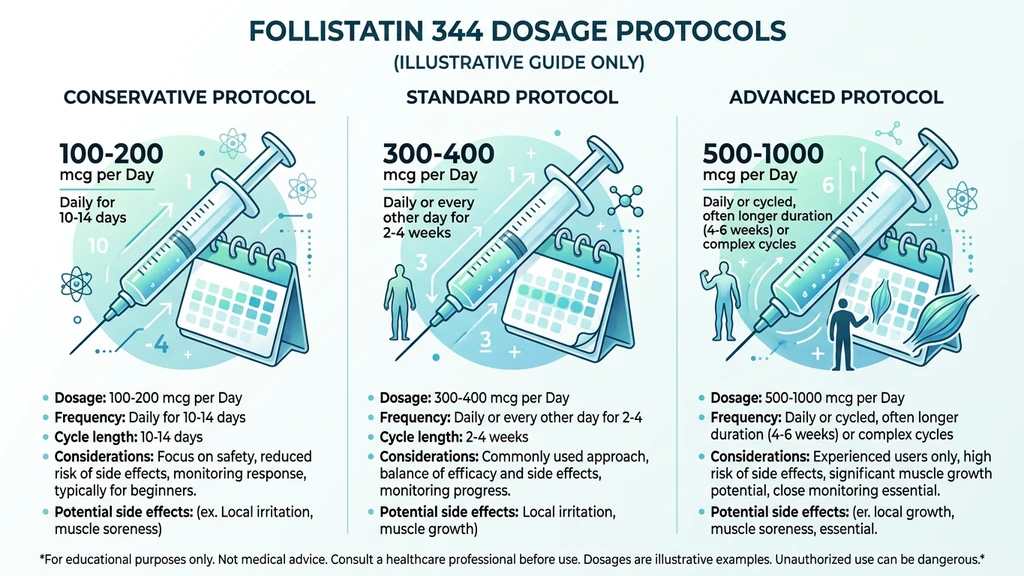
You have been training hard for months. Nutrition is dialed in. Sleep is consistent. And yet the scale and the mirror have stopped changing. You have heard that myostatin, a protein your body produces specifically to cap muscle growth, might be the reason. Follistatin 344 neutralizes it. The standard dosage is 100 to 200 mcg per day, injected subcutaneously for 10 to 30 days, followed by 3 to 4 weeks off.
| Phase | Dose | Frequency | Duration |
|---|---|---|---|
| Conservative | 100 mcg | 1x daily | 10-20 days on, 3-4 weeks off |
| Standard | 100-200 mcg | 1x daily | 20-30 days on, 3-4 weeks off |
| Advanced | 200-300 mcg | 1x daily | 10-20 days on, 4 weeks off |
Unlike most peptides covered on this site, follistatin is a full-length protein (344 amino acids, ~38 kDa), not a short synthetic chain. This distinction matters for reconstitution, storage, and dosing precision. FS-344 has a circulating half-life of approximately 90 minutes, so daily injection is required to maintain effective levels. It is also a precursor: once injected, it undergoes proteolytic cleavage to generate FS-315, the dominant circulating isoform that targets muscle tissue.
Follistatin 344 is not FDA-approved for any indication. No human clinical trials have been completed for the injectable peptide form. All dosing protocols below derive from animal research and practitioner-reported protocols. For the full peptide profile, see the hexarelin page for comparison with other growth-promoting peptides. For a complete reference of dosages across all peptides, see the peptide dosage chart.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is Follistatin 344 and How Does It Build Muscle?
Think of myostatin as a parking brake on a car. Your engine is running: you train, you eat enough protein, you recover properly. But the parking brake limits how fast you can go. No matter how hard you press the accelerator, the car creeps forward instead of surging. Follistatin releases that brake. It does not add horsepower. It removes the constraint that was holding you back.
Here is the literal biology behind that analogy. Follistatin is a naturally occurring glycoprotein produced primarily in your liver, skin, and folliculostellate cells of the pituitary gland. Its primary function is binding and neutralizing members of the TGF-beta superfamily, specifically myostatin (GDF-8), activin A, and activin B.
