You are researching BPC-157, a 15-amino-acid peptide from human gastric juice, and you want to know what the data says for women specifically. The strongest evidence covers gut healing, joint and tendon repair, wound recovery, and interstitial cystitis, where 10 of 12 women reported complete symptom resolution in the only female-specific human trial (Moghalu et al., 2024).
Women carry 60% of autoimmune disease diagnoses. IBS rates run 1.5 to 3 times higher in women than men (Lovell & Ford, 2012). Joint pain affects up to 50% of postmenopausal women as estrogen declines (Magliano, 2010). These are the exact categories where BPC-157 shows its strongest preclinical results.
| Category | Key Finding | Evidence Level |
|---|---|---|
| Gut healing (IBS, leaky gut, IBD) | Repairs intestinal barriers, counters NSAID damage | 20+ animal studies, Phase II IBD trials |
| Interstitial cystitis | 83% complete symptom resolution | Human pilot (12 women) |
| Joint and tendon repair | Accelerates collagen synthesis and fibroblast growth | 36 preclinical studies, 1 human pilot |
| Wound healing and skin | Increases collagen deposition and angiogenesis | 20+ animal models |
| Hormonal interference | No direct effect on estrogen, progesterone, or testosterone | Animal and mechanistic data |
| Pregnancy/breastfeeding safety | Zero data. Both contraindicated. | No studies exist |
BPC-157 is not hormonal. It does not alter estrogen, progesterone, or testosterone. Safety during pregnancy and breastfeeding is unstudied, and both are contraindicated. All evidence beyond three small human pilots comes from animal models. Use the BPC-157 Dosage Calculator once you understand the evidence below.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Gut Healing: The Strongest Evidence for Women
GI disorders hit women harder. IBS prevalence in women runs 1.5 to 3 times higher than in men (Lovell & Ford, 2012). Inflammatory bowel disease flares correlate with menstrual cycle phases. Stress-related gut dysfunction tracks with the higher anxiety prevalence in female populations.
BPC-157 targets multiple GI repair pathways simultaneously. The peptide entered Phase II clinical trials for inflammatory bowel disease under the designations PL-10, PLD-116, and PL 14736 (Sikiric et al., 2006).
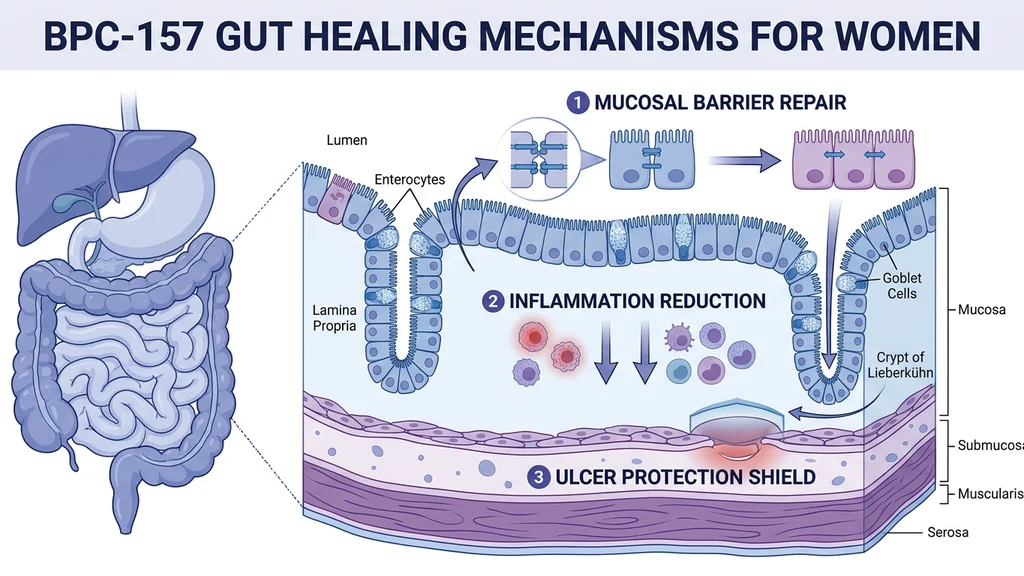
Intestinal Barrier Repair (Leaky Gut)
Think of your intestinal lining like grout between tiles. The tiles are epithelial cells; the grout is tight junction proteins that seal the gaps. NSAIDs dissolve the grout. BPC-157 rebuilds it.
Chang et al. (2020) showed that BPC-157 rescued NSAID-induced cytotoxicity by stabilizing intestinal permeability and restoring cytoprotection (PubMed). The peptide physically reinforces the tight junction proteins that hold gut lining cells together.
This matters if you rely on ibuprofen for menstrual cramps or chronic pain. NSAIDs rank among the most common causes of drug-induced gut barrier damage. BPC-157 counteracted gastrointestinal, liver, and brain lesions caused by diclofenac in animal models (Ilic et al., 2011).
A comprehensive review classified BPC-157 as a broad-spectrum NSAID antidote, counteracting damage from ibuprofen, diclofenac, and other common painkillers (Sikiric et al., 2013). For more options, see our guide to peptides for gut health.
