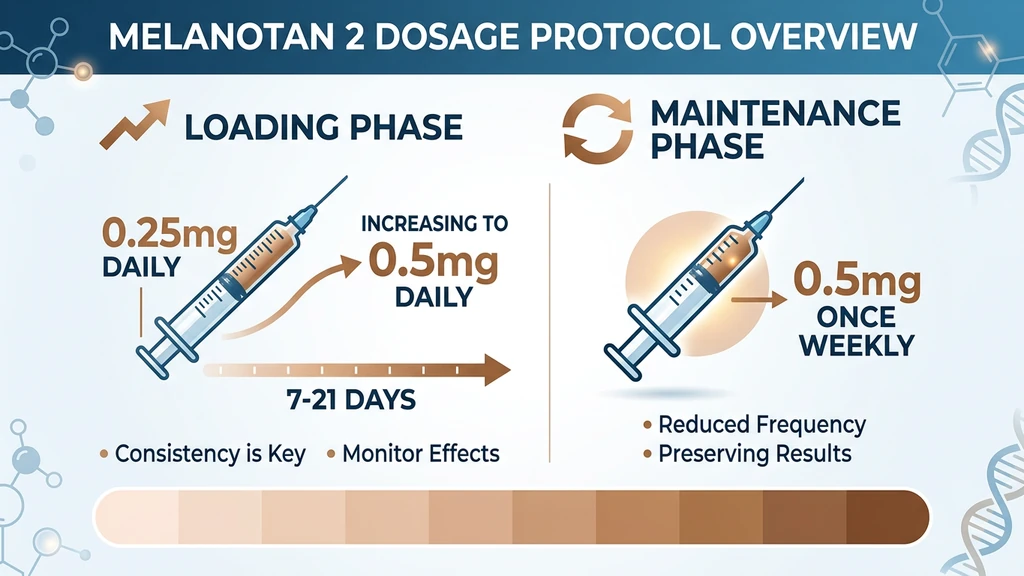
You hold a 10 mg vial of Melanotan II and an insulin syringe. The Reddit guides say "0.25 mg loading dose." The vial says nothing. The standard Melanotan 2 dosage is 0.25 mg daily for a 7 to 14 day loading phase, increasing to 0.5 mg if tolerated, followed by 0.5 mg once or twice weekly for maintenance. Total cumulative dose varies sharply by skin type: Fitzpatrick I-II needs 20 to 50 mg, while III-V often reaches a tan on under 10 mg.
| Phase | Dose | Frequency | Duration |
|---|---|---|---|
| Loading (start) | 0.25 mg | Daily | Days 1 to 3 |
| Loading (full) | 0.5 mg | Daily | Days 4 to 21 (varies by skin type) |
| Maintenance | 0.5 mg | 1 to 2x per week | Ongoing |
Melanotan II (MT-II) was developed at the University of Arizona in the 1980s as a synthetic analog of alpha-melanocyte-stimulating hormone (alpha-MSH). A Phase I clinical trial established the dosing range of 0.01 to 0.03 mg/kg body weight, with significant tanning after five doses (Dorr et al. 1996, PMID: 8637402).
MT-II is not approved by the FDA, TGA, MHRA, or any regulatory body. The melanoma risk is biologically plausible (PMID: 24355990). Use the reconstitution calculator for exact mixing math, and read what do peptides do for skin for the broader skin-peptide landscape.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
How Melanotan 2 Works
MT-II binds to melanocortin receptors on the surface of your cells. Five types of melanocortin receptors exist (MC1R through MC5R), and MT-II activates several of them. Each receptor triggers a different biological response.
MC1R controls skin pigmentation. When MT-II binds to MC1R on melanocytes (the cells that produce pigment), it triggers the cAMP-PKA signaling pathway. This activates the enzyme tyrosinase, which converts tyrosine into melanin. More melanin means darker skin.
MC4R sits in the hypothalamus. Activation here produces two effects: increased sexual arousal (via the oxytocin pathway) and reduced appetite. These are not side effects. They are direct pharmacological actions of the compound.
MC3R and MC5R contribute to energy regulation and sebaceous gland activity. MC5R activation explains why some users notice changes in skin oiliness.
MT-II has a half-life of approximately 33 minutes. This short half-life is why daily dosing is necessary during the loading phase: each injection triggers a burst of melanin production, and cumulative daily doses build a sustained tanning response.
For the complete mechanism and stacking information, see the Melanotan II peptide profile.
