Cagrilintide is a long-acting amylin analogue that mimics a satiety hormone your pancreas releases after every meal. Developed by Novo Nordisk, it targets a fundamentally different hunger pathway than GLP-1 drugs like semaglutide or dual agonists like tirzepatide. When paired with semaglutide as CagriSema, the combination produced 22.7% mean body weight loss at 68 weeks in the Phase 3 REDEFINE 1 trial, with 60% of participants losing 20% or more of their starting weight.
That combination matters because single-pathway drugs appear to be approaching a ceiling. Semaglutide alone delivers roughly 15% weight loss. Tirzepatide reaches 22.5%. CagriSema matches tirzepatide's results by stacking two independent appetite pathways: amylin receptors in the hypothalamus and GLP-1 receptors in the brainstem. Novo Nordisk filed its New Drug Application for CagriSema in December 2025, with an FDA decision expected between late 2026 and mid-2027.
| Quick Reference | Details |
|---|---|
| Drug | Cagrilintide (AM833/NN9838) |
| Developer | Novo Nordisk |
| Class | Long-acting amylin and calcitonin receptor agonist |
| Combination product | CagriSema (cagrilintide + semaglutide 2.4 mg) |
| Half-life | 159-195 hours (6.6-8.1 days) |
| Peak plasma (tmax) | 24-72 hours post-injection |
| CagriSema weight loss | 22.7% at 68 weeks (REDEFINE 1) |
| Cagrilintide monotherapy | 11.8% at 68 weeks (Phase 3) |
| NDA filed | December 18, 2025 |
| FDA status | Under review (decision expected late 2026 to mid-2027) |
This article covers what cagrilintide is, how the amylin pathway drives weight loss, CagriSema clinical trial results, how it compares to tirzepatide and retatrutide, and when it might reach pharmacies.
This is educational content based on published clinical trials and peer-reviewed literature. Consult a healthcare provider before starting any medication.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
What Is Cagrilintide?
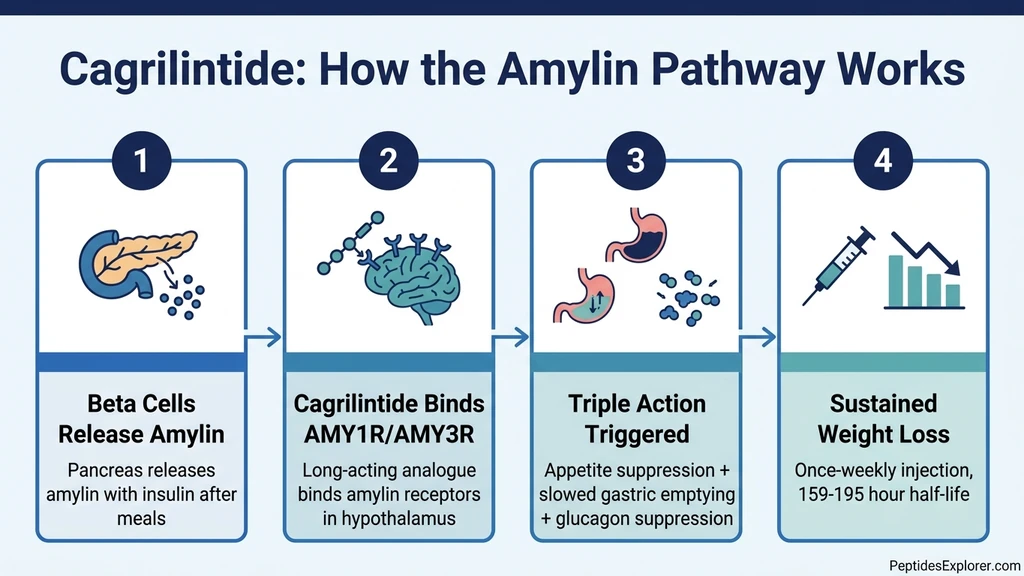
Cagrilintide (developmental codes AM833 and NN9838) is a synthetic, long-acting version of human amylin. Amylin is a 37-amino-acid peptide hormone co-secreted with insulin from pancreatic beta cells every time you eat. Natural amylin tells your brain you are full, slows the rate food leaves your stomach, and suppresses glucagon to keep blood sugar stable. The problem: natural amylin has a half-life of about 13 minutes. It disappears from your bloodstream almost immediately.
