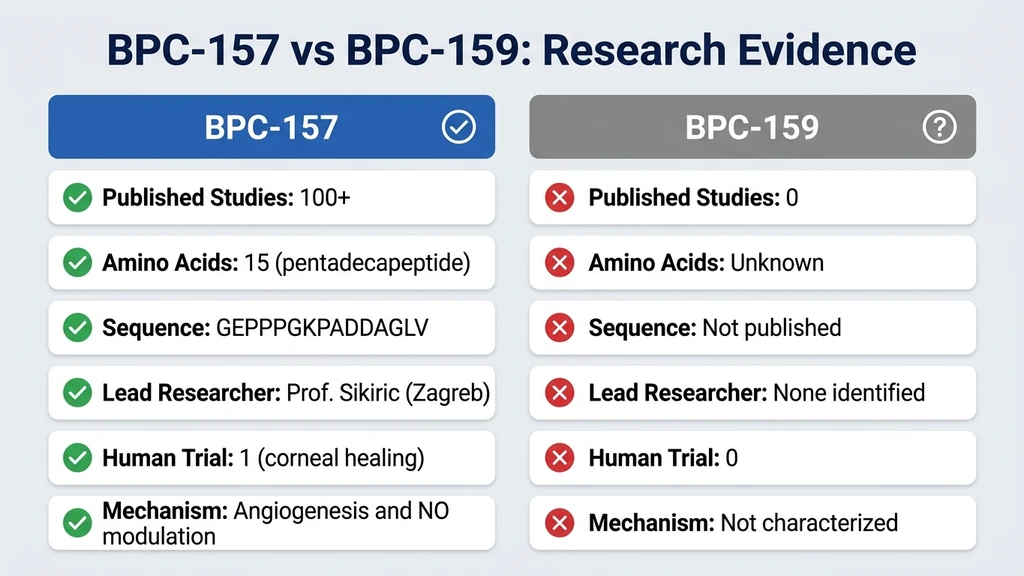
You found a product labeled "BPC-159" while shopping for BPC-157 and are now wondering whether it is the same thing, a newer version, or something else entirely. BPC-157 and BPC-159 are related but distinct peptides. BPC-157 is a 15-amino acid pentadecapeptide backed by over 100 published studies. BPC-159 is an 11-amino acid truncated fragment with virtually no independent published research. BPC-159 products emerged primarily after the FDA placed injectable BPC-157 on its category 2 risk list (Jozwiak et al., Pharmaceuticals, 2025). Consumers should understand that choosing BPC-159 means choosing a peptide with far less scientific evidence supporting its safety and efficacy.
| Feature | BPC-157 | BPC-159 |
|---|---|---|
| Amino acids | 15 (pentadecapeptide) | 11 (truncated fragment) |
| Published studies | 100+ since 1991 | Fewer than 5 independent |
| Human trial data | Phase II (ulcerative colitis), IV pilot | None |
| FDA status | Category 2 risk (injectable) | Not specifically listed |
| Gastric juice stability | 24+ hours (proven) | Unknown |
| Amino acid sequence | GEPPPGKPADDAGLV (confirmed) | Variable between manufacturers |
| Mechanism of action | Extensively characterized | Not established |
| Lead researcher | Predrag Sikiric, Univ. of Zagreb | No dedicated research group |
| Commercial availability | Widely available | Limited, newer products |
BPC-157 is not FDA-approved for any indication. Consult a healthcare provider before using any peptide.
The research gap between these two peptides is enormous. Understanding why BPC-159 exists and what it lacks will protect you from marketing claims disguised as science. For BPC-157 dosing protocols, see our BPC-157 dosage calculator.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is BPC-157?
BPC-157 stands for Body Protection Compound-157. It is a stable gastric pentadecapeptide consisting of 15 amino acids with the sequence GEPPPGKPADDAGLV. The peptide is a partial sequence of a larger protein naturally found in human gastric juice.
Research on BPC-157 began in the early 1990s under Predrag Sikiric at the University of Zagreb. Since then, over 100 preclinical studies have been published, documenting effects on wound healing, tendon repair, gut protection, blood vessel formation, and neurotransmitter modulation (Sikiric et al., Pharmaceuticals, 2024).
