
You started retatrutide last week. The scale has barely moved. You are wondering whether this triple agonist actually lives up to the headlines. Retatrutide begins suppressing appetite within the first week of treatment. Measurable weight loss appears by week 4. By week 24, participants in the Phase 2 trial lost an average of 17.5% of body weight on the 12 mg dose. By week 48, that number reached 24.2%, the largest weight loss ever recorded for any anti-obesity medication at that time point (Jastreboff et al., NEJM, 2023).
The slow start is not a failure of the drug. Retatrutide uses a dose-escalation schedule that begins at 0.5 mg and increases over months to the full therapeutic dose of 8 to 12 mg. Each step up strengthens the triple agonist effect across all three receptor pathways: GLP-1 for appetite suppression, GIP for metabolic signaling, and glucagon for direct fat oxidation. Rushing that process causes severe gastrointestinal side effects that derail treatment entirely.
| Timeframe | What You Feel | Typical Weight Loss | Dose Range |
|---|---|---|---|
| Week 1 | Reduced appetite, possible mild nausea | 0-2 lbs | 0.5 mg |
| Weeks 2-4 | Smaller portions satisfy, food noise quiets | 2-5 lbs | 0.5-1 mg |
| Weeks 5-12 | Strong appetite suppression, cravings fade | 5-12% body weight | 2-4 mg |
| Weeks 13-24 | Visible body composition changes | 12-17% body weight | 4-8 mg |
| Weeks 25-48 | Peak fat burning, waist circumference drops | 17-24% body weight | 8-12 mg |
| Weeks 49-68 | Maximum recorded effect | Up to 28.7% body weight | 12 mg |
For the complete dosing protocol at each escalation step, see our retatrutide dosage guide. To understand why the triple agonist mechanism outperforms earlier drugs, read how does retatrutide work.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Week-by-Week Timeline: What to Expect at Each Stage
The retatrutide experience unfolds in distinct phases. Each one feels different because the drug activates three separate receptor systems that engage at different rates. Understanding this sequence prevents the most common mistake: abandoning treatment before reaching the dose that produces the results from the clinical trials.
Weeks 1-4: The Adaptation Phase (0.5 mg)
The first injection is 0.5 mg, a fraction of the target dose. This is not designed to produce dramatic weight loss. It is designed to let your gastrointestinal tract adapt to simultaneous GLP-1, GIP, and glucagon receptor activation.
Within the first 3 to 7 days, most people notice a subtle shift in appetite. Meals feel more satisfying at smaller portions. The background hum of food-related thoughts, what researchers call "food noise," begins to quiet. You might push away half a plate without conscious effort.
Weight loss at this stage is modest: 2 to 5 pounds over the month, mostly from reduced caloric intake and some water loss. The Phase 2 trial showed minimal separation between retatrutide 0.5 mg and placebo during the first month (Jastreboff et al., NEJM, 2023). That is expected. The therapeutic dose is 16 to 24 times higher than what you are injecting right now.
Nausea is the most common side effect at this stage. In clinical trials, it affected 24 to 43% of participants depending on the escalation protocol. Eating smaller meals, avoiding high-fat food, and staying hydrated minimizes it. The lower starting dose (0.5 mg escalated from 2 mg rather than 4 mg) cut GI side effects substantially in the Phase 2 data.
Weeks 5-12: Appetite Suppression Strengthens (1-4 mg)
The dose escalates through 1 mg, 2 mg, and up to 4 mg during this window. This is where the shift becomes undeniable. All three receptor pathways are now generating meaningful physiological effects.
GLP-1 activation slows gastric emptying, making food sit in the stomach longer and extending the satiety signal after meals. GIP activation improves insulin sensitivity and glucose disposal. Glucagon receptor activation begins mobilizing fat stores, particularly hepatic fat, for energy production. The triple mechanism is now working in concert.
Weight loss accelerates. By week 12, participants on the 4 mg dose in the Phase 2 trial had already lost approximately 7 to 9% of body weight (Jastreboff et al., NEJM, 2023). For a 250 lb person, that translates to 17 to 22 pounds. Cravings for calorie-dense food weaken noticeably. Many users report that the psychological relationship with food shifts from recreational to functional.
Diarrhea can appear during this escalation phase, affecting 18 to 34% of participants in clinical trials. It is usually transient, lasting 3 to 7 days after each dose increase. Maintaining hydration and fiber intake reduces the severity. For complete side effect management, see our retatrutide side effects guide.
