
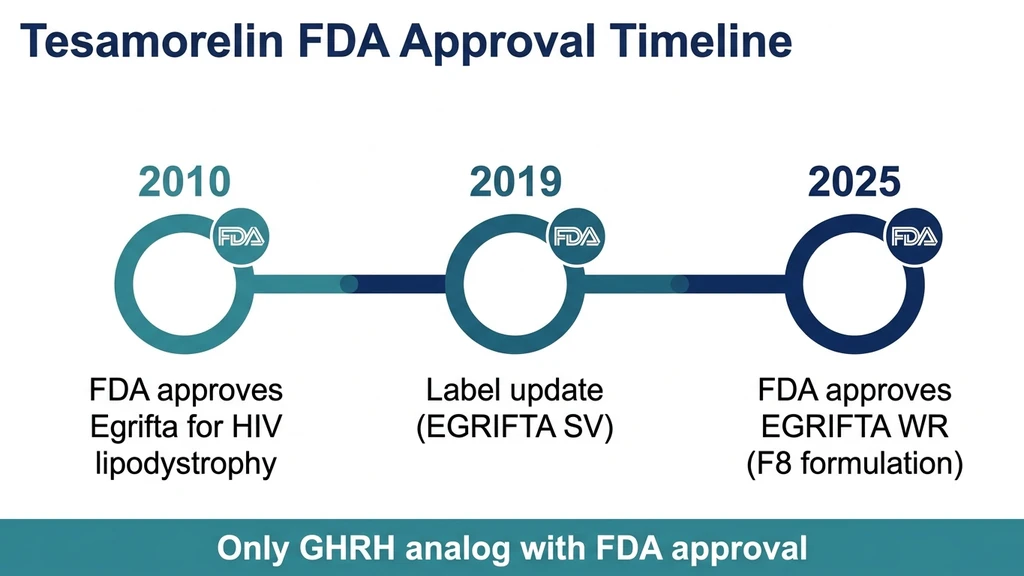
You found a peptide clinic offering tesamorelin for belly fat and want to know if this compound has real regulatory backing or if it is another grey-market research chemical. Yes, tesamorelin is FDA approved. The FDA approved it on November 10, 2010, under the brand name Egrifta, for the reduction of excess abdominal fat in HIV-positive adults with lipodystrophy. It remains the only growth hormone-releasing hormone (GHRH) analog with active FDA approval in the United States.
| Quick Reference | Details |
|---|---|
| FDA approved? | Yes (November 10, 2010) |
| Brand name | Egrifta (original), Egrifta SV, Egrifta WR (2025) |
| Approved indication | Reduction of excess visceral abdominal fat in adults with HIV and lipodystrophy |
| Manufacturer | Theratechnologies Inc. |
| Drug class | Growth hormone-releasing hormone (GHRH) analog |
| Standard dose | 2 mg subcutaneous injection, once daily |
| Pivotal trials | Two Phase 3 trials, 816 total participants |
| Off-label use | Legal with prescription; common for general visceral fat reduction, anti-aging |
| Regulatory comparison | Only GHRH analog with FDA approval; sermorelin, ipamorelin, and CJC-1295 lack current FDA approval |
That approval makes tesamorelin unique among growth hormone peptides. While compounds like sermorelin, ipamorelin, and CJC-1295 are widely used in anti-aging and body composition protocols, none of them hold active FDA approval for any indication. If you are evaluating tesamorelin specifically for fat loss, see the tesamorelin dosage for fat loss guide. For a broader look at peptide legality, that resource covers the current regulatory landscape.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is Tesamorelin and Why Does FDA Approval Matter?
Tesamorelin is a synthetic analog of growth hormone-releasing hormone, the 44-amino-acid peptide your hypothalamus produces to signal the pituitary gland to release growth hormone. Theratechnologies developed tesamorelin by attaching a trans-3-hexenoic acid group to the tyrosine at position 1, which shields the molecule from rapid enzymatic degradation and extends its plasma half-life to approximately 26 minutes after subcutaneous injection (Stanley & Bhatt, Expert Opin Pharmacother 2012).
Unlike direct growth hormone injections, tesamorelin works upstream. It stimulates your pituitary to release GH in a pulsatile, physiological pattern rather than flooding your bloodstream with a flat supraphysiological spike. The pituitary's negative feedback loop stays intact: when GH and IGF-1 levels climb high enough, somatostatin shuts down further release. This self-regulating mechanism is the reason tesamorelin carries fewer metabolic risks than exogenous GH.
FDA approval matters here for three concrete reasons:
- 1.Safety data exists. The approval process required two Phase 3 randomized controlled trials with over 800 participants, plus long-term extension studies. That level of human safety data does not exist for ipamorelin, CJC-1295, or HGH Fragment 176-191.
- 1.Manufacturing standards are enforced. Egrifta is produced under cGMP (current Good Manufacturing Practice) standards that the FDA audits regularly. Compounded peptides from research suppliers are not held to the same standard, which is one reason the FDA peptide crackdown of 2026 focused so heavily on compounding pharmacy oversight.
- 1.Off-label prescribing becomes legal. Once a drug has FDA approval for any indication, licensed physicians can legally prescribe it for other conditions they deem medically appropriate. Without FDA approval, a compound cannot be prescribed at all through traditional pharmacy channels.