Novo Nordisk engineered cagrilintide with structural modifications inspired by salmon calcitonin, a related peptide with superior receptor binding properties. The result is a molecule with a half-life of 159 to 195 hours (6.6 to 8.1 days), reaching peak plasma concentration between 24 and 72 hours after subcutaneous injection (PMID 33894838). This pharmacokinetic profile supports once-weekly dosing. Cagrilintide binds reversibly to albumin in the blood, a design strategy Novo Nordisk also used for semaglutide, which extends its circulation time.
Cagrilintide activates multiple receptor subtypes: AMY1R, AMY2R, AMY3R, and the calcitonin receptor (CTR). These amylin receptor subtypes form when the calcitonin receptor associates with receptor activity-modifying proteins (RAMPs 1-3). A 2025 study published in eBioMedicine confirmed that cagrilintide's weight-lowering effects depend specifically on AMY1R and AMY3R, using knockout mouse models that eliminated each receptor subtype individually (PMID 40609154). Structural data from Nature Communications revealed how cagrilintide binds to G-protein-coupled active states of all four receptor types, explaining its broad pharmacological profile (DOI: 10.1038/s41467-025-58680-y).
The critical distinction from current obesity drugs: cagrilintide works through a brain pathway that semaglutide and tirzepatide do not touch. GLP-1 drugs suppress appetite primarily through receptors in the gut, brainstem, and area postrema. Cagrilintide acts on amylin receptors concentrated in the hypothalamus and area postrema, affecting both homeostatic hunger (your body's energy-balance system) and hedonic hunger (the reward-driven desire to eat palatable food). This mechanistic independence is why combining cagrilintide with semaglutide produces more weight loss than either drug alone.
How the Amylin Pathway Drives Weight Loss
Amylin is sometimes called the "forgotten satiety hormone" because GLP-1 drugs have dominated obesity pharmacology for the past decade. But amylin was actually the first peptide hormone shown to reduce food intake independently of GLP-1 signaling. Pramlintide (Symlin), a first-generation amylin analogue approved in 2005 for diabetes, demonstrated the pathway's therapeutic potential but required multiple daily injections and produced modest effects.
Cagrilintide solved the pharmacokinetic limitations. When it binds to AMY1R and AMY3R receptors in the area postrema and hypothalamus, three overlapping mechanisms activate.
Appetite Suppression Through Satiety Centers
The hypothalamus receives amplified "full" signals after meals. This reduces both the quantity of food consumed per meal and the frequency of food-seeking behavior between meals. Amylin receptors in the area postrema and nucleus tractus solitarius (NTS) mediate this effect (PMID 36883831). Unlike GLP-1 drugs that primarily reduce homeostatic hunger, cagrilintide also modulates the hedonic reward pathways that drive cravings for high-calorie, palatable foods. Early clinical observations suggest CagriSema patients report less "food noise" compared to patients on GLP-1 drugs alone.
Slowed Gastric Emptying
Food moves through the stomach more slowly after cagrilintide administration, extending the duration of post-meal fullness. This mechanism overlaps with GLP-1's effect on gastric motility but acts through a separate receptor pathway. The result: when cagrilintide and semaglutide are combined, gastric emptying slows through two independent signals, which likely contributes to CagriSema's superior weight loss results compared to either drug alone.
Glucagon Suppression
Cagrilintide reduces postprandial glucagon secretion, preventing blood sugar spikes after meals. This is particularly relevant for patients with type 2 diabetes. In the REDEFINE 2 trial, 73.5% of CagriSema-treated patients with type 2 diabetes reached HbA1c levels at or below 6.5%, compared to 15.9% on placebo (PMID 40544432). For context on how GLP-1 drugs manage blood sugar through a parallel mechanism, see our guide on semaglutide before and after results.