Weeks 13-24: The Major Fat Loss Phase (4-8 mg)
This is the inflection zone. Weight loss that was steady becomes dramatic. The dose reaches 4 to 8 mg, and the glucagon receptor pathway is now producing its full thermogenic effect. Your body is burning stored fat at an elevated rate, particularly visceral fat around the organs and liver.
By week 24, the Phase 2 trial data showed clear dose-dependent separation:
| Dose Group | Weight Loss at 24 Weeks |
|---|---|
| Placebo | -1.6% |
| 1 mg | -7.2% |
| 4 mg (from 2 mg start) | -12.9% |
| 8 mg (from 2 mg start) | -17.3% |
| 12 mg (from 2 mg start) | -17.5% |
Source: Jastreboff et al., NEJM, 2023
These numbers are averages. Some participants exceeded 20% at 24 weeks on the higher doses. At this stage, changes are visible. Faces look leaner. Waistbands loosen. People who have not seen you in two months notice. The average waist circumference reduction in the retatrutide groups ranged from 6.5 to 14 cm by this point.
Energy levels often improve during this phase despite the caloric deficit. The glucagon-mediated fat oxidation provides a steady fuel source that compensates for reduced food intake. This is a key advantage of the triple agonist mechanism over pure GLP-1 drugs like semaglutide, which rely entirely on appetite suppression without the direct fat-burning component.
Weeks 25-48: Peak Results Period (8-12 mg)
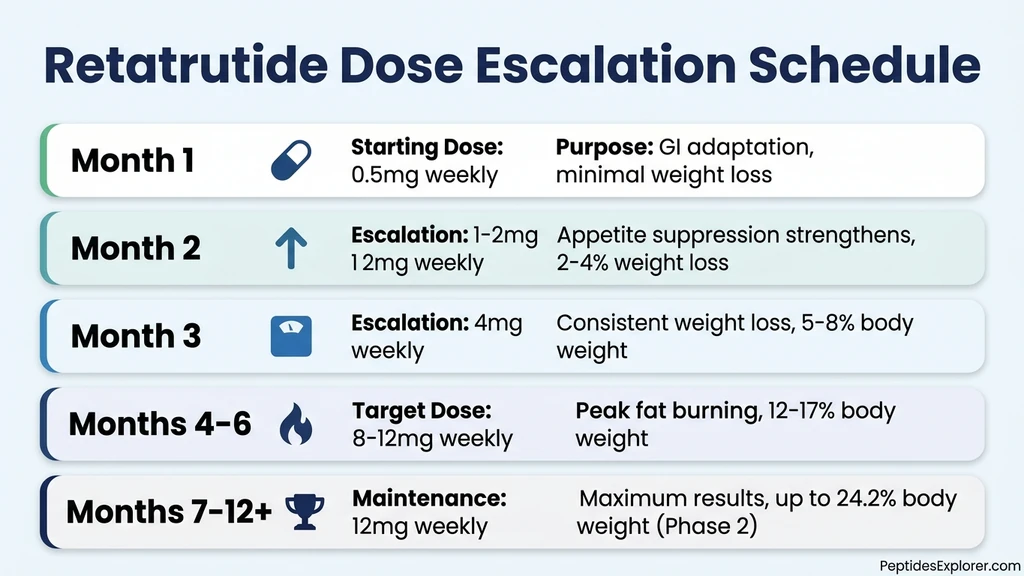
The dose reaches the full therapeutic range of 8 to 12 mg. All three receptor systems are operating at maximum pharmacological effect. This is the period that produced the headline-making Phase 2 results.
At 48 weeks on the 12 mg dose, participants achieved:
- Average weight loss: 24.2% of body weight
- Lost 5% or more: 100% of participants
- Lost 10% or more: 93% of participants
- Lost 15% or more: 83% of participants
- Waist circumference reduction: up to 19.6 cm
Source: Jastreboff et al., NEJM, 2023
For a 250 lb person, 24.2% represents 60.5 pounds. For a 300 lb person, 72.6 pounds. These are not outlier results. They are population averages from a controlled clinical trial with 338 participants.
The 8 mg dose group reached 22.8% weight loss at 48 weeks, only 1.4 percentage points behind the 12 mg group. This suggests that much of the benefit is captured at 8 mg, with diminishing returns at 12 mg that must be weighed against a higher side effect burden.