Myostatin is a negative regulator of muscle mass. Animals with myostatin gene knockouts develop dramatically increased musculature. The famous Belgian Blue cattle carry a natural myostatin mutation and pack roughly twice the muscle of normal cattle. In humans, a child with a myostatin loss-of-function mutation was documented with visible muscular hypertrophy at birth (Schuelke et al. 2004, PubMed 15215484).
Follistatin works by trapping myostatin in a high-affinity complex (Kd ~5-10 pM), preventing it from binding to the activin type II receptor (ActRIIB) on muscle cells. With the myostatin signal blocked, satellite cell proliferation increases and protein synthesis tilts toward net accretion.
Why "344" specifically? The number refers to amino acid count. The FST gene produces a 344-amino-acid precursor protein. After secretion, FS-344 undergoes C-terminal cleavage to produce shorter isoforms. FS-315 (the main circulating form) targets muscle. FS-288 (tissue-bound) concentrates in reproductive organs. When you inject recombinant FS-344, your body converts most of it to FS-315 in the bloodstream.
Follistatin Isoform Comparison
Understanding the three isoforms clarifies why FS-344 is the preferred injectable form.
| Isoform | Amino Acids | Location | Myostatin Binding | Notes |
|---|---|---|---|---|
| FS-344 | 344 | Precursor (injected) | Strong | Cleaved to FS-315 in vivo |
| FS-315 | 315 | Circulation | Strong | Main muscle-targeting form |
| FS-288 | 288 | Tissue-bound (gonads) | Moderate | Concentrates in reproductive tissue |
FS-344 is preferred for injection because it is more stable as a recombinant protein and converts to FS-315 systemically. Injecting FS-315 directly is possible but offers no clear advantage and is less commonly available.
A key 2009 study demonstrated that AAV-delivered follistatin 344 produced the greatest effects on muscle size and function in nonhuman primates, with increases in muscle fiber size of 15-36% in treated quadriceps (Kota et al. 2009, PubMed 19208403). The study also confirmed tolerability with no adverse effects on cardiac pathology or reproductive function in either sex.
Dosage Protocols by Goal
Follistatin 344 dosing differs from standard peptide protocols. There is no loading phase. Cycle length, not daily dose, is the main variable you adjust.
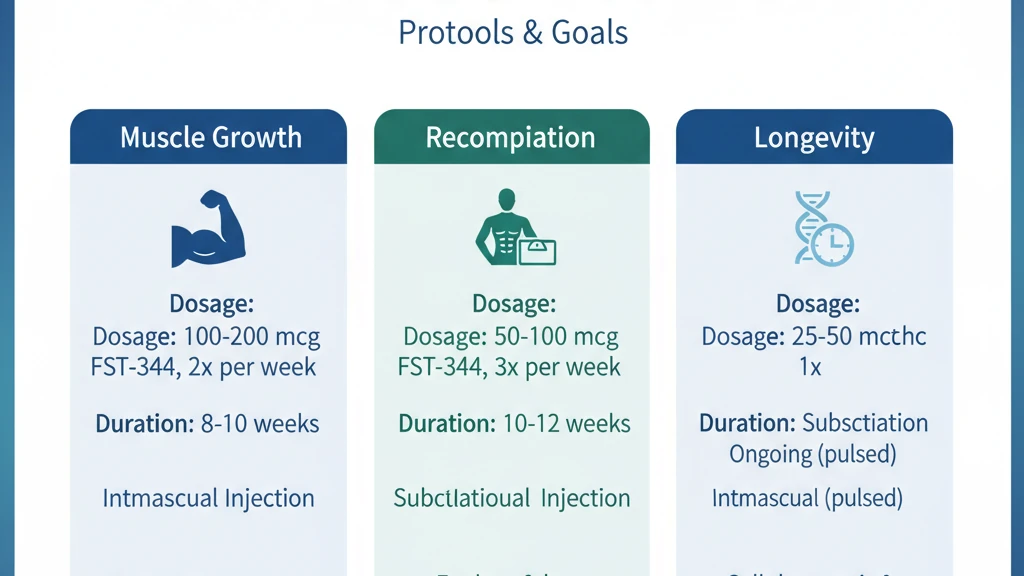
Muscle Growth Protocol
The most commonly reported protocol for hypertrophy:
| Parameter | Value |
|---|---|
| Daily dose | 100-200 mcg |
| Injection route | Subcutaneous (abdomen or thigh) |
| Frequency | 1x daily, post-workout preferred |
| Cycle length | 20-30 days |
| Time off | 3-4 weeks minimum |
| Typical vial size | 1 mg (1,000 mcg) |
| Injections per vial | 5-10 at 100-200 mcg/day |
Post-workout timing aligns the peptide's activity with the natural elevation of protein synthesis after resistance training. For proper peptide injection technique, inject subcutaneously in the abdomen or thigh. Some practitioners inject directly into target muscle groups (intramuscular), though evidence for localized effect beyond systemic distribution remains anecdotal.