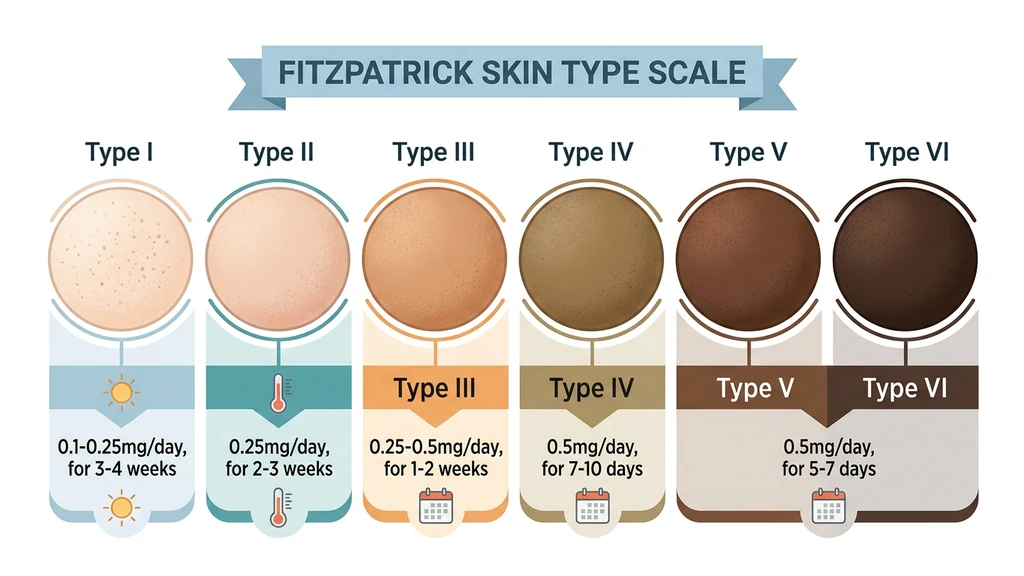
Dosage by Fitzpatrick Skin Type
This is the most important section of this guide. Your Fitzpatrick skin type determines how much Melanotan 2 you need, how long loading takes, and what results to expect. The difference is substantial: a person with very fair skin (Type I) may need 10 times the total dose of someone with olive skin (Type IV).
Why skin type matters at the molecular level: MC1R receptor variants differ between skin types. People with very fair skin often carry MC1R gene variants (like R151C or R160W) that produce a receptor less responsive to melanocortin signaling. Their melanocytes need stronger, more sustained stimulation to switch from producing pheomelanin (red/yellow pigment) to eumelanin (brown/black pigment) (PubMed 17223853).
| Fitzpatrick Type | Description | Starting Dose | Loading Duration | Total Load | Maintenance | Results Timeline |
|---|---|---|---|---|---|---|
| Type I | Very fair, always burns, red/blonde hair | 0.1-0.25 mg/day | 3-4 weeks | 30-50 mg | 0.5 mg 2x/week | 3-6 weeks for visible tan |
| Type II | Fair, usually burns, light hair | 0.25 mg/day | 2-3 weeks | 20-30 mg | 0.5 mg 1-2x/week | 2-4 weeks for visible tan |
| Type III | Medium, sometimes burns | 0.25-0.5 mg/day | 1-2 weeks | 10-20 mg | 0.5 mg 1x/week | 1-2 weeks for visible tan |
| Type IV | Olive, rarely burns | 0.5 mg/day | 7-10 days | 5-10 mg | 0.5 mg every 10-14 days | 5-10 days for visible tan |
| Type V-VI | Dark to very dark | 0.5 mg/day | 5-7 days | Under 5 mg | 0.5 mg every 2-3 weeks | 3-7 days for visible deepening |
How to identify your Fitzpatrick type: Consider how your skin reacts to 30 minutes of midday summer sun without sunscreen. Type I always burns and never tans. Type II usually burns and tans minimally. Type III sometimes burns and tans gradually. Type IV rarely burns and tans easily. Type V very rarely burns and is naturally brown. Type VI never burns and is deeply pigmented.
Special Considerations for Very Fair Skin (Type I)
Type I individuals face the most challenging dosing because their MC1R receptors are the least responsive. Three adjustments make the protocol more effective and tolerable.
Start lower. Begin at 0.1 mg rather than 0.25 mg. Type I users report more intense nausea and flushing at standard starting doses. Building tolerance over the first week matters more here than accelerating results.
Expect longer timelines. Visible tanning may take 3-6 weeks rather than 1-2 weeks. The process is building eumelanin in cells that preferentially produce pheomelanin. This is a biochemical shift, not just an increase in existing pigment.
