Peptide therapeutics crossed $52.6 billion in global revenue last year. One in eight American adults now takes a GLP-1 drug. Google searches for "are peptides safe" jumped 652% in twelve months.
Every figure on this page comes from a verified source: pharmaceutical earnings reports, KFF tracking polls, ClinicalTrials.gov, and DataForSEO search data. The peptide industry left its niche years ago. The numbers below document how far it has traveled.
We compiled 60+ statistics across market size, drug revenue, consumer behavior, regulatory shifts, clinical pipelines, and economic impact. Bookmark this page. We update it quarterly.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Key Takeaways
The ten statistics cited most often from this resource:
- $52.6 billion: Global peptide therapeutics market in 2025 (Precedence Research)
- $62.8 billion: GLP-1 receptor agonist market alone in 2025 (Fortune Business Insights)
- 1 in 8 U.S. adults (12.4%) now taking a GLP-1 drug, doubled from 5.8% in 18 months (KFF, 2025)
- $26 billion: Combined 2024 revenue for Ozempic and Wegovy (Novo Nordisk earnings)
- +652%: 12-month growth in U.S. searches for "are peptides safe" (DataForSEO)
- 28.7%: Average body weight loss from retatrutide in Phase 3 trials (Eli Lilly, 2025)
- 37.0%: U.S. adult obesity rate, down from 39.9% in early 2024 (Gallup)
- ~100 peptide drugs approved worldwide, with 170+ in active clinical development
- 3.4 million: Medicare beneficiaries projected to access GLP-1 drugs under the BALANCE model (CMS, July 2026)
- 42%: Online peptide products that contained inaccurate quantities of the listed compound (JAMA, 2023)
Peptide Market Size and Revenue
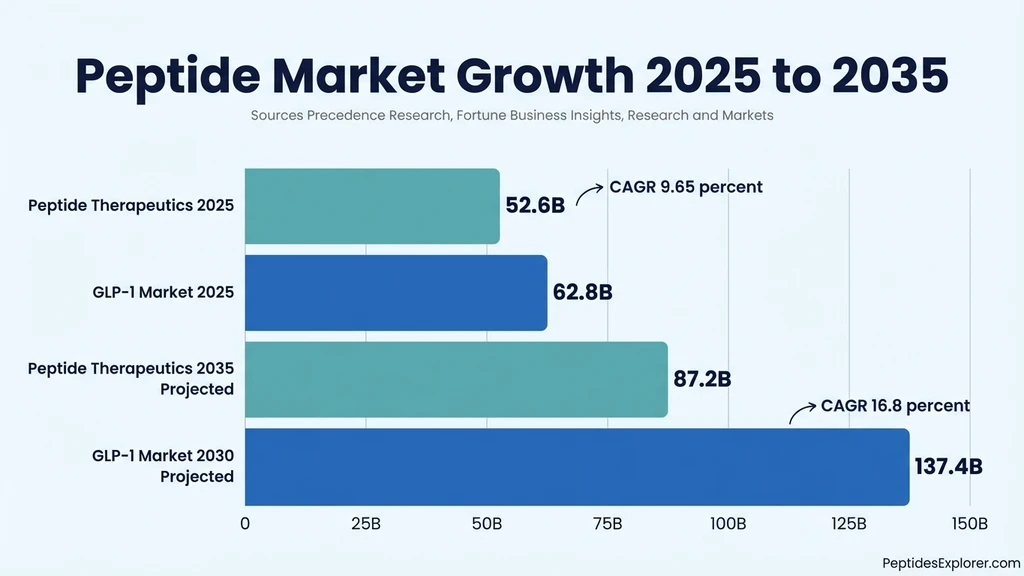
The global peptide therapeutics market reached $52.6 billion in 2025 (Precedence Research), growing at a compound annual rate of 9.65% with projections through 2034. Grand View Research placed the figure at $140.9 billion using a broader definition that includes diagnostics and cosmeceuticals.
