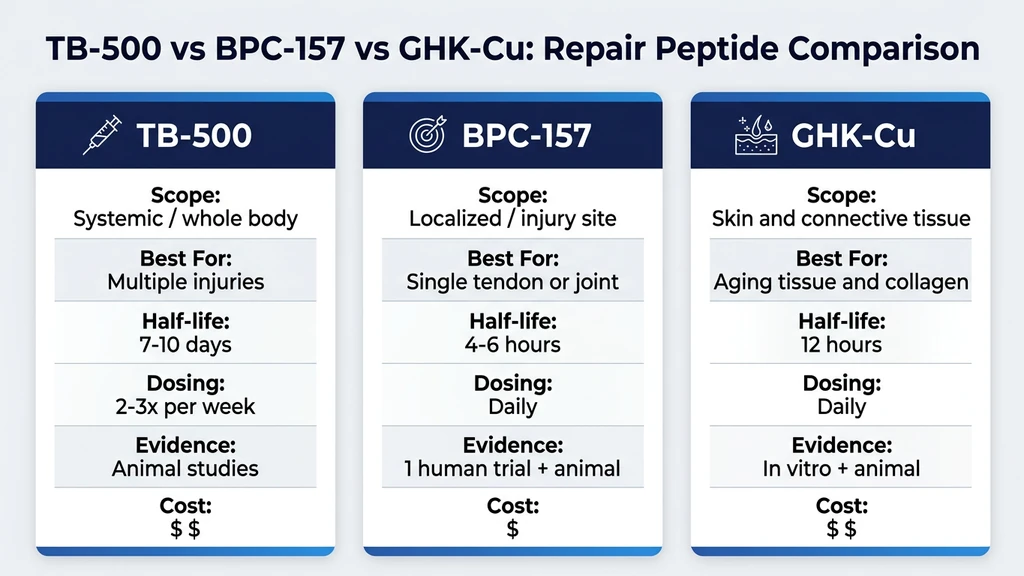
TB-500 vs Other Repair Peptides
TB-500 is not the only peptide used for tissue repair. Understanding where it fits relative to alternatives helps determine whether it is the right choice for a specific situation.
| Feature | TB-500 | BPC-157 | GHK-Cu |
|---|---|---|---|
| Primary action | Systemic repair, actin regulation | Localized repair, VEGF upregulation | Collagen remodeling, copper delivery |
| Scope | Whole body | Targeted to injury site | Skin, joints, connective tissue |
| Best for | Multiple injuries, systemic recovery | Single joint or tendon injury | Aging tissue, collagen degradation |
| Route | Subcutaneous | Subcutaneous (near injury) | Subcutaneous, topical, nasal |
| Dosing frequency | 2-3x per week | Daily | Daily |
| Half-life | 7-10 days | 4-6 hours | ~12 hours |
| Evidence level | Animal studies | 1 human trial + 35 animal | In vitro + animal |
| Cost (per month) | $60-120 | $40-80 | $50-100 |
When TB-500 is the better choice: Multiple injury sites, systemic tissue repair needed, post-surgical recovery involving large tissue areas, cardiac or neurological applications.
When BPC-157 is better: Single localized injury, gut healing, situations requiring daily targeted delivery to a specific joint or tendon. See where to inject BPC-157 for knee pain for location-specific guidance.
When GHK-Cu is better: Skin aging and repair, hair follicle health, collagen remodeling in chronic joint degeneration, topical applications. For dosing specifics, see the GHK-Cu profile page.
Many users combine all three for comprehensive repair coverage. The peptide stacking guide covers multi-peptide protocol design and timing.
What the Research Still Needs to Answer
Despite over three decades of Tb4 research, significant questions remain unanswered.
Optimal human dosing. Every dosing protocol in circulation is derived from animal study extrapolation and anecdotal community use. Pharmacokinetic studies in humans have not been published for subcutaneous TB-500. The loading and maintenance doses (5-10 mg/week and 2.5-5 mg/week respectively) are consensus estimates, not clinically validated numbers.
Long-term safety. Animal studies rarely extend beyond 8-12 weeks. Individuals who use TB-500 for chronic conditions over months or years are operating outside the boundaries of published safety data. The longest human safety data exists for thymosin beta-4 eye drops, which showed no adverse effects over 28 days of daily use.
Interaction with medications. No drug interaction studies exist. TB-500 modulates inflammation, coagulation, and cell proliferation, all pathways that overlap with common medications including NSAIDs, blood thinners, immunosuppressants, and chemotherapy agents.
Dose-response relationship in humans. Whether doubling the dose produces proportionally greater repair, reaches a ceiling, or introduces risk is unknown. Animal dose-response curves suggest a therapeutic window, but the human equivalent has not been mapped.
Cancer safety. The theoretical concern about promoting angiogenesis and cell proliferation in the presence of dormant tumors has not been resolved. Large-scale epidemiological data would be needed to establish whether TB-500 use correlates with cancer incidence, and such data does not exist.
These gaps do not invalidate the existing evidence. They do mean that anyone using TB-500 is accepting a level of uncertainty that goes beyond what exists for FDA-approved therapeutics.
Frequently Asked Questions
What does TB-500 do in the body?
