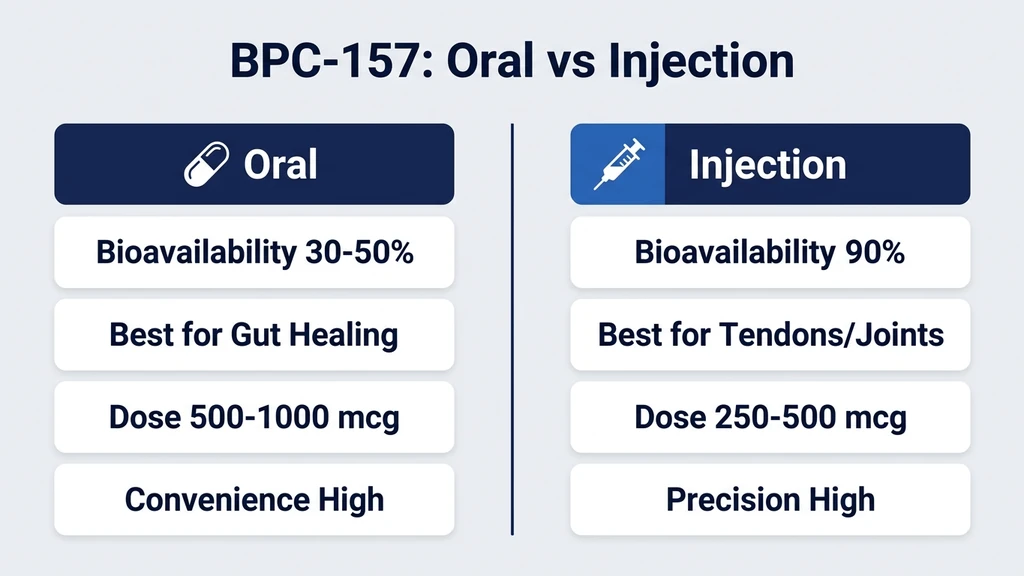
You have a bottle of BPC-157 capsules in one hand and a vial of lyophilized BPC-157 in the other, and you need to decide which route fits your situation. Injectable BPC-157 delivers 80-95% bioavailability and works best for localized musculoskeletal injuries, with effects starting within 1-2 weeks. Oral BPC-157 capsules offer 3-90% bioavailability (depending on formulation) and are superior for gastrointestinal healing, with easier daily use and no needles (He et al., Front Pharmacol, 2022). Choose injectable for tendon tears and joint pain. Choose oral for gut repair and systemic inflammation. Combine both routes for comprehensive recovery.
| Factor | Oral (Capsules) | Injectable (SubQ) |
|---|---|---|
| Bioavailability | ~3% (acetate), up to ~90% (arginate) | 80-95% |
| Best for | GI healing, systemic inflammation | Localized injuries, tendons, joints |
| Onset of effects | 2-4 weeks | 1-2 weeks |
| Side effects | GI (nausea, bloating) | Injection-site reactions, headaches |
| Daily convenience | High (swallow a pill) | Low (reconstitute, draw, inject) |
| Typical dose | 250-500 mcg twice daily | 250-500 mcg once or twice daily |
| Approximate monthly cost | $40-80 | $60-120 (peptide + supplies) |
| FDA status | Available as supplement (not approved) | Category 2 risk compound |
BPC-157 is not FDA-approved for any indication. No completed human clinical trial exists. Consult a healthcare provider before using any peptide.
For dosing protocols, use our BPC-157 dosage calculator. If you are new to taking BPC-157 orally, start with our oral guide.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
How Bioavailability Differs Between Routes
Bioavailability measures how much of a compound reaches systemic circulation in active form. Think of it like fuel efficiency in two different engines. A diesel truck (injectable) delivers 85% of its fuel energy to the wheels. A vintage carbureted car (oral acetate) loses most of its fuel to heat and exhaust, putting only 3% toward motion. A modern direct-injection engine (oral arginate) recovers much of that loss, reaching up to 90%.
Injectable BPC-157 Pharmacokinetics
Subcutaneous injection places BPC-157 directly into tissue beneath the skin, where it diffuses into local capillaries and enters systemic circulation. He et al. (2022) measured the pharmacokinetics in rats and dogs and found subcutaneous bioavailability exceeding 80% (He et al., Front Pharmacol, 2022).