How Tesamorelin Differs from Other GHRH Analogs
The trans-3-hexenoic acid modification at position 1 is not cosmetic chemistry. Natural GHRH(1-44) has a plasma half-life under 7 minutes because dipeptidyl peptidase IV (DPP-IV) rapidly cleaves it. Sermorelin (GRF 1-29) improved on this slightly but still has a half-life of roughly 10 to 12 minutes. Tesamorelin's chemical shield extends that to 26 minutes, long enough for a single daily injection to produce a full GH pulse.
CJC-1295 with DAC took a different approach: it attaches a Drug Affinity Complex that binds to albumin, extending the half-life to roughly 6 to 8 days. The trade-off is that CJC-1295 with DAC produces a sustained, non-pulsatile GH elevation that does not mimic natural physiology. Tesamorelin occupies a middle ground: long enough to work, short enough to maintain pulsatility.
| Peptide | Half-Life | GH Release Pattern | FDA Status |
|---|---|---|---|
| Natural GHRH(1-44) | < 7 min | Pulsatile | N/A (endogenous) |
| Sermorelin (GRF 1-29) | 10-12 min | Pulsatile | Withdrawn 2008 (commercial) |
| Tesamorelin (GHRH analog) | ~26 min | Pulsatile | FDA approved (2010) |
| CJC-1295 with DAC | 6-8 days | Sustained/blunted | Not approved |
| Ipamorelin (GHS) | ~2 hours | Pulsatile | Not approved |
| HGH Fragment 176-191 | ~30 min | Does not stimulate GH | Not approved |
For detailed comparisons, see tesamorelin vs CJC-1295, tesamorelin vs ipamorelin, and the full tesamorelin vs sermorelin vs ipamorelin breakdown.
The Complete FDA Approval Timeline for Tesamorelin
Tesamorelin's regulatory history spans over 15 years of development, clinical trials, and post-market updates. Understanding this timeline provides context for the strength of its evidence base and why no other GH peptide has achieved the same regulatory standing.
2003-2007: Phase 2 and Phase 3 Clinical Trials
Theratechnologies (originally Theralogics) began clinical development of tesamorelin (then designated TH9507) in the early 2000s. The target indication was HIV-associated lipodystrophy, a disfiguring condition where antiretroviral therapy causes pathological redistribution of body fat, particularly accumulating visceral fat around abdominal organs.
Phase 2 dose-ranging studies established that 2 mg/day subcutaneous injection produced optimal visceral fat reduction with an acceptable safety profile. Higher doses did not significantly improve efficacy, consistent with the pituitary's self-limiting GH release mechanism.
Two pivotal Phase 3 trials enrolled a combined 816 HIV-positive adults with documented lipodystrophy:
- Trial 1 (SEROST-0502): 412 participants, 26 weeks, tesamorelin 2 mg vs. placebo. Visceral adipose tissue (VAT) decreased by 15.2% in the tesamorelin group versus 5.0% in the placebo group (P < 0.001) (Falutz et al., NEJM 2007).
- Trial 2 (SEROST-0504): 404 participants, 26 weeks, similar design. VAT decreased by 18.4% with tesamorelin versus 2.2% with placebo (P < 0.001) (Falutz et al., J Clin Endocrinol Metab 2010).
Combined analysis showed that 69% of tesamorelin-treated participants achieved a clinically meaningful VAT reduction of 8% or greater, compared to 33% of placebo recipients. Trunk fat, waist circumference, and waist-to-hip ratio all improved. Subcutaneous fat and limb fat did not change, confirming that tesamorelin selectively targets visceral deposits.
November 10, 2010: FDA Grants Original Approval (Egrifta)
The FDA approved tesamorelin for injection (brand name Egrifta) on November 10, 2010, under BLA (Biologics License Application) 022505. The approved indication: reduction of excess abdominal fat in HIV-infected patients with lipodystrophy.
The original formulation required daily reconstitution of two vials (one containing tesamorelin powder, one containing sterile water for injection) to achieve the 2 mg dose. Patients mixed the solution fresh each morning and injected subcutaneously into the abdomen.
Key label details from the original approval:
- Dosing: 2 mg subcutaneous injection once daily
- Contraindications: Disruption of the hypothalamic-pituitary axis due to hypophysectomy, hypopituitarism, or pituitary tumor/surgery; active malignancy; pregnancy; known hypersensitivity to tesamorelin or mannitol
- Warnings: Fluid retention, potential glucose metabolism changes, hypersensitivity reactions, injection site reactions
- Monitoring: IGF-1 levels at baseline and during treatment; discontinuation recommended if no improvement in lipodystrophy after 26 weeks
The approval was a milestone for the peptide therapy field. Before Egrifta, the only option for HIV lipodystrophy was cosmetic procedures or switching antiretroviral regimens, neither of which addressed the metabolic consequences of visceral fat accumulation.
2019: Label Update (Egrifta SV)
In 2019, the FDA approved a reformulation called Egrifta SV (Single Vial). This version simplified the reconstitution process by packaging tesamorelin as a single vial requiring only one reconstitution step instead of two. The dose remained 2 mg/day. The clinical data, efficacy, and safety profile were unchanged.
Egrifta SV reduced preparation time from approximately 5 minutes (two-vial mixing) to under 2 minutes. For patients injecting daily, that convenience improvement mattered for long-term adherence.
March 2025: FDA Approves Egrifta WR (F8 Formulation)
On March 25, 2025, the FDA approved the supplemental Biologics License Application for Egrifta WR (Weekly Reconstitution), also called the F8 formulation. This represented the most significant formulation change since original approval.