Potential Lean Mass Preservation
One emerging area of research involves body composition during weight loss. Amylin's satiety mechanism differs from GLP-1's mechanism at the neural level, which may influence the ratio of fat loss to lean mass loss. Preclinical data suggest amylin agonists could lead to favorable body composition outcomes compared to GLP-1 monotherapy, though this has not yet been confirmed in large human trials. Lean mass preservation during rapid weight loss is a major clinical concern. For more on this topic, see our analysis of tirzepatide and muscle loss.
CagriSema: The Combination That Changes the Equation
CagriSema is not a single molecule. It is two separate drugs administered together: cagrilintide 2.4 mg and semaglutide 2.4 mg, both given as once-weekly subcutaneous injections. Novo Nordisk developed CagriSema as a fixed-combination product with a single injection device, though the Phase 3 REDEFINE trials used co-administered separate injections.
The rationale is straightforward: amylin and GLP-1 suppress appetite through independent receptor systems. Blocking one pathway leaves the other intact. Blocking both simultaneously produces additive weight loss that neither drug achieves alone. A network meta-analysis (PMC11642503) quantified this: CagriSema produces approximately 9% additional weight loss compared to semaglutide alone, confirming that the amylin pathway delivers meaningful incremental benefit on top of GLP-1 therapy.
This dual-pathway approach distinguishes CagriSema from tirzepatide, which targets GLP-1 and GIP receptors. Tirzepatide uses two incretin pathways. CagriSema uses one incretin pathway (GLP-1) plus one non-incretin pathway (amylin). The clinical implications are significant: patients who plateau on GLP-1/GIP therapy may respond to GLP-1/amylin therapy, and vice versa.
For patients considering the switch between existing drugs, our semaglutide to tirzepatide switching guide covers the practical details.
REDEFINE Clinical Trial Results
Novo Nordisk tested CagriSema through the REDEFINE clinical programme, which includes four major trials. The results establish cagrilintide as a serious contender in the obesity drug landscape. For the latest peptide research statistics, including how these results compare to other investigational drugs, see our 2026 data roundup.
REDEFINE 1: CagriSema in Adults Without Diabetes
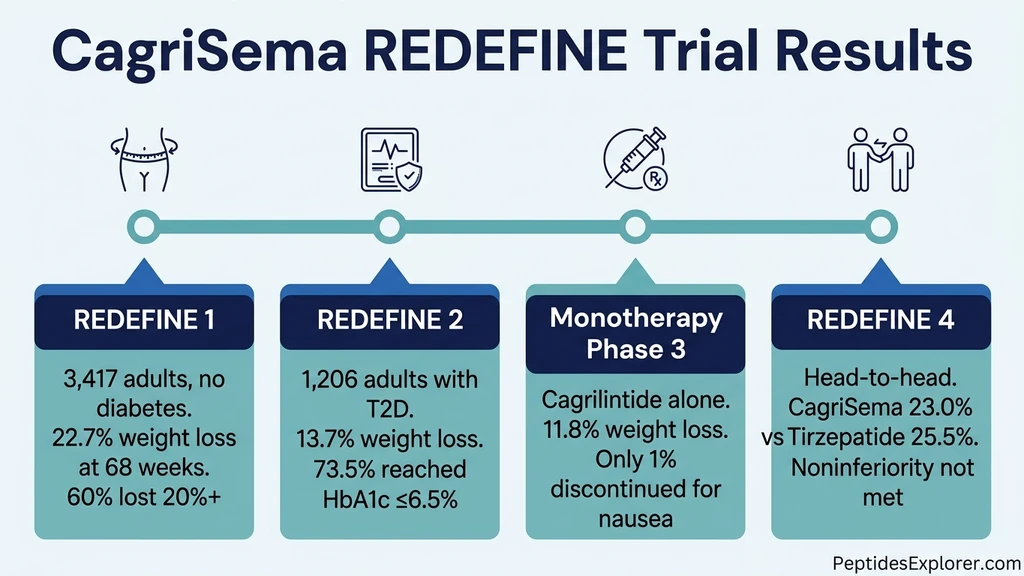
The headline trial enrolled 3,417 adults with obesity (BMI 30+) or overweight with comorbidities (BMI 27+) who did not have type 2 diabetes. Participants were randomized to receive CagriSema, semaglutide alone, cagrilintide alone, or placebo for 68 weeks. Results were published in the New England Journal of Medicine (DOI: 10.1056/NEJMoa2502081).
