Someone built a weight loss protocol that stacks three different mechanisms in one patient: amylin (cagrilintide), GLP-1 (inside tirzepatide), and GIP (also inside tirzepatide). The typical compounded protocol runs cagrilintide from 0.25 mg to 2.4 mg per week, titrated in parallel with tirzepatide from 2.5 mg to 15 mg per week. Both injected on the same day, 4-week titration steps, never escalated together in the same week. This combination is not FDA-approved. The only published human data is from CagriSema trials (cagrilintide + semaglutide, not tirzepatide), and no registered Phase 3 exists for cagrilintide + tirzepatide. Everything you are reading about this stack comes from anecdotal compounding-pharmacy protocols, not controlled trials.
| Quick Reference | Detail |
|---|---|
| FDA approval status | Not approved as a combination |
| Published human trial data | None for this combination (CagriSema = cagri + semaglutide, separate drug) |
| Cagrilintide dose range | 0.25 to 2.4 mg/week |
| Tirzepatide dose range | 2.5 to 15 mg/week |
| Injection frequency | Both weekly, same day, separate injections |
| Titration interval | 4 weeks per step |
| Rule | Never escalate both drugs in the same week |
| Primary side effect | Cumulative GI (nausea 30-50%, diarrhea 20-30%) |
| Origin | Extrapolated from CagriSema protocols + tirzepatide monotherapy |
The logic behind stacking these two is straightforward but unproven in controlled trials. Cagrilintide targets amylin receptors (hunger suppression via hypothalamus), while tirzepatide targets both GLP-1 (gastric emptying, satiety, insulin) and GIP (adipocyte metabolism). Combining them in theory hits three independent pathways of appetite and energy balance. In practice, side effect accumulation may outweigh the additive benefit. For our comprehensive cagrilintide monotherapy guide including the CagriSema trial data, see cagrilintide weight loss dosage.
This is educational content. Consult a healthcare provider before combining any medications.
Get your custom peptide protocol:
- Tailored to your body and goals
- Precise dosing and cycle length
- Safe stacking combinations
- Backed by peer-reviewed studies
- Ready in under 2 minutes
Why People Stack Cagrilintide With Tirzepatide
Both cagrilintide and tirzepatide treat obesity. Each works through different receptors. The rationale for combining them rests on the triple-mechanism hypothesis: if semaglutide + cagrilintide (CagriSema) produced 20.4% weight loss in REDEFINE 1 over 68 weeks (Garvey et al., 2025), substituting tirzepatide for semaglutide should in theory match or exceed that result because tirzepatide is a dual GLP-1/GIP agonist that achieved 20.9% weight loss at the 15 mg dose in SURMOUNT-1 (Jastreboff et al., 2022).
Mechanism map: - Cagrilintide is a long-acting amylin analogue engineered with a fatty acid albumin-binding chain to extend half-life to support once-weekly dosing (Kruse et al., 2021). It binds amylin and calcitonin receptors, suppresses glucagon, slows gastric emptying, and reduces food intake via central hypothalamic signaling. - Tirzepatide is a dual GLP-1 and GIP receptor agonist. GLP-1 activation slows gastric emptying, increases satiety, and promotes insulin secretion. GIP activation increases adipocyte lipolysis and may reduce the nausea burden compared to GLP-1 alone. - The combined theory: hunger suppression from amylin, slowed gastric emptying from GLP-1, fat-cell metabolism signaling from GIP, and insulin sensitization across the board.
The missing evidence: no Phase 3 trial. The only published human trial data on this combination does not exist. The closest approximation is REDEFINE 1 (cagri + sema) and SURMOUNT-1 (tirzepatide alone), neither of which tested the cagri + tirz pair head-to-head. Anyone selling you a cagri + tirz protocol is extrapolating from these adjacent trials.
The current real-world picture: compounding pharmacies that offer both drugs sometimes market a co-administration protocol. This is a regulatory grey zone. Tirzepatide is FDA-approved and patented (Eli Lilly). Cagrilintide NDA was filed December 2025 and has not been approved. Compounded cagrilintide sits in the same "research peptide" category as retatrutide did before approval. For the legal and supply context, see FDA peptide crackdown and are peptides legal.