Egrifta WR contains 11.6 mg of tesamorelin per vial, enough for seven daily doses after a single reconstitution. The daily injection dose is 1.28 mg (0.16 mL of the reconstituted solution) rather than the original 2 mg, reflecting improved bioavailability from the concentrated formulation. Once reconstituted, the solution remains stable for seven days at room temperature (20 to 25 degrees Celsius).
The practical benefits are substantial:
- Weekly reconstitution instead of daily: one preparation session per week
- 50% smaller injection volume compared to Egrifta SV
- Room temperature storage after reconstitution (no refrigeration needed)
- Four single-patient-use vials per monthly supply
For patients and clinicians managing tesamorelin dosage for fat loss, the WR formulation maintains the same clinical efficacy while reducing treatment burden. The availability of Egrifta WR through specialty pharmacies began in September 2025.
What Exactly Did the FDA Approve Tesamorelin For?
The FDA approved tesamorelin for one specific, narrow indication: the reduction of excess abdominal fat in HIV-infected adult patients with lipodystrophy. Understanding the boundaries of this approval is critical for anyone considering tesamorelin, because the gap between what the FDA approved and how the compound is actually used in practice is significant.
The On-Label Indication: HIV-Associated Lipodystrophy
HIV-associated lipodystrophy is a metabolic syndrome that develops in a substantial percentage of patients on long-term antiretroviral therapy (ART). It manifests as abnormal fat redistribution: visceral fat accumulates around abdominal organs while subcutaneous fat wastes from the face, limbs, and buttocks. The visceral fat accumulation is not merely cosmetic. It drives insulin resistance, dyslipidemia, elevated cardiovascular risk, and non-alcoholic fatty liver disease.
Before tesamorelin, physicians had limited options. Switching ART regimens sometimes helped but risked viral resistance. Metformin and thiazolidinediones showed modest effects on visceral fat in this population. Growth hormone itself reduced visceral fat but caused unacceptable rates of insulin resistance and fluid retention at the doses needed.
Tesamorelin solved the GH problem. By stimulating endogenous pulsatile GH release rather than delivering supraphysiological GH directly, it reduced visceral fat by 15 to 18% over 26 weeks without the metabolic penalties of exogenous GH. The Phase 3 data was clean enough that the FDA approved it as the first and still only medication specifically indicated for HIV lipodystrophy.
The label specifies that treatment should be reassessed after 26 weeks. If a patient shows no meaningful improvement in lipodystrophy by that point, the prescribing information recommends discontinuation. In practice, most responders see measurable VAT reduction within 12 weeks, which aligns with the timeline data from both pivotal trials.
What the FDA Did Not Approve Tesamorelin For
The FDA did not approve tesamorelin for:
- General weight loss or obesity
- Bodybuilding or athletic performance enhancement
- Anti-aging or longevity
- Non-alcoholic fatty liver disease (NAFLD) in non-HIV patients
- Cognitive enhancement or neuroprotection
- Growth hormone deficiency (adult or pediatric)
This distinction matters because every single one of those use cases has published research supporting tesamorelin's potential. A randomized trial in Lancet HIV showed tesamorelin reduced liver fat by 37% in HIV patients with NAFLD, with 35% of treated patients dropping below the 5% hepatic fat threshold that defines NAFLD (Stanley et al., Lancet HIV 2019). A controlled trial published in Archives of Neurology found that 20 weeks of GHRH analog administration improved executive function in both healthy older adults and those with mild cognitive impairment (Baker et al., Arch Neurol 2012).
The research is promising. But "promising research" and "FDA approved" are different regulatory categories, and conflating them is a common source of confusion in the peptide space. For a broader perspective on peptide legality and the difference between approved and research compounds, that guide covers the full landscape.
Off-Label Use: What Is Legal and What Is Not
The most common question after "is tesamorelin FDA approved?" is "can I get it for fat loss even though I do not have HIV?" The answer involves understanding off-label prescribing, which is both more common and more legally straightforward than most people realize.
How Off-Label Prescribing Works
Once the FDA approves a drug for any indication, a licensed physician can legally prescribe it for any condition they judge medically appropriate for their patient. This is called off-label prescribing, and it accounts for roughly 20% of all prescriptions written in the United States.
Off-label prescribing is not a loophole. It is a recognized, legal component of medical practice. The FDA regulates drug approval and marketing, but it does not regulate the practice of medicine. A physician who prescribes tesamorelin for visceral fat reduction in a non-HIV patient is exercising clinical judgment within the bounds of their medical license.
Here is what off-label prescribing requires:
- 1.A valid patient-physician relationship. Telemedicine consults qualify, but the relationship must be genuine and documented.
- 2.Medical justification. The physician must have a reasonable clinical basis for believing the drug will benefit the patient. For tesamorelin, the Phase 3 visceral fat reduction data provides that basis.
- 3.Informed consent. The patient must understand that the use is off-label and that the specific indication has not been evaluated by the FDA.
- 4.A legitimate prescription. The prescription must be filled through a licensed pharmacy (retail or specialty).
What off-label prescribing does not cover: purchasing tesamorelin from research chemical suppliers without a prescription, self-administering without medical oversight, or importing from overseas pharmacies without FDA authorization.
The Off-Label Landscape in 2026
Following the FDA peptide crackdown of 2026, the distinction between FDA-approved and non-approved peptides became sharper. The FDA moved several popular peptides, including BPC-157 and various GH secretagogues, off the Category 2 compounding list, restricting their availability through compounding pharmacies.