| Outcome | CagriSema | Semaglutide alone | Cagrilintide alone | Placebo |
|---|---|---|---|---|
| Mean weight loss (%) | 22.7% | ~15% | ~10% | 3.0% |
| Achieved 5%+ loss | 91.9% | 31.5% | ||
| Achieved 20%+ loss | 60% | Not reported | ||
| Achieved 30%+ loss | 23% | Not reported | ||
| Reached BMI below 30 | 50.7% | 10.2% |
The 22.7% mean weight loss is the primary result. For a 250-pound person, that translates to roughly 57 pounds at 68 weeks. The proportion achieving 20%+ loss (60%) is particularly notable because it suggests most patients experience clinically transformative results, not just modest reductions.
REDEFINE 2: CagriSema in Type 2 Diabetes
REDEFINE 2 enrolled 1,206 participants with type 2 diabetes, BMI 27+, and HbA1c between 7% and 10%. Participants were randomized 3:1 to CagriSema or placebo for 68 weeks across 12 countries. Results were published in the New England Journal of Medicine (PMID 40544432).
| Outcome | CagriSema | Placebo |
|---|---|---|
| Mean weight loss | 13.7% | 3.4% |
| HbA1c at or below 6.5% | 73.5% | 15.9% |
| GI adverse events | 72.5% | 34.4% |
The 13.7% weight loss in patients with type 2 diabetes follows the established pattern: patients with diabetes consistently lose less weight on anti-obesity drugs than patients without diabetes. The glycemic benefit was substantial. Nearly three-quarters of CagriSema patients reached an HbA1c target of 6.5%, a result that positions CagriSema as both an obesity and a diabetes drug.
REDEFINE 4: Head-to-Head Against Tirzepatide
REDEFINE 4 was the most anticipated trial. It compared CagriSema directly against tirzepatide 15 mg in 809 adults with BMI 30+ (mean baseline weight 114.2 kg). This was an open-label design, meaning participants knew which drug they received.
| Outcome | CagriSema | Tirzepatide 15 mg |
|---|---|---|
| Weight loss (efficacy estimand) | 23.0% | 25.5% |
| Weight loss (treatment regimen estimand) | 20.2% | 23.6% |
The primary endpoint was noninferiority of CagriSema to tirzepatide, and this endpoint was not met. Tirzepatide produced numerically greater weight loss by both statistical estimands. Two important caveats: the open-label design can influence adherence and outcome reporting, and the drugs work through entirely different mechanisms (amylin + GLP-1 versus GLP-1 + GIP).
For a deeper comparison of the GLP-1/GIP approach, see our retatrutide vs tirzepatide analysis.
Cagrilintide Monotherapy Phase 3
A separate Phase 3 trial tested cagrilintide as a standalone drug, independent of CagriSema. Results were reported in September 2025.
| Outcome | Cagrilintide | Placebo |
|---|---|---|
| Mean weight loss | 11.8% (12.5 kg) | 2.3% (2.5 kg) |
| Achieved 15%+ loss | 31.6% | 4.7% |
| Discontinued due to nausea | 1.0% | Not reported |
The 11.8% result confirms that the amylin pathway alone drives meaningful weight loss. The 1.0% nausea discontinuation rate is noteworthy because GLP-1 drugs typically produce higher discontinuation rates from GI side effects. Based on these results, Novo Nordisk launched the RENEW Phase 3 programme in Q4 2025 to pursue regulatory approval for standalone cagrilintide. If approved, it would become the first pure amylin-based obesity treatment.