The weight loss trajectory at this stage is still declining but at a slower rate. The body approaches a new equilibrium where reduced caloric intake matches reduced caloric expenditure from a smaller body mass. The drug is still active. The mathematics have simply rebalanced. For strategies to optimize results during this phase, see how to take retatrutide.
Weeks 49-68: Maximum Recorded Effect (12 mg)
The Phase 3 TRIUMPH-4 trial, which reported results in December 2025, extended the treatment window to 68 weeks. At the 12 mg dose, participants lost an average of 28.7% of body weight, equivalent to 71.2 pounds for the average participant (Lilly Press Release, Dec 2025). The 9 mg dose produced 26.4% weight loss.
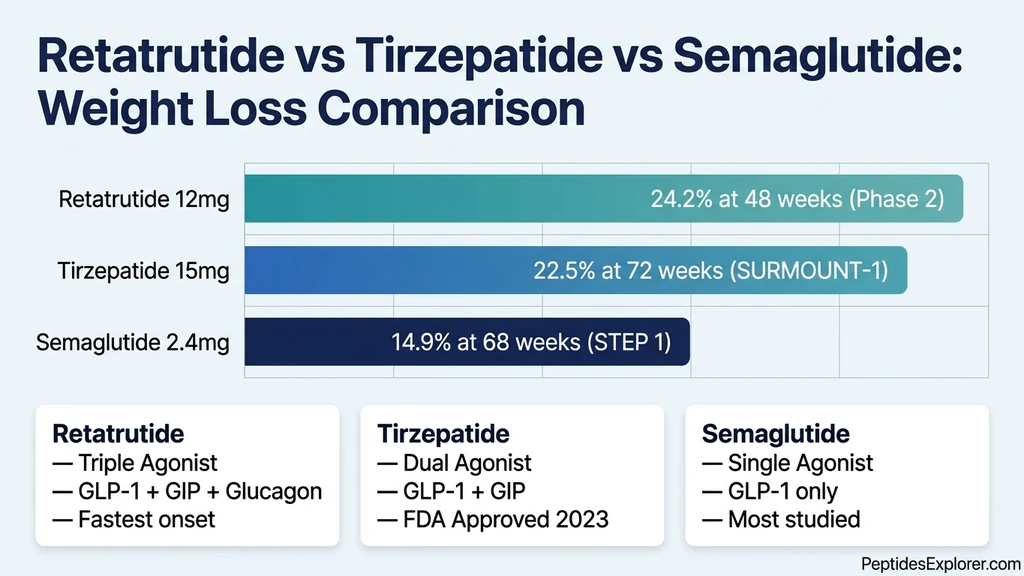
These numbers surpass every other anti-obesity medication tested in clinical trials. Tirzepatide at its maximum dose produced 22.5% at 72 weeks in SURMOUNT-1. Semaglutide at 2.4 mg produced 14.9% at 68 weeks in STEP 1. Retatrutide roughly doubled semaglutide's efficacy over the same duration.
Weight loss continued beyond week 48 but at a reduced rate. The additional 3 to 5 percentage points gained between weeks 48 and 68 represent the tail end of the response curve. Most participants had reached 80 to 90% of their total weight loss by week 48. The remaining weeks contributed meaningful but incremental additional loss.
The TRIUMPH-4 trial also demonstrated a separate benefit: patients with knee osteoarthritis experienced substantial pain relief alongside the weight loss, with clinically meaningful improvements in joint function scores. Reducing 70 pounds of mechanical load on arthritic knees produces predictable orthopedic benefits.
Why Retatrutide Works Faster Than Semaglutide and Tirzepatide
Retatrutide is not simply a stronger version of the same drug. It activates a receptor that neither semaglutide nor tirzepatide touches: the glucagon receptor. This third pathway creates a fundamentally different weight loss profile.
Semaglutide activates only GLP-1 receptors. The entire weight loss mechanism depends on appetite suppression and delayed gastric emptying. When you eat less, you lose weight. But the body's compensatory mechanisms, reduced metabolic rate and increased hunger hormones, fight back over time.
Tirzepatide adds GIP receptor activation to GLP-1. The dual mechanism improves insulin sensitivity and glucose disposal beyond what GLP-1 alone achieves. It produces roughly 50% more weight loss than semaglutide. For a detailed comparison, see our retatrutide vs. tirzepatide analysis.