Peak plasma concentration arrives within 15-30 minutes. The peptide reaches injured tissue at therapeutic levels quickly, which is why users with tendon tears and joint injuries often report noticeable improvement within 1-2 weeks. The half-life is relatively short, supporting twice-daily dosing for sustained tissue levels.
Injecting near the injury site concentrates BPC-157 in the local tissue. A user injecting subcutaneously in the abdomen to heal a knee tendon will still see benefit (systemic circulation delivers the peptide everywhere), but local concentration at the knee will be lower than a direct peritendinous injection. Learn where to inject BPC-157 for knee injuries for optimal placement.
Oral BPC-157 Pharmacokinetics
Oral BPC-157 faces three barriers before reaching systemic circulation: gastric acid degradation, enzymatic breakdown in the small intestine, and first-pass hepatic metabolism.
Standard acetate form: gastric acid destroys most of the peptide. He et al. (2022) measured oral bioavailability at approximately 3% in rats. The remaining 97% is broken down in the GI tract. This sounds catastrophic, but it has one advantage: the GI tract itself receives a massive local dose. For gut healing, this apparent weakness becomes a strength.
Arginate form: the arginine salt stabilizes BPC-157 at stomach pH, protecting it from acid degradation. Patent data (WO2014142764A1) claims oral bioavailability near 90%. Independent peer-reviewed verification of this figure is limited, but the mechanism (pH-buffered salt protection) is pharmacologically sound.
Peak plasma levels for oral BPC-157 arrive 60-90 minutes after ingestion, roughly 3-4x slower than subcutaneous injection. This gradual absorption produces a lower peak but longer duration of systemic exposure. For side effect implications, see BPC-157 capsule side effects.
What About Nasal Spray and Sublingual?
Two alternative routes exist. Nasal spray BPC-157 bypasses first-pass metabolism by absorbing through nasal mucosa directly into systemic circulation. Bioavailability estimates range from 20-40%, placing it between oral acetate and injectable. It may be useful for users who cannot inject but need higher bioavailability than capsules provide. See our BPC-157 nasal spray guide for protocols.
Sublingual administration (dissolving BPC-157 under the tongue) also bypasses the GI tract partially. Absorption through the sublingual mucosa is faster than capsules but less reliable than injection. No pharmacokinetic study has specifically measured sublingual BPC-157 bioavailability.
When to Choose Oral BPC-157
Oral BPC-157 is the right choice in three primary scenarios.
Gut Healing and GI Conditions
Oral BPC-157 delivers the peptide directly to the tissue you want to heal. For gastric ulcers, intestinal permeability ("leaky gut"), IBS symptoms, and NSAID-induced GI damage, the oral route is pharmacologically optimal.
Sikiric et al. demonstrated that oral BPC-157 heals experimental gastric ulcers, cytoprotects against ethanol-induced mucosal damage, and stabilizes intestinal tight junctions (Sikiric et al., Curr Pharm Des, 2011). Chang et al. showed that BPC-157 rescues NSAID-induced intestinal permeability disruption (Chang et al., Curr Pharm Des, 2020).
The low systemic bioavailability of acetate capsules (~3%) is irrelevant for GI healing because the therapeutic target is the gut lining itself. The 97% that gets "destroyed" in the GI tract is actually performing local work on mucosal tissue along the way.
Systemic Inflammation and General Wellness
Users seeking general anti-inflammatory support without a specific injury site benefit from oral BPC-157's sustained, low-level systemic absorption. The arginate form provides higher bioavailability for this purpose.
Oral BPC-157 interacts with the brain-gut axis, modulating neurotransmitter systems (dopamine, serotonin, NO) through enteric nervous system signaling (Sikiric et al., Curr Neuropharmacol, 2017). This makes oral administration particularly relevant for conditions where gut-brain signaling plays a role.
When Injections Are Not Available or Practical
Injectable BPC-157 was classified as a category 2 risk compound by the FDA, restricting compounding pharmacy access. Oral BPC-157 supplements remain available without this restriction.
Capsules also suit users who travel frequently (no syringes to carry or explain), have needle phobia, or prefer the simplicity of swallowing a pill. Compliance is higher with oral dosing: a capsule with breakfast requires no preparation time, no sterile technique, and no injection site management. For details on BPC-157 oral pills, see our product guide.