Inflammatory Bowel Disease and Ulcer Protection
BPC-157 healed colocutaneous fistulas in rats with IBD, outperforming sulfasalazine (a standard IBD drug) and beating corticosteroids, which worsened outcomes (Cesarec et al., 2013). It accelerated healing of ileoileal anastomosis, the surgical reconnection of intestinal segments (Sever et al., 2009).
In short bowel syndrome models, BPC-157 increased villus height, crypt depth, and muscle thickness while promoting consistent weight gain above preoperative values (Lojo et al., 2009).
| GI Condition | BPC-157 Effect (Animal Models) | Source |
|---|---|---|
| Leaky gut / intestinal permeability | Stabilized tight junctions, reversed NSAID damage | Chang et al., 2020 |
| Inflammatory bowel disease | Healed fistulas, outperformed sulfasalazine | Cesarec et al., 2013 |
| Gastric ulcers | Anti-ulcer effect across multiple models | Sikiric et al., 2011 |
| Short bowel syndrome | Increased villus height, improved anastomosis strength | Lojo et al., 2009 |
| NSAID-induced GI lesions | Counteracted diclofenac and ibuprofen toxicity | Sikiric et al., 2013 |
Joint Pain, Tendons, and Musculoskeletal Recovery
Joint pain is one of the most common complaints during perimenopause and postmenopause. Estrogen decline reduces synovial fluid production, accelerates cartilage degradation, and weakens tendons. Up to 50% of women report new or worsened joint symptoms during the menopausal transition.
BPC-157 targets several of these pathways. Animal studies show it accelerates tendon and ligament repair through enhanced fibroblast proliferation, collagen synthesis, and activation of the FAK-paxillin signaling pathway (Chang et al., 2011). For a full comparison, see our guide to peptides for joint pain.
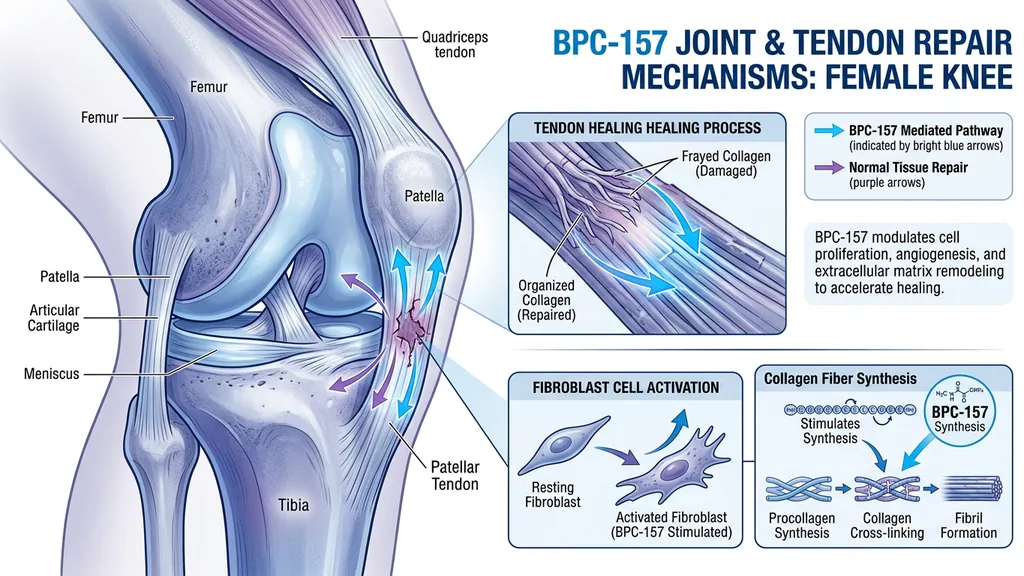
Tendon and Ligament Repair
Staresinic et al. (2003) showed BPC-157 accelerated healing of transected rat Achilles tendons and stimulated tendocyte growth in vitro (PubMed). Treated tendons showed higher load to failure, better Young's modulus of elasticity, and superior fibroblast formation with increased collagen deposition.
A follow-up study confirmed BPC-157 enhances growth hormone receptor expression in tendon fibroblasts, providing a mechanism for the observed collagen production increases (Chang et al., 2014).
A 2025 systematic review of 36 studies spanning 1993 to 2024 concluded that BPC-157 consistently improved outcomes in muscle, tendon, ligament, and bone injury models (Vasireddi et al., 2025). These findings are relevant for women dealing with rotator cuff tears, ACL injuries (women have 2 to 8 times higher ACL tear rates than men in comparable sports), plantar fasciitis, and postpartum connective tissue laxity.
Human Knee Pain Data
The only musculoskeletal human study tested intra-articular BPC-157 for chronic knee pain. Of 16 patients, 14 (87.5%) experienced significant pain relief. Among the 12 who received BPC-157 alone, 11 (91.6%) improved. Seven of those 12 maintained relief for over six months (Lee et al., 2021).
Small retrospective study, no control group. Encouraging but preliminary. No breakdown by sex was published.
| Study | Population | Intervention | Result |
|---|---|---|---|
| Lee et al., 2021 | 16 patients, chronic knee pain | Intra-articular BPC-157 injection | 87.5% significant improvement |
| Moghalu et al., 2024 | 12 women, ages 39-76 | Intravesical BPC-157 (10 mg) | 83% complete symptom resolution |
Skin, Wound Healing, and Collagen Production
BPC-157 accelerates wound healing through multiple overlapping mechanisms: increased collagen deposition, enhanced angiogenesis (new blood vessel formation), faster granulation tissue development, and improved epithelial repair (Seiwerth et al., 1997).