Retatrutide adds glucagon receptor activation on top of both. Glucagon directly stimulates hepatic fat oxidation, forcing the liver to burn stored triglycerides for energy. It increases thermogenesis, the body's baseline energy expenditure from heat production. These are not appetite effects. They are direct metabolic effects that burn fat regardless of what or how much you eat.
| Medication | Receptors | Avg Weight Loss | Trial Duration | Mechanism |
|---|---|---|---|---|
| Semaglutide 2.4 mg | GLP-1 | 14.9% | 68 weeks (STEP 1) | Appetite suppression |
| Tirzepatide 15 mg | GLP-1 + GIP | 22.5% | 72 weeks (SURMOUNT-1) | Appetite + metabolic signaling |
| Retatrutide 12 mg | GLP-1 + GIP + Glucagon | 24.2% | 48 weeks (Phase 2) | Appetite + metabolic + fat oxidation |
The 24.2% at 48 weeks is particularly striking because tirzepatide needed 72 weeks to reach 22.5%. Retatrutide achieved more weight loss in less time. The glucagon component accelerates the onset and magnitude of fat burning, producing visible results earlier in the treatment course.
For a deeper explanation of the triple agonist mechanism, read how does retatrutide work.
The Phase 2 Trial Data: What the Numbers Actually Show
The pivotal Phase 2 trial (NCT04881706) enrolled 338 adults with obesity (BMI 30+) or overweight (BMI 27+) with at least one weight-related comorbidity. Participants were randomized to placebo or one of six retatrutide dose groups: 0.5 mg, 4 mg (starting from 2 mg), 4 mg (starting from 4 mg), 8 mg (starting from 2 mg), 8 mg (starting from 4 mg), or 12 mg (starting from 2 mg). Treatment lasted 48 weeks (Jastreboff et al., NEJM, 2023).
The dose escalation starting point mattered. Groups that started at 2 mg and escalated to their target dose had fewer GI side effects than groups that started at 4 mg. Weight loss was comparable between the two escalation speeds. This finding shaped the Phase 3 protocol, which uses the slower 2 mg starting dose.
Weight Loss by Dose Group at 48 Weeks
The full 48-week results paint a clear dose-response picture:
| Dose Group | Weight Loss (%) | Weight Loss (kg) | Lost 15%+ |
|---|---|---|---|
| Placebo | -2.1% | -2.1 kg | 9% |
| 0.5 mg | -8.7% | -8.7 kg | 19% |
| 4 mg (from 2 mg) | -17.1% | -17.4 kg | 60% |
| 8 mg (from 2 mg) | -22.8% | -23.6 kg | 75% |
| 12 mg (from 2 mg) | -24.2% | -24.8 kg | 83% |
Source: Jastreboff et al., NEJM, 2023
The jump from 0.5 mg to 4 mg nearly doubled the weight loss. The jump from 4 mg to 8 mg added another 5 percentage points. The jump from 8 mg to 12 mg added only 1.4 percentage points. This diminishing return at the highest dose is a key consideration for individual risk-benefit decisions.
Waist Circumference and Body Composition
Waist circumference reductions mirrored the weight loss pattern:
- Placebo: -2.6 cm
- 4 mg group: -12.8 cm
- 8 mg group: -16.4 cm
- 12 mg group: -19.6 cm
A 19.6 cm (7.7 inch) reduction in waist circumference represents a dramatic reshaping of the torso. Visceral fat, the metabolically dangerous fat surrounding organs, appears to be preferentially targeted by the glucagon component. This is consistent with glucagon's known role in hepatic fat mobilization.
For context, a 5 cm reduction in waist circumference is associated with meaningful reductions in cardiovascular risk. The retatrutide 12 mg group achieved nearly four times that threshold.
Safety Profile and Discontinuation Rates
The most common adverse events were gastrointestinal:
- Nausea: 24-43% (dose-dependent)
- Diarrhea: 18-34%
- Vomiting: 8-18%
- Constipation: 6-16%
These side effects were mostly mild to moderate and concentrated in the first 4 to 8 weeks of treatment, coinciding with dose escalation. Starting from a 2 mg dose rather than a 4 mg dose reduced the incidence of nausea by approximately 30%.