At 100 mcg/day for 30 days, your cycle requires 3 mg total. At 200 mcg/day for 20 days, your cycle requires 4 mg. Cost per cycle typically ranges from $200 to $600 depending on source and purity.
Body Recomposition Protocol
For simultaneous lean mass gain and fat reduction:
| Parameter | Value |
|---|---|
| Daily dose | 100 mcg |
| Cycle length | 10-20 days |
| Time off | 3-4 weeks |
| Combined with | Caloric deficit (-300 to -500 kcal) |
| Training focus | Resistance training 4-5x/week |
The gene therapy data from Minicircle (500+ patients treated with follistatin plasmid injection) reported average lean mass increase of 2 pounds and body fat reduction of 0.87% at the 3-month mark. These modest but measurable changes occurred with a single injection rather than daily dosing. The recombinant peptide protocol may produce comparable or greater effects over a full cycle, given the repeated daily exposure.
Longevity and Anti-Aging Protocol
Follistatin gene therapy has attracted attention in the longevity community. Minicircle's data on aged participants showed a 12-year reduction in epigenetic age as measured by TruDiagnostic testing. In animal models, follistatin plasmid delivery extended mouse lifespan by 32.5%.
The anti-aging peptide protocol uses lower doses over shorter cycles:
| Parameter | Value |
|---|---|
| Daily dose | 50-100 mcg |
| Cycle length | 10-14 days |
| Cycles per year | 3-4 |
| Time off between cycles | 6-8 weeks |
These protocols remain speculative for the peptide form. The longevity data comes from gene therapy (sustained follistatin expression over months), not short peptide cycles. The translation between the two approaches is not established.
Weight-Based Dosing Reference
For precision dosing, some practitioners calculate by body weight:
| Body Weight | Conservative (1.0 mcg/kg) | Standard (1.5 mcg/kg) | Upper Range (2.5 mcg/kg) |
|---|---|---|---|
| 60 kg (132 lb) | 60 mcg | 90 mcg | 150 mcg |
| 70 kg (154 lb) | 70 mcg | 105 mcg | 175 mcg |
| 80 kg (176 lb) | 80 mcg | 120 mcg | 200 mcg |
| 90 kg (198 lb) | 90 mcg | 135 mcg | 225 mcg |
| 100 kg (220 lb) | 100 mcg | 150 mcg | 250 mcg |
Most practitioners default to flat dosing of 100 mcg for simplicity. Weight-based dosing adds precision, but the therapeutic window is wide enough that flat dosing works for most adults.
Myostatin Pathway: Follistatin vs Other Inhibitors
Follistatin 344 is not the only approach to myostatin inhibition. Several compounds target the same pathway through different mechanisms.
| Compound | Mechanism | Route | Status | Muscle Effect | Key Limitation |
|---|---|---|---|---|---|
| Follistatin 344 | Binds myostatin/activin directly | SC injection | Research only | 15-36% fiber size increase (primates) | Short half-life, requires daily dosing |
| ACE-031 | Soluble ActRIIB decoy receptor | IV infusion | Failed Phase II | Significant lean mass gains | Nosebleeds, telangiectasia in trials |
| Bimagrumab | Anti-ActRII antibody | IV infusion | Phase III (sarcopenia) | 5-7% lean mass increase | Limited to clinical settings |
| YK-11 | Upregulates follistatin expression | Oral | Research only | Indirect myostatin inhibition | No human clinical trials, steroidal |
| Decorin | Binds myostatin, TGF-beta | SC injection | Preclinical | Modest effect vs follistatin | Very limited human data |
| FS gene therapy | Sustained follistatin expression | Single injection | Experimental (Minicircle) | 2 lb lean mass at 3 months | Expensive, limited access |
ACE-031 (developed by Acceleron Pharma) showed promising muscle-building results in Phase I trials but was discontinued after Phase II due to adverse events including nosebleeds and small dilated blood vessels. The problem: ACE-031 blocks activin broadly, affecting pathways beyond muscle (Campbell et al. 2017, PubMed 27779229).