UV exposure helps. Brief sun exposure (10-15 minutes) during the loading phase accelerates results for Type I skin specifically because UV radiation independently activates melanocyte proliferation. This supplements the MC1R stimulation from MT-II. Do not sunburn. Short, controlled exposures are the goal.
Loading Phase Protocol: Step by Step
The loading phase builds your melanin reserves. Skipping or shortening it produces weaker, less durable results. Follow this day-by-day protocol and adjust based on your skin type from the table above.
Days 1-3: Starting Low Inject 0.25 mg subcutaneously in the evening (or 0.1 mg for Type I skin). Evening injection is recommended because nausea and flushing peak 30-90 minutes after injection, and sleeping through the side effects is the simplest management strategy.
Expect nausea on the first 1-3 injections. This is normal and affects 80-90% of users. It diminishes significantly by day 4-5. See the side effects section below for the full nausea management protocol.
Days 4-7: Increasing the Dose If 0.25 mg is tolerated, increase to 0.5 mg daily. Freckles and moles begin darkening. This is the mechanism working: MT-II activates all melanocytes, not just those in sun-exposed skin. Brief UV exposure (10-15 minutes of natural sunlight or a short tanning bed session) during this phase accelerates visible results.
Weeks 2-3: Building the Tan Continue 0.5 mg daily. The tan becomes visible during this window for most skin types. Monitor all moles and freckles. Photograph them weekly for comparison. Any irregular changes warrant stopping MT-II and seeing a dermatologist.
Transition to Maintenance When you have reached your desired base color, switch to the maintenance protocol. For Type III-V skin, this transition typically happens at days 7-14. For Type I-II skin, it may take 3-4 weeks.
Maintenance Phase Protocol
Once your desired tan is established, reduce dosing frequency to maintain the color without continued daily injections.
Standard maintenance: - Type I-II: 0.5 mg twice per week - Type III-IV: 0.5 mg once per week - Type V-VI: 0.5 mg every 2-3 weeks
Alternative approach: Some users prefer a single larger weekly dose (1.0 mg once per week instead of 0.5 mg twice per week). Both approaches maintain the tan effectively. The single larger dose may cause more pronounced side effects on the injection day.
How long does the tan last without maintenance? After stopping MT-II, the tan persists 4-8 weeks and fades gradually as melanocytes turn over naturally. If you restart after a break, the loading phase is shorter the second time because residual melanin remains in the skin.
Seasonal use: Many users only run MT-II during spring and summer. A common approach is to load in March-April, maintain through summer, and stop in September. The tan fades by late October.
Cycling recommendation: Run MT-II for 8-12 weeks, then take 4 weeks off. This break serves two purposes: it prevents melanocortin receptor desensitization (diminishing returns), and it creates a window for thorough mole monitoring without the confounding variable of active melanocyte stimulation.
Administration Methods Compared
MT-II can be administered by subcutaneous injection or nasal spray. Oral administration does not work: digestive enzymes destroy the peptide before absorption, leaving bioavailability below 5%.
| Method | Bioavailability | Onset to Visible Results | Practical Advantage |
|---|---|---|---|
| Subcutaneous injection | ~100% | 7-21 days (skin type dependent) | Most effective, consistent dosing |
| Nasal spray | 30-40% | 17-30+ days | No needles, but requires 2-3x the dose |
| Oral | Below 5% | Not viable | Destroyed by digestive enzymes |
Subcutaneous Injection (Recommended)
Subcutaneous injection delivers MT-II directly into the fat layer beneath the skin, bypassing the digestive system entirely. Bioavailability is approximately 100%.
Injection sites: Abdomen (2 inches from the navel), outer thigh, or back of the upper arm. The abdomen is preferred because it has consistent subcutaneous fat depth and is easy to access.
Technique: Pinch a fold of skin, insert the needle at a 45-degree angle, inject slowly over 3-5 seconds, withdraw, and apply light pressure. Use 29-31 gauge insulin syringes (0.5 mL or 1 mL).
Site rotation: Alternate injection sites daily to prevent lipodystrophy (localized fat tissue changes). A simple pattern: alternate between left and right sides of the abdomen, then rotate to thighs every few days.
For the complete injection walkthrough, see our peptide injection guide. If you are new to peptide self-administration, start with our getting started with peptides guide.
Nasal Spray: Why It Is Less Effective
Nasal MT-II has become popular on social media because it avoids needles. The trade-off is significant: 60-70% of the peptide is lost during nasal absorption, meaning you need 2-3 times the dose to achieve equivalent melanocortin receptor activation.