When to Choose Injectable BPC-157
Injectable BPC-157 outperforms oral for three categories of localized healing.
Tendon and Ligament Injuries
Gwyer et al. (2019) reviewed BPC-157's musculoskeletal effects and found strong evidence for tendon, ligament, and bone healing in animal models (Gwyer et al., J Complement Integr Med, 2019). BPC-157 upregulates growth hormone receptor expression in tendon fibroblasts, accelerating collagen synthesis and structural repair (Chang et al., J Mol Histol, 2011).
Injecting subcutaneously near the injured tendon delivers a high local concentration directly to the repair site. A user with an Achilles tendon injury benefits from injecting 250-500 mcg into the tissue above the tendon rather than taking a capsule that delivers 3% systemically and near-zero concentration at the ankle.
Joint Pain and Arthritis
Joint capsule inflammation responds to local peptide delivery. Subcutaneous injection near an inflamed joint (not into the joint itself) creates a gradient of BPC-157 concentration that diffuses into the synovial space over hours.
Users with knee osteoarthritis, shoulder bursitis, or hip joint pain report faster onset with injectable BPC-157 (1-2 weeks) compared to oral (3-4 weeks). The difference is concentration-dependent: the injured joint receives orders of magnitude more peptide via local injection than via oral absorption and systemic distribution.
Muscle Tears and Post-Surgical Recovery
Post-operative healing and acute muscle tears benefit from the high bioavailability and rapid onset of injectable BPC-157. The peptide promotes angiogenesis (new blood vessel formation) at the injection site, which accelerates nutrient delivery to damaged tissue.
For post-surgical recovery, some clinicians suggest injecting BPC-157 near (not into) the surgical wound starting 48-72 hours after the procedure. This approach is based on animal wound-healing studies, not human surgical trials. Consult your surgeon before adding any peptide to a post-operative protocol.
Combination Protocol: Using Both Routes Together
The most comprehensive approach uses oral BPC-157 for systemic and GI support while injecting near a specific injury site for localized healing.
Morning: 250-500 mcg oral BPC-157 (capsule with breakfast). This provides GI cytoprotection, brain-gut axis modulation, and baseline systemic peptide levels throughout the day.
Evening: 250-500 mcg injectable BPC-157 subcutaneously near the injury site. This delivers concentrated local healing to the specific tissue that needs repair.
Total daily dose: 500-1,000 mcg across both routes. This stays within the range supported by animal safety data. The 2025 human IV pilot used 20 mg (20,000 mcg) with no adverse events, providing a large safety margin (Altern Ther Health Med, 2025).
| Protocol Phase | Oral Dose | Injectable Dose | Duration |
|---|---|---|---|
| Loading (week 1-2) | 250 mcg AM | 500 mcg PM near injury | 14 days |
| Standard (week 3-6) | 500 mcg AM | 250 mcg PM near injury | 28 days |
| Maintenance (week 7-8) | 500 mcg AM | None | 14 days |
| Washout | None | None | 14-28 days |
This combination protocol has no published clinical validation. It is based on pharmacological logic (different routes serving different therapeutic targets) and community experience.
Cost Comparison: Oral vs Injectable
Cost varies by formulation, source, and dosing protocol. These estimates reflect typical 2026 market pricing for research-grade products.
| Cost Factor | Oral Capsules | Injectable |
|---|---|---|
| Peptide cost (30-day supply) | $40-80 | $60-100 |
| Bacteriostatic water | Not needed | $8-15 |
| Syringes (30-day supply) | Not needed | $10-20 |
| Alcohol swabs | Not needed | $5-8 |
| Total monthly cost | $40-80 | $83-143 |
| Cost per mcg delivered systemically (acetate) | High (~$0.08/mcg reaching blood) | Low (~$0.002/mcg reaching blood) |
| Cost per mcg delivered systemically (arginate) | Lower (~$0.003/mcg reaching blood) | Low (~$0.002/mcg reaching blood) |
The raw capsule price is lower, but cost-per-effective-mcg tells a different story for acetate capsules. With 3% bioavailability, you need roughly 8,300 mcg orally to deliver the same 250 mcg systemically that a single injection provides. Arginate capsules close this gap dramatically.
For GI healing, cost-per-effective-mcg is misleading because systemic absorption is not the goal. The local effect on gut tissue occurs at full oral dose regardless of bioavailability. If your target is the GI tract, oral capsules are both cheaper and more effective.