BPC-157 reached Phase II clinical trials for ulcerative colitis under the drug code PL14736, developed by Diagen d.o.o. A 2025 human safety pilot administered 20 mg intravenously with no adverse events (Safety of IV BPC157, Altern Ther Health Med, 2025). A systematic review confirmed the growing body of clinical evidence supporting its safety and biological activity (Pickering, Biomedicines, 2025).
Proven Mechanisms of BPC-157
Cytoprotection. BPC-157 protects cells against damage from NSAIDs, alcohol, and other toxic insults. It maintains organ integrity across the GI tract, liver, and brain (Sikiric et al., Curr Pharm Des, 2012).
Angiogenesis. The peptide promotes new blood vessel formation through VEGF upregulation and VEGFR2 pathway activation. This is the primary mechanism behind its tissue-healing effects.
Nitric oxide modulation. BPC-157 interacts with the NO system bidirectionally, capable of correcting both hypertension and hypotension in animal models. The Src-Caveolin-1-eNOS pathway is one specific route of action (Hsieh et al., Sci Rep, 2020).
Neurotransmitter effects. BPC-157 modulates dopamine D2 receptors, serotonin pathways, and GABAergic signaling (Sikiric et al., J Physiol Paris, 1999).
Gastric stability. The 15-amino acid sequence resists enzymatic degradation in gastric juice for over 24 hours. This stability is what makes oral administration viable and is the reason the peptide carries "stable" in its full name: stable gastric pentadecapeptide BPC-157.
Oral Bioavailability of BPC-157
BPC-157's stability in gastric juice sets it apart from most peptides, which degrade within minutes of oral ingestion. Research on the arginate salt form (BPC-157-arginate) has demonstrated oral activity in animal models for gut healing, liver protection, and tendon repair.
This oral bioavailability is a direct result of the 15-amino acid structure. The specific sequence creates a compact, enzymatically resistant conformation. For more on oral versus injectable BPC-157, see our BPC-157 oral vs injection comparison.
What Is BPC-159?
BPC-159 is a truncated fragment derived from the same parent Body Protection Compound protein as BPC-157. It consists of 11 amino acids, 4 fewer than BPC-157. The specific sequence removes residues from the BPC-157 chain, potentially eliminating functional domains that contribute to biological activity.
The peptide has no dedicated PubMed-indexed research. There are no independent studies testing BPC-159's effects on healing, cytoprotection, or any other endpoint. No human clinical trial data exists. No toxicology data exists. No stability data has been published.
Claims about BPC-159 benefits, particularly cognitive enhancement, cannot be traced to peer-reviewed literature. These claims appear exclusively in marketing materials from supplement companies selling BPC-159 products.
Why BPC-159 Has Almost No Research
The entire BPC-157 research program was built by Sikiric's group over three decades. They identified the 15-amino acid sequence as the minimal fragment that retains biological activity from the parent protein. BPC-159, being shorter, was either not tested or tested and found less effective. Neither scenario has been published.
No research group has adopted BPC-159 as a subject of investigation. No pharmaceutical company has filed an IND (Investigational New Drug) application for it. No patent protects a specific therapeutic use. The peptide exists commercially without a scientific foundation.
Do Fewer Amino Acids Mean Less Effective?
Not necessarily. Shorter peptides can retain activity if the critical binding domains remain intact. Some truncated peptides are actually more potent per residue than their longer counterparts.
The problem with BPC-159 is specific. BPC-157's 15-amino acid sequence includes the GKPADD motif, which is thought to contribute to receptor binding and biological activity. Removing 4 amino acids from a 15-residue chain means losing roughly 27% of the structural information. Whether the removed residues are critical depends on their position and role, and no published study answers this question for BPC-159.
Without comparative efficacy data, choosing BPC-159 is choosing an unknown. The assumption that "similar structure means similar function" has failed many times in pharmacology. Single amino acid substitutions can eliminate peptide activity entirely.
Structural and Stability Differences
The structural comparison between BPC-157 and BPC-159 reveals why the 4-amino acid difference matters for practical use.
Amino Acid Sequence Comparison
BPC-157 sequence: GEPPPGKPADDAGLV (15 residues)
BPC-159 has a different, shorter sequence of 11 residues. The exact published sequence for BPC-159 varies between commercial sources, which itself raises quality control questions. When manufacturers cannot agree on the sequence of the product they are selling, the research basis is clearly insufficient.