Women lose roughly 30% of their skin collagen in the first five years after menopause. The rate then stabilizes at about 2% per year. BPC-157 has not been studied specifically for anti-aging skin benefits, but its collagen-stimulating properties in wound models suggest potential relevance.
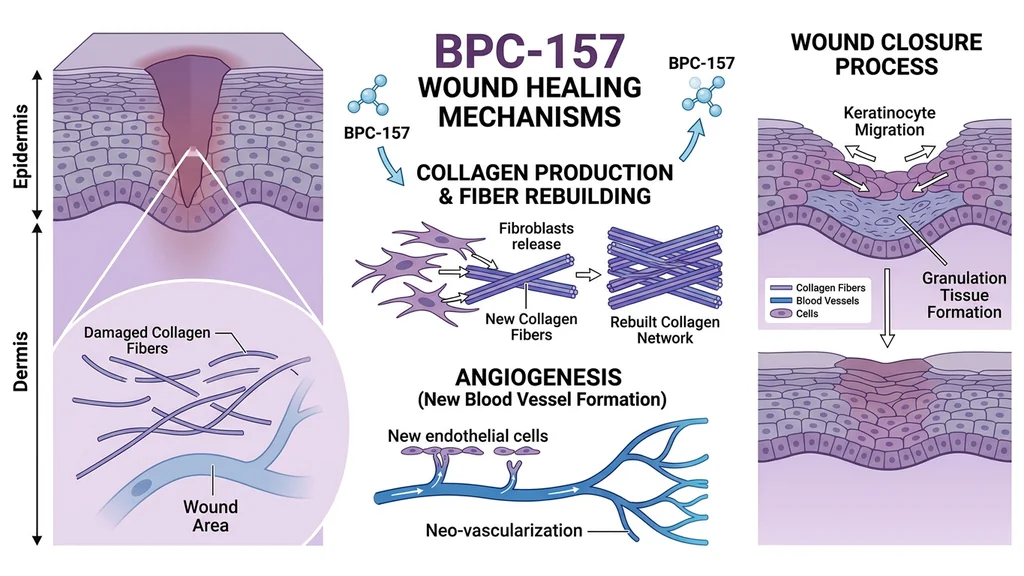
Wound Healing Evidence
Seiwerth et al. (1997) tested BPC-157 across three rat wound models: skin incisions, colon anastomoses, and sponge implants for angiogenesis assessment. The peptide improved collagen formation, angiogenesis, and tensile strength in all three (PubMed).
Hsieh et al. (2017) demonstrated that BPC-157 promotes proliferation, migration, and angiogenesis in vitro while enhancing alkali-burn wound healing in vivo. The peptide increased VEGF expression in wounded skin tissue (PMC).
A comprehensive 2021 review catalogued BPC-157's wound healing effects across burns, diabetic wounds, surgical incisions, and fistulas. The consistent finding: faster closure, reduced scarring, and improved tissue quality (Sikiric et al., 2021). Practical applications for women include C-section recovery, scar reduction after dermatological procedures, chronic wound healing in diabetic patients, and recovery from burns or trauma.
Post-Surgical Healing
Cesarean sections account for roughly 32% of all U.S. deliveries. Breast augmentation and reduction rank among the most common elective surgeries. Hysterectomy remains one of the most frequently performed gynecological procedures.
BPC-157's demonstrated ability to accelerate incisional wound healing, improve tensile strength, and reduce scar tissue formation in animal models makes it a candidate for post-surgical recovery. No human surgical recovery trial has been completed.
Clinicians who prescribe BPC-157 off-label for post-surgical use typically recommend subcutaneous injection near the surgical site at 250 to 500 mcg per day, beginning once the wound is closed. See our injection guide for administration details. Use the BPC-157 Dosage Calculator to determine exact volumes based on your reconstitution concentration.
Bladder Health and Interstitial Cystitis
The interstitial cystitis (IC) pilot study is the single most relevant human trial for women considering BPC-157. IC affects an estimated 3 to 8 million women in the United States, compared to 1 to 4 million men. The condition causes chronic bladder pain, urinary urgency, and frequency, with limited effective treatments.
Twelve women between ages 39 and 76 (mean age 58.3) who had failed pentosan polysulfate received a single intravesical injection of BPC-157 (10 mg total) around areas of bladder inflammation during cystoscopy (Moghalu et al., 2024).
The results: 10 of 12 patients (83.3%) reported complete symptom resolution, rating their success at 100%. The remaining 2 patients rated success at 80%. Zero adverse events. Zero dropouts.

All 12 participants had previously failed standard therapy. An 83% complete resolution rate in treatment-resistant patients is unusual. The study was unblinded and lacked a placebo control, which limits conclusions. Larger controlled trials are needed before BPC-157 becomes a standard IC treatment.
IC pathology involves chronic inflammation of the bladder wall, damage to the glycosaminoglycan layer, and impaired tissue repair. BPC-157's anti-inflammatory, cytoprotective, and wound-healing properties align directly with these mechanisms.