For supplies needed with injectable protocols, see our peptide reconstitution calculator and guide on how to reconstitute peptides.
Side Effect Comparison by Route
Each route produces a distinct side effect profile. The differences trace directly to pharmacokinetics.
| Side Effect | Oral | Injectable | Why |
|---|---|---|---|
| Nausea | Common (20-30%) | Rare (<5%) | Direct GI mucosal contact |
| Diarrhea | Common (first week) | Rare | Local intestinal effect |
| Headache | Uncommon (5-10%) | More common (15-20%) | Faster vasodilation via NO system |
| Injection-site pain | N/A | Common (25%) | Needle trauma |
| Injection-site redness | N/A | Common (15-20%) | Local immune response |
| Dizziness | Rare | Uncommon (10%) | Higher peak plasma levels |
| Bloating | Common (first week) | Rare | GI motility changes |
| Insomnia | Possible | Possible | Dopamine modulation (both routes) |
Oral users experience more GI effects that resolve within 5-7 days. Injectable users deal with site reactions that recur with each injection but are typically mild. For capsule-specific details, see our BPC-157 capsule side effects guide. For headache concerns, see does BPC-157 cause headaches.
Neither route has produced serious adverse events in published research (Sikiric et al., Pharmaceuticals, 2024).
Condition-by-Condition Recommendation Matrix
This matrix matches specific conditions to the optimal BPC-157 route based on pharmacological reasoning and available evidence.
| Condition | Recommended Route | Rationale |
|---|---|---|
| Gastric ulcer | Oral | Direct mucosal contact, local cytoprotection |
| Leaky gut / intestinal permeability | Oral | Tight junction stabilization requires local delivery |
| IBS / IBD symptoms | Oral | GI-specific mechanisms, brain-gut axis |
| NSAID-induced GI damage | Oral | Direct counteraction at mucosal level |
| Achilles tendon tear | Injectable (near site) | Local concentration critical for tendon healing |
| Knee osteoarthritis | Injectable (near joint) | Periarticular delivery, faster onset |
| Rotator cuff injury | Injectable (near site) | Growth hormone receptor upregulation at tendon |
| Post-surgical wound | Injectable (near wound) | Local angiogenesis, accelerated repair |
| Muscle strain | Injectable (near site) | Direct delivery to damaged fibers |
| General inflammation | Oral (arginate preferred) | Systemic bioavailability, convenience |
| Gut + musculoskeletal (both) | Oral AM + Injectable PM | Dual-route covers both targets |
These recommendations extrapolate from animal studies. No human clinical trial has compared oral vs injectable BPC-157 for any specific condition. Consult a physician before starting any peptide protocol. For comprehensive information on BPC-157 benefits, see our detailed guide.
Regulatory Status: Why the Route Matters Legally
The FDA classified injectable BPC-157 as a category 2 risk compound, restricting compounding pharmacies from producing it. This regulatory action reduced the availability of injectable BPC-157 through legal clinical channels.
Oral BPC-157 supplements fall under a different regulatory framework. Sold as dietary supplements rather than compounded drugs, capsule-form BPC-157 remains available for purchase without a prescription. This regulatory asymmetry is the primary reason many users have shifted from injectable to oral BPC-157 since 2024.
The distinction is legal, not pharmacological. The BPC-157 molecule is identical in both forms. Regulatory status does not reflect a safety judgment: the FDA has not approved BPC-157 for any indication regardless of administration route. Neither form is banned outright. The regulatory difference affects where you can legally obtain each form, not whether you can possess it.
Frequently Asked Questions
Is oral BPC-157 as effective as injectable?
For GI healing, oral may be more effective because it delivers the peptide directly to the gut lining. For musculoskeletal injuries (tendons, joints, muscles), injectable is significantly more effective due to 80-95% bioavailability vs 3% for oral acetate. Oral arginate (up to 90% bioavailability) narrows this gap for systemic conditions. The best route depends on your specific goal. See the BPC-157 dosage calculator for protocol planning.
Can I switch from injectable to oral BPC-157 mid-cycle?
Yes. No washout period is needed between routes. If switching to oral for the remaining cycle, start at 250 mcg once daily to assess GI tolerance before increasing. Expect a 1-2 week adjustment period where systemic peptide levels may be lower (if using acetate capsules) until the oral dose stabilizes. Effects on localized injuries may slow temporarily. See our how to take BPC-157 guide.