BPC-157's sequence has been confirmed across hundreds of publications and is registered under CAS number 137525-51-0. This level of standardization does not exist for BPC-159.
Solubility and Enzymatic Resistance
BPC-157 is water-soluble and stable in gastric juice at pH 1-2 for over 24 hours. This gastric stability is the reason it works orally. The compact 15-residue structure resists pepsin, trypsin, and other digestive enzymes that rapidly destroy most peptides.
BPC-159's solubility and enzymatic resistance have not been characterized in published research. Shorter peptides are generally less stable because they have fewer intramolecular interactions to maintain structural integrity. If BPC-159 degrades faster in gastric juice, oral formulations would deliver less active peptide to the target tissue.
This is not speculation about which peptide is "better." It is a statement of what has been measured versus what has not. BPC-157's stability is proven. BPC-159's is unknown.
Research Evidence: 100+ Studies vs Nearly Zero
The evidence gap between BPC-157 and BPC-159 is the single most important factor in this comparison.
BPC-157 Research Base
Preclinical studies: Over 100 published papers across wound healing, tendon repair, gut protection, liver protection, brain injury, bone healing, and vascular function.
Human data: Phase II clinical trial for ulcerative colitis (PL14736). A 2025 IV safety pilot at 20 mg showing no adverse events. Systematic reviews published in 2025 confirming the evidence base (Pickering, Biomedicines, 2025).
Mechanism characterization: Multiple signaling pathways identified and confirmed across independent studies. VEGFR2, PI3K-Akt-eNOS, Src-Cav-1-eNOS, dopaminergic, serotonergic, GABAergic, and NO systems.
Safety data: No LD1 achieved in animal toxicity studies. Phase II trial showed no significant adverse events. Human IV pilot confirmed tolerability at doses far exceeding typical use (Sikiric et al., Gut Liver, 2020).
BPC-159 Research Base
Preclinical studies: No PubMed-indexed studies specifically examining BPC-159 as a distinct therapeutic entity.
Human data: None.
Mechanism characterization: None.
Safety data: None.
Oral activity: Unknown.
Cognitive enhancement claims: Not supported by any peer-reviewed publication. These claims appear exclusively in marketing materials and cannot be verified against scientific literature.
The marketing of BPC-159 relies entirely on borrowing credibility from BPC-157 research. Product pages cite BPC-157 studies to support BPC-159 claims. This is scientifically invalid. A different peptide, even a related one, requires its own evidence.
Why Does BPC-159 Exist? The Regulatory Context
Understanding why BPC-159 products appeared on the market requires understanding the regulatory pressure on BPC-157.
In 2024, the FDA placed injectable BPC-157 on its category 2 risk list for bulk drug substances used in compounding. This designation did not ban BPC-157 outright, but it signaled regulatory scrutiny that made compounding pharmacies and supplement companies nervous about their supply chain.
BPC-159 products began appearing shortly after. The marketing pitch: BPC-159 is a "next-generation" or "improved" version of BPC-157 that falls outside the FDA's specific category 2 listing. Some companies market it as a dietary supplement rather than a compounded drug, exploiting a regulatory gray zone.
This is regulatory arbitrage, not science. BPC-159 is not "next-generation" because no generation of research has produced it. It is structurally different enough from BPC-157 to avoid specific regulatory restrictions while being marketed using BPC-157's research data.
The consumer risk: paying premium prices for a peptide with no independent evidence, based on the assumption that proximity to a well-researched peptide confers the same benefits. For context on peptide legality, see our legality guide.
Fact-Checking BPC-159 Marketing Claims
Several claims appear repeatedly in BPC-159 marketing. Here is what the evidence actually supports.
Claim: BPC-159 Enhances Cognition
Verdict: Unverified. No published study has measured BPC-159's effects on cognition, memory, focus, or any neurological endpoint. BPC-157 has documented interactions with dopaminergic and serotonergic systems (Sikiric et al., 1999), but these findings cannot be transferred to a structurally different peptide without testing.