That discrepancy matters. "Peptide market" figures vary by tens of billions depending on whether the scope covers pharmaceutical drugs only or the full spectrum of research reagents, cosmetic peptides, and agricultural applications. The $52.6 billion figure used throughout this page refers to therapeutic peptides.
| Market Segment | 2025 Value | Projected Value | CAGR | Source |
|---|---|---|---|---|
| Peptide therapeutics (narrow) | $52.6B | $87.2B (2035) | 9.65% | Precedence Research |
| Peptide market (broad) | $140.9B | — | — | Grand View Research |
| GLP-1 receptor agonists | $62.8B | $137.4B (2030) | 16.8% | Fortune Business Insights |
GLP-1 Revenue Dominates the Picture
The GLP-1 receptor agonist class generated $62.83 billion in 2025 (Fortune Business Insights). That single drug class now produces more revenue than the entire peptide therapeutics market did five years ago. Its projected CAGR of 16.8% through 2032 outpaces every other peptide segment.
Semaglutide (branded as Ozempic and Wegovy) produced roughly $26 billion in combined revenue for Novo Nordisk in fiscal year 2024. Tirzepatide (Mounjaro and Zepbound) generated $16.47 billion for Eli Lilly over the same period.
By Q3 2025, tirzepatide became the world's best-selling drug, surpassing Merck's Keytruda. Eli Lilly reported $10.1 billion in tirzepatide revenue for that single quarter and raised its full-year 2025 guidance to $63 billion. Two companies now control virtually all GLP-1 revenue. No other pharmaceutical segment shows this level of duopoly concentration.
Market Projections Through 2030
JPMorgan analysts project 25 to 30 million Americans will take GLP-1 drugs by 2030, up from roughly 15 million today. Research and Markets forecasts the GLP-1 market alone will reach $137.4 billion by that year.
The broader peptide therapeutics market is projected to hit $87.2 billion by 2035 (GlobeNewsWire, December 2025). Cancer, diabetes, and metabolic disorders remain the primary growth drivers. For dosage planning on the most prescribed peptide drug, see our semaglutide dosage calculator.
GLP-1 Drug Statistics
GLP-1 receptor agonists are the largest and fastest-growing peptide segment. Originally developed for type 2 diabetes, they crossed into mainstream weight management after Wegovy's FDA approval in 2021 and Zepbound's in 2023. Five branded products now generate over $44 billion in combined annual revenue.
Revenue by Drug
| Drug (Brand) | Company | 2024 Revenue | Primary Indication |
|---|---|---|---|
| Semaglutide (Ozempic) | Novo Nordisk | ~$18B | Type 2 diabetes |
| Semaglutide (Wegovy) | Novo Nordisk | ~$8B | Obesity / weight management |
| Tirzepatide (Mounjaro) | Eli Lilly | ~$11.6B | Type 2 diabetes |
| Tirzepatide (Zepbound) | Eli Lilly | ~$4.9B | Obesity / weight management |
| Liraglutide (Saxenda) | Novo Nordisk | ~$1.5B | Obesity / weight management |
Sources: Novo Nordisk FY2024 earnings; Eli Lilly FY2024 earnings; BioSpace; BioPharma Dive.
Ozempic alone outsells most pharmaceutical companies' entire portfolios. Its $18 billion in annual revenue exceeds the total revenue of Regeneron, Biogen, or Moderna. The weight-management indication (Wegovy, Zepbound) is the faster-growing segment, but the diabetes indication still generates the larger share of revenue.