Results take 10-21 days longer to appear with nasal spray compared to injection. Absorption varies between uses depending on nasal congestion, humidity, and spray technique. This inconsistency makes precise dosing difficult.
The Cleveland Clinic has issued specific warnings about nasal tanning sprays, citing unpredictable absorption and the inability to control dosing accurately.
If you choose nasal spray despite these limitations, use approximately 1.0-1.5 mg per application (equivalent to 0.5 mg injected). Spray into each nostril while inhaling gently. Do not blow your nose for 15 minutes afterward.
Reconstitution and Mixing Guide
MT-II comes as a lyophilized (freeze-dried) powder in vials, typically containing 10 mg. You must reconstitute it with bacteriostatic water before use.
Standard reconstitution (recommended): - 10 mg vial + 2 mL bacteriostatic water = 5 mg/mL concentration - At this concentration: 0.1 mL (10 units on an insulin syringe) = 0.5 mg MT-II - 0.05 mL (5 units) = 0.25 mg MT-II
Alternative (simpler math): - 10 mg vial + 1 mL bacteriostatic water = 10 mg/mL concentration - At this concentration: 0.05 mL (5 units) = 0.5 mg MT-II - More concentrated, but each unit mark on the syringe represents a larger dose, making small dose adjustments harder
Step-by-step mixing: 1. Clean the rubber stoppers on both the MT-II vial and the bacteriostatic water vial with alcohol swabs 2. Draw 2 mL of bacteriostatic water into a syringe 3. Insert the needle into the MT-II vial and inject the water slowly down the inside wall of the vial (never squirt directly onto the powder) 4. Remove the needle and gently roll the vial between your palms until the powder dissolves completely. Do not shake. 5. Label the vial with the date and concentration
Storage after reconstitution: - Refrigerate at 2-8 degrees C (standard refrigerator) - Use within 30-40 days - Unreconstituted powder stored frozen at -20 degrees C remains stable for 3-5 years
Use our reconstitution calculator for exact measurements based on your specific vial size and water volume. For general peptide storage advice, see how to store peptides. For the full reconstitution process, see how to reconstitute peptides. Need to know how long reconstituted peptides last? MT-II is among the most stable at 30-40 days refrigerated.
Side Effects and How to Manage Them
MT-II produces more noticeable side effects than most peptides because it activates multiple melanocortin receptors simultaneously. The good news: most side effects diminish or disappear within the first week as tolerance develops.
Nausea (80-90% of Users)
Nausea is the most common side effect and the primary reason people abandon MT-II early. It peaks 30-90 minutes after injection and resolves within 2-4 hours. Severity is worst during the first 3-5 injections and diminishes significantly by the end of week one.
Nausea management protocol:
- 1.Start at 0.1-0.25 mg, not 0.5 mg. Dose-dependent nausea is dramatically worse at higher starting doses.
- 2.Inject in the evening, 30-60 minutes before bed. Sleep through the worst of it.
- 3.Take an antihistamine 45 minutes before injection. Fexofenadine 180 mg (Allegra) or loratadine 10 mg (Claritin) reduces nausea by blocking histamine release triggered by MC1R activation. Use non-drowsy formulations if injecting during the day.
- 4.Alternative: ginger root 500 mg, taken 30 minutes before injection.
- 5.Avoid eating 1-2 hours before injection. An empty stomach reduces nausea intensity for most users.
- 6.Increase dose gradually. Add 0.1 mg every 2-3 days rather than jumping from 0.25 mg to 0.5 mg in one step.
Most users find nausea manageable after day 5. If nausea persists beyond two weeks at the same dose, reduce by 0.1 mg and hold there.
Facial Flushing and Warmth
A warm flush across the face, chest, and sometimes arms occurs 10-30 minutes after injection. It is caused by vasodilation triggered by melanocortin receptor activation. The flushing is typically mild, resolves within 1 hour, and is more pronounced at higher doses.
The same antihistamine pre-treatment that reduces nausea also reduces flushing. Evening injection avoids the social inconvenience of visible facial redness.
Spontaneous Erections (Men)
MC4R activation in the hypothalamus triggers oxytocin release, which can produce erections 1-5 hours after injection. This effect is dose-dependent and more common during the loading phase when daily doses are at their highest.
This is the same mechanism behind PT-141 (bremelanotide), which is an FDA-approved derivative of MT-II specifically designed for sexual dysfunction. For some users, this is a welcome effect. For others, evening injection timing avoids practical issues. See peptides for libido for more on this pathway.
Priapism warning: An erection lasting longer than 4 hours is a medical emergency. This is rare with MT-II at standard doses but possible. Seek immediate medical attention if it occurs.