Follistatin's advantage is specificity. It binds myostatin with extremely high affinity while its isoform distribution (FS-315 to muscle, FS-288 to gonads) provides some tissue selectivity. The trade-off is a short circulating half-life requiring daily injection, versus gene therapy approaches that produce sustained expression from a single treatment.
For a broader view of peptides for muscle growth, follistatin sits at the most direct end of the myostatin inhibition spectrum. Growth hormone secretagogues like ipamorelin, GHRP-2, or MK-677 work through entirely different pathways (GH/IGF-1 axis) and can be stacked with follistatin without overlapping mechanisms.
Reconstitution and Storage
Follistatin 344 is more fragile than standard synthetic peptides. It is a full-length protein that can denature with rough handling, excessive heat, or contamination.
Reconstitution steps:
- 1.Allow the lyophilized vial to reach room temperature (5-10 minutes).
- 2.Wipe the stopper of both the follistatin vial and the bacteriostatic water vial with an alcohol swab.
- 3.Draw 0.5 mL of bacteriostatic water per 1 mg of follistatin powder. For a 1 mg vial, use 0.5 mL. This produces a concentration of 200 mcg per 0.1 mL (10 IU on an insulin syringe).
- 4.Inject the water slowly down the inside wall of the vial at a 45-degree angle. Do not squirt directly onto the powder.
- 5.Gently swirl the vial until the powder dissolves completely. Never shake.
- 6.The solution should be clear. Discard if cloudy or if particulates are visible.
Use our reconstitution calculator to determine exact volumes for your vial size and desired dose per injection.
Storage requirements:
| State | Temperature | Duration |
|---|---|---|
| Lyophilized (powder) | 2-8°C (refrigerator) | Months |
| Lyophilized (powder) | -20°C to -80°C (freezer) | Years |
| Reconstituted | 2-8°C (refrigerator) | 7-14 days |
| Reconstituted | -20°C (freezer) | Up to 30 days |
| Room temperature (any form) | 15-25°C | Days (powder), hours (reconstituted) |
Reconstituted follistatin degrades faster than most peptides. The protein's large size and complex tertiary structure make it susceptible to thermal denaturation. Use your reconstituted vial within 7 days for best potency. If you cannot use the full vial in 7 days, aliquot into multiple sterile vials and freeze the extras at -20°C. For full guidelines on reconstituted shelf life, see how long do reconstituted peptides last. Proper peptide storage is critical for follistatin given its sensitivity.
For detailed reconstitution technique, see our guide on how to reconstitute peptides. Injection technique is covered in the peptide injections guide.
Side Effects and Safety Considerations
Follistatin 344 has a limited human safety profile. The gene therapy data (Minicircle, 500+ patients) reported zero serious adverse events at 3 months. The most common effect was a slight LDL cholesterol increase of 8 mg/dL in approximately one-third of patients.
Reported side effects from practitioner protocols:
| Side Effect | Frequency | Severity | Management |
|---|---|---|---|
| Injection site redness/swelling | Common | Mild | Rotate injection sites |
| LDL cholesterol elevation | ~33% | Mild (8 mg/dL average) | Monitor lipids, consider diet adjustment |
| Mild fatigue during cycle | Occasional | Mild | Transient, resolves off-cycle |
| Joint discomfort | Rare | Mild | May indicate rapid muscle growth outpacing connective tissue |
The danger you cannot see in the vial. Sourcing peptides safely is critical. For guidance on evaluating suppliers, see where to buy peptides in 2026. You buy follistatin online. Nearly half of tested products (9 out of 17 in a WADA-funded analysis) contained degraded or mislabeled material (PubMed 31758732). You inject what might be nothing, or worse, an unknown protein. Sourcing from reputable suppliers with third-party certificates of analysis is not optional; it is the only way to know what is actually in your syringe.