User Adoption and Demographics
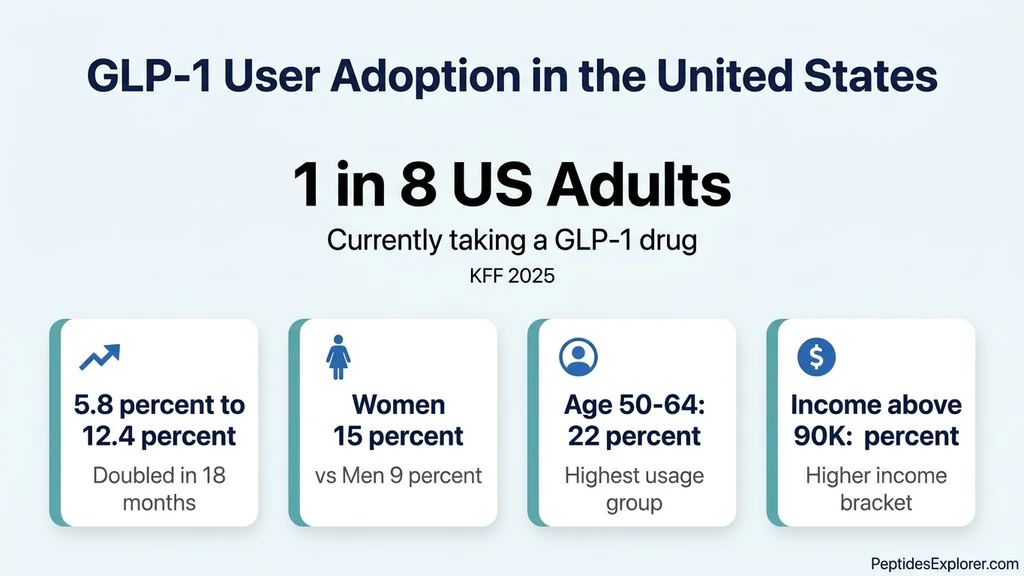
A KFF tracking poll from late 2025 found that 1 in 8 U.S. adults (12.4%) reported currently taking a GLP-1 drug for weight loss, diabetes, or another condition. That figure doubled from 5.8% just 18 months earlier.
| Demographic | GLP-1 Usage Rate | Source |
|---|---|---|
| All U.S. adults | 12.4% | KFF, 2025 |
| Women | 15% | KFF, 2025 |
| Men | 9% | KFF, 2025 |
| Adults aged 50-64 | 22% | KFF, 2025 |
| Household income above $90K | 16% | KFF, 2025 |
| Ever used a GLP-1 drug | 12% | RAND Corporation, Aug 2025 |
| Currently using a GLP-1 drug | 7% | RAND Corporation, Aug 2025 |
Medicaid GLP-1 prescriptions surged from 1.3 million in 2019 to 8.4 million in 2024 (KFF Medicaid spending data). The growth is concentrated in the obesity indication, not diabetes. That shift reflects the cultural transformation of GLP-1 drugs from diabetes treatment to weight management tool.
Oral GLP-1s Change the Access Equation
The FDA approved oral semaglutide (Wegovy) for weight management in December 2025. It was the first GLP-1 pill approved for obesity. Novo Nordisk set the list price at $149 per month without insurance, undercutting the injectable version by more than 70%.
Clinical trials showed 16.6% body weight loss at 64 weeks with the oral formulation. Patients who avoided injections now have a daily pill option. Analysts expect oral GLP-1s to expand the total addressable market by 30 to 40%.
The injection barrier kept millions of potential patients on the sideline. A daily pill eliminates that barrier. For injection guidance on current formulations, see our tirzepatide dosage calculator.