What is BPC-157 arginate and is it better than standard capsules?
BPC-157 arginate is an arginine salt formulation that resists gastric acid degradation. Patent data claims oral bioavailability near 90% vs ~3% for standard acetate capsules. If accurate, arginate delivers systemic peptide levels closer to injectable BPC-157. Independent peer-reviewed PK data is limited. Arginate capsules cost roughly 2-3x more than acetate and may cause fewer GI side effects due to less gastric degradation.
How long does oral BPC-157 take to work compared to injectable?
Injectable BPC-157 users typically report noticeable effects within 1-2 weeks for localized injuries. Oral BPC-157 (acetate) takes 2-4 weeks for systemic effects and 1-2 weeks for GI-specific improvements. Oral arginate may match injectable timelines for systemic conditions. Individual response varies considerably. Some users feel nothing for 3 weeks, then notice rapid improvement.
Can I take oral BPC-157 and inject on the same day?
Yes. The combination protocol uses oral BPC-157 in the morning for systemic and GI support, plus injectable BPC-157 near an injury site in the evening. Total daily dose ranges from 500-1,000 mcg across both routes. The 2025 human IV pilot used 20 mg (20,000 mcg) with no adverse events, suggesting substantial safety margin above these combined doses.
Does injectable BPC-157 hurt?
Subcutaneous injection with a 29-31 gauge insulin syringe produces mild, brief discomfort comparable to a light pinch. About 25% of users report some injection-site pain, and 15-20% notice redness lasting 1-2 hours. Proper technique reduces both. Injecting slowly (5-10 seconds per injection) and rotating sites minimizes discomfort over multi-week protocols. See how to inject BPC-157 for technique.
Why is injectable BPC-157 harder to get than capsules?
The FDA classified injectable BPC-157 as a category 2 risk compound, restricting compounding pharmacies from producing it. Oral BPC-157 supplements are regulated as dietary supplements, not compounded drugs, and remain available without a prescription. The regulatory distinction is legal, not based on safety data. The molecule is identical in both forms.
What is the cheapest effective BPC-157 route?
For GI healing, oral acetate capsules at $40-80/month are cheapest and most effective (low systemic bioavailability is irrelevant for gut targets). For systemic or localized healing, injectable BPC-157 at $83-143/month delivers the most peptide per dollar spent. Oral arginate at $80-150/month approaches injectable effectiveness at similar cost. Use our BPC-157 dosage calculator to model your specific protocol costs.
The Bottom Line
The route you choose should match the tissue you want to heal. Oral BPC-157 excels for GI conditions: the low systemic bioavailability of acetate capsules is a feature when the gut lining is the target. Injectable BPC-157 excels for localized injuries: tendons, joints, and muscles benefit from direct, high-concentration delivery near the damage site. Combining both routes covers both targets simultaneously.
Bioavailability data from He et al. (2022) and patent filings for arginate formulations give a clear pharmacokinetic framework for this decision. But no human clinical trial has directly compared oral vs injectable BPC-157 outcomes for any condition. Every recommendation here extrapolates from animal research and mechanism-based reasoning.
Plan your oral or injectable protocol with our BPC-157 dosage calculator. For injection technique, see how to inject BPC-157. For reconstitution volumes, use the peptide reconstitution calculator. Review how to take BPC-157 for a complete overview of all administration routes. For stacking with TB-500 across both routes, see our BPC-157 + TB-500 dosage guide.
Related Articles
How to Take BPC-157: Dosage and Timing
How to take BPC-157: inject 250-500 mcg subcutaneously near the injury once daily for 4-8 weeks. Reconstitution, dosing, and cycle length.
BPC-157 Capsule Side Effects: Oral User Guide
BPC-157 capsule side effects: nausea in 20-30% of users, diarrhea days 1-5, fewer headaches than injectable. Acetate vs arginate compared.
BPC-157 Oral Pills & Capsules: Do They Work?
BPC-157 oral pills deliver 250-500 mcg to the gut. Oral vs injectable vs nasal comparison, dosing protocols, and sourcing guidance.
Peptides for Bodybuilding (2026)
Best peptides for bodybuilding: BPC-157, TB-500, MK-677, CJC-1295/Ipamorelin, HGH Fragment 176-191. Dosages and stacking protocols.