Tesamorelin was not affected by this crackdown because it holds active FDA approval. This is the practical benefit of regulatory status: when enforcement tightens, approved compounds remain accessible through standard prescription channels while unapproved compounds face supply disruptions.
The current off-label prescribing landscape for tesamorelin includes:
- Anti-aging clinics prescribing it for visceral fat reduction in non-HIV patients
- Hormone optimization practices combining it with other GH secretagogues (commonly ipamorelin) for synergistic effects
- Hepatology research exploring its effects on NAFLD in non-HIV populations
- Neurology research investigating cognitive protection in aging populations
For patients comparing tesamorelin to other GH peptides that may be harder to access, see tesamorelin vs ipamorelin and tesamorelin vs sermorelin vs ipamorelin. The sermorelin for fat loss guide covers another GHRH analog with a different regulatory history.
Insurance Coverage and Cost Implications
FDA approval does not automatically mean insurance coverage. Most insurers cover Egrifta only for the on-label indication (HIV lipodystrophy) and require prior authorization demonstrating:
- Confirmed HIV diagnosis on antiretroviral therapy
- Documented lipodystrophy with excess visceral fat
- CT or MRI evidence of visceral adiposity
- Failure of other interventions (in some plans)
Off-label prescriptions for non-HIV patients are almost universally denied by insurance. Out-of-pocket cost for brand-name Egrifta runs approximately $1,500 to $2,500 per month at retail pricing. Some specialty pharmacies and manufacturer assistance programs reduce this for qualifying patients.
Compounded tesamorelin, available through 503B outsourcing facilities, costs significantly less (typically $200 to $600 per month) but does not carry the brand-name label. The compounded version uses the same active molecule but is not FDA-verified for bioequivalence. For a cost comparison across GH peptides, use our peptide cost calculator.
The Clinical Evidence Behind FDA Approval
The FDA does not approve drugs based on mechanism theory or animal studies. Tesamorelin's approval rested on a specific body of human clinical evidence. Reviewing the pivotal trials in detail explains why the FDA was convinced and why no other GH peptide has matched this regulatory achievement.
Phase 3 Visceral Fat Reduction Trials
The two Phase 3 trials were randomized, double-blind, placebo-controlled studies conducted across multiple centers. Both used CT scan measurements of visceral adipose tissue area at the L4-L5 vertebral level, the gold standard for quantifying visceral fat.
Combined results (816 participants):
| Outcome | Tesamorelin 2 mg | Placebo | P-value |
|---|---|---|---|
| VAT change (26 weeks) | -15 to -18% | -0.6 to -5% | < 0.001 |
| VAT reduction (absolute) | -21 to -34 cm2 | -1 to +8 cm2 | < 0.001 |
| Responders (>=8% VAT reduction) | 69% | 33% | < 0.001 |
| Trunk fat change | Significant decrease | No change | < 0.001 |
| Waist circumference | -2.1 cm | -0.3 cm | 0.02 |
| Subcutaneous fat | No change | No change | NS |
| Limb fat | No change | No change | NS |
The selectivity of the fat reduction was notable. Tesamorelin reduced visceral fat without depleting the subcutaneous fat or limb fat that HIV patients were already losing from lipodystrophy. This selectivity comes from the higher density of beta-adrenergic receptors in visceral adipose tissue, which makes it more responsive to GH-mediated lipolysis (Moller & Jorgensen, Endocr Rev 2009).
For detailed dosing from these trials, see the tesamorelin dosage for fat loss guide. To understand how these results compare to the CJC-1295 + ipamorelin dosage calculator protocols, that tool provides side-by-side dosing estimates.
Long-Term Extension Data: 52 Weeks
The Phase 3 trials included 52-week extension phases where participants who responded at 26 weeks continued treatment. This extension data was critical for FDA reviewers assessing sustained efficacy and long-term safety.
Key findings from the 52-week data:
- VAT reduction was maintained through week 52 in participants who continued tesamorelin. Those who switched to placebo at week 26 regained visceral fat, returning to near-baseline levels by week 52, confirming that the effect requires ongoing treatment (Falutz et al., J Clin Endocrinol Metab 2010).
- Metabolic markers improved in sustained responders. Triglycerides decreased, adiponectin increased, and glucose homeostasis remained stable over the full year.
- IGF-1 levels stabilized. The initial increase in IGF-1 (expected with any GH-stimulating therapy) plateaued within normal range by week 12 and remained stable through week 52, confirming the pituitary feedback loop was functioning correctly.
- No new safety signals emerged between week 26 and week 52. The adverse event profile in the extension phase matched the initial 26-week data.
The fat regain after discontinuation is an important clinical detail. Tesamorelin does not permanently restructure fat distribution. It maintains reduced visceral fat as long as treatment continues, similar to how statin therapy maintains cholesterol reduction only during active use.
NAFLD and Liver Fat Data
Although not part of the original FDA approval package, subsequent controlled trials expanded the evidence for tesamorelin's metabolic benefits. The most significant was a randomized, double-blind, multicenter trial examining tesamorelin's effect on non-alcoholic fatty liver disease in HIV patients.
Stanley et al. (Lancet HIV, 2019): 61 HIV-infected men and women with hepatic fat fraction of 5% or greater (defined as NAFLD) were randomized to tesamorelin 2 mg/day or placebo for 12 months.