The danger you will notice too late. Eleven bodybuilders using high-dose follistatin 344 developed central serous chorioretinopathy (CSCR), a condition where fluid builds up under the retina, distorting and blurring your vision. Average recovery: 2.3 months of impaired sight. Recurrence was observed in patients who resumed follistatin. The authors concluded that follistatin-344 injection should be considered a risk factor for CSCR (Dag et al. 2020, PubMed 32671599). If you experience any visual changes, stop immediately and see an ophthalmologist.
Fertility impact: Follistatin inhibits pituitary FSH (follicle-stimulating hormone) release. In women, FSH drives ovarian follicle development. In men, FSH supports spermatogenesis. Artificially elevated follistatin may suppress FSH and impair your fertility during the cycle. This effect should reverse off-cycle, but if you and your partner are actively trying to conceive, avoid follistatin entirely. The Kota 2009 primate study found no permanent reproductive effects, but short-term suppression was not measured (PubMed 19208403).
Activin suppression beyond muscle: Activin regulates wound healing, immune function, and erythropoiesis (red blood cell production). Broadly suppressing activin signaling could have effects beyond muscle. Follistatin's isoform-specific distribution provides some protection, but high-dose exogenous follistatin may override this selectivity.
WADA status: Follistatin is listed on the World Anti-Doping Agency (WADA) Prohibited List under S4.3 (Metabolic Modulators). If you are subject to anti-doping testing, you must not use follistatin in any form. Testing methods can detect both the recombinant protein and evidence of gene therapy delivery.
Follistatin 344 should not be used by individuals with active cancer, as myostatin inhibition may theoretically promote tumor growth in certain contexts. If you have cardiovascular disease, liver disease, or hormonal conditions, consult a physician before use. For a broader overview of peptide safety considerations, see our peptide safety guide.
Cycling and Long-Term Use
Unlike GHRPs that require cycling due to receptor desensitization (see our hexarelin dosage guide for a detailed example), follistatin cycling is primarily about cost management and safety margin rather than receptor dynamics.
Myostatin does not develop resistance to follistatin binding. The reason for cycling is threefold:
- 1.Cost: At $200-600 per cycle, continuous use is expensive. Cycling allows your body to consolidate gains during off periods.
- 2.FSH recovery: Off-cycle periods allow your pituitary FSH to normalize, protecting reproductive function.
- 3.Unknown long-term effects: With no long-term human data, periodic breaks are the conservative approach.
Recommended cycling schedule:
| Pattern | On Period | Off Period | Best For |
|---|---|---|---|
| Short cycles | 10 days | 3 weeks | First-time users, cost-conscious |
| Standard cycles | 20-30 days | 4 weeks | Muscle growth, experienced users |
| Quarterly cycles | 14 days | 10-12 weeks | Longevity protocols |
During off-cycle periods, muscle gains from follistatin tend to be well-retained. The underlying mechanism (satellite cell activation, myonuclear addition) creates structural changes in your muscle fibers. This contrasts with gains from growth hormone secretagogues, which may partially reverse when the GH stimulus is removed.
Stacking follistatin with growth-promoting peptides during the same period is practiced but increases both cost and the complexity of monitoring. Common stacking partners include IGF-1 LR3 for synergistic muscle growth or CJC-1295 + ipamorelin for GH axis support. For general peptide stacking principles and compatibility checking, see our peptide stack calculator.
Follistatin 344 vs Follistatin 315: Which to Choose?
This question comes up frequently, and the answer is straightforward. For injectable use, FS-344 and FS-315 are functionally equivalent because FS-344 converts to FS-315 in your bloodstream.
| Factor | FS-344 | FS-315 |
|---|---|---|
| Amino acids | 344 | 315 |
| Stability as recombinant | Higher | Lower |
| Availability | Common | Rare |
| In vivo conversion | Converts to FS-315 | Already active form |
| Myostatin binding affinity | High (direct + via FS-315) | High |
| Tissue distribution | Systemic (via FS-315) | Primarily muscle/circulation |
| Research base | Extensive (gene therapy studies) | Limited as standalone |
| Recommended for injection | Yes (standard choice) | Acceptable if available |
The practical reason FS-344 dominates the market: it is easier to produce as a stable recombinant protein and has the larger evidence base from gene therapy research. FS-315 is the bioactive downstream product, but injecting the precursor (FS-344) and letting your body do the conversion is the established approach.