Mole and Freckle Darkening
This is not a side effect in the traditional sense. It is the mechanism working. MT-II activates all melanocytes in the body, including those in existing moles and freckles. Expect them to become darker and more prominent. New moles (nevi) can also appear.
Why this matters for safety: Melanoma arises from melanocytes. Stimulating melanocyte activity while simultaneously making it harder to visually distinguish normal mole changes from dangerous ones creates a genuine monitoring challenge. See the safety section below for the mandatory mole monitoring protocol.
Other Side Effects
- Appetite suppression: Mediated by MC4R in the hypothalamus. Many users consider this a benefit rather than a side effect.
- Fatigue and drowsiness: Common in the first 2-3 hours post-injection. Evening dosing avoids daytime impairment.
- Mild water retention: Some users report slight puffiness, particularly facial. Resolves within a few days of dose stabilization.
- Increased skin oiliness: MC5R activation affects sebaceous glands. Usually mild.
- Rare serious events: Published case reports include rhabdomyolysis (PubMed 23121206) and renal infarction. These are rare but underscore that MT-II carries real medical risks.
Safety Warnings and Melanoma Risk
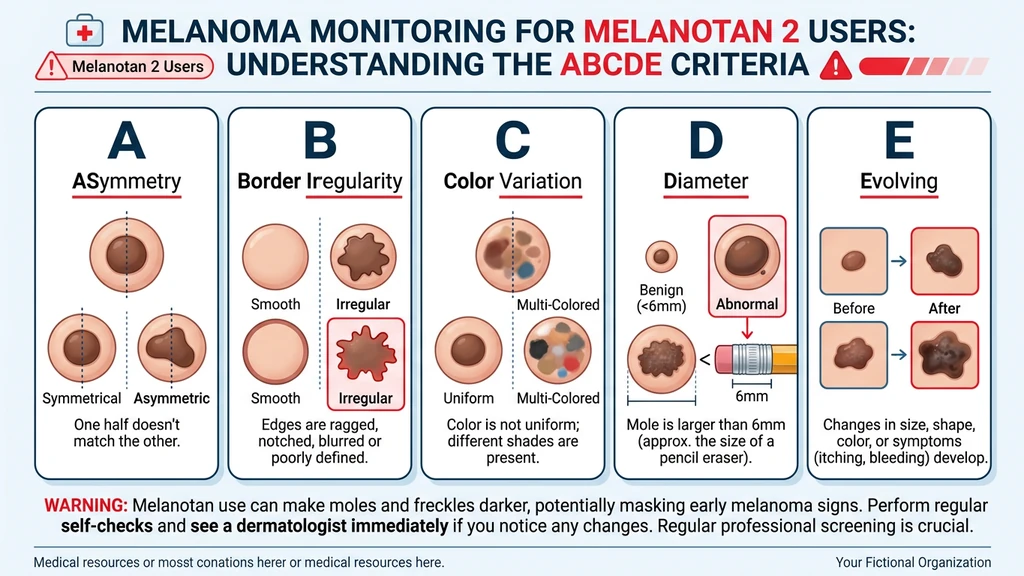
This section contains information that could affect your health. Read it completely.
MT-II is not approved by any regulatory body. The FDA (US), TGA (Australia), MHRA (UK), and EMA (EU) have all issued warnings about melanotan products. The Australian TGA specifically warns consumers not to use tanning products containing melanotan.
Melanoma case reports. At least four published case reports describe melanoma emerging during or after MT-II use (PubMed 24355990). A comprehensive safety review confirmed the association between MT-II and atypical nevi changes (PubMed 25929579). No direct causal link is established, but the biological plausibility is clear: MT-II stimulates the same cells from which melanoma originates.
Who should NOT use Melanotan 2: - Anyone with a personal or family history of melanoma - People with many atypical or dysplastic nevi (irregular moles) - Skin type I with extensive sun damage history - Pregnant or breastfeeding women - People with cardiovascular disease (MC4R activation causes transient blood pressure elevation)
Mandatory mole monitoring protocol:
1. Before starting: Get a full-body mole check from a dermatologist. Establish a photographic baseline. 2. Monthly during use: Self-examine all moles using the ABCDE criteria: - Asymmetry: One half does not match the other - Border: Edges are irregular, ragged, or blurred - Color: Uneven color with shades of brown, black, red, white, or blue - Diameter: Larger than 6 mm (the size of a pencil eraser) - Evolving: Any change in size, shape, color, elevation, or any new symptom (bleeding, itching, crusting) 3. At 3 months and 6 months: Follow-up dermatology check 4. Immediate stop: If any mole shows ABCDE changes, discontinue MT-II and see a dermatologist immediately
Melanotan 1 vs Melanotan 2: Which to Choose
Both compounds stimulate melanin production, but their pharmacological profiles differ significantly.