Some BPC-157 research shows neuroprotective effects in brain injury models. BPC-159 marketing extrapolates from these studies, which is not valid science. The specific amino acids removed from BPC-157 to create BPC-159 may include residues critical for blood-brain barrier penetration or receptor binding.
Claim: BPC-159 Is Safer Because It Is Shorter
Verdict: Unsupported. Peptide safety is not correlated with length. Shorter peptides can be more toxic, equally safe, or less safe than longer ones depending on their specific interactions. BPC-157's safety has been demonstrated across toxicology studies, a Phase II trial, and a human IV pilot. BPC-159 has zero safety data.
Choosing BPC-159 for "safety" reasons means choosing a peptide with no safety testing over one with extensive safety documentation. The logic inverts the evidence.
Claim: BPC-159 Is the 'Next Generation' of BPC-157
Verdict: Marketing language. "Next generation" implies iterative improvement based on research. BPC-159 was not developed through systematic optimization of BPC-157. No research group published findings showing BPC-159 outperforms BPC-157 on any endpoint. The term is commercial positioning, not a scientific designation.
True next-generation development would involve: identifying BPC-157's active site, engineering modifications to improve potency or stability, testing the modified peptide in preclinical models, and publishing results. None of these steps have occurred for BPC-159.
Should You Choose BPC-157 or BPC-159?
The decision framework is straightforward when framed by the evidence.
When BPC-157 Is the Clear Choice
For any researched indication. Gut healing, tendon repair, wound healing, joint support, liver protection. BPC-157 has published data for all of these. BPC-159 has data for none.
When oral bioavailability matters. BPC-157's gastric stability is proven. If you plan to take capsules rather than injections, BPC-157 (particularly the arginate form) has demonstrated oral activity. BPC-159's oral survival is unknown. For more on this topic, see can you take BPC-157 orally and our BPC-157 capsule side effects guide.
When safety data matters. Over 30 years of preclinical research, Phase II trial data, and a human IV safety pilot support BPC-157. Choosing the peptide with established safety data is the rational default.
When quality control matters. BPC-157 has a confirmed amino acid sequence (GEPPPGKPADDAGLV) and CAS number (137525-51-0). Testing and verification are standardized. BPC-159's sequence varies between manufacturers, making quality verification difficult.
When BPC-159 Might Be Considered
Only if BPC-157 is genuinely unavailable in your jurisdiction. Some regions may restrict BPC-157 access following FDA category 2 classification while BPC-159 remains available.
With the understanding that evidence is minimal. You are accepting a peptide with no published efficacy data, no safety data, and no stability data.
As part of a blend product where BPC-159 is the only available component. Some supplement companies offer combination products containing BPC-159. If BPC-157 alternatives exist, choose them first.
The price comparison matters too. BPC-159 products are sometimes marketed at a premium as "advanced" or "enhanced" formulations. Paying more for less evidence is a poor trade.
The Bottom Line on Evidence Quality
Science is not a spectrum of opinions. Either a peptide has been tested and the results published, or it has not. BPC-157 has been tested. BPC-159 has not.
A comprehensive review of BPC-157's multifunctionality cataloged its effects across wound healing, GI protection, musculoskeletal repair, neurological protection, and cardiovascular function (Jozwiak et al., Pharmaceuticals, 2025). The breadth of documented activity reflects decades of systematic investigation.
BPC-159 cannot point to a single entry in that catalog. Every claim made about BPC-159 is either borrowed from BPC-157 data or generated by marketing teams. When a company cites "BPC research" to sell BPC-159, ask which specific BPC-159 study they are referencing. The answer, as of 2026, is that none exists.
This does not mean BPC-159 is dangerous or ineffective. It means we do not know. In a field where YMYL (Your Money or Your Life) decisions are involved, "we do not know" is the honest answer that should guide your choice.
Frequently Asked Questions
Is BPC-159 the same as BPC-157?
No. BPC-157 is a 15-amino acid peptide with over 100 published preclinical studies, Phase II clinical trial data, and a confirmed amino acid sequence (GEPPPGKPADDAGLV, CAS 137525-51-0). BPC-159 is an 11-amino acid truncated fragment from the same parent protein with virtually no independent published research. They share an origin but are structurally and evidentially different. See our BPC-157 benefits for what the research actually supports.
Is BPC-159 safer than BPC-157?
