GLP-3 does not exist in human biology. Your body produces exactly two glucagon-like peptides from the proglucagon gene: GLP-1 and GLP-2. There is no third. The only organism with a true GLP-3 is the cartilaginous fish (sharks and rays), where it regulates ketone metabolism.
What clinics and social media call "GLP-3" is retatrutide (LY3437943), an investigational weight loss drug made by Eli Lilly. Retatrutide is a triple receptor agonist that targets GLP-1, GIP, and glucagon receptors simultaneously. The "3" in the nickname refers to the three receptors it activates, not a third glucagon-like peptide.
| Detail | Information |
|---|---|
| What people mean by "GLP-3" | Retatrutide (LY3437943) |
| Manufacturer | Eli Lilly and Company |
| Real mechanism | Triple agonist: GLP-1 + GIP + Glucagon |
| Does GLP-3 exist in humans? | No. Only GLP-1 and GLP-2 |
| Max weight loss in trials | 28.7% body weight at 68 weeks |
| Liver fat reduction | Up to 82% in MASLD substudy |
| FDA approved? | No (Phase 3 clinical trials, as of March 2026) |
| Expected FDA decision | Mid-to-late 2027 |
| Available at clinics? | Not legally (investigational only) |
This article explains why the term "GLP-3" spread, what GLP-1 and GLP-2 actually do, what retatrutide is and how it works, and what the clinical trial data shows. For the full drug overview, see our complete retatrutide guide. For side effect data, see our retatrutide side effects breakdown.
This is educational content. Consult a healthcare provider before starting any medication.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Why People Search for GLP-3
The term "GLP-3" started appearing on social media and telehealth marketing sites in late 2024. It gained traction for two reasons.
First, the naming pattern. Ozempic and Wegovy use semaglutide, a GLP-1 receptor agonist. That was the first generation. Mounjaro and Zepbound use tirzepatide, a dual GLP-1/GIP agonist. That was the second generation. Retatrutide targets three receptors. Calling it "GLP-3" follows the pattern of generational naming, even though the name is biochemically incorrect.
Second, marketing. Some compounding pharmacies and wellness clinics adopted the term because it sounds like a natural progression. "GLP-3" implies there is a third peptide in the GLP family, which makes the drug sound like a biological molecule rather than a synthetic pharmaceutical. This framing is misleading.
The correct classification: retatrutide is a synthetic triple receptor agonist. It is not a naturally occurring peptide. It is not the third member of the GLP family. It is a drug that activates three different receptors, one of which happens to be the GLP-1 receptor.
What GLP-1 and GLP-2 Actually Do
Your body produces both GLP-1 and GLP-2 from the same precursor protein, proglucagon, in the L-cells of your small intestine. They are released after you eat. Despite sharing a source, they do very different things.
GLP-1 (Glucagon-Like Peptide 1)
GLP-1 controls appetite and blood sugar. When released after a meal, it slows gastric emptying (food stays in your stomach longer, so you feel full), stimulates insulin secretion, suppresses glucagon release (which lowers blood sugar), and signals satiety to the brain.
This is the receptor targeted by every major weight loss drug on the market. Semaglutide (Ozempic, Wegovy) is a GLP-1 receptor agonist. Tirzepatide (Mounjaro, Zepbound) also activates GLP-1 alongside GIP. Retatrutide activates GLP-1 alongside GIP and glucagon.
Natural GLP-1 has a half-life of about 2 minutes. Your body breaks it down almost immediately through the enzyme DPP-4. Drug versions like semaglutide are engineered to resist DPP-4 breakdown, giving them half-lives of 5 to 7 days. That is why a single weekly injection works.
For a deeper look at how GLP-1 drugs work in practice, see our guide on how semaglutide works and our semaglutide dosage chart.
GLP-2 (Glucagon-Like Peptide 2)
GLP-2 has nothing to do with appetite or weight loss. It controls intestinal growth and repair. When released, it stimulates the growth of intestinal villi (the finger-like projections that absorb nutrients), increases blood flow to the gut, reduces intestinal inflammation, and enhances nutrient absorption.