| Feature | Melanotan I (Afamelanotide) | Melanotan II |
|---|---|---|
| Receptor selectivity | MC1R selective | MC1R, MC3R, MC4R, MC5R (non-selective) |
| Tanning effect | Slower, more gradual | Faster, more pronounced |
| Libido effect | Minimal | Significant |
| Appetite suppression | Minimal | Moderate to strong |
| Nausea incidence | Lower | Very common (80-90%) |
| New mole risk | Lower | Higher (activates more melanocyte pathways) |
| Regulatory status | Approved in EU as Scenesse for EPP | Not approved anywhere |
| Half-life | ~30 minutes | ~33 minutes |
| Cost | Higher (pharmaceutical product) | Lower (research chemical) |
Melanotan I (afamelanotide) is sold under the brand name Scenesse and is approved in the European Union for erythropoietic protoporphyria (EPP), a condition where sunlight exposure causes severe pain. Because MT-I only targets MC1R, it produces tanning without the sexual side effects, appetite changes, or nausea associated with MT-II.
Melanotan II is the more popular choice for tanning because it works faster and costs less. The trade-off is a broader side effect profile caused by non-selective receptor activation.
PT-141 (bremelanotide) is a derivative of MT-II that was specifically modified to target MC4R for sexual dysfunction. It is FDA-approved under the brand name Vyleesi for hypoactive sexual desire disorder in women. If your primary interest is the libido-enhancing effects rather than tanning, PT-141 is the targeted, approved option. See the PT-141 profile for details, or our comprehensive peptides for libido guide for a full comparison.
Results Timeline: What to Expect Week by Week
Individual results vary by skin type, dose, and UV exposure during loading. Below is a general timeline based on clinical data and community reporting. The table assumes subcutaneous injection at standard dosing (0.25-0.5 mg/day during loading).
| Timeline | What Happens | Notes |
|---|---|---|
| Days 1-3 | Nausea, flushing, possible erections. No visible tanning. Freckles may darken slightly. | Side effects at their peak. Antihistamine protocol recommended. |
| Days 4-7 | First visible changes. Freckles and moles become noticeably darker. Slight warming of overall skin tone. Libido increase common. | Brief UV exposure (10-15 min) accelerates tanning. |
| Week 2 | Noticeable tan developing. Appetite suppression becomes evident. Nausea diminishing substantially. | Type III-V skin may be ready for maintenance transition. |
| Week 3-4 | Significant tan for most skin types. Loading phase complete for types III+. Type I-II continuing to build. | Monitor moles closely. New freckles may appear. |
| Week 5-8 | Deep, sustained tan. All skin types on maintenance. Results plateau. | Reduce to maintenance frequency. |
| Post-cessation | Tan persists 4-8 weeks, fading gradually. | Can restart with a shorter loading phase. |
UV exposure note: MT-II stimulates melanin production at the cellular level, but UV radiation triggers additional melanin distribution and darkening in the skin. Brief sun exposure (10-15 minutes) during loading produces noticeably better results than MT-II alone. Extended sunbathing or deliberate sunburning is unnecessary and counterproductive.
Restarting after a break: If you have used MT-II before and take time off, the second loading phase is typically 30-50% shorter. Residual melanin and primed melanocytes respond faster to renewed stimulation.
Frequently Asked Questions
What is the standard Melanotan 2 dosage for beginners?
Start with 0.25 mg subcutaneously once daily. After 3-5 days, increase to 0.5 mg if tolerated. Continue daily during the loading phase (7-21 days depending on your Fitzpatrick skin type), then switch to 0.5 mg once or twice weekly for maintenance. Very fair skin (Type I) should start even lower at 0.1 mg.
How much bacteriostatic water do I add to a 10 mg vial?
Add 2 mL of bacteriostatic water for a concentration of 5 mg/mL. At this concentration, 0.1 mL on an insulin syringe (10 units) equals 0.5 mg of Melanotan 2, and 0.05 mL (5 units) equals 0.25 mg. Use our reconstitution calculator for exact measurements with any vial size.
Can I use Melanotan 2 nasal spray instead of injections?
Nasal spray has only 30-40% bioavailability compared to approximately 100% for subcutaneous injection. You need 2-3 times the dose, and visible results take 10-21 days longer to appear. Injection is significantly more effective and consistent. The Cleveland Clinic has issued warnings about nasal tanning sprays due to unpredictable absorption and dosing difficulty.