The Phase 3 TRIUMPH-4 trial reported discontinuation rates of 12 to 18% due to adverse events in the retatrutide groups, compared to 4% in placebo (Lilly Press Release, Dec 2025). This is higher than tirzepatide's Phase 3 discontinuation rate (approximately 7%) but must be weighed against the greater weight loss achieved.
For managing nausea on retatrutide and diarrhea on retatrutide, we have dedicated management guides.
Phase 3 TRIUMPH Trial Results: What We Know in 2026
Eli Lilly's Phase 3 program for retatrutide includes multiple trials under the TRIUMPH and TRANSCEND names. As of early 2026, the first Phase 3 results have been reported.
TRIUMPH-4 (December 2025): This trial enrolled adults with obesity or overweight and knee osteoarthritis. At 68 weeks, participants on retatrutide 12 mg lost an average of 28.7% of body weight (71.2 lbs). The 9 mg dose produced 26.4% weight loss. Both doses met primary and key secondary endpoints (Lilly Press Release, Dec 2025).
TRANSCEND-T2D-1 (2025): This trial assessed retatrutide in people with type 2 diabetes. The drug met primary and key secondary endpoints, delivering superior HbA1c reduction and weight loss at 40 weeks. Participants lost up to an average of 16.8% of body weight (36.6 lbs), which is lower than the non-diabetic population but still exceeds what semaglutide or tirzepatide achieve in diabetic cohorts.
Seven additional Phase 3 trials are expected to complete in 2026, covering obesity without comorbidities, obstructive sleep apnea, and additional diabetes populations. For the latest on regulatory timeline and availability, see when will retatrutide be available.
These Phase 3 results confirm and extend the Phase 2 data. The 28.7% weight loss at 68 weeks exceeds the 24.2% seen at 48 weeks in Phase 2, indicating that the drug continues producing weight loss well beyond the Phase 2 trial endpoint. For those researching how to obtain retatrutide, see our guide to getting retatrutide.
Factors That Speed Up or Slow Down Your Results
The clinical trial numbers are population averages. Your individual timeline will vary based on several controllable and uncontrollable factors.
Starting Weight and Body Composition
Heavier individuals lose more total pounds but a similar percentage of body weight. A person starting at 300 lbs who loses 24% drops 72 lbs. A person starting at 200 lbs loses 48 lbs at the same percentage. The absolute number differs, but the metabolic and health impact is proportionally equivalent.
Higher starting weights also produce faster initial scale movement because the caloric deficit created by appetite suppression is proportionally larger when baseline intake is higher. A person eating 3,500 calories daily who cuts to 2,000 creates a 1,500-calorie deficit. A person eating 2,200 who cuts to 1,500 creates only a 700-calorie deficit.
Insulin Resistance and Type 2 Diabetes
People with type 2 diabetes lose weight more slowly on all GLP-1 based medications, including retatrutide. The TRANSCEND-T2D-1 Phase 3 trial showed 16.8% weight loss in diabetic participants, compared to 28.7% in the non-diabetic TRIUMPH-4 population. Insulin resistance impairs the body's ability to mobilize and oxidize fat stores, even with glucagon receptor activation.
The good news: blood sugar control improves rapidly, often within the first 4 weeks. HbA1c reductions appear before significant weight loss occurs because GLP-1 and GIP directly improve beta cell function and insulin secretion. Weight loss is slower but still substantial and medically meaningful.
Diet Quality and Protein Intake
Retatrutide reduces how much you eat. It does not control what you eat. People who prioritize protein intake (0.7 to 1.0 g per pound of lean body mass) preserve muscle mass during rapid weight loss. Those who eat calorie-restricted but protein-poor diets lose more muscle, which slows metabolism and increases regain risk after treatment ends.
The caloric deficit from retatrutide is substantial, often 800 to 1,200 fewer calories daily at full dose. Without deliberate protein intake, the body catabolizes muscle tissue to meet amino acid demands. Resistance training combined with adequate protein produces the best body composition outcomes during treatment.
Physical Activity and Exercise
Exercise does not dramatically change the weight number on retatrutide because the drug already creates a large caloric deficit. However, exercise changes what that weight loss consists of. Resistance training preserves lean mass, improves insulin sensitivity independently of the drug, and accelerates visceral fat reduction. Cardiovascular exercise improves the metabolic and cardiovascular benefits that accompany weight loss.
The practical challenge is that many participants feel reduced energy during the first 4 to 8 weeks of treatment as the body adapts to lower caloric intake. Moderate exercise that respects energy levels is more sustainable than ambitious workout programs during dose escalation.