Results: - Hepatic fat fraction decreased by 37% (absolute effect: -4.1%, P = 0.02) in the tesamorelin group versus placebo - 35% of tesamorelin-treated patients dropped below the 5% threshold, resolving their NAFLD classification, versus 4% of placebo patients - Fibrosis progression was substantially attenuated in the tesamorelin group - Hepatic expression of genes involved in oxidative phosphorylation increased, while genes for inflammation and tissue repair decreased
(Stanley et al., Lancet HIV 2019)
These findings are relevant because visceral fat and liver fat are metabolically linked. Tesamorelin's ability to reduce both suggests that its mechanism operates at the level of hepatic and visceral lipid metabolism, not just peripheral fat mobilization. This data supports the off-label use case but has not led to a label expansion because the study was conducted exclusively in HIV patients.
Cognitive Function Research
A separate line of research, unrelated to the HIV lipodystrophy indication, has explored GHRH analog effects on brain function. Growth hormone and IGF-1 have established roles in neurogenesis, synaptic plasticity, and neuroprotection, and GHRH receptors are expressed throughout the central nervous system.
Baker et al. (Archives of Neurology, 2012): 152 adults (76 healthy older adults and 76 with mild cognitive impairment) were randomized to 20 weeks of tesamorelin or placebo. Tesamorelin improved executive function (task switching accuracy, Stroop interference, verbal fluency) in both groups. Adults with MCI showed preserved cognitive scores where the placebo group declined. GABA levels increased across multiple brain regions in treated participants (Baker et al., Arch Neurol 2012).
A follow-up MRI study found that tesamorelin preserved hippocampal volume compared to placebo, consistent with IGF-1's known role in promoting neurogenesis and reducing neuronal apoptosis in the hippocampus.
These findings have not been pursued for FDA approval. The trial was relatively small and short, and the FDA would require larger, longer Phase 3 trials with defined cognitive endpoints to consider an approval for this indication. But the data adds another dimension to tesamorelin's evidence base that no other GH peptide can claim.
Safety Profile: What the FDA Reviewed
The FDA's safety review for tesamorelin was based on the combined Phase 3 data plus extension studies. The adverse event profile is well-characterized because the sample size (816 participants) and duration (up to 52 weeks) exceed what is available for any non-approved GH peptide.
Common Adverse Events
The most frequently reported adverse events in clinical trials, compared to placebo:
| Adverse Event | Tesamorelin (%) | Placebo (%) | Clinical Significance |
|---|---|---|---|
| Injection site reactions (erythema, pruritus, pain) | 8.5% | 3.0% | Mild, self-limiting |
| Arthralgia (joint pain) | 13.3% | 10.9% | Usually transient, resolves weeks 2-4 |
| Peripheral edema | 6.1% | 2.8% | Fluid retention from GH effect |
| Myalgia (muscle pain) | 5.5% | 3.1% | Mild, first 2 weeks |
| Paresthesia (numbness/tingling) | 4.8% | 2.0% | Carpal tunnel risk if persistent |
| Nausea | 4.5% | 3.5% | Typically resolves within days |
| Rash/urticaria | 3.6% | 1.5% | Monitor for hypersensitivity |
The overall discontinuation rate due to adverse events was 6.8% for tesamorelin versus 3.2% for placebo. No treatment-related deaths occurred in any clinical trial.
For context on how these rates compare to other growth hormone peptides, see is sermorelin safe, which shows similar adverse event patterns at lower frequencies (consistent with sermorelin's shorter half-life and lower potency).
Glucose Metabolism: The Key Safety Concern
The FDA's primary safety concern with tesamorelin was its effect on glucose metabolism. Growth hormone is a counter-regulatory hormone that opposes insulin action, and any therapy that raises GH levels carries theoretical risk of insulin resistance and diabetes.
The data showed a real but modest signal. In Phase 3 trials, 5% of tesamorelin-treated patients developed HbA1c levels of 6.5% or greater (the diabetes threshold) compared to 1% of placebo patients, yielding a hazard ratio of 3.3 for incident diabetes. However, the absolute risk increase was 4%, and most cases were in patients with pre-existing glucose impairment at baseline.
The label includes a specific warning: "EGRIFTA may cause glucose intolerance. Evaluate glucose status before initiating and monitor periodically during therapy."
In practice, this means:
- Baseline HbA1c and fasting glucose before starting treatment
- Repeat testing at 12 and 26 weeks
- Closer monitoring for patients with pre-existing insulin resistance, metabolic syndrome, or family history of type 2 diabetes
- Consider discontinuation if HbA1c exceeds 6.5% without pre-existing diabetes
The 52-week extension data showed that glucose changes stabilized after the initial 26-week period. Long-term tesamorelin users who maintained normal glucose at 26 weeks generally remained stable through 52 weeks (Falutz et al., 2010).
This glucose signal is one reason tesamorelin is not a casual addition to a body composition protocol. It requires monitoring, which brings us back to the importance of physician oversight for off-label use.
Contraindications and Drug Interactions
The FDA label lists the following contraindications:
- Active malignancy. GH and IGF-1 can promote cell proliferation. Any active cancer is an absolute contraindication. Patients with a history of cancer should wait until cleared by their oncologist. This concern applies equally to all GH-stimulating peptides, not just tesamorelin.
- Disruption of the hypothalamic-pituitary axis. Patients who have had pituitary surgery, radiation to the pituitary region, or diagnosed hypopituitarism will not respond to tesamorelin because the mechanism requires a functioning pituitary. These patients need exogenous GH, not a GH secretagogue.