Consumer Search Trends
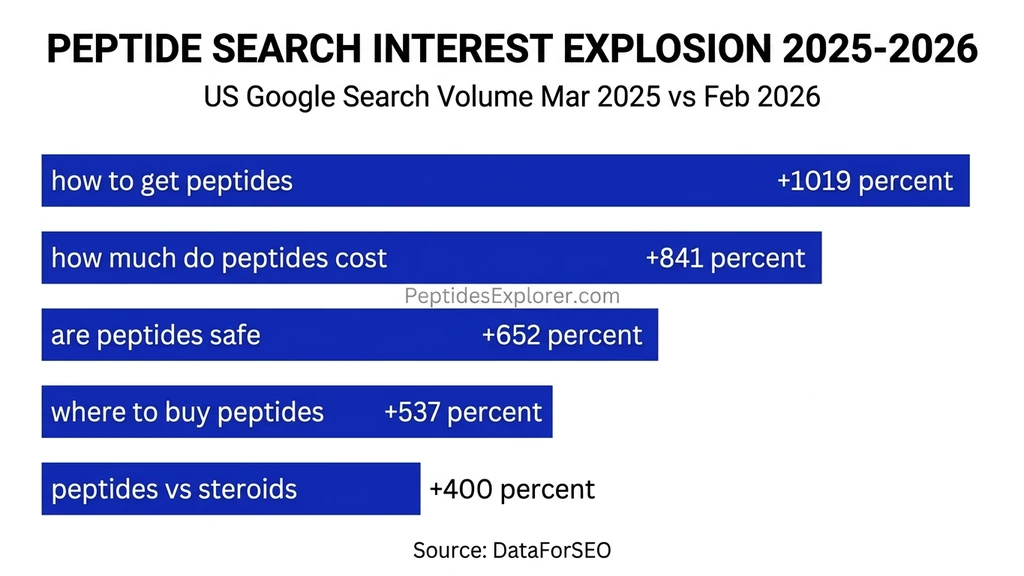
Google search data quantifies how fast public interest in peptides is accelerating. We analyzed U.S. search volumes from DataForSEO comparing March 2025 to February 2026. The growth rates are extraordinary across every category of peptide query.
Fastest-Growing Peptide Searches (12-Month Change)
| Search Term | Mar 2025 | Feb 2026 | Growth |
|---|---|---|---|
| "how to get peptides" | 590/mo | 6,600/mo | +1,019% |
| "how much do peptides cost" | 170/mo | 1,600/mo | +841% |
| "GLP-1 usage statistics" | 30/mo | 260/mo | +767% |
| "are peptides steroids" | 2,400/mo | 18,100/mo | +654% |
| "are peptides safe" | 3,600/mo | 27,100/mo | +652% |
| "peptide market" | 50/mo | 320/mo | +540% |
| "where to buy peptides" | 1,900/mo | 12,100/mo | +537% |
| "are peptides legal" | 1,600/mo | 9,900/mo | +519% |
| "peptides vs steroids" | 720/mo | 3,600/mo | +400% |
| "peptide therapy near me" | 9,900/mo | 12,100/mo | +22% |
Source: DataForSEO (U.S. Google Ads data). Analysis by PeptidesExplorer.
What the Search Data Reveals
Three patterns stand out in this data.
Safety and legality queries are growing fastest. "Are peptides safe" and "are peptides steroids" both exceeded 650% growth. This signals a wave of first-time consumers entering the market. People searching "are peptides steroids" have heard the word but do not yet understand what peptides are. That is new-market behavior.
Purchase-intent searches show explosive commercial demand. "How to get peptides" grew 1,019% in twelve months. "Where to buy peptides" grew 537%. These searchers have moved past curiosity into active purchasing decisions.
The comparison query "peptides vs steroids" grew 400%. Fitness communities are evaluating peptides as alternatives to anabolic compounds. The question itself implies familiarity with both categories and a desire to choose between them.
FDA and Regulatory Statistics
The regulatory landscape for peptides shifted more between 2024 and 2026 than in the previous decade. Three developments reshaped access to both pharmaceutical and compounded peptides.
FDA Approvals and Pipeline
Approximately 100 peptide-based drugs have received regulatory approval worldwide as of 2025. Between 2016 and 2024, the FDA approved 34 new peptide therapeutics (MDPI Pharmaceuticals, 2025 review).
Over 200 peptide-related clinical trials were active on ClinicalTrials.gov during 2024 and 2025, spanning oncology, metabolic disease, neurology, and infectious disease. Peptide vaccines represented one of the fastest-growing trial categories.
The total number of peptide molecules in active clinical development exceeds 170 globally (Precedence Research, 2025). For context on how this expanding pipeline affects individual peptide users, see our getting started guide.