There is no evidence that BPC-159 is safer. BPC-157 has extensive safety data: no LD1 in animal toxicity studies, a Phase II trial with no significant adverse events, and a 2025 human pilot at 20 mg IV with zero reported side effects. BPC-159 has no published safety data of any kind. Choosing BPC-159 for safety means choosing an untested peptide over a well-documented one.
Does BPC-159 really enhance cognition?
No published study supports this claim. BPC-159 cognitive enhancement marketing cannot be traced to peer-reviewed research. BPC-157 has documented neurotransmitter effects (dopamine D2, serotonin modulation), but these findings do not automatically apply to BPC-159. Removing 4 amino acids from a 15-residue peptide may eliminate the domains responsible for neurological activity.
Is BPC-159 legal?
BPC-159 is not specifically listed on the FDA's category 2 risk list, unlike injectable BPC-157. Some companies market it as a dietary supplement. Legal status varies by jurisdiction and may change as regulators evaluate newer peptide products. Legality does not equal safety or efficacy. For current peptide regulations, see our peptide legality guide.
Can I use BPC-159 and BPC-157 together?
No published research has studied this combination. Since BPC-159's mechanism of action is unknown, potential interactions with BPC-157 are entirely unpredictable. If you have access to BPC-157, adding BPC-159 offers no documented benefit. The rational approach: use BPC-157 alone at established doses (250-500 mcg daily) where the safety and efficacy data actually exists.
Why is BPC-159 cheaper than BPC-157 at some vendors?
An 11-amino acid peptide costs less to synthesize than a 15-amino acid peptide. Lower synthesis costs translate to lower retail prices. Some vendors market BPC-159 at a premium by labeling it as 'advanced' or 'next-generation,' but the manufacturing cost is objectively lower. Price does not reflect efficacy when one peptide has 100+ studies and the other has near zero.
Will BPC-159 replace BPC-157?
Highly unlikely based on current evidence. BPC-157's research base took 30+ years and hundreds of studies to build. BPC-159 would need to duplicate that effort independently to be considered a replacement. No research group has undertaken this work. BPC-159 exists as a commercial product, not a scientifically validated therapeutic candidate.
Where can I find BPC-159 research?
As of April 2026, searching PubMed for 'BPC-159' returns no dedicated studies on this specific peptide. Results that appear reference the broader BPC family or BPC-157 specifically. Marketing materials citing 'research' are referencing BPC-157 studies, not BPC-159 data. This absence of dedicated research is the defining characteristic of BPC-159.
The Bottom Line
BPC-157 and BPC-159 are not interchangeable. BPC-157 is a 15-amino acid peptide supported by over 100 published studies, Phase II clinical trial data, and a 2025 human safety pilot. BPC-159 is an 11-amino acid truncated fragment with no independent published research, no human data, and no established mechanism of action.
BPC-159 products exist because of regulatory pressure on BPC-157, not because of scientific advancement. The "next-generation" label is marketing. The cognitive enhancement claims are unverified. The safety profile is unknown.
Choose BPC-157 whenever it is available. Use our BPC-157 dosage calculator to plan your protocol. For information on BPC-157 benefits, BPC-157 side effects, and oral vs injection routes, explore our comprehensive BPC-157 content library. If you have questions about where to obtain BPC-157 from verified sources, see our sourcing resources.
Helpful Tools
Related Articles
BPC-157 TB-500 Side Effects: Stack Safety Data
BPC-157 TB-500 stack side effects: headaches, fatigue weeks 1-2, anaphylaxis risk, cancer screening tips. Week-by-week timeline inside.
Peptide Therapy Side Effects: Complete Guide (2026)
Peptide therapy side effects by category: GLP-1, GH-releasing, healing, and immune. Severity matrix, incidence rates, and management strategies.
Tesamorelin vs CJC-1295: FDA GHRH vs Research
Tesamorelin vs CJC-1295: FDA GHRH (-18% visceral fat, Phase III) vs research peptide (2-10x GH, Phase I). Cost, dosing, and who picks which.
BPC-157 vs TB-500: Which Healing Peptide Wins?
BPC-157 vs TB-500 compared: mechanism, dosage, cost, half-life, and best use cases for tendon injuries, muscle tears, gut healing, and post-surgery recovery.