- Pregnancy. Tesamorelin is classified as pregnancy category X. It should not be used during pregnancy, and pregnancy testing is recommended before initiation in women of reproductive age.
- Known hypersensitivity to tesamorelin or mannitol (used as an excipient in the formulation).
Drug interactions of clinical significance:
- Glucocorticoids (prednisone, dexamethasone): May blunt the GH response to tesamorelin
- Insulin and oral hypoglycemics: Dose adjustment may be needed due to tesamorelin's effect on glucose metabolism
- 11beta-hydroxysteroid dehydrogenase type 1 (11beta-HSD-1): GH inhibits this enzyme, potentially reducing cortisone-to-cortisol conversion. Patients on cortisone replacement may need dose adjustments.
For broader drug interaction screening across peptides, use our peptide interaction checker.
How Tesamorelin Compares to Non-Approved GH Peptides
The FDA approval question is not purely academic. It affects what you can access, how safely it is manufactured, and how much clinical data supports your decision. Here is how tesamorelin stacks up against the most common alternatives in terms of regulatory standing and evidence quality.

Tesamorelin vs Sermorelin
Sermorelin (GRF 1-29) had FDA approval from 1997 to 2008 under the brand name Geref, indicated for diagnosing GH deficiency in children. EMD Serono voluntarily withdrew it in 2008 for commercial reasons, not safety concerns.
Sermorelin is still available through compounding pharmacies but has no active FDA approval. Its clinical safety record is strong (decades of human use data, multiple controlled trials), but the evidence base is smaller than tesamorelin's for fat reduction.
The practical difference: tesamorelin can be prescribed through standard specialty pharmacies with an active FDA-approved label. Sermorelin requires compounding pharmacy sourcing, which is subject to varying state regulations and the evolving FDA peptide crackdown enforcement landscape.
For a full comparison, see tesamorelin vs sermorelin vs ipamorelin and sermorelin for fat loss.
Tesamorelin vs CJC-1295
CJC-1295 is a modified GHRH analog that has never received FDA approval for any indication. It exists in two forms: CJC-1295 with DAC (Drug Affinity Complex) and CJC-1295 without DAC (also called modified GRF 1-29 or MOD-GRF 1-29).
The DAC version extends the half-life to 6 to 8 days, producing sustained GH elevation rather than pulsatile release. The FDA reviewed CJC-1295 with DAC for its Pharmacy Compounding Advisory Committee in 2024 and it was removed from the Category 2 compounding list, making it unavailable through standard compounding channels in the US.
CJC-1295 has no Phase 3 trial data, no long-term human safety studies, and no FDA-verified manufacturing standards. The evidence for its efficacy comes from Phase 1/2 data and clinical observation.
For detailed head-to-head analysis, see tesamorelin vs CJC-1295. To estimate dosing for CJC-1295 combinations, use the CJC-1295 + ipamorelin dosage calculator.
Tesamorelin vs Ipamorelin
Ipamorelin is a growth hormone secretagogue (GHS) that works through a different receptor than tesamorelin. While tesamorelin stimulates GHRH receptors, ipamorelin activates ghrelin receptors (GHS-R) on pituitary somatotrophs. The two mechanisms are complementary, which is why some protocols combine them.
Ipamorelin has never received FDA approval. It was on the FDA's Category 2 compounding list but was removed in September 2024, similar to CJC-1295. Its availability through compounding pharmacies is uncertain and varies by state.
The evidence base for ipamorelin consists of Phase 1/2 studies primarily focused on GI motility (post-surgical ileus), not body composition. No randomized controlled trials have measured ipamorelin's effect on visceral fat with CT scan endpoints comparable to the tesamorelin Phase 3 program.
For a direct comparison, see tesamorelin vs ipamorelin.
Tesamorelin vs HGH Fragment 176-191
HGH Fragment 176-191 is a modified fragment of the growth hormone molecule (amino acids 176-191) that is purported to stimulate lipolysis without the full spectrum of GH effects. It has never received FDA approval, has no published Phase 3 data, and is available only through research suppliers and some compounding pharmacies.
The fragment does not stimulate GH release from the pituitary. It does not raise IGF-1 levels. Its mechanism is fundamentally different from tesamorelin: rather than working through the GH axis, it is thought to interact directly with fat cell receptors. The evidence for its efficacy is limited to in vitro studies and animal models, with no controlled human trials published in peer-reviewed journals.
From a regulatory perspective, HGH Fragment 176-191 occupies the weakest position among commonly discussed fat loss peptides: no FDA approval, no clinical trial data, and uncertain legal status.
The 2026 Regulatory Landscape: Why FDA Approval Matters More Than Ever
The peptide regulatory environment has changed dramatically since 2024. Understanding where tesamorelin sits within this evolving landscape explains why its FDA approval status carries increasing practical significance.
The FDA Peptide Crackdown and Its Impact
Starting in late 2024 and accelerating through 2025 and into 2026, the FDA tightened enforcement on compounded peptides. The agency removed several popular compounds from the Category 2 bulk drug substance list, which governs what 503A and 503B compounding pharmacies can legally produce.
Compounds affected include BPC-157, CJC-1295, ipamorelin, and several others commonly prescribed in anti-aging and body composition protocols. The full FDA peptide crackdown analysis covers the specific compounds, timelines, and implications.