Dose Adherence and Escalation Timing
Missing doses disrupts the steady-state blood levels that produce consistent appetite suppression and fat oxidation. Retatrutide has a half-life of approximately 6 days, meaning levels drop meaningfully after a missed weekly injection. Consistent weekly dosing on the same day maintains the pharmacological pressure needed for continuous weight loss.
Advancing through the dose escalation on schedule, rather than staying at lower doses out of caution or fear of side effects, is also critical. The difference between 4 mg and 12 mg at 48 weeks is 7 percentage points of body weight, roughly 17 additional pounds for a 250 lb person. Dose optimization matters. Use our retatrutide dosage calculator to plan your escalation protocol.
When Retatrutide Seems Like It Is Not Working
Not everyone responds to retatrutide on the expected timeline. Before concluding the drug has failed, check these common causes of slow response.
1. You have not reached the therapeutic dose. If you are still at 0.5 mg or 1 mg, you are on a fraction of the dose that produced the clinical trial results. The 24.2% average was achieved at 12 mg. Most significant weight loss occurs after reaching 4 mg or higher. The first 8 to 12 weeks are adaptation, not the real treatment.
2. You are eating through the appetite suppression. Retatrutide reduces hunger signals, but it does not prevent eating. Emotional eating, habitual snacking, and social eating can override the pharmacological appetite suppression. If you are eating when not hungry, the drug is working but behavior is counteracting it.
3. Liquid calories are bypassing the mechanism. Retatrutide slows gastric emptying for solid food, extending satiety. Liquid calories (alcohol, sugary coffee drinks, smoothies, juice, soda) bypass this mechanism entirely. A daily 400-calorie coffee habit offsets a substantial portion of the caloric deficit the drug creates.
4. A concurrent medication is promoting weight gain. Insulin, sulfonylureas, beta-blockers, certain antidepressants (mirtazapine, olanzapine, paroxetine), and corticosteroids all promote weight gain through pathways that retatrutide does not fully counteract. A medication review with your prescriber may identify adjustable factors.
5. Your expectations exceed the clinical data. The average loss at 48 weeks is 24.2% at the highest dose. That means half of participants lost less. Some achieved 15 to 18%. That is still medically transformative, representing 40+ pounds for most people. Non-linear patterns are normal: 2 weeks of no change followed by a sudden 4-pound drop is typical. Track monthly trends, not daily fluctuations.
For the complete troubleshooting approach to maximizing your results, see our guide on how to take retatrutide.
What Happens When You Stop Retatrutide?
The clinical trial data from GLP-1 based medications consistently shows weight regain after discontinuation. The STEP 1 extension study for semaglutide showed participants regained two-thirds of lost weight within one year of stopping. No published discontinuation data exists for retatrutide yet because the Phase 3 trials are still ongoing, but the same pharmacological principles apply.
Retatrutide's half-life is approximately 6 days, similar to semaglutide. After the last injection, drug levels decline over 4 to 5 weeks. As levels drop, appetite returns to baseline. The glucagon-mediated fat oxidation ceases. The GIP-mediated metabolic improvements fade. Without the drug maintaining the caloric deficit and enhanced fat burning, the body's weight-regulatory systems push toward the pre-treatment set point.
This does not mean treatment is futile. The weight loss period provides a window to establish exercise habits, dietary patterns, and metabolic improvements (reduced liver fat, improved insulin sensitivity) that partially persist after stopping. However, the evidence across all GLP-1 medications suggests that most people will need ongoing treatment to maintain the full weight loss.
For context on what long-term treatment looks like with the current generation of drugs, see tirzepatide maintenance dose after weight loss. The principles of maintenance dosing will likely apply to retatrutide when it receives FDA approval.
How to Track Your Retatrutide Progress Accurately
The scale is unreliable for measuring daily progress. Body weight fluctuates by 2 to 5 pounds daily based on water retention, sodium intake, bowel contents, and hormonal cycles. Retatrutide's effect on gastric emptying can temporarily increase stomach contents, masking fat loss on the scale. Use these more reliable tracking methods.
Weekly weigh-ins under consistent conditions. Weigh yourself once per week, same day, same time (morning, after urination, before eating). Record the number without reacting to it. Evaluate trends over 4-week blocks, not individual readings.