Hormonal Considerations for Women
BPC-157 does not directly alter estrogen, progesterone, testosterone, or other reproductive hormones. It works through tissue repair pathways: nitric oxide modulation, VEGF-mediated angiogenesis, and growth hormone receptor upregulation in local tissue. Unlike hormone replacement therapy or selective estrogen receptor modulators, it has no hormonal mechanism.
Indirect interactions still deserve consideration.
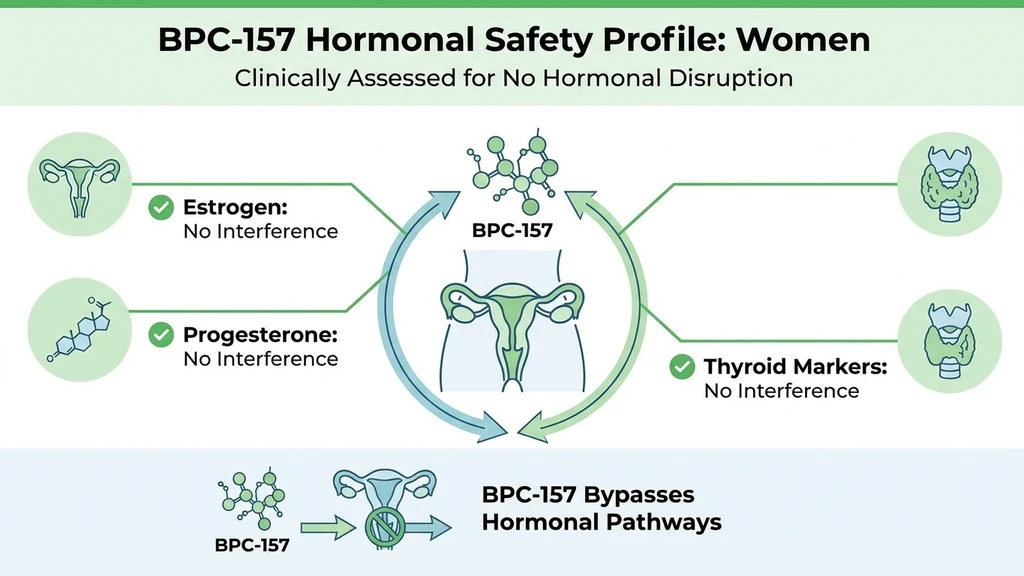
BPC-157 and Estrogen-Sensitive Tissue
BPC-157 promotes angiogenesis and reduces local inflammation. Both effects could theoretically influence estrogen-responsive tissues: breast tissue, endometrial lining, ovarian follicles.
No study has examined BPC-157's effects on estrogen receptor signaling or estrogen metabolism. The theoretical concern is that BPC-157's pro-angiogenic activity could alter the microenvironment of hormone-sensitive tissues. This is the same mechanism that raises questions about cancer risk (discussed in the safety section below).
Women with a history of estrogen receptor-positive breast cancer, endometriosis, or uterine fibroids should discuss these theoretical risks with their healthcare provider before using BPC-157.
Neurotransmitter Effects Relevant to Women
BPC-157 modulates dopamine, serotonin, GABA, and noradrenaline systems (Sikiric et al., 2021). It counteracts disturbances in these systems rather than simply pushing levels up or down.
Klicek et al. (2004) measured regional serotonin synthesis changes after BPC-157 administration in rats. Synthesis increased in the substantia nigra and nucleus accumbens while decreasing in the hypothalamus and hippocampus (PubMed).
Why this matters for women: serotonin dysregulation contributes to PMS, PMDD, perimenopausal mood changes, and postpartum depression. BPC-157's serotonin-modulating effects, observed only in animals, suggest a possible mechanism for the mood improvements some women report anecdotally. This is not clinical evidence for treating mood disorders. BPC-157 also blocks amphetamine-induced stereotypy, suggesting a stabilizing rather than stimulating effect on dopaminergic pathways (Jelovac et al., 1999).
Fertility Considerations
No study has tested BPC-157 in women trying to conceive. Animal data raises both potential benefits and concerns.
Theoretical benefits from animal models include improved blood flow to reproductive organs via angiogenesis, reduced pelvic inflammation that can impair fertility, and tissue repair at endometrial and ovarian levels. Theoretical concerns include unknown effects on follicle microenvironment from angiogenesis modulation, unstudied effects on enzyme systems metabolizing steroid hormones, and zero safety data in early pregnancy.
Women actively trying to conceive should avoid BPC-157 until human reproductive safety data exists. Embryonic development is acutely vulnerable to biochemical signals, and the potential upside does not justify unknown risks during conception and implantation.
Quantified Risk Scenarios
Two concrete scenarios show what happens when women use BPC-157 without full information.