Cagrilintide Dosage and Titration
The standard cagrilintide titration follows the REDEFINE trial protocol. Dose escalation occurs every four weeks to minimize gastrointestinal side effects, the same gradual approach used for semaglutide and tirzepatide.
| Week | Cagrilintide Dose | Administration |
|---|---|---|
| 1-4 | 0.25 mg | Once weekly, subcutaneous |
| 5-8 | 0.5 mg | Once weekly, subcutaneous |
| 9-12 | 1.0 mg | Once weekly, subcutaneous |
| 13-16 | 1.7 mg | Once weekly, subcutaneous |
| 17+ | 2.4 mg (maintenance) | Once weekly, subcutaneous |
When used as CagriSema, the semaglutide component follows its own titration schedule simultaneously, reaching the same 2.4 mg maintenance dose used in Wegovy. Both injections are given on the same day. Novo Nordisk is developing a single-injection device for the commercial product.
An alternative rapid titration schedule used in some clinical trials starts at 0.6 mg and escalates every two weeks to reach a 4.5 mg maintenance dose. This higher dose produced 10.8% weight loss in the Phase 2 dose-finding study (PMID 34798060), but the 2.4 mg dose was selected for Phase 3 development as the optimal balance of efficacy and tolerability.
For detailed dosing information on the semaglutide component, see our cagrilintide weight loss and dosage guide. For semaglutide-specific dosing, use our semaglutide dosage calculator.
How Cagrilintide Compares to Other Weight Loss Drugs
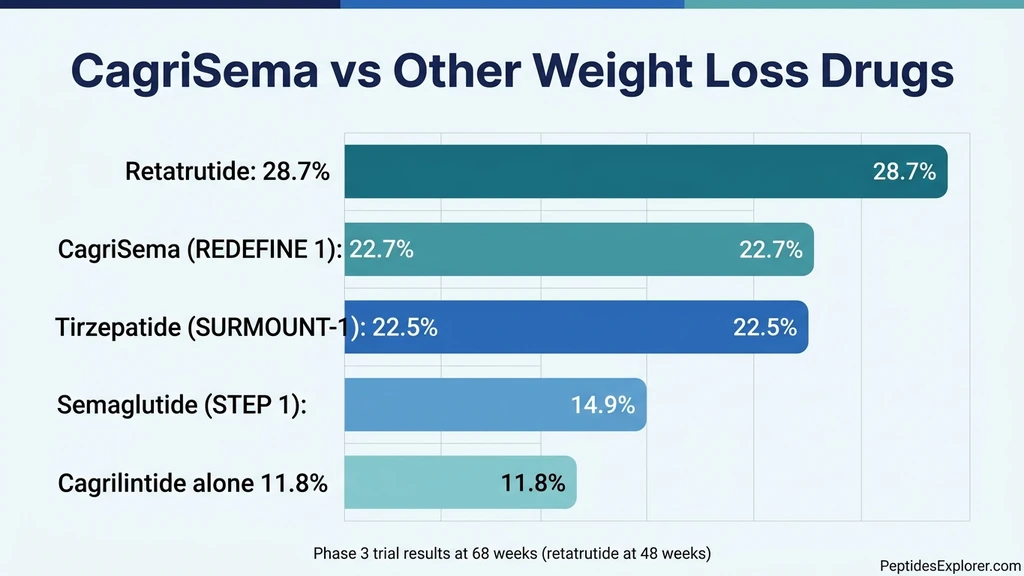
The obesity drug landscape has expanded rapidly since semaglutide's approval. Each drug targets different receptor combinations, which matters for patients who do not respond adequately to one mechanism.
| Drug | Mechanism | Receptors | Max Weight Loss | FDA Status |
|---|---|---|---|---|
| Semaglutide (Wegovy) | GLP-1 agonist | GLP-1R | ~15% | Approved |
| Tirzepatide (Zepbound) | Dual incretin agonist | GLP-1R + GIPR | ~22.5-25.5% | Approved |
| CagriSema | GLP-1 + amylin agonist | GLP-1R + AMY1R/AMY3R | ~22.7% | NDA filed |
| Cagrilintide (mono) | Amylin agonist | AMY1R/AMY3R/CTR | ~11.8% | Phase 3 (RENEW) |
| Retatrutide | Triple agonist | GLP-1R + GIPR + GCGR | ~28.7% | Phase 3 |
| Survodutide | Dual agonist | GLP-1R + GCGR | ~19% | Phase 3 |
Several patterns emerge from this comparison. First, combination approaches consistently outperform single-pathway drugs. Second, CagriSema and tirzepatide achieve similar results through entirely different mechanisms. Third, retatrutide's triple-agonist approach produces the highest weight loss reported in any Phase 2 trial, though its Phase 3 programme is still ongoing.