The only FDA-approved GLP-2 drug is teduglutide (Gattex), used to treat short bowel syndrome. It helps patients who have lost large portions of their intestine absorb enough nutrients to survive without intravenous feeding.
GLP-2 is not relevant to weight loss. It is not targeted by retatrutide, tirzepatide, or semaglutide. No obesity drug in development targets the GLP-2 receptor.
Why There Is No GLP-3
The proglucagon gene encodes five peptides: glucagon, GLP-1, GLP-2, glicentin, and oxyntomodulin. That is the complete list in humans. There is no GLP-3 gene, no GLP-3 receptor, and no GLP-3 protein in mammalian biology.
The only true GLP-3 exists in cartilaginous fish (sharks, skates, and rays). In these animals, GLP-3 appears to regulate ketone body metabolism. It has no known function in humans because humans do not produce it.
When someone offers you "GLP-3" treatment, they are selling retatrutide under a made-up name.
What Retatrutide Actually Is
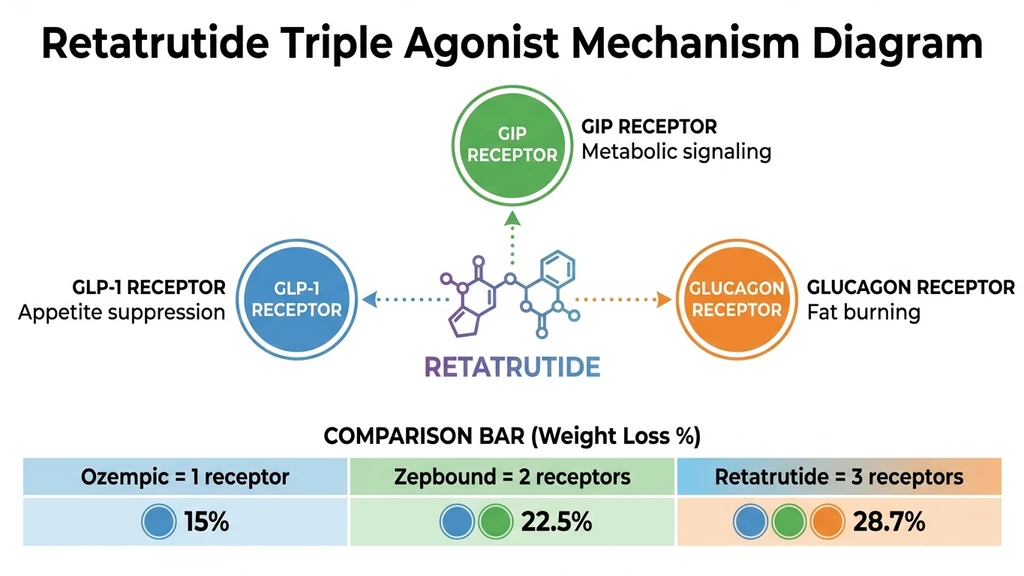
Retatrutide (LY3437943) is an investigational once-weekly injectable drug developed by Eli Lilly. It is a synthetic peptide made of 39 amino acids with a fatty acid chain attached to extend its half-life to approximately 6 days.
The drug activates three hormone receptors simultaneously. This triple agonist mechanism is what produced the highest drug-induced weight loss ever recorded in clinical trials. For a detailed mechanism breakdown, see our guide on how retatrutide works.
| Receptor | What It Does | Drug Examples |
|---|---|---|
| GLP-1 only | Suppresses appetite, slows digestion | Semaglutide (Ozempic, Wegovy) |
| GLP-1 + GIP | Appetite + metabolic signaling | Tirzepatide (Mounjaro, Zepbound) |
| GLP-1 + GIP + Glucagon | Appetite + metabolism + fat burning | Retatrutide (investigational) |
Each generation added a receptor. Each additional receptor produced roughly 6 to 8 extra percentage points of weight loss in clinical trials. One receptor averages about 15% weight loss. Two receptors average about 22.5%. Three receptors produced 28.7%.
The glucagon receptor is the new addition. Glucagon is traditionally known for raising blood sugar, which seems counterproductive. But glucagon also increases energy expenditure (your body burns more calories at rest) and drives fat oxidation, particularly in the liver. This is why retatrutide shows the strongest liver fat reduction data of any obesity drug.