The Compounding Reclassification
In late 2023, the FDA placed 19 widely used peptides on its Category 2 ("difficult to compound") list, restricting access through 503A and 503B compounding pharmacies. BPC-157, AOD-9604, and several others lost their compounding pathway.
On February 27, 2026, following advocacy from clinicians and patients, 14 of those 19 peptides were reclassified back to Category 1 (eligible for compounding). The reclassification was announced but had not yet been formally published in the Federal Register as of March 2026.
The practical impact: peptides that were only available through grey-market vendors can now be legally prescribed by a physician and prepared by a licensed pharmacy. For the full regulatory timeline, see our analysis of the Peptide Sciences shutdown.
Medicare GLP-1 Coverage
The Centers for Medicare & Medicaid Services (CMS) announced the BALANCE model in early 2026, launching July 2026. Medicare Part D beneficiaries with BMI above 30 (or 27 with comorbidities) will access GLP-1 drugs at a $50 per month copay.
Medicare previously excluded weight-loss drugs from coverage entirely. The BALANCE model is projected to cover 3.4 million beneficiaries in its first year. That single policy change could add billions in GLP-1 revenue while expanding access to the population segment most likely to benefit.
Obesity and Public Health Impact
GLP-1 adoption is producing measurable changes in U.S. population health data for the first time in over a decade.
U.S. Obesity Rate Is Declining
Gallup tracking data showed the U.S. adult obesity rate dropping from 39.9% in early 2024 to 37.0% by late 2025. That 2.9 percentage point decline translates to roughly 7.6 million fewer obese adults.
The CDC's National Center for Health Statistics confirmed the trend in Data Brief #537, noting a statistically significant decline in adult obesity prevalence beginning in late 2024. This is the first sustained decline in the U.S. obesity rate in over a decade. Researchers cautioned that the trend coincides with both GLP-1 adoption and updated measurement methodologies.
Behavioral and Economic Shifts Among Users
JPMorgan's 2026 healthcare research report found that GLP-1 users consume 21% fewer daily calories and spend 31% less on groceries compared to pre-treatment levels. The bank estimated that sustained adoption could reduce U.S. food and beverage industry revenue by $30 to $55 billion annually by 2030 to 2034.
Morgan Stanley published a parallel finding: bariatric surgery volumes have declined 15% in markets with high GLP-1 adoption. The surgical weight-loss industry is watching its patient pipeline shrink in real time.
A Pew Research Center analysis from January 2026 found that 53% of Americans view GLP-1 drugs favorably, up from 42% in 2024. Support is highest among adults who personally know someone taking the drugs. Public perception has shifted from skepticism ("celebrity diet drug") toward acceptance ("legitimate medical treatment").
Clinical Pipeline: Next-Generation Peptides
The next wave of peptide drugs is in late-stage clinical trials. Several could reach market by 2027 or 2028, with efficacy data that surpasses every current option.
Retatrutide (Eli Lilly)
Retatrutide is a triple-agonist peptide targeting GLP-1, GIP, and glucagon receptors simultaneously. In Phase 3 trials, participants lost an average of 28.7% of their body weight (71.2 lbs) at 68 weeks. That result exceeds anything achieved by semaglutide or tirzepatide in comparable studies.
Eli Lilly has seven additional Phase 3 readouts expected throughout 2026, covering NASH (metabolic liver disease), sleep apnea, and cardiovascular outcomes. If approved, retatrutide would be the most effective weight-loss drug ever brought to market. For current efficacy and safety data, see our retatrutide side effects analysis.
Orforglipron (Eli Lilly)
Orforglipron is a small-molecule oral GLP-1 receptor agonist in Phase 3 development. Unlike oral semaglutide (still a peptide requiring fasting), orforglipron is a non-peptide compound that can be taken without food restrictions. Phase 2 data showed 14.7% body weight loss at 36 weeks.
If approved, orforglipron would simplify daily GLP-1 use and reach patients who refuse both injections and fasting-dependent pills. The convenience advantage could pull tens of millions of additional patients into the GLP-1 market.