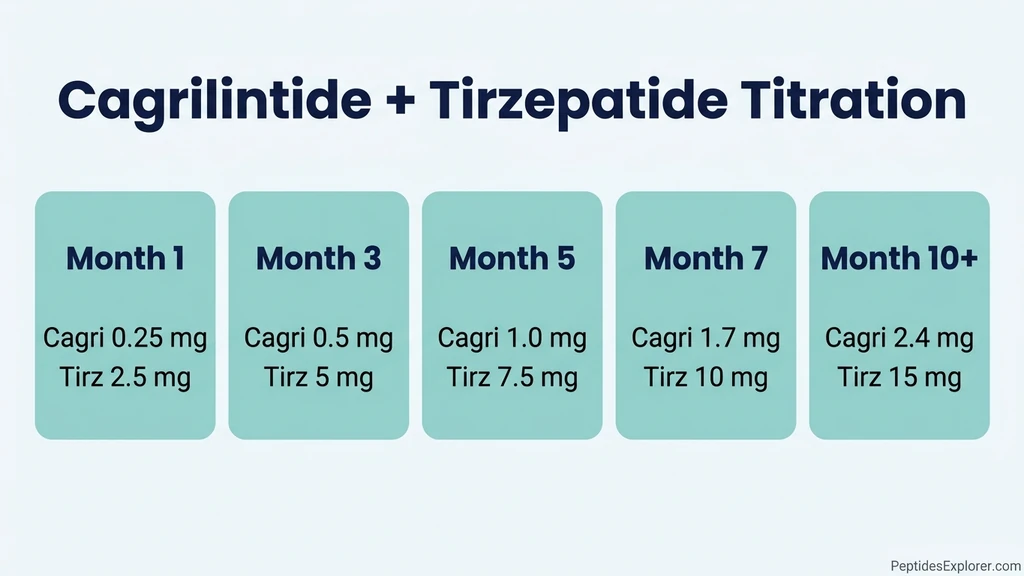
Standard Titration Protocol (Compounded)
The most widely-used compounded protocol stacks the two weekly injections on the same day, escalates each on alternating months, and holds at maintenance doses indefinitely.
| Month | Cagrilintide weekly | Tirzepatide weekly | Notes |
|---|---|---|---|
| 1 | 0.25 mg | 2.5 mg | Starter. Both drugs at lowest dose. |
| 2 | 0.25 mg | 5 mg | Tirzepatide bumps to 5 mg. Hold cagri. |
| 3 | 0.50 mg | 5 mg | Cagri bumps. Hold tirz. |
| 4 | 0.50 mg | 7.5 mg | Tirz bumps. Hold cagri. |
| 5 | 1.0 mg | 7.5 mg | Cagri bumps. Hold tirz. |
| 6 | 1.0 mg | 10 mg | Tirz bumps. Hold cagri. |
| 7 | 1.7 mg | 10 mg | Cagri approaches target. |
| 8 | 1.7 mg | 12.5 mg | Tirz approaches target. |
| 9 | 2.4 mg | 12.5 mg | Cagri at target. |
| 10+ | 2.4 mg | 15 mg | Both at target or stop one step below based on tolerance. |
The core rule: never escalate both drugs in the same week. Each dose increase carries a 2 to 3-week window of elevated GI side effects. Stacking two increases in one week produces cumulative nausea that most patients cannot tolerate, and many discontinue the stack entirely.
Titration flexibility: Slower is always safer. If you reach a dose level that produces 5 to 7 days of nausea, hold that level for an extra 4 weeks before the next step. The CagriSema trial documented 79.6% GI adverse events at similar dose levels in the cagri + sema combination; tirzepatide monotherapy already carries 30 to 50% nausea. The combined rate is not published but logically sits in the 60 to 80% range during titration.
Injection day logistics: Both drugs are given subcutaneously, once weekly. Use two separate injection sites (for example, left abdomen for one, right thigh for the other). Never mix them in the same syringe. See best injection sites for tirzepatide and the tirzepatide dosage calculator for site rotation guidance.
How This Differs From CagriSema
CagriSema is Novo Nordisk's investigational drug: cagrilintide co-formulated with semaglutide, injected as a single weekly dose. Cagrilintide + tirzepatide is a compounded stack, not an FDA-approved product.
Key differences:
| Attribute | CagriSema (cagri + sema) | Cagri + Tirz stack |
|---|---|---|
| Approval status | NDA filed Dec 2025 | None |
| Formulation | Single co-formulated vial | Two separate drugs |
| Mechanism | Amylin + GLP-1 | Amylin + GLP-1 + GIP |
| Trial data | REDEFINE 1 (22.7% WL at 68 weeks) | None published |
| Weekly injection | One shot | Two shots |
| Evidence strength | Phase 3 completed | Anecdotal |
Why the GIP angle may (or may not) matter: Tirzepatide's GIP receptor activation is thought to contribute to its superior efficacy over semaglutide in SURMOUNT-1 vs STEP-1 (22.5% vs 14.9% mean weight loss). If that mechanism additively stacks with cagrilintide's amylin effect, the cagri + tirz combination could theoretically produce greater weight loss than CagriSema. But this is speculation until a head-to-head trial runs.