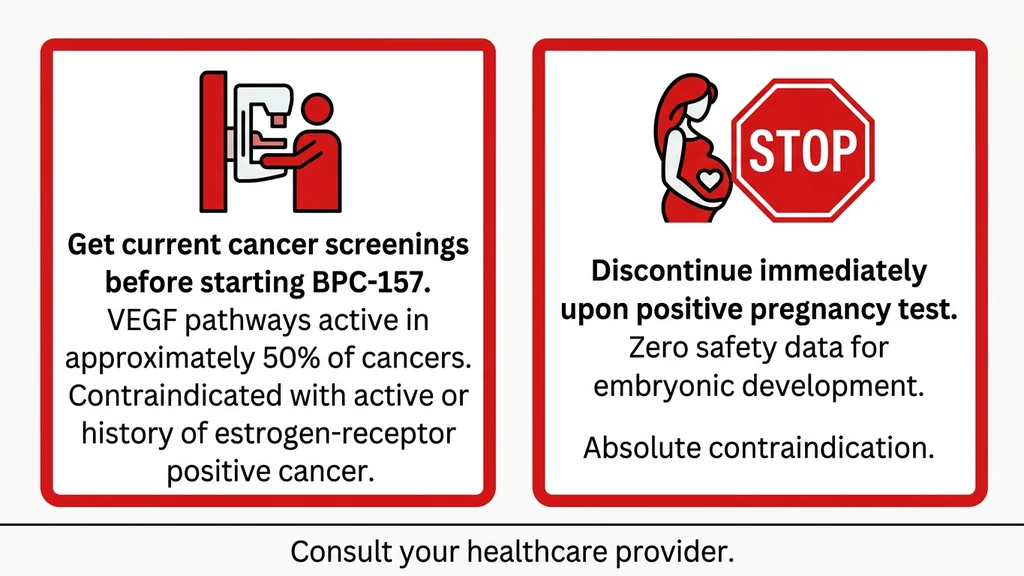
Scenario 1: Using BPC-157 with undiagnosed estrogen receptor-positive breast cancer.
A 47-year-old woman starts BPC-157 at 500 mcg/day for knee pain during perimenopause. She has not had a mammogram in three years. BPC-157 upregulates VEGF and VEGFR2 expression. VEGF/VEGFR2 pathways are active in approximately 50% of human cancers. The same FAK signaling that BPC-157 enhances for tendon repair is used by aggressive tumors to invade tissue (Vasireddi et al., 2025). If an undetected tumor is present, BPC-157's pro-angiogenic activity could theoretically accelerate its blood supply. No study has proven this happens, but no study has ruled it out.
The fix: get current cancer screenings before starting any peptide that promotes angiogenesis. Women over 40 should have a recent mammogram. BRCA1/BRCA2 carriers should consult their oncologist.
Scenario 2: Continuing BPC-157 after discovering pregnancy.
A woman on a 4-week BPC-157 cycle at 250 mcg/day discovers she is 5 weeks pregnant. BPC-157 promotes angiogenesis, modulates nitric oxide, and influences growth factor pathways. All three systems play active roles in embryonic development and placental formation. No data exists on placental transfer. No data exists on effects during organogenesis, the 3-to-8-week window when organs form and the embryo is most vulnerable. The half-life is under 30 minutes, but even brief disruptions to angiogenic signaling during organogenesis can cause irreversible developmental effects.
The fix: discontinue immediately upon a positive pregnancy test. Inform your obstetrician about the peptide, the dose, and the duration of use.
Safety, Side Effects, and Contraindications
BPC-157's safety profile in limited human data is clean. Zero adverse events in the knee pain study (16 patients), the interstitial cystitis study (12 patients), and the IV pharmacokinetics study. Animal toxicity studies using doses far exceeding typical human protocols have not identified a lethal dose (Sikiric et al., 2011).
Forty total human subjects is a thin dataset. Absence of reported problems does not guarantee safety at population scale. For a complete breakdown of known and theoretical risks, see our BPC-157 side effects guide.
Pregnancy and Breastfeeding
BPC-157 should not be used during pregnancy or while breastfeeding. No safety data exists for either population.
The concerns are concrete. BPC-157 promotes angiogenesis, modulates nitric oxide signaling, and influences growth factor pathways. All of these systems drive embryonic development, placental formation, and fetal growth.
Specific unknowns: no data on BPC-157 transfer across the placental barrier, no data on transfer into breast milk, unstudied pharmacokinetics in pregnancy despite a known sub-30-minute half-life with hepatic metabolism and renal clearance, and completely uncharacterized effects on fetal organogenesis and neural development. Women who become pregnant during a BPC-157 protocol should discontinue immediately and inform their obstetrician.
Cancer Risk and Angiogenesis Concerns
BPC-157's pro-angiogenic properties cut both ways. In wound healing, new blood vessels deliver oxygen and nutrients to repair sites. In cancer, new blood vessels feed tumors and facilitate metastasis.
BPC-157 upregulates VEGF and VEGFR2 expression. VEGF/VEGFR2 pathways are active in approximately half of human cancers. The FAK signaling that BPC-157 enhances for tendon repair is also exploited by aggressive tumors to invade tissue (Vasireddi et al., 2025).
No study has shown BPC-157 directly causes cancer or accelerates tumor growth. No study has ruled it out. Women who should avoid BPC-157 or use it only under oncologist supervision include those with active malignancy, history of estrogen receptor-positive breast cancer, BRCA1/BRCA2 carrier status, or family history of cancers where angiogenesis is a known driver.
Common Side Effects
Reported side effects from clinical use and community reports are generally mild.
| Side Effect | Frequency | Notes |
|---|---|---|
| Injection site redness/swelling | Common | Resolves within 24 to 48 hours |
| Mild nausea (oral route) | Occasional | Usually transient, first few days |
| Lightheadedness | Rare | Typically with first injection |
| Fatigue | Rare | Usually resolves within a week |
| GI discomfort | Occasional | More common with oral administration |
Women using hormonal birth control, HRT, or thyroid medication should note: no drug interactions have been documented, but no interaction studies have been conducted. Monitor for changes after starting BPC-157 and report them to your prescriber.