Tesamorelin was unaffected by this crackdown because its regulatory pathway is different. As an FDA-approved biologic with an active BLA, it is manufactured by Theratechnologies under cGMP standards and distributed through legitimate specialty pharmacy channels. The compounding restrictions do not apply to FDA-approved products.
This creates a practical two-tier system:
| Category | Examples | Availability (2026) | Quality Assurance |
|---|---|---|---|
| FDA-approved GH peptides | Tesamorelin (Egrifta) | Stable, prescription through specialty pharmacies | cGMP, FDA-audited |
| Non-approved GH peptides | Ipamorelin, CJC-1295, HGH Frag | Variable, subject to enforcement | Compounding standards vary |
For patients who relied on non-approved GH secretagogues that are now harder to source, tesamorelin (via off-label prescription) represents the most pharmacologically similar alternative with a secure supply chain.
What FDA Approval Guarantees (and Does Not Guarantee)
FDA approval guarantees four things:
- 1.Efficacy for the approved indication has been demonstrated in controlled trials meeting pre-specified endpoints.
- 2.Safety has been characterized across a defined patient population with documented adverse events, contraindications, and monitoring requirements.
- 3.Manufacturing is controlled. Every batch meets identity, purity, potency, and sterility standards verified by the FDA.
- 4.Post-market surveillance continues. Adverse events after approval are tracked through the FDA Adverse Event Reporting System (FAERS), and the manufacturer is required to update the label if new safety signals emerge.
What FDA approval does not guarantee:
- Efficacy for off-label uses. The Phase 3 data applies to HIV lipodystrophy. Extrapolating to general visceral fat reduction is reasonable (the biology is the same) but not FDA-verified.
- Safety in all populations. The trials enrolled HIV-positive adults. Safety in non-HIV populations, pediatric patients, or elderly populations with comorbidities was not specifically studied in the approval program.
- Superiority over alternatives. FDA approval means the drug works better than placebo for its indication. It does not mean it is the best option for every patient.
For a comprehensive understanding of peptide legality across different regulatory categories, that resource provides the current framework.
Practical Guide: How to Access Tesamorelin in 2026
Knowing that tesamorelin is FDA approved is the starting point. Actually obtaining it requires navigating the prescription, pharmacy, and cost landscape.
Prescription Pathways
On-label (HIV lipodystrophy): An HIV specialist or infectious disease physician documents the lipodystrophy diagnosis, submits prior authorization to the patient's insurer, and writes the prescription for Egrifta WR. Specialty pharmacies like CuraScript, Accredo, or Optum handle fulfillment. Insurance typically covers this after prior authorization approval.
Off-label (general visceral fat reduction): An endocrinologist, anti-aging physician, or hormone optimization clinic evaluates the patient, orders baseline labs (IGF-1, HbA1c, fasting glucose, comprehensive metabolic panel), and writes the prescription. The physician must document the clinical rationale. The prescription goes to a specialty pharmacy or a 503B outsourcing facility that compounds tesamorelin.
Telemedicine clinics: Several national telemedicine platforms now prescribe tesamorelin off-label for visceral fat reduction. These typically require lab work (either provided by the patient or ordered through a partner lab), a video consultation with a licensed prescriber, and ongoing monitoring labs at defined intervals.
Regardless of the pathway, physician oversight is non-negotiable for tesamorelin. The glucose metabolism effects alone require baseline and follow-up monitoring that self-directed use cannot safely provide.
Brand vs Compounded: What You Need to Know
Brand-name Egrifta WR is manufactured by Theratechnologies under FDA-audited cGMP conditions. Each vial is verified for potency, purity, and sterility. The cost is higher ($1,500 to $2,500/month without insurance) but the product reliability is guaranteed.
Compounded tesamorelin is produced by 503B outsourcing facilities using bulk tesamorelin active pharmaceutical ingredient (API). Compounded versions are legal because tesamorelin has FDA approval (the compound itself is not restricted, only the brand-name product has exclusivity for the specific formulation). Compounded tesamorelin typically costs $200 to $600/month.
The trade-off is quality assurance. 503B facilities are FDA-registered and inspected, but the inspection frequency and depth is less rigorous than for branded pharmaceutical manufacturing. API sourcing varies. Some 503B facilities use U.S.-sourced API with certificates of analysis; others source from overseas with less transparent supply chains.
For patients choosing compounded tesamorelin, look for: - 503B registration (not just 503A) - U.S.-sourced API with third-party testing certificates - Sterility testing on every batch (not just periodic) - A reconstitution calculator to verify accurate dosing: use our peptide reconstitution calculator
For broader guidance on sourcing peptides safely, the is sermorelin safe article covers similar considerations for another GH-releasing peptide.
Frequently Asked Questions
Is tesamorelin FDA approved?
Yes. The FDA approved tesamorelin on November 10, 2010, under the brand name Egrifta, for the reduction of excess abdominal fat in HIV-infected adults with lipodystrophy. It was the first and remains the only GHRH analog with active FDA approval in the United States. The most recent formulation, Egrifta WR, received supplemental FDA approval in March 2025. For a full comparison of how tesamorelin's approval status differs from other GH peptides, see tesamorelin vs sermorelin vs ipamorelin.
Can I get tesamorelin without having HIV?
Yes, through off-label prescribing. Once a drug has FDA approval for any indication, a licensed physician can legally prescribe it for other conditions they deem medically appropriate. Many anti-aging clinics and hormone optimization practices prescribe tesamorelin off-label for visceral fat reduction in non-HIV patients. However, insurance will not cover off-label use, so expect to pay out of pocket. For dosing protocols used in off-label settings, see tesamorelin dosage for fat loss.