For patients currently on GLP-1 therapy who are considering their options, the practical difference between CagriSema and tirzepatide may come down to tolerability and individual response rather than aggregate weight loss numbers. Some patients respond better to amylin-based appetite suppression than incretin-based approaches. Having drugs with different mechanisms gives physicians more therapeutic flexibility.
For a complete overview of peptide-based weight loss options, see our guide on best peptides for weight loss.
Side Effects and Safety Profile
Cagrilintide and CagriSema produce side effects consistent with other injectable weight loss drugs. Gastrointestinal events dominate the adverse event profile and are dose-dependent, peaking during titration and resolving at stable maintenance doses.
| Side Effect | CagriSema (REDEFINE 1) | Cagrilintide Alone (Phase 3) | Semaglutide Alone |
|---|---|---|---|
| Any GI event | 79.6% | Lower | ~74% |
| Nausea | ~45% | ~20-47% (dose-dependent) | ~44% |
| Diarrhea | ~25% | Moderate | ~30% |
| Vomiting | ~22% | Low | ~24% |
| Constipation | ~15% | Moderate | ~24% |
| Injection site reactions | ~10% | ~5% | ~5% |
| Discontinued for nausea | Not separately reported | 1.0% | ~3-5% |
The 79.6% GI adverse event rate for CagriSema sounds alarming in isolation, but context matters. Most events were classified as mild and transient. The pattern mirrors semaglutide and tirzepatide: nausea peaks during the dose-escalation phase (weeks 1-16) and diminishes significantly once patients reach stable maintenance doses.
Cagrilintide monotherapy shows a notably favorable tolerability profile. Only 1.0% of participants discontinued due to nausea in the Phase 3 trial, which is lower than typical discontinuation rates for GLP-1 drugs. This suggests the amylin pathway itself produces less severe GI distress than GLP-1 signaling. The higher GI rates seen with CagriSema likely reflect the additive effect of combining both pathways.
Serious adverse events were rare across all REDEFINE trials. Acute pancreatitis, gallbladder events, and renal events occurred at rates similar to those seen with semaglutide monotherapy. No new safety signals emerged from adding cagrilintide to semaglutide.
For guidance on managing GI side effects with currently available drugs, see our articles on relieving semaglutide nausea and tirzepatide constipation.
The Amylin Drug Class: Beyond Cagrilintide
Cagrilintide is the most advanced amylin analogue in development, but it is not the only one. The success of the REDEFINE programme has validated amylin as a therapeutic target, prompting multiple pharmaceutical companies to develop their own amylin-based drugs.
Pramlintide (Symlin), approved in 2005, was the first synthetic amylin analogue. It requires two to three daily injections and produces modest weight loss of 1-2 kg, far less than cagrilintide's 12.5 kg. Pramlintide proved the concept; cagrilintide made it clinically relevant through its extended half-life.
The broader trend in obesity pharmacology is moving toward multi-pathway approaches. Retatrutide targets three incretin-related pathways (GLP-1, GIP, and glucagon receptors). CagriSema targets one incretin pathway plus amylin. Future combinations may incorporate even more pathways. The research community increasingly views obesity as a condition requiring multi-receptor intervention, similar to how hypertension and HIV are treated with combination therapy.
This trajectory has direct implications for patients. Within the next two to three years, physicians may have five or more mechanistically distinct obesity drugs to choose from. Patients who do not respond to one mechanism will have alternatives. For the latest data on investigational peptides, see our 2026 peptide statistics roundup.
FDA Approval Timeline and Availability
Novo Nordisk filed its New Drug Application (NDA) with the FDA for CagriSema on December 18, 2025. The submission includes data from REDEFINE 1, REDEFINE 2, and supporting trials. Under standard FDA review (10-12 months from acceptance), approval could come in late 2026 to early 2027. If the FDA grants priority review, the timeline could accelerate to late 2026.