Beyond Weight Loss
Peptide research extends well beyond GLP-1 and obesity. Peptide-drug conjugates (molecules that deliver chemotherapy directly to tumor cells) represent one of the fastest-growing segments in cancer therapeutics, with over 30 candidates in active clinical trials as of 2025.
Antimicrobial peptides are advancing through development as antibiotic resistance worsens. Neurodegenerative disease programs are testing peptide approaches for Alzheimer's and Parkinson's. The total number of peptide molecules in active clinical development exceeds 170 globally (Precedence Research, 2025).
Economic Ripple Effects
The GLP-1 boom generates consequences that reach far beyond pharmaceutical revenue and healthcare budgets. Multiple industries are adjusting to a population that eats less, weighs less, and spends healthcare dollars differently.
Food and Beverage Industry Impact
JPMorgan estimated that GLP-1 drugs could reduce caloric consumption across the U.S. population by 1.5 to 2.5% by 2030. For the food and beverage industry, that translates to $30 to $55 billion in lost annual revenue. Snack companies, fast food chains, and alcohol producers face the greatest exposure.
The impact is already visible. Restaurant chains reported lower average check sizes in markets with high GLP-1 adoption during Q3 2025 earnings calls. Consumer packaged goods companies are reformulating product lines toward smaller portions and higher protein content. A drug designed for diabetes is quietly reshaping the American food economy.
Telehealth and Compounding Pharmacy Growth
Peptide therapy fueled a parallel boom in telehealth clinics and compounding pharmacies. Search volume for "peptide therapy near me" holds steady at 12,100 monthly searches. "Peptide clinic" grew from 590 to 1,600 searches per month over 12 months (+171%).
503B outsourcing facilities that compound peptides saw revenue growth of 25 to 40% between 2024 and 2025 (industry trade publications). The FDA's evolving stance on compounding eligibility creates both opportunity and regulatory risk for this segment. For background on regulatory shifts, see our FDA peptide crackdown analysis.
Research Peptide Statistics
Not all peptides are GLP-1 drugs. A substantial portion of search volume and consumer interest centers on research peptides used in recovery, longevity, and performance contexts.
Most Searched Peptides in the U.S.
| Peptide | Primary Use | Monthly U.S. Search Volume |
|---|---|---|
| Semaglutide | Weight loss, diabetes | 1,500,000+ |
| Tirzepatide | Weight loss, diabetes | 823,000 |
| BPC-157 | Tissue repair, gut healing | 74,000 |
| Retatrutide | Weight loss (investigational) | 40,500 |
| TB-500 | Recovery, wound healing | 33,100 |
| GHK-Cu | Skin repair, anti-aging | 18,100 |
| Ipamorelin | Growth hormone release | 14,800 |
Source: DataForSEO, February 2026. Approximate U.S. monthly averages.
The search hierarchy is steep. Semaglutide generates 20 times the search volume of BPC-157, the most-searched research peptide. Pharmaceutical GLP-1 drugs dominate public attention. Research peptides like TB-500 and GHK-Cu maintain dedicated audiences within fitness, biohacking, and longevity communities, but the scale difference is dramatic.
Quality and Safety Concerns
A 2023 study published in JAMA found that 42% of peptide products purchased online from non-pharmaceutical sources contained inaccurate quantities of the listed compound. Some contained no active peptide at all. Others contained unlisted contaminants.
This data point drives much of the regulatory scrutiny around grey-market peptide sales. The Peptide Sciences shutdown in March 2026 underscored the instability of unregulated supply chains. Independent testing by Finnrick showed quality scores ranging from A to E across the same vendor's product line.
For guidance on evaluating peptide sources, see our peptide safety guide and 2026 buyer's guide.
Methodology
Market and revenue figures come from pharmaceutical company earnings reports and SEC filings (Novo Nordisk, Eli Lilly) and published market research (Precedence Research, Fortune Business Insights, Grand View Research, Research and Markets). Adoption and demographic data come from federal surveys: KFF Health Tracking Poll, RAND Corporation, CDC National Center for Health Statistics, and Gallup.