Waist circumference. Measure at the navel with a flexible tape measure. Retatrutide targets visceral fat aggressively through the glucagon pathway, making waist circumference one of the most responsive metrics. A decrease in waist circumference with stable scale weight means you are losing fat and retaining or building muscle.
Progress photos. Take standardized front, side, and back photos every 4 weeks in the same lighting and clothing. Visual changes, particularly facial and abdominal, often precede scale changes because body recomposition is not captured by weight alone.
Lab work. Request bloodwork at baseline, 12 weeks, and 6 months. Track fasting glucose, HbA1c (if diabetic or pre-diabetic), lipid panel, liver enzymes (ALT, AST), and inflammatory markers (CRP). These numbers quantify metabolic improvement even when the scale stalls. Liver enzymes are particularly relevant because retatrutide's glucagon component directly reduces hepatic fat.
Clothing fit. The most underrated metric. If your belt moves in a notch, waistbands loosen, or shirts fit differently through the midsection, the drug is working. Inches lost around the waist are clinically more meaningful than pounds on the scale.
For proper preparation and injection technique, see our reconstitution guide for retatrutide.
Retatrutide vs. Other GLP-1 Medications: Speed of Results
All GLP-1 based medications suppress appetite within the first week. The differences appear in the magnitude and speed of weight loss over subsequent months. Retatrutide's glucagon component accelerates fat loss, producing more weight loss in less time than its predecessors.
| Metric | Retatrutide 12 mg | Tirzepatide 15 mg | Semaglutide 2.4 mg |
|---|---|---|---|
| Receptors activated | GLP-1 + GIP + Glucagon | GLP-1 + GIP | GLP-1 only |
| Weight loss at ~24 wks | 17.5% | ~12% | ~10% |
| Weight loss at ~48 wks | 24.2% | ~20% | ~13% |
| Peak weight loss (trial) | 28.7% (68 wks) | 22.5% (72 wks) | 14.9% (68 wks) |
| Time to 15% loss | ~24 weeks | ~36 weeks | ~52 weeks |
| GI side effect rate | 24-43% nausea | 18-33% nausea | 44% nausea |
Sources: Jastreboff et al., NEJM, 2023, Jastreboff et al., NEJM, 2022, Wilding et al., NEJM, 2021
The 15% weight loss threshold, often considered the benchmark for metabolic surgery-level results, is reached at approximately 24 weeks on retatrutide 12 mg, 36 weeks on tirzepatide 15 mg, and 52 weeks on semaglutide 2.4 mg. Retatrutide achieves this milestone roughly twice as fast as semaglutide.
For a detailed head-to-head comparison with tirzepatide, including cost, availability, and side effect profiles, see our retatrutide vs. tirzepatide comparison.
Frequently Asked Questions
How long does retatrutide take to work for weight loss?
Retatrutide suppresses appetite within the first week of treatment. Measurable weight loss appears by week 4. Significant results (10%+ body weight) are typically seen by weeks 12 to 16 as the dose escalates toward the therapeutic range of 8 to 12 mg. The Phase 2 trial showed 24.2% average weight loss at 48 weeks on the 12 mg dose. For the complete dosing protocol, see our retatrutide dosage guide.
How much weight can you lose on retatrutide in 3 months?
At 12 weeks (approximately 3 months), participants in the Phase 2 trial on higher doses had lost 7 to 12% of body weight. For a 250 lb person, that is 17 to 30 lbs. The exact amount depends on your dose, starting weight, diet, and metabolic status. By 3 months you are still in the dose escalation phase, so weight loss continues accelerating. Learn more about the triple agonist mechanism.
Is retatrutide faster than tirzepatide for weight loss?
Yes. Retatrutide reaches the 15% weight loss threshold in approximately 24 weeks, compared to 36 weeks for tirzepatide. At 48 weeks, retatrutide 12 mg produced 24.2% weight loss, while tirzepatide needed 72 weeks to reach 22.5%. The glucagon receptor component in retatrutide directly burns fat, accelerating results beyond what dual agonism achieves. See our full retatrutide vs. tirzepatide comparison.
Why am I not losing weight on retatrutide after 2 weeks?
At 2 weeks, you are on the lowest starting dose (0.5 mg), which is roughly one-twenty-fourth of the full 12 mg therapeutic dose. This starter dose exists for GI adaptation, not weight loss. The Phase 2 trial showed minimal separation from placebo during the first month at low doses. Weight loss accelerates as the dose escalates to 4 mg and above. For side effect management during escalation, see our retatrutide nausea guide.