Key milestones:
- December 18, 2025: NDA filed for CagriSema (weight management indication)
- Q1 2026: FDA acceptance of NDA expected
- Late 2026 to mid-2027: FDA decision (standard review timeline)
- Q4 2025: RENEW Phase 3 programme launched for standalone cagrilintide
- 2027-2028: Potential RENEW programme readout and separate NDA for cagrilintide monotherapy
If approved, CagriSema would be the first combination obesity drug targeting both amylin and GLP-1 pathways. It would compete directly with tirzepatide (Zepbound) and expand the treatment options for patients who have not achieved adequate results on existing medications.
Pricing is not yet announced, but Novo Nordisk's pricing for Wegovy ($1,349/month list price) provides a reference point. CagriSema, as a combination product with potentially superior efficacy, will likely carry a premium. Insurance coverage negotiations will determine real-world accessibility for most patients.
For patients seeking treatment now, semaglutide and tirzepatide are the FDA-approved options. Use our semaglutide dosage calculator for dosing guidance on currently available medications.
Frequently Asked Questions
What is cagrilintide and how does it work?
Cagrilintide is a long-acting amylin analogue developed by Novo Nordisk. It mimics amylin, a hormone co-released with insulin from pancreatic beta cells after meals. By binding to AMY1R and AMY3R receptors in the hypothalamus and area postrema, cagrilintide suppresses appetite, slows gastric emptying, and reduces glucagon secretion (PMID 40609154). Its half-life of 159-195 hours allows once-weekly injection. See our cagrilintide dosage guide for titration details.
What is CagriSema?
CagriSema is a combination of cagrilintide 2.4 mg and semaglutide 2.4 mg given as once-weekly injections. It targets two independent appetite pathways: amylin receptors (via cagrilintide) and GLP-1 receptors (via semaglutide). In the REDEFINE 1 trial, CagriSema produced 22.7% mean weight loss at 68 weeks, with 60% of participants losing 20% or more. Novo Nordisk filed for FDA approval in December 2025.
How much weight can you lose on cagrilintide?
Cagrilintide monotherapy produced 11.8% weight loss (12.5 kg) at 68 weeks in the Phase 3 trial. Combined with semaglutide as CagriSema, weight loss nearly doubles to 22.7% at 68 weeks. For a 250-pound person, 22.7% equals roughly 57 pounds. See our guide on best peptides for weight loss for how this compares to other options.
Is cagrilintide FDA-approved?
No. As of April 2026, cagrilintide is not FDA-approved. Novo Nordisk filed an NDA for CagriSema on December 18, 2025. A standard FDA review takes 10-12 months, putting the expected decision between late 2026 and mid-2027. Standalone cagrilintide is in the separate RENEW Phase 3 programme. Currently available FDA-approved options include semaglutide and tirzepatide.
How does cagrilintide differ from semaglutide and tirzepatide?
Cagrilintide targets amylin receptors (AMY1R/AMY3R) in the hypothalamus. Semaglutide targets GLP-1 receptors in the gut and brainstem. Tirzepatide targets both GLP-1 and GIP receptors. These are three distinct pathways. Cagrilintide uniquely affects hedonic hunger (cravings), not just homeostatic hunger. This mechanistic independence is why CagriSema (cagrilintide + semaglutide) outperforms semaglutide alone.
How does CagriSema compare to tirzepatide in clinical trials?
In the head-to-head REDEFINE 4 trial, CagriSema produced 23.0% weight loss versus 25.5% for tirzepatide 15 mg. CagriSema did not meet the noninferiority endpoint. The drugs work through different mechanisms: amylin + GLP-1 (CagriSema) versus GLP-1 + GIP (tirzepatide). See our retatrutide vs tirzepatide comparison for more on these distinctions.
What are the side effects of cagrilintide?
GI events are most common: nausea (20-47% depending on dose), diarrhea, vomiting, and constipation. Most are mild and transient, peaking during dose escalation. Only 1.0% of participants discontinued cagrilintide monotherapy due to nausea, lower than typical GLP-1 discontinuation rates. For managing similar side effects, see our guide on relieving semaglutide nausea.