Search volumes were collected through DataForSEO, which aggregates Google Ads keyword data. All figures reflect approximate U.S. monthly averages. Year-over-year growth comparisons use March 2025 vs. February 2026 data points. Clinical data references published results from ClinicalTrials.gov and pharmaceutical company press releases.
Every statistic on this page cites its primary source. We update this page quarterly and verify each figure against its original source before republication.
Summary of All Key Statistics
| Category | Statistic | Value | Source |
|---|---|---|---|
| Market | Global peptide therapeutics (2025) | $52.6B | Precedence Research |
| Market | GLP-1 receptor agonist market (2025) | $62.8B | Fortune Business Insights |
| Market | GLP-1 market projected (2030) | $137.4B | Research and Markets |
| Market | Peptide therapeutics projected (2035) | $87.2B | GlobeNewsWire |
| Revenue | Ozempic + Wegovy combined (2024) | ~$26B | Novo Nordisk earnings |
| Revenue | Mounjaro + Zepbound combined (2024) | $16.5B | Eli Lilly earnings |
| Revenue | Tirzepatide single quarter (Q3 2025) | $10.1B | Eli Lilly earnings |
| Revenue | Eli Lilly full-year 2025 guidance | $63B | Eli Lilly earnings |
| Adoption | U.S. adults currently on GLP-1 | 12.4% (1 in 8) | KFF, 2025 |
| Adoption | Women vs. men GLP-1 usage | 15% vs. 9% | KFF, 2025 |
| Adoption | Adults aged 50-64 usage | 22% | KFF, 2025 |
| Adoption | Medicaid GLP-1 prescriptions (2024) | 8.4 million | KFF |
| Adoption | Projected U.S. GLP-1 users (2030) | 25-30 million | JPMorgan |
| Search | "are peptides safe" 12-month growth | +652% | DataForSEO |
| Search | "how to get peptides" 12-month growth | +1,019% | DataForSEO |
| Search | "where to buy peptides" 12-month growth | +537% | DataForSEO |
| Clinical | Retatrutide Phase 3 weight loss | 28.7% body weight | Eli Lilly |
| Clinical | Orforglipron Phase 2 weight loss | 14.7% body weight | Eli Lilly |
| Clinical | Oral semaglutide weight loss | 16.6% body weight | Novo Nordisk |
| Clinical | Peptide drugs approved worldwide | ~100 | MDPI Pharmaceuticals |
| Clinical | Peptides in active development | 170+ | Precedence Research |
| Regulatory | Peptides reclassified to Category 1 | 14 of 19 | HHS, Feb 2026 |
| Regulatory | Medicare BALANCE model launch | July 2026 | CMS |
| Regulatory | Medicare BALANCE projected enrollees | 3.4 million | CMS |
| Health | U.S. obesity rate decline (2024-2025) | 39.9% to 37.0% | Gallup |
| Health | GLP-1 user calorie reduction | 21% fewer | JPMorgan |
| Health | GLP-1 user grocery spending reduction | 31% less | JPMorgan |
| Health | Americans viewing GLP-1 favorably | 53% (up from 42%) | Pew, Jan 2026 |
| Economic | Food industry revenue impact (by 2030) | $30-55B/year | JPMorgan |
| Economic | Bariatric surgery volume decline | 15% | Morgan Stanley |
| Safety | Online peptides with inaccurate content | 42% | JAMA, 2023 |
Frequently Asked Questions
How big is the peptide market in 2026?
The global peptide therapeutics market was valued at $52.6 billion in 2025 (Precedence Research), with a 9.65% CAGR projected through 2034. The GLP-1 segment alone reached $62.8 billion. Broader definitions including diagnostics and cosmeceuticals put the total above $140 billion (Grand View Research).
How many Americans take GLP-1 drugs?
Approximately 1 in 8 U.S. adults (12.4%) reported taking a GLP-1 drug as of late 2025 (KFF tracking poll). That doubled from 5.8% in 18 months. Women use them at higher rates than men (15% vs. 9%). Adults aged 50 to 64 report the highest usage at 22%.