Is tesamorelin the same as Egrifta?
Egrifta is the brand name for tesamorelin. Theratechnologies manufactures three formulations: the original Egrifta (2010, two-vial reconstitution), Egrifta SV (2019, single-vial), and Egrifta WR (2025, weekly reconstitution with smaller injection volume). All contain tesamorelin as the active ingredient at the FDA-approved dose. Compounded tesamorelin available from 503B pharmacies uses the same molecule but is not manufactured by Theratechnologies. To verify your dosing with compounded formulations, use the peptide reconstitution calculator.
Is ipamorelin FDA approved like tesamorelin?
No. Ipamorelin has never received FDA approval for any indication. It works through a different mechanism (ghrelin receptor agonist) than tesamorelin (GHRH receptor agonist). Ipamorelin was removed from the FDA's Category 2 compounding list in September 2024, further restricting its availability. Tesamorelin is the only GHRH analog and one of the very few GH-stimulating peptides with active FDA approval. For a detailed comparison, see tesamorelin vs ipamorelin.
Is tesamorelin safe for long-term use?
The Phase 3 extension studies documented safety through 52 weeks of continuous use, and some patients in real-world clinical practice have used Egrifta for several years. The primary long-term concern is glucose metabolism: 5% of tesamorelin users developed HbA1c levels in the diabetic range versus 1% of placebo users. Regular monitoring of HbA1c and fasting glucose is essential. No treatment-related deaths or serious cardiovascular events were reported in clinical trials. For context on GH peptide safety, see is sermorelin safe.
Does tesamorelin require a prescription?
Yes. As an FDA-approved prescription biologic, tesamorelin requires a valid prescription from a licensed healthcare provider. There is no legal over-the-counter or direct-to-consumer pathway. Any vendor selling tesamorelin without requiring a prescription is operating outside FDA regulations. The FDA peptide crackdown of 2026 has increased enforcement against unauthorized sellers.
Will tesamorelin help with belly fat if I do not have HIV?
The mechanism of visceral fat reduction is the same regardless of HIV status. Tesamorelin stimulates pulsatile GH release, which increases lipolysis preferentially in visceral fat depots. The Phase 3 trials proved this in HIV patients, and off-label clinical experience supports similar effects in non-HIV patients, though controlled trial data in this specific population is limited. Tesamorelin does not reduce subcutaneous fat. For a broader look at how GH peptides target different fat compartments, see sermorelin for fat loss and the CJC-1295 + ipamorelin dosage calculator.
Is tesamorelin legal in the United States?
Yes. Tesamorelin is fully legal in the United States as an FDA-approved prescription drug. It can be prescribed on-label for HIV lipodystrophy and off-label for other indications by any licensed physician. It can be dispensed by licensed specialty pharmacies and compounded by FDA-registered 503B outsourcing facilities. For the current legal status of other peptides, see are peptides legal.
The Bottom Line
Tesamorelin holds a regulatory position that no other growth hormone-releasing peptide can claim: active FDA approval based on two Phase 3 randomized controlled trials totaling over 800 participants. That approval, granted in November 2010 for HIV-associated lipodystrophy and updated as recently as March 2025 with the Egrifta WR formulation, provides a level of clinical validation that sermorelin, ipamorelin, CJC-1295, and HGH Fragment 176-191 do not have.
The practical implications in 2026 are significant. As the FDA peptide crackdown continues reshaping access to compounded peptides, tesamorelin's FDA-approved status provides supply chain stability, manufacturing quality assurance, and a legal prescribing framework that survives regulatory tightening. For patients seeking visceral fat reduction through GH-axis stimulation, it is the only option with Phase 3 efficacy data, characterized safety data, and an uninterrupted path from prescription to pharmacy.
Off-label prescribing makes tesamorelin accessible beyond its HIV lipodystrophy indication, but that accessibility comes with responsibilities: physician oversight, baseline and ongoing metabolic monitoring, and informed consent about the limits of the evidence for non-approved uses. The compound works. The data is strong. The regulatory foundation is solid. Use it under medical guidance.
For dosing information, see the tesamorelin dosage for fat loss guide. For head-to-head comparisons with other GH peptides, see tesamorelin vs CJC-1295, tesamorelin vs ipamorelin, and tesamorelin vs sermorelin vs ipamorelin. To calculate reconstitution volumes and compare costs, use our peptide reconstitution calculator and peptide cost calculator.
Helpful Tools
Related Articles
Tesamorelin vs Ipamorelin: Key Differences
Tesamorelin vs ipamorelin: GHRH analog vs ghrelin mimetic. Fat loss data, dosing, side effects, cost, and stacking protocols. PubMed-cited guide.
Tesamorelin Dosage for Fat Loss
Tesamorelin dosage guide for fat loss. FDA-approved 2 mg/day protocol, visceral fat reduction data, cycling strategies, and GH peptide comparisons.
Tesamorelin vs CJC-1295: FDA GHRH vs Research
Tesamorelin vs CJC-1295: FDA GHRH (-18% visceral fat, Phase III) vs research peptide (2-10x GH, Phase I). Cost, dosing, and who picks which.
Does Sermorelin Increase Testosterone?
Does sermorelin increase testosterone? Not directly. It boosts GH, which supports testosterone via IGF-1 and improved sleep. Evidence reviewed.