Why the stack might be worse, not better: Side effects from GLP-1, GIP, and amylin all converge on similar GI pathways. Nausea receptors stack nonlinearly. A 30% nausea rate from tirzepatide + 40% from cagrilintide does not equal 70%; the combined rate often exceeds 80% at peak titration, driving discontinuation. CagriSema's co-formulation at lower effective doses may have a better side effect profile than the stack.
For a complete rundown of cagrilintide monotherapy and CagriSema data, see cagrilintide weight loss dosage. For the tirzepatide-only protocol, see how to inject tirzepatide and tirzepatide dosage chart in units. For the survodutide alternative that also uses a triple-receptor mechanism, see survodutide vs tirzepatide.
Side Effect Profile and Safety Limits
The cumulative side effect burden is the single biggest reason to think twice about this stack.
Documented side effects (extrapolated from CagriSema + tirzepatide data): - Nausea: 40 to 60% during titration, declining to 10 to 20% at maintenance - Diarrhea: 20 to 30% at peak dose levels - Constipation: 15 to 25% (alternates with diarrhea for many patients) - Vomiting: 10 to 20% during dose increases - Injection site reactions: 5 to 10% per site - Pancreatitis (rare): same risk as either drug alone; not demonstrably additive - Thyroid C-cell tumors: boxed warning on tirzepatide applies
What to stop the stack for immediately: - Persistent severe abdominal pain (rule out pancreatitis) - Vomiting for more than 48 hours - Signs of severe dehydration - Neck mass or dysphagia (rule out thyroid pathology) - New onset mood changes or suicidal ideation (both drugs carry this warning)
Dose de-escalation rule: If you cannot tolerate the planned titration step, drop one level, hold 4 additional weeks, and retry. Never skip a dose of either drug at maintenance; a missed cagrilintide dose for more than 10 days essentially restarts the titration clock for that drug (tirzepatide has a 5-day half-life and is more forgiving).
Drug interactions: Both drugs slow gastric emptying, which can dramatically reduce absorption of oral medications taken within 2 hours of injection. This is particularly problematic for oral contraceptives, thyroid medications, and warfarin. See tirzepatide drug interactions for the complete interaction list that applies here.
For the full safety framework on GLP-1 stacking, see can tirzepatide cause anxiety and tirzepatide long-term side effects.
When This Stack Makes Sense (And When It Does Not)
Reasonable candidates: - Patients who have plateaued on tirzepatide monotherapy at 15 mg for 6+ months and still have significant weight to lose - Patients under specialist care who understand this is an unapproved combination - Patients willing to tolerate slower titration than either drug alone would require
Poor candidates: - Someone starting their first GLP-1 agent (start with tirzepatide or semaglutide monotherapy first) - Anyone with BMI under 30 without metabolic comorbidities (risk/benefit does not justify a triple-agent stack) - History of pancreatitis, medullary thyroid carcinoma, or MEN-2 syndrome (both drugs contraindicated) - Pregnancy, breastfeeding, or planning conception within 2 months (both drugs contraindicated) - Anyone seeking this because online content called it the "next level". Marketing is not evidence
What the stack does not do: - Accelerate weight loss beyond what tirzepatide alone produces (unproven) - Reduce side effects compared to either drug alone (it increases them) - Replace the need for lifestyle change, protein intake, resistance training - Work without consistent weekly dosing
If plateau is your concern, first optimize the current protocol. Common fixes: increase protein to 1.6 to 2.0 g/kg lean body mass, add resistance training 3x weekly, ensure 7 to 9 hours of sleep, and audit hidden calorie sources. See why am I not losing weight on tirzepatide for the full plateau checklist before adding a second drug.
Frequently Asked Questions
Is cagrilintide + tirzepatide FDA-approved?
No. Neither drug is approved for use in combination. Tirzepatide is FDA-approved as monotherapy (Mounjaro, Zepbound). Cagrilintide NDA was filed December 2025 as part of CagriSema (cagri + semaglutide); it has no standalone approval. The cagri + tirz combination is compounded only and has no Phase 3 trial data. See cagrilintide weight loss dosage for the approval timeline.
What is the typical dose of cagrilintide when stacked with tirzepatide?
0.25 mg weekly to start, titrating up every 4 weeks through 0.5, 1.0, 1.7, and 2.4 mg as tolerated. The target maintenance dose is 2.4 mg weekly, matching the CagriSema trial protocol. Never escalate cagrilintide in the same week you escalate tirzepatide; side effects stack.
Can I mix cagrilintide and tirzepatide in the same syringe?
No. Use two separate syringes, two separate injection sites, same day. Mixing can destabilize either peptide and changes absorption kinetics unpredictably. Standard practice is two abdominal injections at least 2 inches apart, or abdomen + thigh. See best injection sites for tirzepatide.