How much revenue do GLP-1 drugs generate?
Semaglutide products (Ozempic and Wegovy) generated roughly $26 billion for Novo Nordisk in 2024. Tirzepatide products (Mounjaro and Zepbound) generated $16.5 billion for Eli Lilly. By Q3 2025, tirzepatide became the world's best-selling drug with $10.1 billion in a single quarter.
Are peptide searches growing on Google?
U.S. Google search volume for peptide-related queries grew between 400% and 1,019% from March 2025 to February 2026 (DataForSEO). "How to get peptides" grew 1,019%. "Are peptides safe" grew 652%. "Where to buy peptides" grew 537%. The growth reflects mainstream consumers entering the peptide market for the first time.
How many peptide drugs are FDA-approved?
Approximately 100 peptide-based drugs have received regulatory approval worldwide. The FDA approved 34 new peptide therapeutics between 2016 and 2024 (MDPI Pharmaceuticals, 2025). Over 170 peptide molecules are in active clinical development across oncology, metabolic disease, neurology, and infectious disease.
Has the U.S. obesity rate dropped because of GLP-1 drugs?
Gallup data shows the U.S. adult obesity rate dropped from 39.9% to 37.0% between early 2024 and late 2025, the first sustained decline in over a decade. That translates to roughly 7.6 million fewer obese adults. The CDC confirmed the trend in Data Brief #537, though updated measurement methods may also contribute.
What is retatrutide?
Retatrutide is a triple-agonist peptide (GLP-1, GIP, glucagon) developed by Eli Lilly. Phase 3 trials showed 28.7% average body weight loss at 68 weeks, the highest of any weight-loss drug tested. Seven additional Phase 3 readouts covering liver disease, sleep apnea, and cardiovascular outcomes are expected in 2026.
Where do these peptide statistics come from?
All statistics are sourced from pharmaceutical company earnings reports (Novo Nordisk, Eli Lilly), federal surveys (KFF, RAND, CDC, Gallup), ClinicalTrials.gov for pipeline data, market research firms (Precedence Research, Fortune Business Insights), and DataForSEO for search volume analysis. Each figure cites its specific source.
The Bottom Line
The peptide industry in 2026 is shaped by three converging forces: GLP-1 adoption that doubled in 18 months, regulatory shifts that restricted grey-market vendors while expanding clinical access, and a pipeline of next-generation drugs with unprecedented efficacy data.
Revenue is measured in tens of billions. The U.S. obesity rate declined for the first time in a decade. Consumer search interest grew by hundreds of percent. Medicare coverage for weight-loss drugs begins in July 2026. Peptides are one of the largest and fastest-moving segments in global pharmaceuticals.
We update this page quarterly. If you use these figures in your own research, cite this source for the most current data. New to peptides? Start with our beginner's guide or take the personalized peptide quiz to find the right protocol for your goals. For dosage planning, explore our semaglutide and tirzepatide dosage calculators.
Helpful Tools
Related Articles
FDA Peptide Crackdown 2026
Timeline of FDA actions against peptide vendors in 2025-2026. Why Peptide Sciences shut down, which peptides are banned, and what options remain.
Is Peptide Sciences Coming Back?
Is Peptide Sciences coming back? No. FDA regulatory analysis, company status update, and where to get research peptides now. Updated March 2026.
BPC-157 Dosage Per Body Weight (Chart)
BPC-157 dosage per body weight: 1.6-10 mcg/kg. Full chart for 120-280 lbs, injection vs oral doses, gender data, and why most practitioners prescribe flat 250-500 mcg.
Hims Weight Loss Reviews 2026: Pricing & Verdict
Hims weight loss review 2026: GLP-1 medications from $149/mo plus $149 membership. Branded Wegovy and Ozempic via Novo Nordisk partnership. Trustpilot 3.5/5. Full pricing, pros, cons, and Reddit sentiment.