What are the most common side effects of retatrutide?
The most common side effects are gastrointestinal: nausea (24-43%), diarrhea (18-34%), vomiting (8-18%), and constipation (6-16%). These are dose-related and concentrated during the escalation phase. Starting at a lower dose (2 mg escalation) reduces severity by approximately 30%. Most GI side effects are mild to moderate and resolve within 3 to 7 days after each dose increase. See our full retatrutide side effects guide.
Does retatrutide work better than semaglutide?
In clinical trials, retatrutide produced roughly double the weight loss of semaglutide in similar timeframes. Retatrutide 12 mg achieved 24.2% at 48 weeks, while semaglutide 2.4 mg reached 14.9% at 68 weeks. The difference comes from retatrutide's glucagon receptor activation, which directly burns fat through thermogenesis and hepatic fat oxidation, a pathway semaglutide does not engage. Compare all three drugs in our retatrutide dosage guide.
When will retatrutide be FDA approved?
Seven Phase 3 trials are expected to complete in 2026. Based on the typical FDA review timeline, approval could come in late 2026 or 2027, pending successful completion of all trials and the submission of a New Drug Application by Eli Lilly. For the latest regulatory updates and current access options, see when will retatrutide be available and how to get retatrutide.
How do I prepare and inject retatrutide?
Research-grade retatrutide requires reconstitution with bacteriostatic water before injection. The peptide is injected subcutaneously (under the skin) once weekly, typically in the abdomen. Proper reconstitution, storage, and injection technique are critical for maintaining potency and safety. See our how to reconstitute retatrutide guide for step-by-step instructions and our how to take retatrutide guide for administration best practices.
The Bottom Line
Retatrutide works in phases: appetite suppression within the first week, measurable weight loss by week 4, significant body composition changes by weeks 12 to 16, and peak results at 48 to 68 weeks. The Phase 2 trial documented 24.2% average body weight loss at 48 weeks on the 12 mg dose. The Phase 3 TRIUMPH-4 trial extended that to 28.7% at 68 weeks, the largest drug-induced weight loss ever recorded in clinical trials.
The single most important factor is patience through the dose escalation. The 0.5 mg starting dose is for GI adaptation, not weight loss. Each step up activates the triple agonist mechanism more fully: GLP-1 for appetite suppression, GIP for metabolic signaling, and glucagon for direct fat oxidation. The results from the clinical trials were achieved at 8 to 12 mg, not at the starting dose.
Use our retatrutide dosage calculator to plan your escalation protocol. For side effect management during the early weeks, see our guides on nausea and diarrhea. For reconstitution and injection technique, use our reconstitution guide. If you are comparing options, see our retatrutide vs. tirzepatide analysis.
Related articles: - How Does Retatrutide Work? — detailed triple agonist mechanism breakdown - Retatrutide Dosage Guide — complete dosing protocol with escalation schedule - Retatrutide Side Effects — full safety profile from Phase 2 and Phase 3 trials - Retatrutide vs. Tirzepatide — head-to-head comparison of dual vs. triple agonist - How to Get Retatrutide — current access options and procurement guide - When Will Retatrutide Be Available? — FDA timeline and Phase 3 trial updates - How to Reconstitute Retatrutide — step-by-step preparation instructions - How to Take Retatrutide — injection technique and administration guide - Retatrutide Nausea — managing the most common side effect - Retatrutide Diarrhea — GI side effect management strategies
Helpful Tools
Related Articles
How Does Retatrutide Work?
Retatrutide activates 3 receptors (GLP-1, GIP, glucagon) to reduce appetite, improve metabolism, and burn fat. 28.7% weight loss in trials.
GLP-3 Peptide: What It Really Is
GLP-3 does not exist. What clinics call 'GLP-3' is retatrutide (LY3437943), Eli Lilly's triple agonist targeting GLP-1, GIP, and glucagon receptors.
Retatrutide Availability & Timeline
Retatrutide is expected to receive FDA approval in mid-2027 with pharmacy availability in late 2027. Full timeline and Phase 3 trial status.
Retatrutide: Mechanism & Trial Results
Retatrutide (LY3437943) is a triple agonist weight loss drug by Eli Lilly. 28.7% weight loss in trials, 82% liver fat reduction. Not yet FDA-approved.