Will stacking cagrilintide with tirzepatide produce more weight loss than tirzepatide alone?
Unknown. No controlled trial has tested this. The CagriSema trial (cagri + sema) produced 22.7% weight loss at 68 weeks vs 14.9% for semaglutide alone. If the same additive logic applies to tirzepatide (22.5% alone), the stack could theoretically produce 25 to 30% weight loss, but this is extrapolation. For published results, see cagrilintide weight loss dosage and retatrutide vs tirzepatide for alternative triple-mechanism drugs.
What side effects should I expect with the cagri + tirz stack?
Nausea (40 to 60% during titration), diarrhea (20 to 30%), constipation (15 to 25%), and vomiting (10 to 20%). Side effects peak during dose escalation and decline at maintenance. Combined GI side effect rates are higher than either drug alone. See tirzepatide long-term side effects and can tirzepatide cause anxiety for monoagent baselines.
How is this different from CagriSema?
CagriSema is cagrilintide + semaglutide co-formulated in one injection; it has Phase 3 data and an NDA filed. Cagri + tirzepatide is compounded from two separate drugs; no Phase 3 data. The triple-agonist advantage of cagri + tirz (adding GIP mechanism) is theoretical until tested. For CagriSema specifics, see cagrilintide weight loss dosage.
Do I need higher doses of either drug when stacking?
No. Use the standard titration for each drug individually (cagrilintide to 2.4 mg, tirzepatide to 10 to 15 mg). Higher doses do not produce proportional benefit and dramatically increase side effects. If a combination of 2.4 mg cagri + 15 mg tirz does not produce results, a different mechanism class (like retatrutide) is more useful than higher doses. See retatrutide dosage.
What should I do if I cannot tolerate the stack?
Drop back to the previous tolerated dose level and hold 4 weeks before retrying the escalation. If you still cannot tolerate it, stop cagrilintide and continue tirzepatide monotherapy. Tirzepatide is the primary effective drug; cagrilintide is the addition. Dropping the addition is always preferable to dropping both. For reintroduction protocols after a stop, see how to inject tirzepatide.
The Bottom Line
Cagrilintide + tirzepatide is an unapproved combination with no Phase 3 trial data. The dosing protocols floating in compounding-pharmacy communities are extrapolated from CagriSema (which uses semaglutide, not tirzepatide) and from tirzepatide monotherapy. The triple-mechanism rationale is theoretically sound: amylin + GLP-1 + GIP stacks three independent appetite pathways. Whether this converts to better outcomes than tirzepatide alone is unknown.
If you and your prescriber decide to run this stack, the key rules are simple. Escalate one drug per 4-week window, never both. Use separate injection sites. Expect cumulative GI side effects especially during the first 4 to 6 months. Drop cagrilintide first if tolerance becomes a problem; tirzepatide is the primary effective agent.
Most people who think they need this stack actually need plateau troubleshooting instead. Protein intake, resistance training, sleep, and hidden calorie audits resolve more stalled weight loss than adding a second drug. Before adding cagrilintide, see why am I not losing weight on tirzepatide and tirzepatide maintenance dose after weight loss. If plateau persists despite optimization, retatrutide (which natively combines GLP-1, GIP, and glucagon) may be a more evidence-backed alternative; see retatrutide vs tirzepatide.
Related Articles: - Cagrilintide Weight Loss Dosage - Tirzepatide Dosage Chart in Units - Retatrutide vs Tirzepatide - Survodutide vs Tirzepatide - Why Am I Not Losing Weight on Tirzepatide - How to Inject Tirzepatide
Related Articles
CagriSema Dosing Chart (Cagrilintide + Semaglutide)
CagriSema dosing chart and titration. Cagrilintide 0.25-2.4 mg + semaglutide 0.25-2.4 mg, weekly. Not FDA-approved (NDA filed Dec 2025). REDEFINE-1 trial data.
Cagrilintide: Weight Loss & Dosage
Cagrilintide weight loss and dosage guide. CagriSema produced 22.7% loss at 68 weeks. Titration from 0.25mg to 2.4mg weekly. Not yet FDA-approved.
BPC-157 Dosage Per Body Weight (Chart)
BPC-157 dosage per body weight: 1.6-10 mcg/kg. Full chart for 120-280 lbs, injection vs oral doses, gender data, and why most practitioners prescribe flat 250-500 mcg.
How Much Sermorelin Per Day: Complete Dosage Guide
Sermorelin dosage: 200-500 mcg/day subcutaneous before bedtime. Starting dose, titration, gender-specific protocols, and IGF-1 monitoring.