How do I stop the nausea from Melanotan 2?
Take fexofenadine 180 mg (Allegra) or loratadine 10 mg (Claritin) 45 minutes before injection. Start at 0.1-0.25 mg rather than 0.5 mg. Inject before bedtime to sleep through the worst of it. Avoid eating 1-2 hours before injection. Increase the dose by 0.1 mg every 2-3 days instead of jumping up quickly. Nausea affects 80-90% of users but typically resolves after 3-5 days.
Does Melanotan 2 work on fair skin (Fitzpatrick Type I-II)?
Yes, but fair skin requires longer loading periods (3-4 weeks instead of 1-2 weeks for darker skin types) and higher total cumulative doses (20-50 mg instead of 5-10 mg). MC1R receptor variants in very fair-skinned individuals are less responsive to melanocortin signaling, requiring more sustained stimulation to switch from pheomelanin to eumelanin production.
Is Melanotan 2 safe? Can it cause melanoma?
MT-II is not approved by any regulatory body. At least four published case reports associate melanoma with MT-II use, though no direct causal link is established (PubMed 24355990). MT-II stimulates the same cells (melanocytes) from which melanoma originates. Get a baseline full-body mole check from a dermatologist before starting and monitor monthly using the ABCDE criteria.
How long does a Melanotan 2 tan last after stopping?
The tan typically persists 4-8 weeks after the last injection, fading gradually as melanocytes turn over naturally. Maintenance doses of 0.5 mg once or twice weekly can sustain the color indefinitely. If you restart after a break, the loading phase is shorter because residual melanin and primed melanocytes respond faster.
What is the difference between Melanotan 1 and Melanotan 2?
Melanotan 1 (afamelanotide, brand name Scenesse) selectively targets MC1R for tanning with fewer side effects and is approved in the EU for erythropoietic protoporphyria. Melanotan 2 is non-selective, activating MC1R through MC5R, which adds libido enhancement and appetite suppression but also produces more side effects including nausea, spontaneous erections, and higher rates of new mole formation.
Can women use Melanotan 2?
Yes. Dosing protocols are identical regardless of sex. Women experience the same tanning, appetite suppression, and libido enhancement effects. The main additional consideration: MT-II should not be used during pregnancy or breastfeeding due to insufficient safety data.
Do I need UV exposure for Melanotan 2 to work?
Not strictly required, but 10-15 minutes of sun exposure during the loading phase significantly accelerates and deepens results. MT-II stimulates melanin production at the cellular level. UV radiation triggers additional melanin distribution and darkening in the visible skin layers. The two processes are complementary. Extended sunbathing or deliberate sunburning is not necessary and is not recommended.
Frequently Asked Questions
What is the standard Melanotan 2 dosage for beginners?
Start with 0.25 mg subcutaneously once daily. After 3 to 5 days, increase to 0.5 mg if tolerated. Continue daily during loading (7 to 21 days depending on skin type), then switch to 0.5 mg once or twice weekly for maintenance. Very fair skin (Type I) should start lower at 0.1 mg. Run the reconstitution calculator before drawing.
How much bacteriostatic water do I add to a 10 mg vial?
Add 2 mL of bacteriostatic water for a 5 mg/mL concentration. At this concentration, 0.1 mL on an insulin syringe (10 units) equals 0.5 mg of Melanotan II. For 0.25 mg draw 5 units. Confirm volumes with the reconstitution calculator if you mixed differently.
Can I use Melanotan 2 nasal spray instead of injections?
Nasal spray has only 30 to 40 percent bioavailability compared to roughly 100 percent for subcutaneous injection. You need 2 to 3 times the dose and results take 10 to 21 days longer. Injection is more effective and consistent. The Cleveland Clinic has issued warnings on nasal tanning sprays. For nasal-route pros and cons in other peptides, see the BPC-157 nasal spray guide.
How do I stop the nausea from Melanotan 2?
Take fexofenadine 180 mg (Allegra) or loratadine 10 mg (Claritin) 45 minutes before injection. Start at 0.1 to 0.25 mg, inject before bedtime, and avoid eating 1 to 2 hours prior. Nausea affects 80 to 90 percent of users but typically resolves after 3 to 5 days. For broader peptide side-effect management, see the peptide safety guide.
Does Melanotan 2 work on fair skin (Fitzpatrick Type I-II)?