If a vendor offers "follistatin 315" at a significantly lower price, verify purity and source carefully. The less common product carries higher risk of quality issues simply due to smaller production volumes and less standardized manufacturing.
Clinical Evidence Summary
Follistatin research spans gene therapy, animal models, and limited human observational data. No randomized controlled trial has tested injectable FS-344 in humans.
| Study | Model | Intervention | Key Finding | Reference |
|---|---|---|---|---|
| Kota et al. 2009 | Macaques | AAV1-FS344 gene delivery | 15-36% increase in muscle fiber size, no adverse effects on heart or reproduction | PubMed 19208403 |
| Lee & McPherron 2001 | Mice | Follistatin overexpression | Dramatic increase in muscle mass, 117-327% vs controls | PubMed 11440715 |
| Haidet et al. 2008 | Mice (mdx) | AAV-FS344 | Improved muscle mass and strength in muscular dystrophy model | PMC2393740 |
| Rodino-Klapac et al. 2009 | Review | Multiple | Follistatin as promising therapy for muscle disease | PMC2717722 |
| Schuelke et al. 2004 | Human (case) | Natural myostatin mutation | Child born with myostatin loss-of-function showed extreme musculature | PubMed 15215484 |
| Mendell et al. 2015 | Humans (6 BMD patients) | AAV1-FS344 gene therapy | Up to 125m improvement in 6-minute walk test, no serious adverse events | PMC4426808 |
| Dag et al. 2020 | Humans (11 bodybuilders) | FS-344 injection | CSCR eye condition, 2.3-month avg recovery | PubMed 32671599 |
| Minicircle (ongoing) | Humans (500+) | FS gene therapy | +2 lb lean mass, -0.87% body fat, -12 yr epigenetic age at 3 months | minicircle.io |
The strongest evidence comes from the primate gene therapy study (Kota 2009), which demonstrated durable muscle increases with a single treatment and no safety concerns over the monitoring period. Translating this to injectable peptide protocols requires assumptions about bioavailability and dosing equivalence that have not been validated in controlled settings.
For comparison, IGF-1 LR3 has a somewhat stronger human evidence base for injection protocols, though it works through a different mechanism (direct anabolic signaling rather than myostatin inhibition).
Frequently Asked Questions
What is the best follistatin 344 dosage for muscle growth?
The standard dosage is 100 to 200 mcg per day injected subcutaneously for 20 to 30 days, followed by 3 to 4 weeks off. Most practitioners start at 100 mcg/day for the first cycle. Exceeding 200 mcg/day has not been shown to produce additional benefit in any available data.
How long does a follistatin 344 cycle last?
A standard cycle runs 10 to 30 days, with 20 days being the most common for hypertrophy. You should take at least 3 to 4 weeks off between cycles to allow FSH recovery. First-time users typically start with a shorter 10-day cycle to assess tolerance before committing to 20+ days.
What is the difference between follistatin 344 and follistatin 315?
FS-344 is a 344-amino-acid precursor that converts to FS-315 in your bloodstream. FS-315 is the active circulating form that targets muscle. For injection purposes, they are functionally equivalent. FS-344 is preferred because it is more stable as a recombinant protein, more widely available, and backed by a larger research base.
Can follistatin 344 affect fertility?
Yes, temporarily. Follistatin suppresses pituitary FSH release, which drives ovarian follicle development in women and spermatogenesis in men. The Kota 2009 primate study found no permanent reproductive effects. These changes should reverse off-cycle, but couples actively trying to conceive should avoid follistatin entirely.
How do you reconstitute follistatin 344?