Can cagrilintide be used without semaglutide?
Yes. Cagrilintide monotherapy produced 11.8% weight loss at 68 weeks in a dedicated Phase 3 trial, compared to 2.3% for placebo. Novo Nordisk launched the RENEW Phase 3 programme in Q4 2025 to pursue separate regulatory approval for standalone cagrilintide. It may become an option for patients who cannot tolerate GLP-1 drugs. For current weight loss peptide options, several alternatives exist.
When will CagriSema be available?
Novo Nordisk filed the NDA on December 18, 2025. Under standard FDA review, a decision is expected between late 2026 and mid-2027. If approved, CagriSema would be the first amylin/GLP-1 combination obesity drug. For patients seeking treatment now, the FDA-approved options are semaglutide and tirzepatide. Use our semaglutide dosage calculator for current dosing guidance.
What is the difference between amylin and GLP-1?
Amylin is released from pancreatic beta cells alongside insulin after meals. It acts primarily on AMY1R and AMY3R receptors in the hypothalamus and area postrema, reducing both physical hunger and food cravings. GLP-1 is released from intestinal L-cells and acts on GLP-1 receptors in the gut and brainstem, primarily slowing gastric emptying and reducing homeostatic hunger. They are complementary pathways, which is why CagriSema targets both. Learn more about the GLP-1 pathway in our semaglutide before and after guide.
The Bottom Line
Cagrilintide represents the first clinically validated drug to harness the amylin pathway for obesity treatment at scale. As a standalone molecule, it produced 11.8% weight loss with lower GI discontinuation rates than GLP-1 drugs. Combined with semaglutide as CagriSema, it reached 22.7% weight loss at 68 weeks in REDEFINE 1, placing it in the same efficacy tier as tirzepatide. The head-to-head REDEFINE 4 trial showed a modest advantage for tirzepatide (25.5% vs. 23.0%), but the drugs attack obesity through fundamentally different receptor systems.
The REDEFINE 2 trial confirmed dual benefits in type 2 diabetes: 13.7% weight loss and 73.5% of patients reaching HbA1c at or below 6.5% (PMID 40544432). Novo Nordisk filed for FDA approval in December 2025. The RENEW programme will determine whether standalone cagrilintide earns its own regulatory path.
What matters most is that amylin agonism is now a proven pathway. Patients who do not respond adequately to GLP-1 or GLP-1/GIP drugs will have a mechanistically distinct option. The obesity pharmacology toolkit is expanding from single-target to multi-target combination therapy, and cagrilintide is at the center of that shift.
For patients seeking treatment today, semaglutide and tirzepatide remain the FDA-approved options. Use our semaglutide dosage calculator for dosing guidance, explore semaglutide before and after results, or read our retatrutide vs tirzepatide comparison for a broader view of the pipeline.
This is educational content based on published Phase 3 clinical trial data, peer-reviewed research, and Novo Nordisk regulatory filings. It is not medical advice. Consult a qualified healthcare provider before starting or changing any medication.
Related Articles
CagriSema Dosing Chart (Cagrilintide + Semaglutide)
CagriSema dosing chart and titration. Cagrilintide 0.25-2.4 mg + semaglutide 0.25-2.4 mg, weekly. Not FDA-approved (NDA filed Dec 2025). REDEFINE-1 trial data.
Cagrilintide: Weight Loss & Dosage
Cagrilintide weight loss and dosage guide. CagriSema produced 22.7% loss at 68 weeks. Titration from 0.25mg to 2.4mg weekly. Not yet FDA-approved.
Is Tresiba a GLP-1?
Tresiba (insulin degludec) is NOT a GLP-1. It is an ultra-long-acting basal insulin analog. Key differences from Ozempic, Mounjaro, and other GLP-1 agonists.
Is Qsymia a GLP-1?
Is Qsymia a GLP-1? No. It is an oral mix of phentermine (appetite suppressant) and topiramate. Mechanism, weight loss, and GLP-1 comparison.