The Three Receptors Explained
GLP-1 receptor: Reduces appetite. Slows gastric emptying. This is the receptor responsible for the "food noise" reduction that patients describe. It is also the receptor that causes nausea in the first weeks of treatment.
GIP receptor: Works alongside GLP-1 to amplify insulin sensitivity and improve post-meal glucose control. The GIP receptor also appears to enhance the weight loss signal from GLP-1, though the exact mechanism is still being studied. Tirzepatide proved that adding GIP to GLP-1 produces more weight loss than GLP-1 alone.
Glucagon receptor: Forces the body to burn stored fat for energy, especially visceral fat around organs. Increases resting metabolic rate by 50 to 100 calories per day. The glucagon receptor is why retatrutide reduced liver fat by 82% in the MASLD substudy, compared to roughly 55% with tirzepatide (Lilly Press Release, Dec 2025).
Clinical Trial Results
Retatrutide has been tested in two major trial phases. Both produced record-breaking results.
Phase 2 Trial (NEJM 2023)
The Phase 2 trial enrolled 338 adults with obesity (BMI 30+) or overweight with comorbidities (BMI 27+). Participants received retatrutide at doses of 1, 4, 8, or 12 mg weekly, or placebo, for 48 weeks.
Results at the 12 mg dose after 48 weeks:
- Average weight loss: 24.2% of body weight
- Weight was still decreasing at week 48 (no plateau reached)
- 100% of participants on the 12 mg dose lost at least 5% body weight
- Over 90% lost at least 10%
- Over 75% lost at least 15%
The trial was published in the New England Journal of Medicine (Jastreboff et al., NEJM 2023). Weight loss was still accelerating at the study's end, which led to the longer Phase 3 trial.
Phase 3 TRIUMPH-4 Results (2025-2026)
The TRIUMPH-4 trial enrolled 445 adults with obesity or overweight plus knee osteoarthritis. Participants received retatrutide at 9 mg or 12 mg weekly, or placebo, for 68 weeks. Eli Lilly announced initial results in December 2025, with full data presented in February 2026.
| Metric | Retatrutide 9 mg | Retatrutide 12 mg | Placebo |
|---|---|---|---|
| Average weight loss | 26.4% (64.2 lbs) | 28.7% (71.2 lbs) | 3.2% |
| Achieved 5%+ loss | ~95% | 97% | 31% |
| Achieved 10%+ loss | ~90% | 93% | 12% |
| Achieved 15%+ loss | ~82% | 85% | 5% |
| Achieved 20%+ loss | ~68% | 72% | 2% |
| Achieved 25%+ loss | ~48% | 54% | <1% |
| Achieved 30%+ loss | N/A | 39.4% | N/A |
| Achieved 35%+ loss | N/A | 23.7% | N/A |
These are the highest weight loss numbers ever recorded for any pharmaceutical. For comparison, semaglutide produces about 15% weight loss at its approved dose, and tirzepatide produces about 22.5%. A pooled meta-analysis of 878 patients across retatrutide trials found a mean difference of -14.33% body weight versus placebo (PMC12026077).
TRIUMPH-4 also measured knee osteoarthritis outcomes. Patients on the 12 mg dose experienced approximately 75% reduction in knee OA pain scores, compared to 40% with placebo. Twelve percent of patients on the 12 mg dose became completely pain-free. The 12 mg group also showed a 14.0 mmHg reduction in systolic blood pressure (Eli Lilly press release, Feb 2026).
For dosing details on how these results were achieved, see our retatrutide dosage guide.