Yes, but fair skin requires longer loading (3 to 4 weeks instead of 1 to 2 weeks) and higher cumulative doses (20 to 50 mg instead of 5 to 10 mg). MC1R receptor variants in fair-skinned individuals are less responsive to melanocortin signaling, so melanocytes need more sustained stimulation to switch from pheomelanin to eumelanin production. See the Melanotan II peptide profile for receptor detail.
Is Melanotan 2 safe? Can it cause melanoma?
MT-II is not approved by any regulatory body. At least four case reports associate melanoma with MT-II use (PMID: 24355990). No direct causal link is proven, but MT-II stimulates the same cells from which melanoma originates. Get a baseline mole check and self-screen monthly using ABCDE criteria. Review the peptide safety guide for full risk framing.
How long does a Melanotan 2 tan last after stopping?
The tan typically persists 4 to 8 weeks after the last injection, fading gradually as melanocytes turn over. Maintenance doses of 0.5 mg once or twice weekly sustain the color indefinitely. If you restart after a break, the loading phase is shorter because residual melanin and primed melanocytes respond faster. For storage during gaps, see how to store peptides.
What is the difference between Melanotan 1 and Melanotan 2?
Melanotan I (afamelanotide, brand Scenesse) selectively targets MC1R for tanning with fewer side effects and is EU-approved for erythropoietic protoporphyria. Melanotan II is non-selective across MC1R through MC5R, adding libido enhancement and appetite suppression but also more nausea and new-mole risk. For libido-only use cases, PT-141 is the targeted MC4R alternative.
Can women use Melanotan 2?
Yes. Dosing is identical regardless of sex. Women experience the same tanning, appetite suppression, and libido enhancement effects. The main extra consideration is that MT-II should not be used during pregnancy or breastfeeding due to insufficient safety data. For female-specific peptide context, see peptides for women over 40 and peptides for libido.
Do I need UV exposure for Melanotan 2 to work?
Not strictly required, but 10 to 15 minutes of sun exposure during loading significantly accelerates results. MT-II stimulates melanin production at the cellular level. UV radiation triggers additional melanin distribution and darkening in visible skin layers. The two processes are complementary. Extended sunbathing or deliberate sunburning is unnecessary and counterproductive. For receptor mechanism detail, see the Melanotan II profile.
The Bottom Line
Melanotan 2 dosing is not one-size-fits-all. Your Fitzpatrick skin type determines your starting dose, loading duration, total cumulative dose, and maintenance frequency. Type I skin may need 30-50 mg over 3-4 weeks. Type IV skin may need under 10 mg over 7-10 days.
The loading phase builds melanin reserves. The maintenance phase sustains them. Subcutaneous injection delivers close to 100% bioavailability. Nasal spray loses 60-70% of the peptide and produces slower, less consistent results.
Nausea is expected. It responds well to antihistamine pre-treatment and resolves within the first week for most users. Mole darkening is the mechanism working, not a complication, but it demands ongoing monitoring.
MT-II is not approved by any regulatory body. The melanoma risk, while unproven causally, is biologically plausible. A dermatologist baseline check before starting and monthly self-exams using the ABCDE criteria are non-negotiable precautions. For a broader overview of peptide safety considerations, see our peptide safety guide.
Use our reconstitution calculator for precise mixing, the peptide dosage chart for cross-referencing other compounds, and the interaction checker if you plan to stack MT-II with other peptides. Not sure which peptide is right for you? Try our peptide quiz for a personalized recommendation. For sourcing guidance, see where to buy peptides in 2026. For stacking strategies across multiple compounds, see our peptide stacking guide.
Related Articles: - Peptides for Libido - How to Store Peptides - Peptide Safety Guide - Getting Started with Peptides - Peptide Stacking Guide
Related Articles
5-Amino-1MQ Dosage: Protocols, Cycling, and Safety
5-Amino-1MQ dosage guide: 50-150 mg/day oral protocols, NNMT inhibition mechanism, cycle length, stacking options, and PubMed-cited research.
How to Microdose Semaglutide: Guide
Learn how to microdose semaglutide at 0.1-0.25 mg for appetite control with fewer side effects. Covers protocols, measuring small doses, evidence, and risks.
GHK-Cu + BPC-157 + TB-500 Blend Dosage
GHK-Cu + BPC-157 + TB-500 blend dosage chart for 70 mg and 42 mg vials. Per-injection breakdown in mcg, reconstitution steps, and cycling protocol.
KPV Peptide Dosage Chart: Oral, Injectable & Topical
KPV peptide dosage is 200-500 mcg/day. Full chart for oral (gut/IBD), injectable, topical, and nasal routes, plus the KPV + BPC-157 gut stack. PubMed-cited.