Add 0.5 mL of bacteriostatic water per 1 mg of powder. Inject the water slowly at a 45-degree angle down the vial wall. Swirl gently (never shake) until dissolved. The solution must be clear; discard if cloudy. Store reconstituted follistatin at 2 to 8°C and use within 7 to 14 days, or freeze aliquots at -20°C for up to 30 days.
Is follistatin 344 better than YK-11 for muscle growth?
They target myostatin through different mechanisms. Follistatin 344 directly binds and neutralizes myostatin with a binding affinity of ~5 to 10 pM. YK-11 is a steroidal SARM that upregulates your body's own follistatin production indirectly. Follistatin provides more predictable inhibition but requires injection. YK-11 is oral but has zero human clinical trials and carries steroidal side effects.
Can you inject follistatin 344 intramuscularly for localized growth?
Some practitioners inject directly into target muscles, claiming localized effects. However, FS-344 converts to FS-315 in your bloodstream and distributes systemically regardless of injection site. Any localized effect is temporary, limited to the initial high concentration before systemic distribution occurs. Subcutaneous injection remains the standard 1x daily approach.
What are the side effects of follistatin 344?
The most common are injection site reactions (redness, swelling) and mild LDL cholesterol elevation of about 8 mg/dL in roughly one-third of users. A case series documented CSCR (an eye condition causing blurred vision) in 11 bodybuilders using high-dose follistatin, with an average recovery of 2.3 months. Stop immediately if you notice visual changes.
How much does a follistatin 344 cycle cost?
A single cycle costs approximately $200 to $600 depending on dosage and source. At 100 mcg/day for 30 days, you need 3 mg total (three 1 mg vials). At 200 mcg/day for 20 days, you need 4 mg. Prices per 1 mg vial range from $60 to $150. Follistatin is among the most expensive peptides due to its complex manufacturing as a full-length recombinant protein.
The Bottom Line
Follistatin 344 occupies a unique position in the peptide landscape. It is the most direct approach to myostatin inhibition available outside of gene therapy. The standard 100 to 200 mcg daily protocol over 10 to 30 day cycles is well-established in practitioner use, and the primate gene therapy data provides the strongest preclinical support for both efficacy and safety.
The key differentiator from other muscle-building peptides: follistatin removes a growth inhibitor rather than adding a growth signal. This mechanism is complementary to GH secretagogues, IGF-1, and anabolic compounds, making follistatin a logical addition to advanced protocols. The trade-offs are real: high cost per cycle, fragile reconstitution requirements, limited human data, and potential fertility effects during use.
Start conservatively at 100 mcg/day for a 10-day cycle. Assess your response and tolerance before extending to longer cycles or higher doses. Monitor your lipids and be alert to any visual changes. Cycle off for at least 3 to 4 weeks between runs.
For reconstitution help, use our reconstitution calculator. To check compatibility with other peptides, try the interaction checker. Explore our complete peptide profiles for information on other compounds that complement follistatin in a growth-focused protocol. New to peptides? Start with our getting started with peptides guide.
Related Articles: - Peptides for Muscle Growth - IGF-1 LR3 Dosage Protocol - Hexarelin Dosage Guide - Peptide Stacking Guide - How to Store Peptides - Tesamorelin Dosage for Fat Loss — complementary GHRH protocol for body recomposition - Sermorelin for Fat Loss — GH-pathway fat loss to pair with muscle-building protocols
Helpful Tools
Related Articles
How Much Sermorelin Per Day: Complete Dosage Guide
Sermorelin dosage: 200-500 mcg/day subcutaneous before bedtime. Starting dose, titration, gender-specific protocols, and IGF-1 monitoring.
CJC-1295 Dosage: Complete Protocol Guide
CJC-1295 dosage guide: DAC and no-DAC protocols. Dosing by goal (anti-aging, fat loss, muscle, sleep), ipamorelin stacking, and injection timing.
Hexarelin Dosage Guide: Protocols
Hexarelin dosage by goal: 100-200 mcg protocols, dose-response data, desensitization timeline, GHRP comparison table, and cycling guide.
5-Amino-1MQ Dosage: Protocols, Cycling, and Safety
5-Amino-1MQ dosage guide: 50-150 mg/day oral protocols, NNMT inhibition mechanism, cycle length, stacking options, and PubMed-cited research.