Retatrutide vs Semaglutide vs Tirzepatide

The three drugs represent three generations of the same approach. Each one activates more receptors and produces more weight loss. The tradeoff is more side effects with each generation.
| Feature | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
| Brand names | Ozempic, Wegovy | Mounjaro, Zepbound | None (investigational) |
| Receptors | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Generation | 1st | 2nd | 3rd |
| Max weight loss | ~15% | ~22.5% | ~28.7% |
| Liver fat reduction | ~30% | ~55% | ~82% |
| Nausea rate | ~44% | ~31% | ~43% |
| Diarrhea rate | ~30% | ~21% | ~33% |
| FDA approved | Yes (2021) | Yes (2022) | No (Phase 3) |
| Monthly cost | $300-$1,350 | $299-$1,086 | Not available |
| Dosing | Once weekly | Once weekly | Once weekly |
| Max dose | 2.4 mg | 15 mg | 12 mg |
A direct head-to-head trial comparing retatrutide and tirzepatide is currently underway (NCT06662383, 800 patients). Results are expected in December 2026. Until then, all comparisons are cross-trial, meaning the patient populations and study designs differ.
For a more detailed analysis, see our full retatrutide vs tirzepatide comparison.
Beyond Weight Loss: Other Clinical Benefits
Retatrutide's triple receptor mechanism produces clinically meaningful effects that extend well beyond weight reduction. Several of these secondary benefits may lead to additional FDA indications.
Liver Disease (MASLD)
In the MASLD substudy, retatrutide reduced liver fat by 82% at 48 weeks. Over 85% of patients on higher doses achieved complete resolution of hepatic steatosis (Nature Medicine, 2024). These results surpass tirzepatide (~55% liver fat reduction) and semaglutide (~30%). The TRIUMPH-5 trial is testing retatrutide specifically for MASLD as a standalone indication.
Type 2 Diabetes
Retatrutide reduced HbA1c by 1.3 to 2.0 percentage points across doses in Phase 2 (Jastreboff et al., NEJM 2023, PMID: 37351564). The TRIUMPH-1 trial is evaluating retatrutide specifically for type 2 diabetes. The triple receptor mechanism improves glucose control through three pathways: GLP-1-driven insulin secretion, GIP-mediated insulin sensitivity, and glucagon-driven hepatic glucose regulation.
Knee Osteoarthritis
TRIUMPH-4 enrolled patients with obesity and knee osteoarthritis. At the 12 mg dose, knee OA pain scores dropped by approximately 75%, compared to 40% with placebo. Twelve percent of patients on 12 mg became completely pain-free. Weight loss alone accounts for part of this improvement, but the magnitude suggests additional anti-inflammatory mechanisms may be involved (Eli Lilly press release, Feb 2026).
Cardiovascular Risk Factors
Retatrutide produced significant improvements across multiple cardiovascular markers. Systolic blood pressure dropped 14.0 mmHg in the 12 mg group. Triglycerides, non-HDL cholesterol, and high-sensitivity C-reactive protein (hsCRP) all decreased substantially. These cardiometabolic benefits mirror and exceed those seen with tirzepatide and semaglutide.
Sleep Apnea
Eli Lilly is investigating retatrutide for obstructive sleep apnea in the TRIUMPH program (NCT05931367). Results have not yet been reported. Given that tirzepatide already demonstrated significant AHI reduction in sleep apnea trials, retatrutide's greater weight loss effect may produce even larger benefits.
Side Effects
Retatrutide side effects follow the same pattern as other GLP-1 drugs but occur at higher rates due to the additional receptor activation. Most are gastrointestinal and dose-dependent, meaning they are worst during dose escalation and improve over time.
Phase 3 TRIUMPH-4 data provided the most detailed safety profile to date. The table below compares rates across doses and against placebo.
| Side Effect | Retatrutide 9 mg | Retatrutide 12 mg | Placebo |
|---|---|---|---|
| Nausea | ~35% | 43.2% | 10.7% |
| Diarrhea | ~25% | 33.1% | 13.4% |
| Vomiting | ~15% | 20.9% | 0% |
| Constipation | ~20% | 25.0% | 8.7% |
| Dysesthesia | ~15% | 20.9% | 0.7% |
| Decreased appetite | ~14% | 17% | N/A |
| Injection site reactions | ~5% | 6% | N/A |
| Heart rate increase | 2-4 bpm | 2-4 bpm | N/A |
Dysesthesia is a new safety signal identified in Phase 3 that was not observed in Phase 2. It presents as abnormal skin sensations: tingling, tenderness, or heightened sensitivity to touch. In TRIUMPH-4, dysesthesia affected 20.9% of patients on the 12 mg dose versus just 0.7% on placebo. The condition was generally mild and did not lead to treatment discontinuation. Researchers suspect it is linked to glucagon receptor activity, as dysesthesia has not been reported with GLP-1-only or dual-agonist drugs (BioSpace/Eli Lilly press release, Feb 2026).