BPC-157 is not FDA-approved for any indication. It is classified as a research compound. Quality and purity vary between suppliers. Third-party testing certificates (COA) are the minimum standard for any product you consider.
Common Mistakes Women Make with BPC-157
Mistake 1: Starting BPC-157 without current cancer screenings.
BPC-157 promotes angiogenesis. If an undetected tumor exists, increased blood vessel formation could theoretically accelerate its growth. VEGF/VEGFR2 pathways are active in roughly 50% of human cancers (Vasireddi et al., 2025). Women over 40 should have current mammography and routine bloodwork before starting any pro-angiogenic peptide.
Mistake 2: Expecting gut healing from subcutaneous injection alone.
Oral BPC-157 delivers the peptide directly to the GI lining. Subcutaneous injection bypasses the gut entirely. For IBS, leaky gut, or IBD, oral administration at 500 to 1000 mcg/day is the preferred route because it places the peptide in direct contact with damaged tissue. Injecting into your abdomen for gut issues sends the peptide into subcutaneous fat, not your intestinal wall.
Mistake 3: Continuing BPC-157 during early pregnancy.
The 3-to-8-week window of organogenesis is when fetal organs form. BPC-157 modulates angiogenesis, nitric oxide, and growth factor pathways, all active in embryonic development. No reproductive safety data exists. Women who discover pregnancy during a cycle should discontinue the same day and inform their obstetrician.
Mistake 4: Treating community anecdotes as equivalent to clinical data.
The interstitial cystitis pilot studied 12 women. The knee pain study enrolled 16 patients. Total human BPC-157 safety data covers fewer than 40 subjects. Online forums contain thousands of reports, but self-selected testimonials carry no statistical weight. Base your expectations on the published data, not Reddit threads.
Dosing, Administration, and Practical Protocol
BPC-157 dosing for women follows the same protocols established in animal research and off-label clinical practice. No sex-specific dosing adjustments have been identified. Body weight, target condition, and administration route determine the protocol.
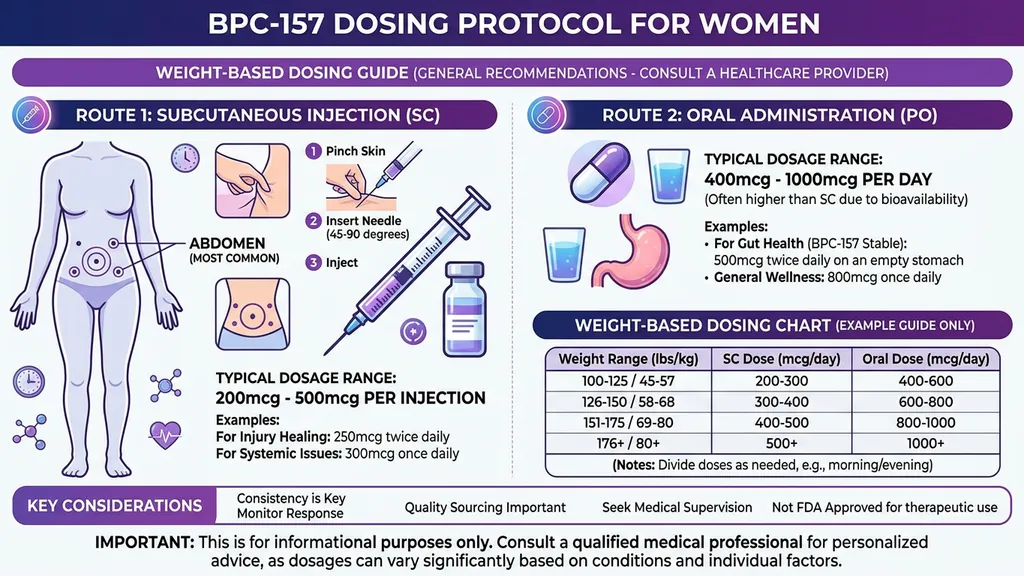
Standard Dosing Protocol
Subcutaneous injection:
- 1.Standard dose: 250 to 500 mcg per day
- 2.Split into 1 to 2 daily injections
- 3.Inject near the target area when possible (near an injured joint, abdomen for systemic use)
- 4.Cycle length: 4 to 8 weeks, followed by 2 to 4 weeks off
Oral administration:
- 1.Standard dose: 500 to 1000 mcg per day (higher dose compensates for lower bioavailability)
- 2.Take on an empty stomach
- 3.Preferred route for gut-specific conditions (direct contact with GI lining)
- 4.Bioavailability: approximately 30 to 50% compared to roughly 90% for subcutaneous injection
Use the BPC-157 Dosage Calculator to calculate exact injection volumes based on your vial size and reconstitution volume. The Peptide Reconstitution Calculator determines how much bacteriostatic water to add. For step-by-step administration, see the peptide injection guide.