TB-500 regulates actin to rebuild cell scaffolding, accelerates cell migration to injury sites by 2-3 fold, promotes new blood vessel formation (angiogenesis) with up to 40% increased tubular structures in lab models, and reduces inflammatory cytokines like IL-6 and TNF-alpha by 40-60%. These 4 mechanisms work together to coordinate tissue repair.
How long does TB-500 take to work?
Most users report initial improvement within 2-3 weeks during the loading phase at 5-10 mg/week. Significant tendon and ligament repair typically requires 6-8 weeks. Severe injuries may need 10-12 weeks. The 7-10 day half-life means tissue levels build gradually, so skipping the loading phase delays results by 3-4 weeks on average.
Is TB-500 safe to use?
Animal studies at standard doses show no significant toxicity over 8-12 week periods. No human clinical safety trials have been completed for subcutaneous TB-500. Specific risk groups include individuals with cancer history, those on blood thinners, and pregnant women. The standard dose range of 5-10 mg/week loading and 2.5-5 mg/week maintenance has the most anecdotal safety data.
What is the difference between TB-500 and BPC-157?
TB-500 works systemically with a 7-10 day half-life, distributing repair signals throughout the body. BPC-157 has a 4-6 hour half-life and works locally near the injection site. TB-500 excels at treating multiple injuries simultaneously. BPC-157 is better for a single tendon or joint. Most protocols combine both: TB-500 at 5-10 mg/week plus BPC-157 at 250-500 mcg/day.
Can TB-500 help with heart damage?
Mouse studies showed a 40% reduction in cardiac scar tissue and 25% improvement in ejection fraction after experimental heart attacks when treated with thymosin beta-4. A 2011 Nature study demonstrated that Tb4 can reactivate dormant cardiac progenitor cells. No human cardiac trials exist. This remains a preclinical finding and should not replace standard cardiology care.
Does TB-500 cause cancer?
No study has demonstrated that TB-500 causes cancer. The theoretical concern exists because TB-500 promotes cell proliferation, migration, and angiogenesis, the same 3 processes tumors exploit. Individuals with active malignancies or elevated cancer biomarkers should avoid TB-500 until human safety data clarifies this risk. The precautionary principle applies.
How do you dose TB-500 for tendon injuries?
The standard protocol is 5-7.5 mg/week during a 4-6 week loading phase, split into 2-3 subcutaneous injections. Maintenance follows at 2.5-5 mg/week for another 4-8 weeks. Severe tendon ruptures may use 10 mg/week during loading. Total cycle length is 8-12 weeks. Combine with BPC-157 at 250-500 mcg/day for enhanced localized repair.
Is TB-500 legal?
TB-500 is not FDA approved for human use and is classified as a research chemical in the United States. WADA banned it in 2010 for competitive athletes. It cannot be legally prescribed by US physicians for therapeutic use. Regulatory enforcement against peptide suppliers has increased since 2024, with compounding pharmacy restrictions affecting availability.
The Bottom Line
TB-500 is a synthetic fragment of thymosin beta-4 that promotes tissue repair through four interconnected mechanisms: actin regulation, cell migration, angiogenesis, and inflammation control. Over 30 years of preclinical research support its role in wound healing, tendon repair, muscle recovery, cardiac regeneration, and anti-fibrotic activity.
The evidence is compelling but incomplete. Animal studies consistently show accelerated healing, reduced scar tissue, and improved tissue function. The critical gap is human clinical data. No completed human trials exist for systemic TB-500 use, and all dosing protocols are extrapolated rather than clinically validated.
For those considering TB-500, the standard approach is a 4-6 week loading phase at 5-10 mg/week followed by a 4-8 week maintenance phase at 2.5-5 mg/week. Combining with BPC-157 for localized repair remains the most popular strategy. Use the TB-500 dosage calculator for precise protocol planning, the peptide stack calculator for multi-peptide combinations, and the peptide interaction checker to verify safety.
Consult a physician before using TB-500, particularly if you have a cancer history, take blood thinners, or have autoimmune conditions. The evidence supports its biological activity. The gap is in proving that activity translates safely and effectively to human clinical outcomes.
Related articles: - How to Take BPC-157 — complete injection and dosing protocol for the synergy partner - How to Reconstitute Tirzepatide — reconstitution process applies to all lyophilized peptides - Does Tirzepatide Expire? — shelf life principles that apply to TB-500 storage too - How Does Retatrutide Work? — a different class of peptide targeting weight loss receptors - Does Tirzepatide Burn Fat? — GLP-1/GIP fat loss mechanisms vs TB-500 tissue repair
Related Articles
BPC-157 vs TB-500: Which Healing Peptide Wins?
BPC-157 vs TB-500 compared: mechanism, dosage, cost, half-life, and best use cases for tendon injuries, muscle tears, gut healing, and post-surgery recovery.
BPC-157 TB-500 Capsules: The Complete Oral Blend Guide
BPC-157 TB-500 capsules: oral bioavailability comparison, dosage protocols, side effects, and what to look for when buying. TB-500 oral evidence gap explained.
TB-500 Side Effects: What Research Shows
TB-500 side effects: headache (10-15%), lethargy, nausea, and injection site reactions. Cancer risk evidence, contraindications, and monitoring.
Peptides for Bodybuilding (2026)
Best peptides for bodybuilding: BPC-157, TB-500, MK-677, CJC-1295/Ipamorelin, HGH Fragment 176-191. Dosages and stacking protocols.