Discontinuation rates due to adverse events were dose-dependent. At 9 mg, 12.2% of patients discontinued. At 12 mg, 18.2% discontinued. Only 4.0% of placebo patients discontinued. These rates are notably higher than those seen with tirzepatide (5-7%) and semaglutide (7%), likely reflecting the additional glucagon receptor activation.
For complete side effect data and management strategies, see our retatrutide side effects guide.
When Will Retatrutide Be Available?
Retatrutide is not FDA-approved and cannot be legally prescribed as of March 2026. Any clinic offering "GLP-3" or "retatrutide" outside of a clinical trial is operating in a gray area at best.
| Milestone | Expected Date |
|---|---|
| Phase 3 TRIUMPH trials ongoing | 2025-2027 |
| FDA NDA submission | Late 2026 or early 2027 |
| FDA approval (if granted) | Mid-to-late 2027 |
| Pharmacy availability | Late 2027 or 2028 |
| Insurance coverage negotiations | 2028+ |
Eli Lilly is running several Phase 3 trials under the TRIUMPH program:
- TRIUMPH-1: Type 2 diabetes (efficacy and safety)
- TRIUMPH-3: Obesity without diabetes
- TRIUMPH-4: Obesity + knee osteoarthritis (results announced Dec 2025, full data Feb 2026)
- TRIUMPH-5: MASLD / fatty liver disease
The only legal way to access retatrutide today is through enrollment in one of these clinical trials. Trial sites are listed at clinicaltrials.gov. For a detailed timeline breakdown, see our guide on when retatrutide will be available.
If a clinic offers you retatrutide or "GLP-3" today, the product is either not retatrutide (it may be compounded semaglutide or tirzepatide labeled misleadingly) or it was obtained outside of legal channels. Exercise extreme caution. For guidance on accessing approved GLP-1 medications, see our guide on how to get retatrutide.
The Bigger Picture: Obesity Drug Pipeline
Retatrutide is not the only next-generation obesity drug in development. The pipeline is accelerating as pharmaceutical companies compete to produce more effective treatments.
| Drug | Mechanism | Developer | Max Weight Loss | Status |
|---|---|---|---|---|
| Semaglutide | GLP-1 | Novo Nordisk | ~15% | FDA approved |
| Tirzepatide | GLP-1 + GIP | Eli Lilly | ~22.5% | FDA approved |
| Retatrutide | GLP-1 + GIP + Glucagon | Eli Lilly | ~28.7% | Phase 3 |
| Orforglipron | Oral GLP-1 | Eli Lilly | ~14.7% | Phase 3 |
| Survodutide | GLP-1 + Glucagon | Boehringer | ~19% | Phase 3 |
| Amycretin | GLP-1 + Amylin | Novo Nordisk | ~22% | Phase 2 |
| CagriSema | GLP-1 + Amylin (combo) | Novo Nordisk | ~22.7% | Phase 3 |
The trend is clear. Each new drug either adds receptors or combines mechanisms to produce more weight loss. Retatrutide currently leads the field in raw efficacy, but direct comparison trials are still needed.
For patients currently seeking treatment, semaglutide and tirzepatide are the FDA-approved options. Use our semaglutide dosage calculator or tirzepatide dosage calculator to understand dosing.
Frequently Asked Questions
Is GLP-3 a real peptide?
No. GLP-3 does not exist in human biology. Humans produce only GLP-1 and GLP-2 from the proglucagon gene. The only organism with a true GLP-3 is cartilaginous fish (sharks and rays). When clinics or social media refer to 'GLP-3,' they mean retatrutide, a synthetic drug that activates three receptors.
What is the difference between GLP-1 and GLP-3?
GLP-1 is a real human hormone that controls appetite and blood sugar. 'GLP-3' is a marketing term for retatrutide, a drug that activates GLP-1, GIP, and glucagon receptors. GLP-1 drugs produce about 15% weight loss. Retatrutide (the so-called 'GLP-3') produces about 28.7% because it activates two additional receptors beyond GLP-1.