Condition-Specific Recommendations
| Condition | Suggested Route | Dose Range | Duration |
|---|---|---|---|
| Gut healing / IBS / leaky gut | Oral preferred | 500-1000 mcg/day | 6-8 weeks |
| Joint pain / tendon injury | Subcutaneous near site | 250-500 mcg/day | 4-8 weeks |
| Post-surgical recovery | Subcutaneous near site | 250-500 mcg/day | 4-6 weeks |
| General recovery / wellness | Subcutaneous (abdomen) | 250 mcg/day | 4 weeks |
| Interstitial cystitis | Clinical setting only | 10 mg intravesical (per study) | Single procedure |
These ranges are derived from animal dose translations and off-label clinical practice. They are not FDA-approved dosing guidelines. Women under 120 lbs (55 kg) may start at the lower end of each range.
The interstitial cystitis study used a single 10 mg dose administered directly into the bladder during cystoscopy. That is a clinical procedure, unrelated to daily self-administration protocols.
Stacking with Other Peptides
BPC-157 is frequently combined with other peptides. Three common stacks for women:
BPC-157 + TB-500: TB-500 (Thymosin Beta-4) promotes cell migration and differentiation through pathways distinct from BPC-157's angiogenesis and growth factor expression. Together they cover more repair mechanisms than either alone. The human knee study that combined BPC-157 and TB-4 showed 75% improvement, though the sample was only 4 patients.
BPC-157 + GHK-Cu: For skin and connective tissue. GHK-Cu stimulates collagen I and III synthesis through a different mechanism than BPC-157. Women interested in both wound healing and skin quality often combine these two peptides.
BPC-157 + KPV: For gut-specific protocols. KPV is an anti-inflammatory tripeptide that targets gut inflammation through melanocortin receptors. Combined with BPC-157's barrier repair properties, this stack addresses both inflammatory and structural components of GI conditions. For interaction details on BPC-157 paired with alcohol, see BPC-157 and alcohol.
Frequently Asked Questions
Is BPC-157 safe for women?
BPC-157 appears safe for non-pregnant, non-breastfeeding women without active cancer. The interstitial cystitis pilot (12 women, ages 39 to 76) reported zero adverse events (Moghalu et al., 2024). Animal toxicity studies have not identified a lethal dose. Total human safety data covers fewer than 40 subjects across all trials. Women with hormone-sensitive cancers or BRCA mutations should consult an oncologist first.
Can I take BPC-157 during pregnancy or breastfeeding?
No. BPC-157 has not been studied in pregnant or breastfeeding women. The peptide promotes angiogenesis, modulates nitric oxide, and influences growth factor pathways, all active in embryonic development and placental function. No data exists on placental transfer or breast milk secretion. Discontinue immediately if you become pregnant and inform your obstetrician about the peptide, dose, and duration.
Does BPC-157 affect estrogen or other female hormones?
Current evidence shows BPC-157 does not directly alter estrogen, progesterone, or testosterone. Its primary mechanisms involve tissue repair through nitric oxide modulation, VEGF-mediated angiogenesis, and local growth factor upregulation (Sikiric et al., 2021). No clinical interaction has been documented with hormonal birth control or HRT. Indirect effects on hormonal systems cannot be fully excluded because BPC-157 modulates enzyme and neurotransmitter pathways.
What is the best BPC-157 dose for women?
The standard protocol is 250 to 500 mcg per day via subcutaneous injection, or 500 to 1000 mcg per day orally. Women under 55 kg (120 lbs) typically start at 250 mcg/day. For gut conditions like IBS, oral administration delivers the peptide directly to the GI lining. For joint or surgical recovery, subcutaneous injection near the target site provides higher local concentration. Use the BPC-157 Dosage Calculator for personalized volumes. Cycles run 4 to 8 weeks.
Can BPC-157 help with menopause-related joint pain?
BPC-157 has not been studied in menopausal women specifically. Its mechanisms align with menopausal joint complaints: accelerated tendon and ligament repair, collagen synthesis promotion, and growth hormone receptor upregulation in connective tissue (Chang et al., 2014). These are mechanistic extrapolations from animal data, not confirmed clinical benefits. Women on HRT should note that no interaction studies with BPC-157 exist.
What happened in the interstitial cystitis BPC-157 study?
Twelve women (ages 39 to 76) with treatment-resistant IC received a single intravesical injection of 10 mg BPC-157 during cystoscopy. Ten of 12 (83.3%) reported complete symptom resolution. The remaining 2 rated success at 80%. Zero adverse events, zero dropouts (Moghalu et al., 2024). The study was unblinded without placebo control. Larger trials are needed.
Should I take BPC-157 orally or by injection?
Route depends on your target condition. Oral BPC-157 delivers the peptide directly to the GI lining, making it preferred for IBS, leaky gut, and IBD at 500 to 1000 mcg/day. Subcutaneous injection near the target site is preferred for joint, tendon, or surgical recovery at 250 to 500 mcg/day. Oral bioavailability runs 30 to 50% compared to roughly 90% for injection, which is why oral doses are higher.
Can BPC-157 cause cancer?
No study has shown BPC-157 causes cancer or accelerates tumor growth. The concern is theoretical: BPC-157 upregulates VEGF and VEGFR2, pathways active in roughly 50% of human cancers (Vasireddi et al., 2025). Women with active malignancy, estrogen receptor-positive breast cancer history, or BRCA mutations should avoid BPC-157 or use it only under oncologist supervision.
The Bottom Line
BPC-157 addresses gut inflammation, joint degeneration, slow wound healing, and neurotransmitter imbalance. These conditions disproportionately affect women. The strongest signal comes from the interstitial cystitis pilot: 10 of 12 treatment-resistant women achieved complete symptom resolution from a single injection (Moghalu et al., 2024).
Gut healing evidence spans two decades of animal research demonstrating barrier repair, NSAID damage reversal, and fistula healing. Joint and tendon repair data is consistent across 36 preclinical studies. The human knee pain pilot, while limited to 16 subjects, aligns with the animal findings.
What remains unknown is substantial. Long-term human safety data does not exist. Effects on estrogen-sensitive tissue are uncharacterized. Cancer risk from pro-angiogenic activity is theoretical but unresolved. Pregnancy and breastfeeding are clear contraindications. The evidence supports cautious, informed use for women with specific conditions, current cancer screenings, and medical guidance.
Calculate your exact volumes with the BPC-157 Dosage Calculator. Prepare your vials with the Peptide Reconstitution Calculator. Review the full safety profile in our BPC-157 side effects guide. Learn proper technique from the peptide injection guide. Compare GI-targeting options in peptides for gut health.
References: 1. Chang CH et al. (2020). BPC 157 rescued NSAID-cytotoxicity via stabilizing intestinal permeability. *Curr Pharm Des.* 26(25):2971-2981. PubMed 2. Chang CH et al. (2011). The promoting effect of BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration. *J Appl Physiol.* 110(3):774-780. PubMed 3. Chang CH et al. (2014). BPC 157 enhances growth hormone receptor expression in tendon fibroblasts. *Molecules.* 19(11):19066-19077. PMC 4. Cesarec V et al. (2013). BPC 157 effective in healing of colocutaneous fistulas. *J Pharmacol Sci.* 108(1):7-17. PubMed 5. Hsieh MJ et al. (2017). BPC-157 enhances wound healing in vivo and promotes proliferation, migration, and angiogenesis in vitro. *Drug Des Devel Ther.* 11:2015-2025. PMC 6. Ilic S et al. (2011). BPC 157 effects on NSAID toxicity model: diclofenac-induced lesions. *Life Sci.* 88(11-12):535-542. PubMed 7. Jelovac N et al. (1999). BPC 157 and chronic amphetamine-induced behaviour disturbances. *Naunyn Schmiedebergs Arch Pharmacol.* 359(4):R38. PubMed 8. Lee JY et al. (2021). Intra-articular injection of BPC 157 for multiple types of knee pain. *Altern Ther Health Med.* 27:8-13. PubMed 9. Lojo N et al. (2009). Gastric pentadecapeptide BPC 157 and short bowel syndrome in rats. *Dig Dis Sci.* 53(12):3302-3306. PubMed 10. Moghalu O et al. (2024). Effect of BPC-157 on symptoms in patients with interstitial cystitis: a pilot study. *Altern Ther Health Med.* PubMed 11. Seiwerth S et al. (1997). BPC 157's effect on healing. *J Physiol Paris.* 91(3-5):173-178. PubMed 12. Sikiric P et al. (2006). BPC 157 in trials for IBD. Full and distended stomach, and vascular response. *J Physiol Pharmacol.* 57(Suppl 2):101-108. PubMed 13. Sikiric P et al. (2011). Stable gastric pentadecapeptide BPC 157: novel therapy in GI tract. *Curr Pharm Des.* 17(16):1612-1632. PubMed 14. Sikiric P et al. (2013). Toxicity by NSAIDs. Counteraction by BPC 157. *Curr Pharm Des.* 19(1):76-83. PubMed 15. Sikiric P et al. (2021). BPC 157 and wound healing. *Front Pharmacol.* 12:627533. PMC 16. Sikiric P et al. (2021). BPC 157 and the central nervous system. *Curr Neuropharmacol.* 19(5):768-789. PubMed 17. Staresinic M et al. (2003). BPC 157 accelerates healing of transected rat Achilles tendon. *J Orthop Res.* 21(6):976-983. PubMed 18. Vasireddi N et al. (2025). Emerging use of BPC-157 in orthopaedic sports medicine: a systematic review. *Curr Rev Musculoskelet Med.* PubMed
*This article is for educational purposes only and does not constitute medical advice. BPC-157 is not FDA-approved for any indication. Always consult a qualified healthcare provider before using any peptides.*
Related Articles
Peptides for Bodybuilding (2026)
Best peptides for bodybuilding: BPC-157, TB-500, MK-677, CJC-1295/Ipamorelin, HGH Fragment 176-191. Dosages and stacking protocols.
BPC-157 Oral vs Injection: Which Route Is Right for Your Goals?
BPC-157 oral vs injection: 80-95% injectable bioavailability vs 3-90% oral. He et al. PK data, condition-by-condition matrix, combination protocol, and cost breakdown.
How Long Does BPC-157 Take to Work?
BPC-157 timelines: gut healing 1-2 weeks, pain relief 1-2 weeks, tendon repair 4-8 weeks, bone healing 6-12 weeks. Factors that speed results.
BPC-157 Oral Pills & Capsules: Do They Work?
BPC-157 oral pills deliver 250-500 mcg to the gut. Oral vs injectable vs nasal comparison, dosing protocols, and sourcing guidance.