Can I get GLP-3 at a clinic right now?
No. Retatrutide (the drug called 'GLP-3') is not FDA-approved and cannot be legally prescribed outside of clinical trials as of March 2026. Any clinic offering 'GLP-3' is either mislabeling a different product or obtaining it outside legal channels. FDA approval is expected in mid-to-late 2027.
How much weight can you lose on GLP-3 (retatrutide)?
In Phase 3 trials, participants on the 12 mg dose of retatrutide lost an average of 28.7% of their body weight over 68 weeks. For a person starting at 250 pounds, that is roughly 71 pounds. Over 50% of participants lost more than 25% of their body weight.
Why do clinics call retatrutide GLP-3?
The nickname follows a generational pattern. Semaglutide targets one GLP receptor (GLP-1). Tirzepatide targets two receptors. Retatrutide targets three. Calling it 'GLP-3' implies a third-generation drug, even though the name is biochemically wrong. Some clinics use the term for marketing purposes because it sounds like a natural hormone.
Is retatrutide better than Ozempic or Zepbound?
Retatrutide produced more weight loss in trials (28.7%) than semaglutide/Ozempic (~15%) or tirzepatide/Zepbound (~22.5%). It also showed stronger liver fat reduction (86% vs 55% vs 30%). The tradeoff is higher side effect rates in some populations: nausea ranged from 26.5% (TRANSCEND-T2D-1) to 43% (TRIUMPH-4), and a tingling sensation called dysesthesia appeared in 4.4 to 20.9% depending on the trial. A direct head-to-head trial versus tirzepatide is underway with results expected in December 2026.
What receptors does retatrutide target?
Retatrutide targets three receptors: GLP-1 (appetite suppression and blood sugar control), GIP (metabolic signaling and insulin sensitivity), and glucagon (fat burning and increased energy expenditure). This triple agonist mechanism is why retatrutide produces more weight loss than drugs targeting one or two receptors.
When will retatrutide be FDA-approved?
Eli Lilly is expected to submit a New Drug Application in late 2026 or early 2027. If approved, the FDA decision could come in mid-to-late 2027, with pharmacy availability in late 2027 or 2028. Phase 3 TRIUMPH trials are still ongoing as of March 2026. The only legal way to access retatrutide now is through clinical trial enrollment.
The Bottom Line
"GLP-3" is a marketing term, not a molecule. No human cell produces it. No human receptor responds to it. The drug people actually mean is retatrutide, Eli Lilly's triple receptor agonist that produced 28.7% weight loss in Phase 3 trials by targeting GLP-1, GIP, and glucagon receptors simultaneously.
The clinical data is genuinely remarkable. Retatrutide outperforms every approved obesity drug by a significant margin. But it remains investigational. It is not FDA-approved. It is not available at legitimate pharmacies. Any offer of "GLP-3 treatment" in 2026 should be met with skepticism.
For patients who need treatment now, semaglutide and tirzepatide are the FDA-approved options with strong clinical evidence. Use our retatrutide dosage calculator to understand how the drug is dosed in clinical trials, or explore our complete retatrutide guide for the full picture.
This is educational content based on published clinical trial data and Eli Lilly press releases. It is not medical advice. Consult a healthcare provider before starting any weight loss medication.
Helpful Tools
Related Articles
How Does Retatrutide Work?
Retatrutide activates 3 receptors (GLP-1, GIP, glucagon) to reduce appetite, improve metabolism, and burn fat. 28.7% weight loss in trials.
How Long Does Retatrutide Take to Work?
Retatrutide suppresses appetite within days, produces weight loss by week 4, and delivers up to 24.2% loss at 48 weeks in trials.
Retatrutide: Mechanism & Trial Results
Retatrutide (LY3437943) is a triple agonist weight loss drug by Eli Lilly. 28.7% weight loss in trials, 82% liver fat reduction. Not yet FDA-approved.
Retatrutide Availability & Timeline
Retatrutide is expected to receive FDA approval in mid-2027 with pharmacy availability in late 2027. Full timeline and Phase 3 trial status.